Abstract
Multiple and mixed valvular heart disease (MMVD) is defined as the combination of stenotic or regurgitant lesions occurring on ≥2 cardiac valves (ie, multiple valvular heart disease [VHD]) or the combination of stenotic and regurgitant lesions on the same valve (ie, mixed VHD). This condition is frequent in clinical routine, with a notable shift in epidemiology over recent years, marked by a decline in rheumatic heart disease and an increase in degenerative causes. Even if echocardiography stands as the cornerstone for diagnosing VHD, practicians need to remain cautious because some echocardiographic parameters might be invalid in the context of MMVD. Considering echocardiography's pitfalls, MMVD needs to be assessed using multiparametric perspective, including cardiovascular magnetic resonance, cardiac computed tomography, biomarkers, and the assessment of comorbidities. Multimodality imaging seems to be a promising approach to assess patient's prognosis, by providing a precise analysis of cardiac remodeling and an accurate evaluation of MMVD severity. Due to the heterogeneity of this clinical scenario, studies are typically conducted in single centers with a limited number of patients. Therefore, there is a lack of dedicated guidelines regarding the severity assessment and the clinical decision-making for these MMVD patients. The aim of this review is to provide a state-of-the-art using the available evidence on the management of MMVD considering the interactions between different valve lesions, the diagnostic pitfalls, and the strategies that should be considered including the role of multimodality imaging in the presence of MMVD.
This is a visual representation of the abstract.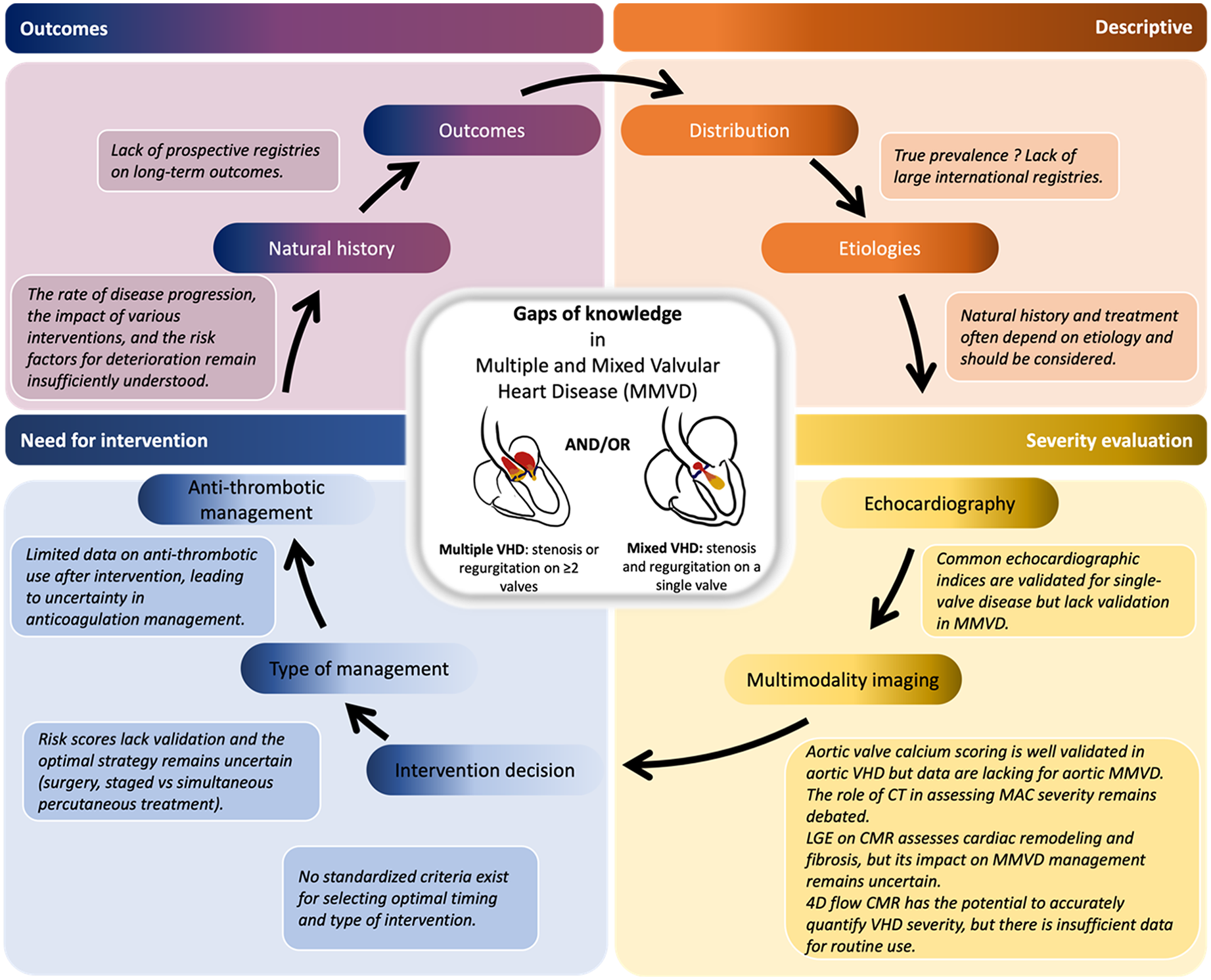
Keywords
Key points
Transthoracic echocardiography is the first-line tool for the assessment of VHD. However, some echocardiographic parameters are unreliable in MMVD.
Cardiac CT plays an important role in AS not only by assessing the aortic valve calcium score but also by measuring the aorta and describing the valve morphology.
2D-flow CMR is a powerful technique for assessing valvular regurgitation but is more limited for valvular stenosis. 4D-flow CMR, through direct quantification of the regurgitant jet, appears to be promising in MMVD. However, further studies are needed to confirm its usefulness.
Ventricular remodeling is associated with the presence of LGE on CMR, which is associated with increased mortality and cardiovascular outcomes in patients with single VHD. The value of LGE in MMVD should be evaluated.
There is a severe lack of evidence regarding diagnostic tools, prognostic elements, and treatment recommendations for patients with MMVD. In this context, EACVI-MMVD, a real-life registry of MMVD patients, is currently conducted to provide unique descriptive and prognostic data. This study will be followed by the MULTIVALVE registry that will use a standardized cardiovascular imaging protocol.
Introduction
Multiple and mixed valvular heart disease (MMVD) is a complex pathology with high morbidity and mortality rates.1,2 Multiple and mixed valvular heart disease is defined as the combination of stenotic or regurgitant lesions occurring on at least 2 cardiac valves (also called multiple valvular heart disease [VHD]) or the combination of stenotic and regurgitant lesions on the same valve (also called mixed VHD). Although this is a highly prevalent condition, there remains a significant lack of data and recommendations, for both diagnosis and treatment. 2 The complexity of MMVD is mainly due to the difficulty to assess its severity, with unreliable echocardiographic parameters, and the multitude of factors influencing his investigation (loading conditions, timing of their onset, ejection fraction…). 3 Thus, many diagnostic challenges remain to be overcome for better therapeutic guiding.
The aim of this review is to provide a state-of-the-art about MMVD, considering etiologies and pathophysiology, multimodality imaging, and treatments.
General Principles
Etiology
Rheumatic heart disease
Despite a big decline in industrialized countries in the past decades,4,5 rheumatic heart disease (RHD) remains the most common type of VHD worldwide with approximately 40.5 million of people affected, primarily impacting middle- and low-income countries. The peak prevalence of RHD is between 20 and 29 years and after 15 years old, women have a higher prevalence. 6 Rheumatic heart disease disproportionately affects the most vulnerable and marginalized populations, highlighting the urgent need to improve access to antibiotics and enhance sanitary conditions. Additionally, active efforts are underway to develop Group A Streptococcus vaccines. 7
Degenerative VHD
By contrast, the burden of degenerative valve disease is constantly increasing in developed countries due to the increase in life expectancy (Figure 1). 8 In the EURObservational Research Programme Valvular Heart Disease II survey, degenerative cause was the most frequent etiology (>60%) of multiple VHD, followed by RHD (<20%). 8 Indeed, in this aging population, a degenerative and calcified aortic valve can coexist with a variable degree of calcifications of the mitral annulus. 9
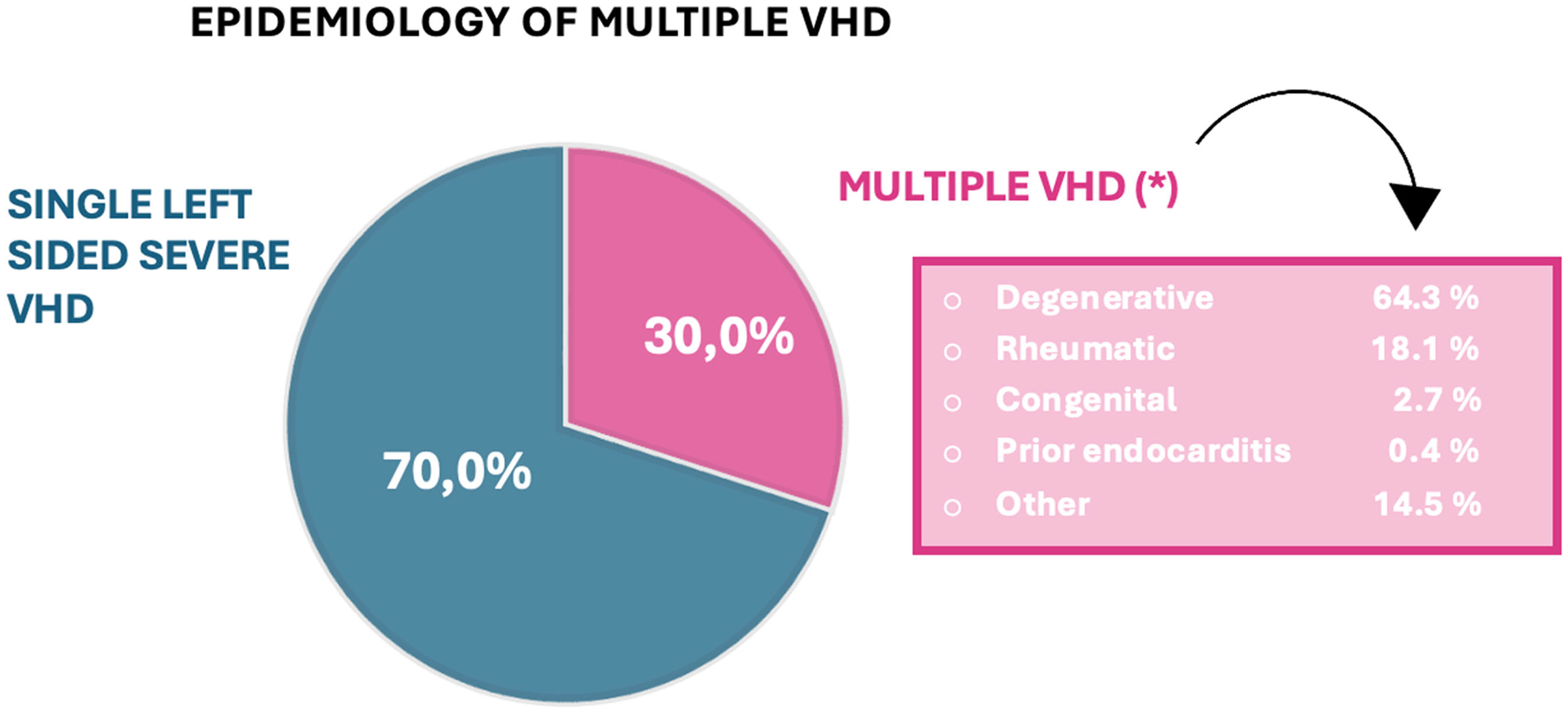
Epidemiology of Multiple Valvular Heart Disease (VHD) (modified from Tribouilloy et al 7 ).
Other etiologies
Other causes of acquired MMVD include the sequelae of infective endocarditis, 10 thoracic radiotherapy, 11 adverse effects of some medications such as ergot-derived agonists or anorectic agents, 12 or Ehlers-Danlos syndromes (Table 1). 13 Bicuspid aortic valve is one of the main causes of mixed aortic valve disease (MAVD) combining aortic regurgitation (AR) and aortic stenosis (AS). 14 Attention should be paid to cases where MMVD is associated with congenital heart disease, as this is a distinct condition requiring specific recommendations.
Etiologies of Multiple and Mixed Valvular Heart Disease.
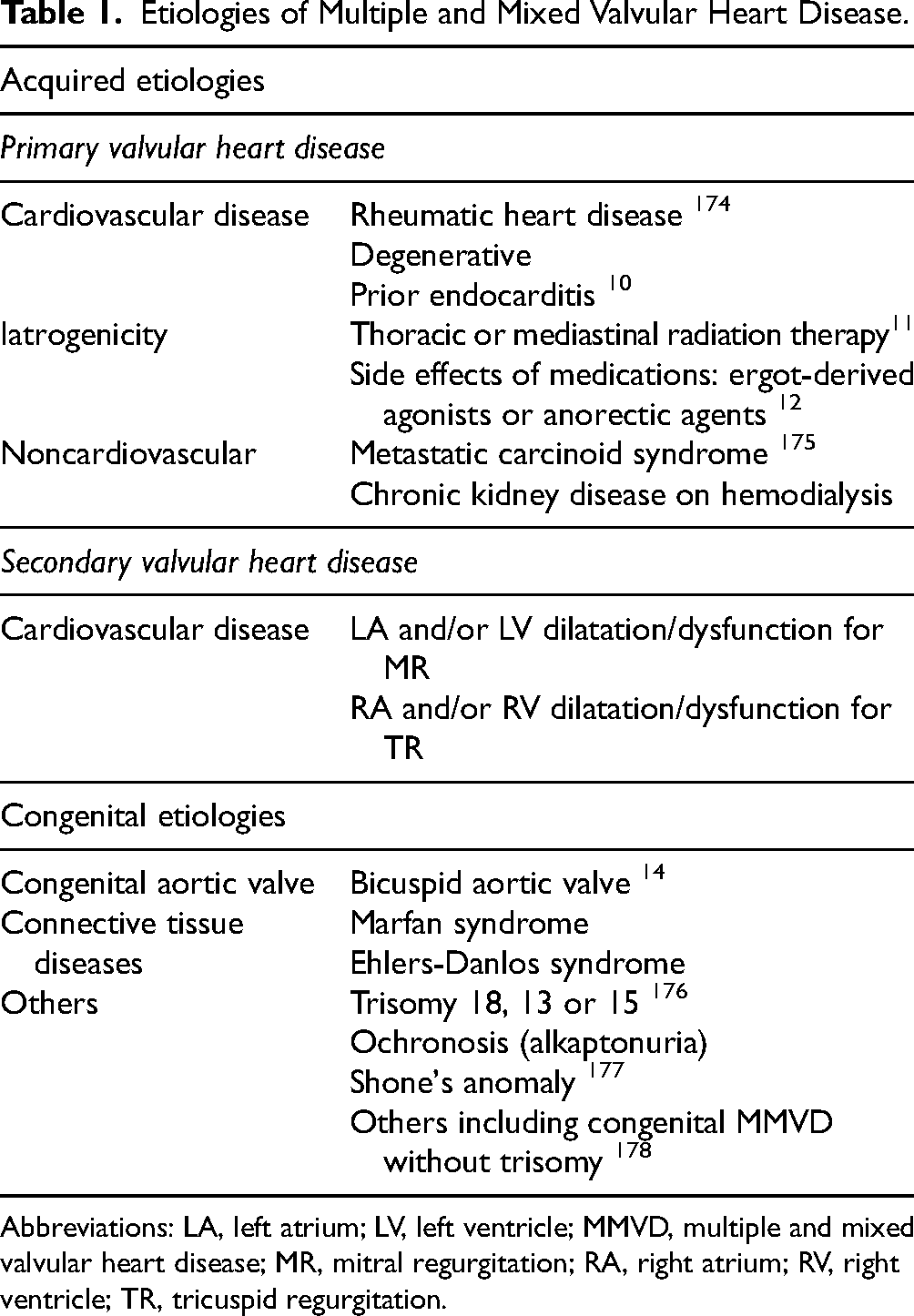
Abbreviations: LA, left atrium; LV, left ventricle; MMVD, multiple and mixed valvular heart disease; MR, mitral regurgitation; RA, right atrium; RV, right ventricle; TR, tricuspid regurgitation.
Finally, secondary mitral regurgitation (MR) and tricuspid regurgitation (TR) are frequently linked to left ventricle (LV) and right ventricle (RV) dilatation and/or dysfunction, respectively.15,16 Among patients referred for transcatheter aortic valve implantation (TAVI), approximately 20% have at least moderate secondary MR. 17 In addition, primary and secondary etiologies of MMVD can coexist. 18
Pathophysiology of MMVD
The hemodynamic consequences and the clinical presentation of a VHD are influenced by a concomitant lesion on the same valve (mixed VHD) or on another valve (multiple VHD). Pathophysiology is complex, depending on affected valves, time of onset, severity of each individual valvular defect, pathogenesis, loading conditions, and overall ventricular function. 19 For example, secondary MR can result from leaflet tethering and mitral annular dilatation due to increased afterload in AS, or from left ventricular dilatation in AR. 3 Similarly, secondary TR can be classified as ventricular, associated with primary or secondary MR, or atrial, associated with concomitant atrial fibrillation, which is common in VHD.
Adaptive mechanisms and ventricular remodeling help to maintain equilibrium until heart failure and first symptoms develop. A key pathophysiological feature is that some valvular lesions may exacerbate or, on the contrary, alleviate a concomitant VHD. For example, the clinical expression of AR is different with mitral stenosis (MS), which limits volume overload, and with MR, which may exacerbate it.13,20 Similarly, the treatment of a valvular lesion can influence another, as seen in the treatment of AS, which often leads to a reduction in the severity of MR.17,18
Global Evaluation and Management
As already mentioned, the hemodynamic impact of MMVD on blood flow, ventricular dimensions, and function can significantly influence the diagnostic process. When valvular lesions exhibit comparable severity, the presence of a proximal VHD may attenuate the full expression of a concurrent distal VHD. For example, in a scenario where a patient presents with severe mitral and tricuspid VHD, the clinical manifestations related to the right-sided lesion will likely be more pronounced. 13 Physical examination findings may be misleading, particularly regarding the timing and intensity of murmurs detected upon auscultation. In this review, we will detail the importance of a multiparametric approach in echocardiography for accurately quantifying stenoses or regurgitation. 21 Besides echocardiography, multimodality imaging has become essential in the evaluation of MMVD, 21 through 2-dimensional (2D) flow and 4-dimensional (4D) flow sequences in cardiovascular magnetic resonance (CMR) for the evaluation of regurgitant lesions, and the quantification of the aortic valve calcifications as marker of AS severity or to detect an aortic root dilatation using cardiac computed tomography (CT). 22
Cardiovascular Multimodality Imaging
Cardiovascular Imaging in MMVD Patients
Echocardiography
Echocardiography is the main tool for assessing VHD, especially given its accessibility and routine use in clinical practice. It plays a crucial role in the diagnosis, assessment of the etiology (Table 1), severity and clinical impact of the disease, as well as in the evaluation of the feasibility of surgical or percutaneous interventions. Moreover, echocardiography is essential in guiding treatment decisions and determining the optimal timing for intervention.
While echocardiography stands as the cornerstone for assessing VHD, some echo techniques routinely employed to assess stenosis or regurgitation have been validated primarily in patients with single-VHD, potentially limiting their validity in the context of MMVD (Tables 2 and 3).
Multimodality Imaging in Multiple Valvular Heart Diseases.
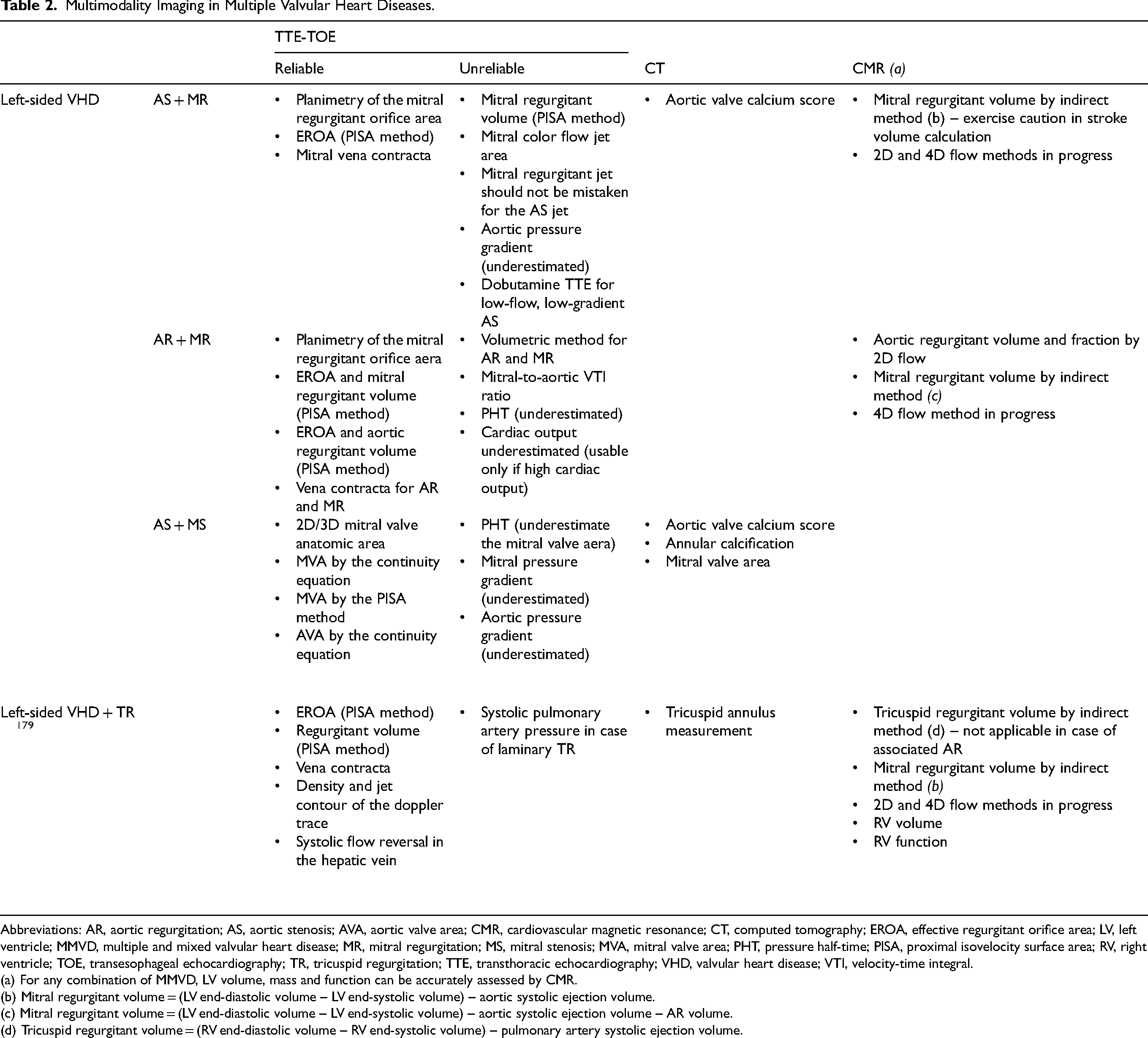
Abbreviations: AR, aortic regurgitation; AS, aortic stenosis; AVA, aortic valve area; CMR, cardiovascular magnetic resonance; CT, computed tomography; EROA, effective regurgitant orifice area; LV, left ventricle; MMVD, multiple and mixed valvular heart disease; MR, mitral regurgitation; MS, mitral stenosis; MVA, mitral valve area; PHT, pressure half-time; PISA, proximal isovelocity surface area; RV, right ventricle; TOE, transesophageal echocardiography; TR, tricuspid regurgitation; TTE, transthoracic echocardiography; VHD, valvular heart disease; VTI, velocity-time integral.
(a) For any combination of MMVD, LV volume, mass and function can be accurately assessed by CMR.
(b) Mitral regurgitant volume = (LV end-diastolic volume – LV end-systolic volume) – aortic systolic ejection volume.
(c) Mitral regurgitant volume = (LV end-diastolic volume – LV end-systolic volume) – aortic systolic ejection volume – AR volume.
(d) Tricuspid regurgitant volume = (RV end-diastolic volume – RV end-systolic volume) – pulmonary artery systolic ejection volume.
Multimodality Imaging in Mixed Valvular Heart Diseases.
Abbreviations: AR, aortic regurgitation; AS, aortic stenosis; AVA, aortic valve area; CMR, cardiovascular magnetic resonance; CT, computed tomography; EROA, effective regurgitant orifice area; LV, left ventricle; MMVD, multiple and mixed valvular heart disease; MR, mitral regurgitation; MS, mitral stenosis; MVA, mitral valve area; PHT, pressure half-time; PISA, proximal isovelocity surface area; RV, right ventricle; TOE, transesophageal echocardiography; TTE, transthoracic echocardiography; VTI, velocity-time integral.
(a) For any combination of MMVD, LV volume and function can be accurately assessed by CMR.
(b) Mitral regurgitant volume = (LV end-diastolic volume – LV end-systolic volume) – aortic systolic ejection volume.
To avoid measurement errors, the assessment can be supplemented with transesophageal echocardiography (TOE) and 3D echocardiography methods, which provide a more reliable analysis of volumes and anatomy. However, it represents a real technical challenge and a recent international survey showed a relatively low level of skills and knowledge in TOE among young cardiologists in this situation. 23
Cardiac CT
Cardiac CT has become a crucial tool for assessing both AS severity and preprocedural work-up of TAVI. Aortic stenosis severity can be assessed by quantifying valvular calcium burden using the Agatston score. 24 Women exhibit less calcification but more fibrosis than men at equivalent levels of AS severity, necessitating distinct thresholds for defining severe AS: >1200 Agatston units (AU) for women, and >2000 AU for men. 25 However, the assessment of AS severity should not solely rely on one parameter. In RHD or in cardiac amyloidosis without severe calcifications, these thresholds may not accurately reflect the severity of AS. 26 Mitral annulus calcification (MAC) is common in patients with MMVD, being observed in 50% of patients with concomitant severe AS. 27 Although there is currently no validated standardized scoring system for its quantification, 28 cardiac CT appears to be a relevant tool for defining the severity of MAC and predicting valve embolization/migration during valve-in-MAC procedures. 29
Cardiovascular Magnetic Resonance
Cardiovascular magnetic resonance uniquely allows for direct measurement of flow using the “phase contrast” method, bypassing the complex equation-based calculations required by echocardiography. It also provides flexibility in the selection of imaging plane positioning, independently of probe placement or of the limitations imposed by the acoustic windows that can restrict echocardiography. Direct measurement of regurgitation volume in AR, performed through 2D-flow measurements at the aortic root, is generally unaffected by concomitant aortic or MS, or MR. However, in cases of concurrent AS, the measurement of aortic flow can be underestimated due to intravoxel signal dephasing caused by the stenosis jet.30,31 Direct phase-contrast imaging at the mitral valve for measuring forward and backward flow is not recommended to assess MR. 32 The preferred indirect method for calculating the MR volume employs the following formula: regurgitant volume = (LV end-diastolic volume – LV end-systolic volume) – aortic systolic ejection volume. This approach remains valid in the presence of AR but the presence of AS may compromise the accuracy of this method in determining systolic ejection volume. 32 Notably, CMR using 2D-flow is not a good method to assess the severity of valvular stenosis, due to the slick image slice, partial volume effects, lower temporal resolution compared to Doppler echo, and signal loss due to turbulence. 33 Indeed, several studies have shown the risk of underestimation of valvular stenoses.34,35
In centers with local expertise, 2D flow can be replaced by 4D-flow to measure transvalvular flow. The latter is a relatively novel CMR technique, which offers time-resolved 3-dimensional (3D) imaging and enables the measurement of blood flow within a 3D volume in a single acquisition, covering the entire heart.36,37 This allows for the quantification of flow across all 4 valves using the same 4D-flow dataset, which may be highly beneficial in assessing MMVD 38 as no assumptions on regurgitant jet morphology are required. However, current literature in 4D flow CMR is mainly related to single valve disease, and there are limited data on the specific additional value of 4D-flow compared to 2D-flow in this context. 39 Cardiovascular magnetic resonance is currently recommended for single VHD when transthoracic echocardiography (TTE) data are unreliable (inadequate echocardiographic quality or discrepant results, eccentric, multiple, and late-systolic regurgitant jets) and for assessing LV/RV volumes and fibrosis. Although CMR appears to reliably assess valvular regurgitation in complex cases such as MMVD (grade I recommendation), 25 its use is currently limited by its accessibility.
Interpretation of Cardiovascular Imaging for Each Combination of MMVD
Aortic stenosis and MR
Aortic stenosis with MR is the most common MMVD scenario, accounting for 54% of left-sided multiple VHD. 8 As with any VHD, the initial assessment is made by TTE. However, the increased afterload resulting from AS complicates the assessment of MR severity. While regurgitant volume and the area of the color flow jet are anticipated to be higher, the effective mitral regurgitant orifice and vena contracta are less affected (Table 2). The advent of CMR using 2D and 4D flow to measure regurgitant volume is a promising method.32,40 However, 2D and 4D flow may also be susceptible to inaccuracies in the setting of AS and requires further investigation.
Mitral regurgitation influences the clinical presentation of AS by reducing forward flow and contributing to a low-flow state. The transaortic pressure gradient is often low despite a small aortic valve area (AVA), resulting in a low-flow low-gradient AS.3,18 Current guidelines recommend dobutamine stress echocardiography to assess low-flow low-gradient AS. 25 However, when resulting from MR, low-flow low-gradient AS is often associated with preserved left ventricular ejection fraction, and dobutamine stress TTE is not recommended in these cases; secondly, inasmuch as even if there is contractile reserve, there will be no significant effect on forward stroke volume. In this setting, cardiac CT can be used to assess the aortic valve calcium score which is correlated with the degree of AS (Figure 2). 25
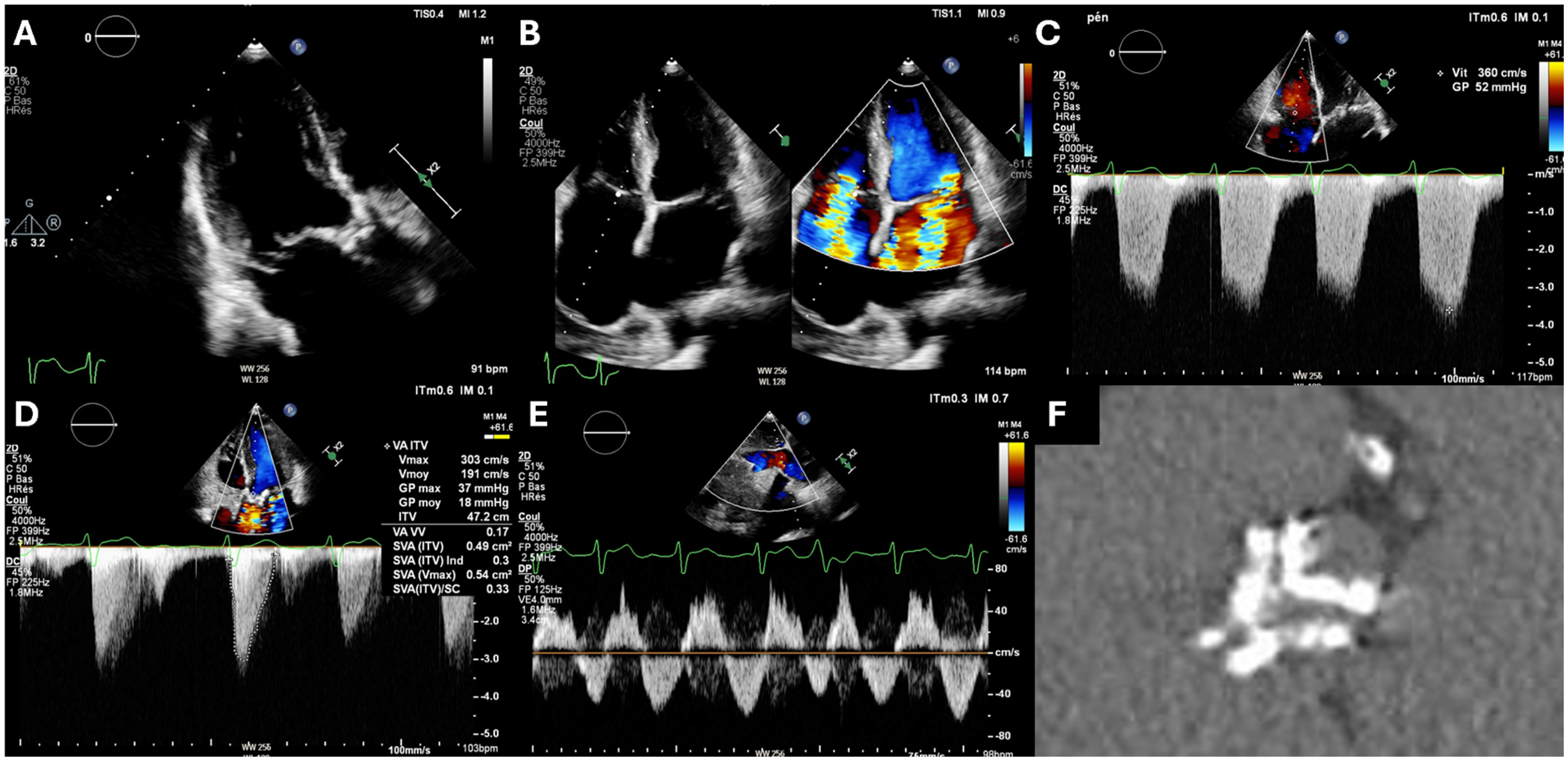
Association of Aortic Stenosis with Mitral and Tricuspid Regurgitation. Clinical context: a 73-year-old female with degenerative aortic stenosis, and mitral and secondary tricuspid regurgitation. (A) Three-chamber transthoracic echocardiography (TTE) showing a calcified aortic valve and P2 prolapse. (B) Four-chamber view with and without color Doppler showing enlargement of the left and right atria, prolapse of the posterior mitral valve with mitral regurgitation and a large vena contracta and secondary tricuspid regurgitation with a large vena contracta. (C) Severe pulmonary hypertension estimated at 55 mm Hg + right atrial pressure. (D) Severe low-gradient aortic valve stenosis (peak jet velocity of 3 m/s and aortic valve area estimated at 0.49 cm2). (E) Reversal of subhepatic vein flow in favor of severe tricuspid regurgitation. (F) Noncontrast computed tomography (CT) scan showing a heavily calcified tricuspid aortic valve (aortic valve calcium score 2300 Agatston units [AU]).
Aortic Regurgitation and MR
Although the diagnosis and quantification of associated AR/MR severity are primarily echocardiography-based, the quantitative assessment of both valve diseases remains challenging (Table 2). Whereas clinicians frequently use the pressure half-time (PHT) for assessing the AR severity, PHT is less reliable in the presence of MR due to the altered LV diastolic properties. Furthermore, the semiquantitative assessment of the severity of isolated MR by the mitral to aortic velocity-time integral ratio cannot be used in the presence of significant AR. 41
Cardiovascular magnetic resonance could be very useful to assess the AR/MR severity (Figure 3). Indeed, it can directly quantify regurgitant volume and fraction of AR using 2D-flow measurements at the aortic root. This method is not affected by the coexisting MR, which is evaluated by subtracting the systolic (forward) flow above the aortic valve from the LV stroke volume.30,31 The MR severity can also be assessed using an indirect method for calculating the regurgitation volume with the following formula: regurgitant volume = (LV end-diastolic volume – LV end-systolic volume) – aortic systolic ejection volume – AR volume. 32 This approach to quantify MR severity remains valid in the presence of AR. Left ventricle dilatation or impaired function are strong indicators of a poor prognosis, 42 and CMR is the gold standard for their assessment. 43
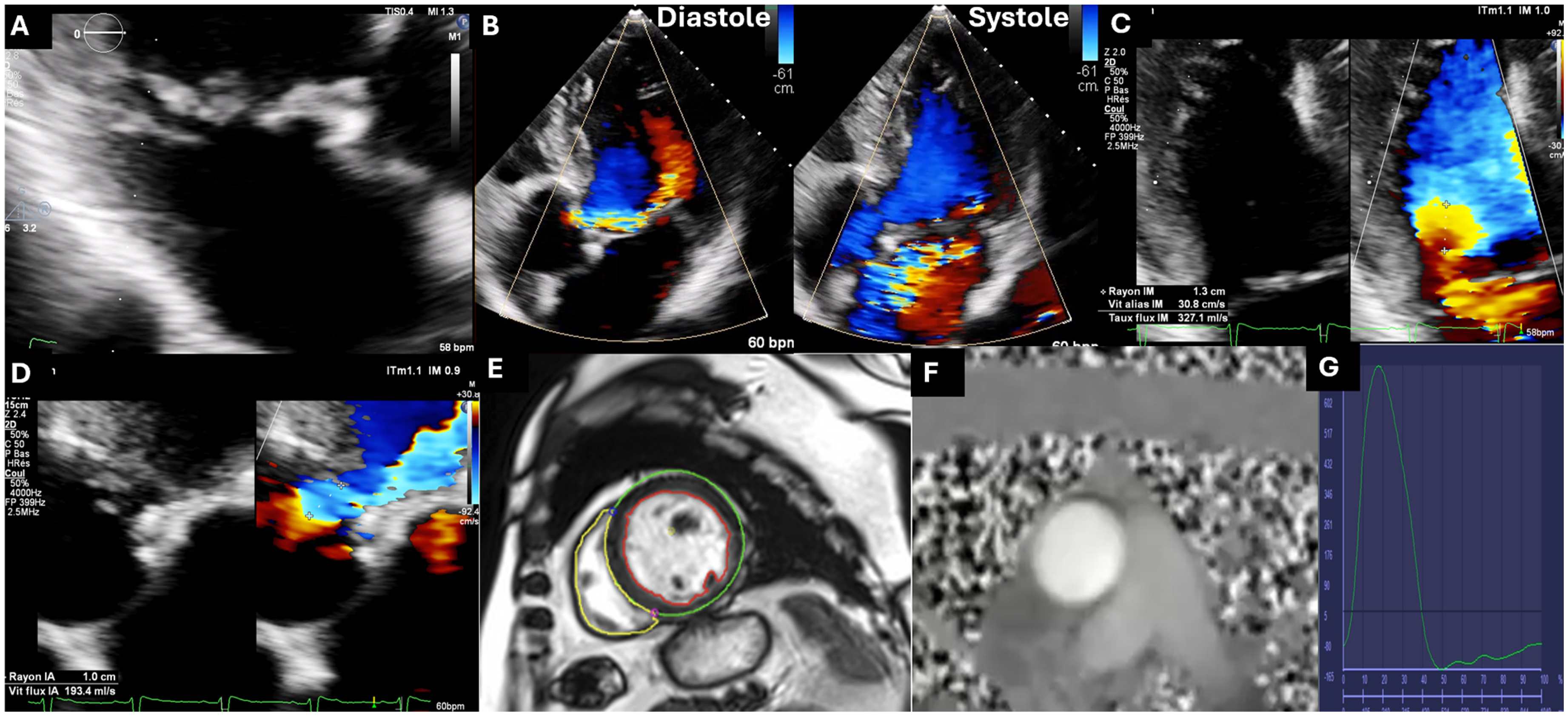
Association of Aortic and Mitral Regurgitation. Clinical context: a 66-year-old patient with severe degenerative aortic and mitral regurgitation. (A) Three-chamber transthoracic echocardiography (TTE) showing P2 prolapse with a flail leaflet. (B) Eccentric jets of aortic and mitral regurgitation. (C) Proximal isovelocity surface area (PISA) radius of the mitral valve of 13 mm at an aliasing velocity of 31 cm/s (effective regurgitant orifice area of 60 mm2, regurgitant volume of 67 mL). (D) Aortic valve PISA radius of 10 mm at an aliasing velocity of 31 cm/s (effective regurgitant orifice area of 45 mm2, regurgitant volume of 65 mL). (E-G) Cardiac MRI assessment of the severity of mitral and aortic regurgitations including: a regurgitant volume of 55 mL (regurgitant fraction 55%) for mitral regurgitation, and 65 mL (regurgitant fraction 39%) for aortic regurgitation (left ventricular stroke volume: 165 mL, aortic flow forward [phase contrast]: 110 mL, and aortic backward flow [phase contrast]: 65 mL).
Aortic Stenosis and MS
The combination of AS and MS is not uncommon (19% of patients with 2 severe left-sided VHD without severe TR 8 ) and represents a real diagnostic challenge. Indeed, in this low-flow situation, both aortic and mitral pressure gradients can be low despite a small surface area. 44 Furthermore, the mitral flow PHT may underestimate the mitral valve area due to the reduced LV compliance caused by AS. The continuity equation and mitral valve area planimetry remain solid methods, but calcifications complicate measurement in cases of degenerative MS (Table 2). The degenerative cause is becoming more prevalent than the rheumatic cause, increasing the role of the cardiac CT. Computed tomography also provides important information on annular calcification and mitral valve area, 45 which correlates with the severity of MS (Figure 4). 46
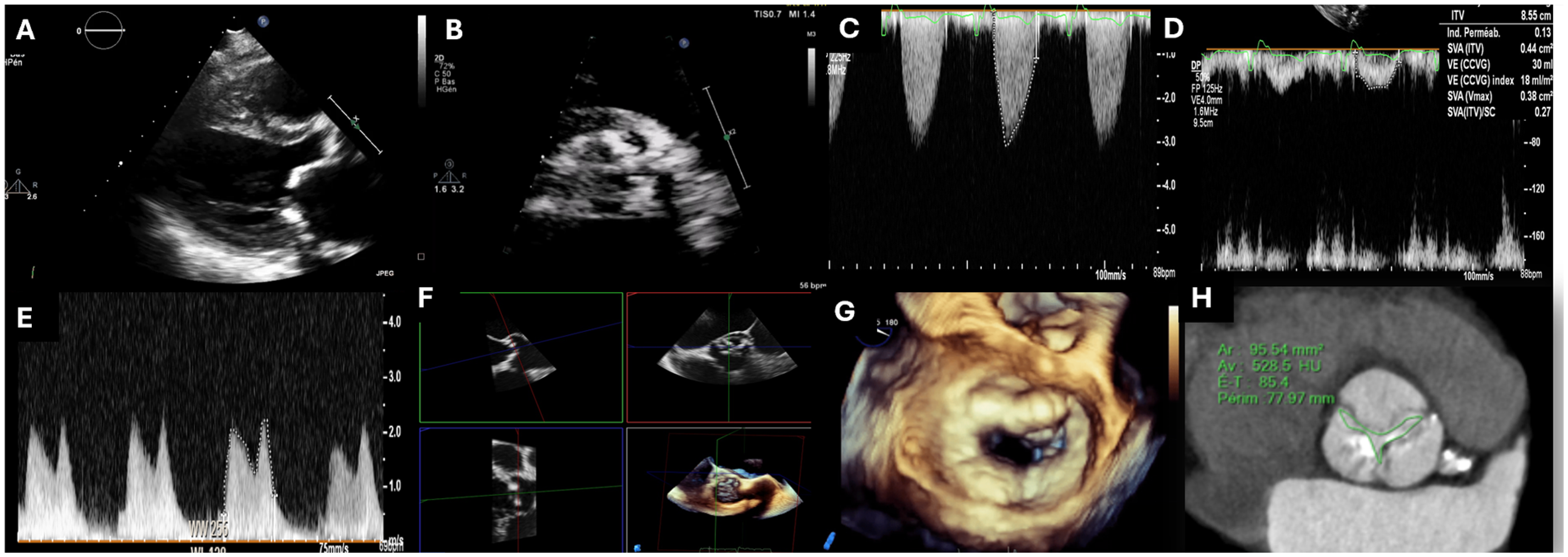
Association of Aortic Stenosis and Mitral Stenosis. Clinical context : a 78-year-old female patient with degenerative calcified aortic stenosis and mitral stenosis. (A and B) Severe calcification of the mitral annulus, mitral valve, and aortic valve. (C and D) Low-flow, low-gradient severe aortic stenosis with a peak aortic jet of 3 m/s, estimated aortic valve area of 0.45 cm2 and a calculated indexed systolic ejection volume of 18 mL/m2. (E) Mean mitral valve gradient elevated to 9 mm Hg. (F) Planimetry of the aortic valve using multiplanar reconstruction in 3D transoesophageal echocardiography, measured at 0.8 cm2. (G) 3D “en-face” view of the mitral valve showing severe annular calcifications. (H) Contrast-enhanced computed tomography (CT) planimetry of the aortic valve, measured at 0.95 cm2.
Aortic Stenosis and AR (Mixed Aortic Valvular Disease)
Echocardiography is a key diagnostic tool for MAVD (Figure 5), but conventional parameters for assessing isolated AS or AR have limitations (Table 3). Peak aortic jet velocity and mean gradient increase not only with AS but also with AR due to a greater stroke volume. Even with an AVA greater than 1 cm2, severe pressure overload occurs. 3 Yet, the continuity equation remains a reliable estimate of AVA in this setting. 47 The impaired LV relaxation secondary to the LV hypertrophy makes the PHT method unreliable for AR assessment. 48 Usually, MAVD results in less pronounced LV dilatation due to AS-induced remodeling. As a result, even a moderate regurgitant volume may be clinically more significant in a normal or slightly dilated LV compared with a markedly dilated LV. For that reason, Unger et al suggested to use the regurgitation fraction rather than the regurgitant volume in this situation. 49 If feasible, the use of vena contracta remains valid, such as femoral diastolic flow reversal or abdominal flow reversal, which are even more specific for AR severity than descending aortic flow reversal.
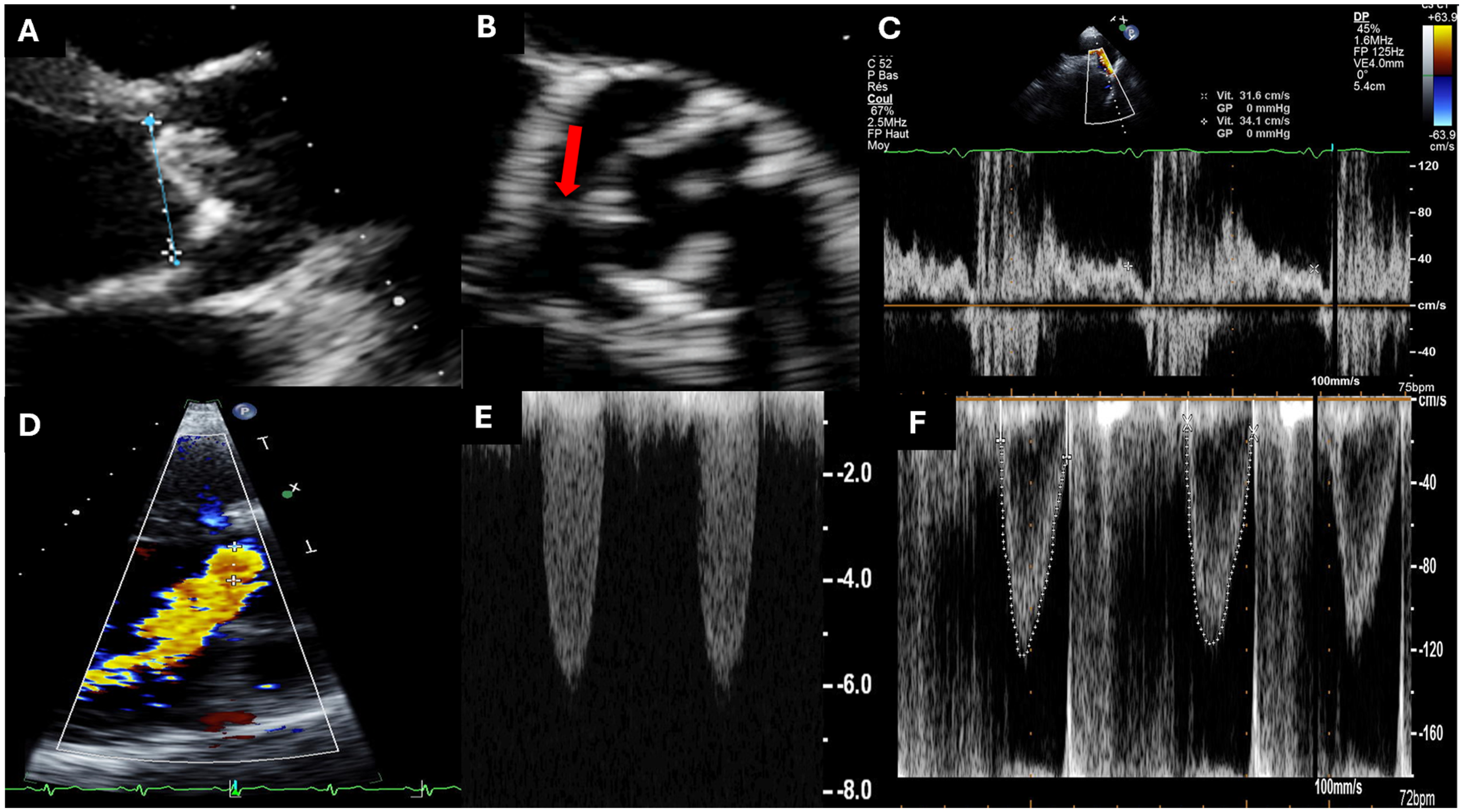
Aortic Stenosis and Aortic Regurgitation (Mixed Aortic Valvular Disease). Clinical context: a 55-year-old patient with severe mixed aortic valve disease. Transthoracic echocardiography shows severe aortic regurgitation and high gradient aortic stenosis despite persistent aortic valve opening. (A) The aortic valve is moderately calcified, and the aortic annulus is large (26 mm). (B) Short-axis view showing a bicuspid aortic valve with a raphe between the right and noncoronary cusps (red arrow) and persistent aortic valve opening. (C and D) Presence of severe aortic regurgitation with a high end-diastolic velocity (31 cm/s) in the descending aorta (C) and a large proximal isovelocity surface area (PISA) radius of 10 mm at an aliasing velocity of 30 cm/s (D). (E) High gradient aortic stenosis with peak jet velocity greater than 5 m/s. (F) Increase in cardiac output estimated at 10.5 l/min. The aortic valve area is calculated at 1.3 cm2 by the continuity equation.
More generally, the aortic mean pressure gradient is a good reflection of the MAVD severity with a value > 40 mm Hg suggesting severe MAVD.
The role of dobutamine stress echocardiography in low-flow low-gradient AS is to discriminate pseudo severe from true severe AS by calculating AVA at a normal flow rate. Aortic valve area <1.0 cm2 at a normal flow rate indicates true severe AS. In some cases, at very high flow rates, the same aortic valve could reach an AVA ≥ 1 cm2, secondary to the residual valvular compliance. 50 The same scenario may apply to MAVD because of greater transaortic flow. Some patients may have an AVA ≥1 cm2, indicating moderate AS, but that would correspond to <1 cm2 at a normal flow rate, in line with severe AS. 49
Cardiac CT may help to differentiate between true severe and pseudo severe AS by assessment of the aortic valve calcium score (true severe if ≥2000 AU in men and ≥1200 AU in women). 51
Mitral Stenosis and MR (Mixed Mitral Valvular Disease)
The association of MS and MR is not frequent representing 5% of VHD patients. 52 Rheumatic heart disease and degenerative etiology with MAC are the main causes. 53 Interestingly, MR elevates the flow rate through the mitral valve, leading to an increase in left atrial (LA) pressure, particularly in individuals with associated MS. 3 Echocardiographic assessment of MR severity is challenging, particularly with severe calcifications that obscure the jet area. 20 Nevertheless, both the proximal isovelocity surface area (PISA) and volumetric method remain applicable (Table 3). For MS assessment, the estimated mitral valve area from PHT is not accurate due to the presence of a significant MR that alters LV compliance. Similarly, the continuity equation is unreliable and may overestimate the severity of MS.3,20 The Doppler mitral gradient reflects the global severity of mixed mitral VHD, and a value greater than 10 mm Hg could be a good criterion of severity. 20 Finally, planimetric area is probably the most reliable technique to evaluate the MS severity if feasible. The impact of exercise echocardiography on clinical decision-making is unclear. Although it has been recommended for isolated MS, 25 there are currently only limited data in patients with mixed mitral valve disease.54,55
Cardiac CT using the latest generation scanners has shown acceptable feasibility, accuracy, and reproducibility in assessing mitral valve orifice area measurements and can be helpful in cases of mixed valvular disease with difficulties in performing planimetric area in echocardiography. 45 Atrial fibrillation, which is common in this population, leads to artifacts limiting its use. In the case of MAC-related mitral valve, the mitral annulus calcium score is not yet recommended in clinical practice.28,29 Finally, the assessment of MR by CMR is helpful using the indirect method, already mentioned above.32,56
Left-Sided VHD with TR
Secondary TR frequently complicates left-sided VHD 25 through pulmonary hypertension, RV dilatation and dysfunction, and annular dilatation.3,18,57 Furthermore, increased RV filling pressure may contribute to the development of right atrial dilatation and atrial fibrillation, both of which can cause and exacerbate TR. 58
Tricuspid regurgitation evaluation is commonly made by echocardiography (Figure 2, Table 2).25,59 However, a multimodal approach could be used, with CMR for the regurgitant volume, RV volume and function, and catheterization to estimate pulmonary pressure. Regurgitant volume calculated with 2D PC CMR by using indirect volumetric assessment has been less evaluated than for the mitral valve and RV stroke volume obtained from the RV delineation is more challenging; 4D flow appears to be an interesting technique with a direct measurement avoiding RV delineation.60,61 Secondary TR may develop late after treatment of the left-sided VHD 62 and is associated with the worst long-term prognosis. 63
In the case of RHD, particular attention should be paid to the assessment of the tricuspid valve and RV function, as well as the pulmonary valve.
Case of MMVD Patients with Prosthetic Valves
Prosthetic dysfunction involving concurrently multiple valves, whether prosthetic and/or native, is not uncommon. 59 A study assessing the risk of redo valve surgery after bioprosthethic valve replacement found that the most common concurrent cardiac procedure was on a valve other than the failed bioprosthesis, occurring in 27% of cases. Aortic valve replacement was performed in 20% of cases of patients referred for mitral prosthesis dysfunction, and tricuspid annuloplasty or replacement in 21% of cases. 64
More recently, real-life registries have provided interesting and novel data. The Valve-In-Valve (ViV) International Data registry examined outcomes of transcatheter ViV procedures in 459 patients, of whom about 30% had mixed lesions, involving either the aortic or mitral valve. 65 Moreover, in a recent study comparing the clinical and echocardiographic outcomes after surgical redo mitral valve replacement and transcatheter mitral ViV therapy among patients with mitral bioprosthesis degeneration, the authors showed that nearly 60% of patients had at least moderate associated MR, 8% had at least moderate AR, and 60% had at least moderate associated TR. 66 Finally, in the VIVA registry, which evaluated patients referred for aortic in-valve surgery, 21% of patients had a mixed VHD combining AR and AS. 67
Cardiac Remodeling Due to MMVD
Several studies using CMR have already identified dynamic changes within the myocardial structure during the progression of VHD. 68 This phenomenon, called ventricular remodeling, not only is necessary to maintain a balance between valve disease severity and cardiac output but also serves as a gateway to heart failure, 69 worsening secondary valve regurgitations, 70 and increased mortality and outcomes. Ventricular remodeling is associated with late gadolinium enhancement (LGE) on CMR,71,72 which is linked to mortality in AS 73 and to ventricular arrhythmias in primary MR. 74 Beyond LGE, T1 mapping, and extracellular volume mapping can be used to better quantify the diffuse interstitial fibrosis.40,68 Interestingly, recent studies have suggested an important role of LA and LV coupling through a combined index in the left remodeling of ischemic heart disease and some cardiomyopathies.75,76 Although several important determinants of left atrioventricular coupling index are linked to VHD, 77 no study has yet evaluated the interest of a combined index evaluating LA and LV coupling in VHD patients. Finally, significant innovations, including high-resolution LGE can better characterize myocardial fibrosis. 78
Treatments
Current Guidelines
Due to the lack of data to support and direct clinical decision-making, current European and American guidelines on MMVD are still limited and primarily based on small retrospective studies or expert consensus opinion and are therefore mostly Level C recommendations.25,59 In most cases, one lesion is predominant, and management should follow recommendations for this lesion. However, when 2 or more lesions are moderate, their combined effect may lead to a worse prognosis, even in the absence of associated severe VHD. This often leads to delays in patient management. In such cases, symptom evolution and global assessment—including clinical assessment, LV/RV dilation and dysfunction, and pulmonary hypertension—are key factors in guiding therapeutic decisions, and intervention may be considered. 3 However, the increased risk of combined intervention should be evaluated for each patient. Some lesions may benefit from a staged approach. The MMVD's relationships are not yet fully understood, and our understanding of their coexistence in terms of current management remains limited, emphasizing the role of the “Heart valve team.” 3 This highlights the unmet need for studies that will help the clinician to tailor the therapeutic strategy in each individual MMVD patients.8,19
Surgical Valve Replacement
A recent study reported that among patients with severe multiple VHD, only 26% had undergone surgery, among which 57% involved more than one valvular procedure (35% valve replacement and 22% valve repair).
58
Based on literature data and expert consensus, the main principles guiding the decision to proceed to surgical management of MMVD patients should be the following:
Multiple surgical valve surgery is associated with an increased operative risk.3,79–81 In particular, triple valve surgery (TVS) remains a high-risk procedure with in-hospital mortality rates ranging from 7% to 11% and significant long-term morbidity, which varies according to population and underlying etiology.82,83 In younger patients (Middle Eastern countries), RHD is the primary cause of TVS, with diabetes and hypertension being important prognostic factors.
84
In older populations (Western countries), degenerative valve disease predominates, with factors such as New York Heart Association class IV, renal failure, and redo sternotomy having a significant impact on surgical outcomes.
85
Furthermore, when feasible, valve repair should be privileged.86,87 The durability of mechanical prosthesis is advantageous for young patients.
88
However, this should be balanced against the higher risk of anticoagulation in patients with multiple mechanical valve prostheses.
89
Assessment of the operative risk is instrumental in the choice of surgical, transcatheter approach, combined, or medical treatment strategy. Some risk scores such as the STS score have been validated for MMVD patients, but lack external validation.81,90 Minimally invasive surgery is usually limited by its inability to address MMVD.
88
Surgery is most often advised when at least 2 VHDs are severe unless the patient is at high surgical risk and all lesions are amenable to transcatheter procedures.
45
When a patient presents a severe VHD requiring surgery, the decision to address another concurrent moderate lesion should be made on an individual basis and should include the likelihood to improvement after treatment of the severe lesion and the natural history of an untreated moderate VHD.
3
Percutaneous Valve Replacement
Few studies have assessed the percutaneous management of MMVD with most data from TAVI registries for AS. 91 While AS and AR may increase the risk of transcatheter heart valve (THV) migration due to the hyperdynamic jet during TAVI procedure, the presence of mitral or tricuspid VHD does not pose a significant challenge to TAVI implantation. Although the clinical impact of concomitant baseline VHD on post-TAVI outcomes varies depending on the specific type of baseline VHD,92,93 there is limited evidence to recommend transcatheter intervention for MMVD. Indeed, while TAVI is becoming a dominant therapeutic option in patients with severe AS and moderate to severe surgical risk, percutaneous valve replacement or transcatheter edge-to-edge repair (TEER) for MR, MS, and TR are still reserved for patients at high surgical risk. Recent TAVI trials emphasize careful preprocedural MR and TR assessment. While functional MR often improves after TAVI due to left ventricular unloading,94,95 primary MR and significant TR may persist, requiring additional interventions. 96 Considering the latest data, the evolution of MR remains unpredictable and the superiority of a staged or concomitant mitral-tricuspid approach over isolated TAVI is not established, warranting close imaging follow-up.97–100 For concomitant severe MS, combining transcatheter mitral valve replacement with TAVI (TMVI) may be an alternative, particularly in cases unsuitable for percutaneous mitral balloon commissurotomy (PMC).101,102 While concomitant TR as an independent predictor of mortality after TAVI remains controversial,96,103 staged interventions for TR may still be warranted if symptomatic. Similarly, the efficacy of additional TR procedures in high-risk patients with severe MR undergoing TMVI or TEER repair still needs further investigation.
Valve Repair
Surgical Valve Repair
Whether valve repair or replacement is the optimal surgical strategy should be considered in the decision-making for MMVD patients. Hence, echocardiography is critical not only in determining the indication and timing for surgery but also in predicting the likelihood of successful valve repair. 3 Between 2001 and 2017, the rate of surgical valve repair increased from 46% to 51% for MR and from 2% to 22% for AR. 2 In the STS registry, 23 404 patients underwent concomitant aortic valve replacement and mitral valve surgery between 1993 and 2007. 104 In the context of AS and MR, mitral valve repair should be preferred, if possible, to replacement as studies have suggested late survival improvement. 105 Therefore, efforts should be directed toward improving rates of mitral valve repair, even if prosthetic valve replacement of another valve decreases the willingness of a surgeon to repair the mitral valve. Controversy exists, however, as to whether repair or replacement of the mitral valve should be preferred in patients undergoing aortic valve replacement who have moderate-to-severe secondary M.In this setting, 2 studies showed no benefit from mitral valve repair with aortic valve replacement over double-valve replacement on survival, and an increased long-term incidence of mitral valve failure in patients undergoing mitral repair was even observed.106,107 Concomitant tricuspid valve repair at the time of left-sided valve surgery is the preferred technique with a lower rate of complications than transcatheter tricuspid valve replacement (TTVR).57,108–110
Percutaneous Mitral Commissurotomy
Commissurotomy can be performed surgically or percutaneously. It can be used in the context of valvular stenosis, often mitral. 25 Double surgical valve replacement is preferable to isolated PMC in patients with severe MS combined with severe aortic valve disease. However, in patients with severe MS and moderate aortic valve disease, percutaneous ballon valvuloplasty can be performed to postpone the surgical treatment of both valves.
Transcatheter Edge-to-Edge Repair
Transcatheter edge-to-edge repair is a minimal-invasive solution that is increasingly used in primary or secondary MR. It has been evaluated against optimal medical therapy, resulting in an upgrade of the recommendation. 25 Regarding the tricuspid valve, experience with transcatheter tricuspid valve interventions suggests a potential role in high-risk patients. 111 In the specific setting of MMVD, multiple transcatheter procedures are feasible, but clinical experience remains limited for robust recommendations. 112 In high-risk patients with severe AS and severe secondary MR, combined or often sequential TAVI and TEER may be feasible. 113 In inoperable with severe primary MR, TEER is a safe alternative and should be considered early if symptoms persist and MR is still severe after TAVI. Finally, the efficacy of more recent TEER system iterations will be investigated in high-risk (MITRA-HR study) and intermediate-risk patients (REPAIR-MR study).114,115
Antithrombotic Management
No international guidelines specify the antithrombotic management for MMVD patients.25,59
Native Valves
In patients with native valves, antithrombotic treatment depends on associated comorbidities. For atrial fibrillation, which is frequent in MMVD, direct oral anticoagulants (DOACs) are the first-line treatment,116,117 except in case of MS. Indeed, the INVICTUS study showed that vitamin K antagonists (VKAs) led to a lower rate of a composite of outcomes than rivaroxaban. 118
Transcatheter Bioprosthesis
There is no difference in antithrombotic management between single VHD and MMVD, possibly due to a lack of evidence in MMVD patients. Indeed, in patients with aortic transcatheter bioprosthesis, aspirin therapy is recommended as recent studies showed a significant decrease in major bleeding with aspirin over dual antiplatelet therapy.119,120 After implantation of transcatheter mitral bioprosthesis (TMVI; eg, ViV, valve-in-ring, valve-in-MAC), anticoagulation for 3 months is commonly prescribed. The use of DOACs after TMVI, compared with VKAs, appears to reduce the risk of bleeding complications,121,122 even though we currently have no data from randomized trials. Vitamin K antagonists are widely used over DOACs for the first 6 months after percutaneous mitral or TTVR (dedicated devices).
Surgical Bioprosthesis or Valve Repair
After aortic valve surgery, if there is no indication of anticoagulation, aspirin is preferable to anticoagulation.123–125 After mitral/tricuspid valve surgery, anticoagulation for at least 3 months is recommended.126,127 Although few data are available on the use of DOACs in the 3 months following surgery, there are no alarm signals even while DOACs are widely prescribed.128,129
Mechanical Valves
Mechanical valves require lifelong treatment with VKAs guided by the international normalized ratio (INR). Direct oral anticoagulants failed to show any benefit and are even harmful.130,131 Target INR varies between 2.5 and 3.5 and should be based upon prosthesis thrombogenicity and patient-related risk factors. The use of self-monitoring INR must be favored to reduce VKA-related complications.
Multiple and Mixed Valvular Heart Disease Associated with Specific Conditions
Atrial and Ventricular Arrhythmia
Valvular heart disease is independently associated with atrial fibrillation with a prevalence of 30%. 132 However, while patients with moderate to severe MS and/or mechanical prosthetic should undergo anticoagulation with VKAs, current evidence does not suggest that the presence of other forms of VHD should influence the choice of oral anticoagulation therapy. 133 According to a recent study, MMVD in patients with atrial fibrillation does not increase thrombotic risk. 134 The decision to use oral anticoagulation to prevent thromboembolic events, using either VKAs or DOACs, should be determined through a shared decision-making process. Vitamin K antagonists remain the preferred anticoagulants for patients with rheumatic MS and/or mechanical heart valves. Importantly, the evaluation of these MMVD patients with atrial fibrillation could be very challenging but feasible using an optimized imaging protocol. 135
Cardiac Implantable Electronic Device–Related VHD
Cardiac implantable electronic device, including pacemakers, implantable cardioverter defibrillator (ICD) may have the potential to induce specific VHDs and may be implanted as a consequence of long-term, untreated VHDs.
Valvular-induced ventricular arrhythmias or even sudden cardiac death, which are more commonly associated with MR, with 136 or without mitral valve prolapse137,138 and with AS, 139 might account for an important number of defibrillator implantations for primary and secondary prevention of sudden cardiac death. Tricuspid regurgitation is the most common complication of device implantation and can be classified into 2 distinct categories: lead-induced TR caused by lead-leaflet interaction, 140 and lead-associated TR, which occurs when preexisting TR is aggravated by devices,141,142 right ventricular apical pacing only might produce MR due to left ventricular dyssynchronous contraction.143,144 On the other hand, LV resynchronization could serve as a tool for lowering the degree of MR. 145 Alternative leadless strategies that have demonstrated no worsening of TR after implantation should be considered. 141 These include leadless pacemakers or subcutaneous ICDs.146,147
Finally, for the evaluation of these patients using CMR, although the presence of a defibrillator causes important artifacts, it is important to mention the good feasibility of the CMR exam including 2D-flow in patients with a pacemaker. 148
Cardiac Amyloidosis
Amyloid deposition can involve any cardiac structure, including the valves. The most common VHD in cardiac amyloidosis is AS, particularly in transthyretin amyloidosis, 149 where it often presents with a low-flow, low-gradient pattern. Mitral regurgitation and MS are the next most prevalent VHD.150,151 The mechanism remains unclear but may involve direct deposit, chronic inflammation, and oxydative stress caused by amyloid infiltration, which ultimately results in calcification, rigidity, fibrosis, and leaflet sclerosis.152,153 Multivalvular involvement places a significant hemodynamic load on a stiff and dysfunctional myocardium, which negatively contributes to the LV remodeling. Mohty et al demonstrated that mitral and/or aortic valve thickening was associated with higher all-cause mortality. 151 The tricuspid valve can also be affected, typically with regurgitation. 154 However, mitral and tricuspid VHD are rarely more than mild or moderate and rarely require valve intervention. Cardiac amyloidosis screening could be interesting in MMVD patients with suggestive signs. 155
Perspectives
Transcatheter Mitral and TVR
Transcatheter mitral valve replacement might serve as an alternative invasive treatment option to overcome the current anatomical constraints of percutaneous repair with the potential to treat mixed mitral valve disease. 156 Multiple devices (using either transseptal or transapical access and using single or multistep approaches depending on devices), specifically designed for this purpose are being evaluated in early feasibility studies as well as pivotal trials. Nonetheless, to date, only one device, the Tendyne (Abbott Structural) system, has received a European conformity mark. The cornerstone of investigating patient eligibility relies on preprocedural planning, which necessitates multimodality imaging assessment. Yet, this process poses a challenge in clinical practice, given the high screening failure rates of around 90%, which emphasize the critical importance of optimizing candidacy. 157 Patients with severe MAC pose a particular challenge and are frequently excluded from most studies evaluating new mitral THVs. This unmet clinical need has been partially addressed by the off-label use of aortic THVs with transseptal access, albeit showing poor outcomes at 1 year therefore raising the question of the futility.158,159
Similarly, TTVR has emerged as an alternative to isolated TR surgery which is associated with high morbidity and mortality. Several novel percutaneous transcatheter methods for tricuspid valve repair and replacement have been extensively studied in recent years, showing promising clinical results in terms of mortality and rehospitalization within the initial year of follow-up. 160 To date, only one device has been recently approved by both the FDA and CE for commercial use, the Evoque Tricuspid Valve (Edwards Lifesciences).
Risk Stratification Using Machine Learning
To improve decision-making for VHD patients, emphasis should be on identifying factors and developing risk models that will guide clinical decisions.161,162 To meet this need, traditional risk assessment tools, such as the STS score, have been tailored for VHD and, to a certain extent, MMVD, demonstrating utility in clinical decision-making.81,163 Nonetheless, many clinical prediction models lack external validation 90 and fail to fully grasp the intricate dynamics of MMVD. This is particularly concerning given the multidimensional nature of multimodality imaging data in MMVD patients. 3 This underscores the need for a paradigm shift toward more personalized decision-making processes. 162
Machine learning emerges as a promising solution, offering sophisticated tools for handling the nonlinearities inherent in cardiology, and enhancing prognostication and decision-making in cardiovascular disease. Supervised ML improves prediction score, 164 while unsupervised learning is used to perform clustering165,166 and provides insights into patient phenotypes and risk continuums. 167 Recent advancements have led to ML-driven scores for AS, MR, and TR that surpass traditional models in predicting outcomes.168–173 However, a specific ML score for MMVD patients, integrating the interactions between valves, remains undeveloped.
Presentation of the EACVI-MMVD Study
Due to this lack of evidence about MMVD, we advocate for the initiation of the first international MMVD registry to: (1) assess the prevalence of MMVD; (2) evaluate management strategies using multimodality cardiovascular imaging; and (3) prognosticate outcomes for MMVD patients. Indeed, the Heart Imagers of Tomorrow of the EACVI is performing the “European Association of Cardiovascular Imaging – Multiple and Mixed Valvular heart Diseases” (EACVI-MMVD) study (ClinicalTrials: NCT06235385) as a large prospective, multicenter, observational “real-life” study including all consecutive patients diagnosed with MMVD using TTE over a 6-month recruitment period in more than 100 centers from more than 20 different countries (Figure 6). The entire spectrum of multimodality imaging data performed in clinical routine will be collected. All patients will be followed to assess the occurrence of cardiovascular events and valvular intervention needed within a year. Notably, 3 dedicated core laboratories (MIRACL.ai, Lariboisiere Hospital, Paris for CMR; CORELAB ECHO, Rennes Hospital for Echo; and VaRMI Core Lab, Laval Hospital, Canada for cardiac CT) will analyze cardiovascular imaging data from participating centers.
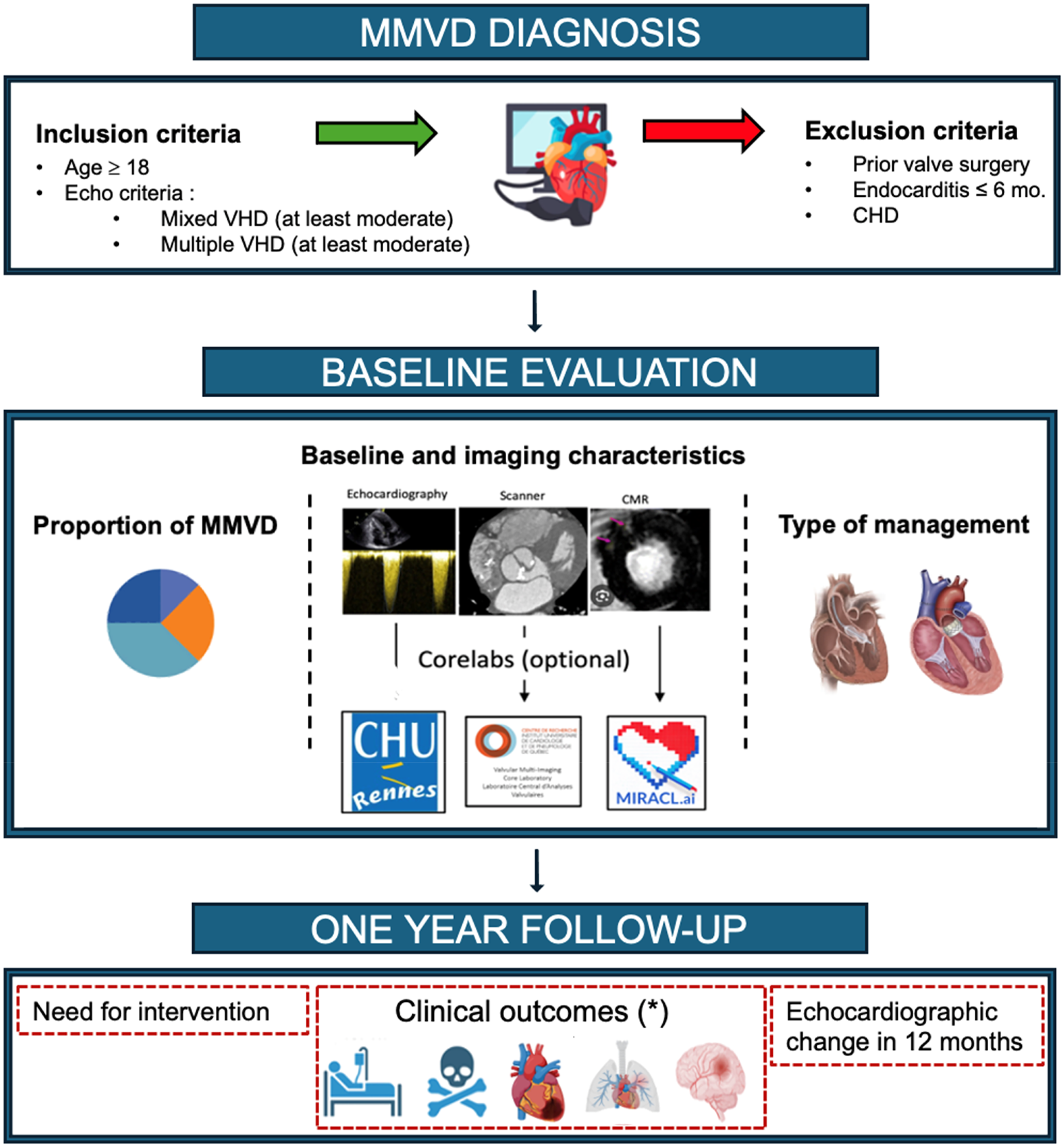
EACVI-MMVD Study Design (ClinicalTrials: NCT06235385). (*) Clinical outcomes: hospitalization for heart failure, cardiac death, myocardial infarction, pulmonary embolism, stroke. MMVD, multiple and mixed valvular heart disease.
Presentation of the MULTIVALVE Study
While the EACVI-MMVD study will provide unique data on a global scale for MMVD patients, we can fear a lack of granularity in the cardiovascular imaging data linked to the absence of a standardized protocol and the heterogeneity of practices across countries. To achieve a highly standardized MMVD registry for cardiovascular imaging, the French Society of Cardiology will begin in 2025 the “MULTIVALVE” registry as a prospective multicenter study in France including all consecutive patients diagnosed with MMVD using TTE over a 12-month recruitment period. The strengths of this study include: (1) carrying out a more interventional study with a standardized cardiovascular imaging protocol for TTE and CMR mandatory for all patients; (2) analysis of all images of TTE and CMR by 2 centralized analysis core lab (MIRACL.ai, Lariboisiere Hospital, Paris for CMR and cardiac CT; and CORELAB ECHO, Rennes Hospital for Echo); and (3) matching with French social security data allowing exhaustive monitoring of valve interventions, drug treatments and outcomes for 5 years.
Conclusion
Multiple and mixed VHD are very common conditions, with an increasing incidence of degenerative cause, especially in developed countries. Considering each patient singularity and echocardiography pitfalls, multiparametric evaluation with multimodality imaging, symptoms, and biomarkers, supported by machine learning score, appears promising to make personalized care approaches. However, given these difficulties, there are a paucity of data concerning MMVD patient and treatment recommendation. To provide adequate answers to this daily clinical and therapeutic challenge, the “EACVI MMVD” (international observational and “real life” study) is underway, and the “MULTIVALVE” study (prospective multicenter study in France) will start in 2025.
Footnotes
Abbreviations
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
