Abstract
Background
Novel cardiac valve prostheses require long-term studies assessing safety in animal models prior to clinical use. Although sheep are well-established preclinical models, the anatomy and morphology of the sheep heart impart unique challenges in the positioning and securement of transcatheter mitral valve replacement (TMVR) devices, resulting in early postoperative mortality. We developed a refined hybrid surgical approach to study the chronic in vivo performance of a novel TMVR device intended for regulatory approval.
Methods
Eleven adult sheep were enrolled in this study. Eight Abbott Next-Generation Cephea (Cephea) TMVR devices were implanted using a hybrid surgical approach, with the valve deployed and secured under direct visualization on cardiopulmonary bypass. For a control, three Medtronic Mosaic bioprostheses (Mosaic) were implanted with standard techniques. Sheep were survived for 20 weeks per regulatory guidance, followed by humane euthanasia and comprehensive necropsy.
Results
All sheep were successfully implanted and recovered from surgery, with 7/8 Cephea and 3/3 Mosaic animals surviving to the 20-week study endpoint. A surgical complication unrelated to TMVR performance resulted in one early death at 58 days post-operatively. At term, all Cephea valves performed comparably to controls, appearing well-healed within the annulus with competent, unobstructed leaflets.
Conclusions
Our novel hybrid surgical approach allowed for a comprehensive evaluation of the long-term performance of the Cephea, with 100% survival at 30 days and 88% at the 20-week endpoint. These results suggest that this approach may improve chronic survival in animal studies evaluating the safety of novel TMVR devices prior to clinical investigation.
This is a visual representation of the abstract.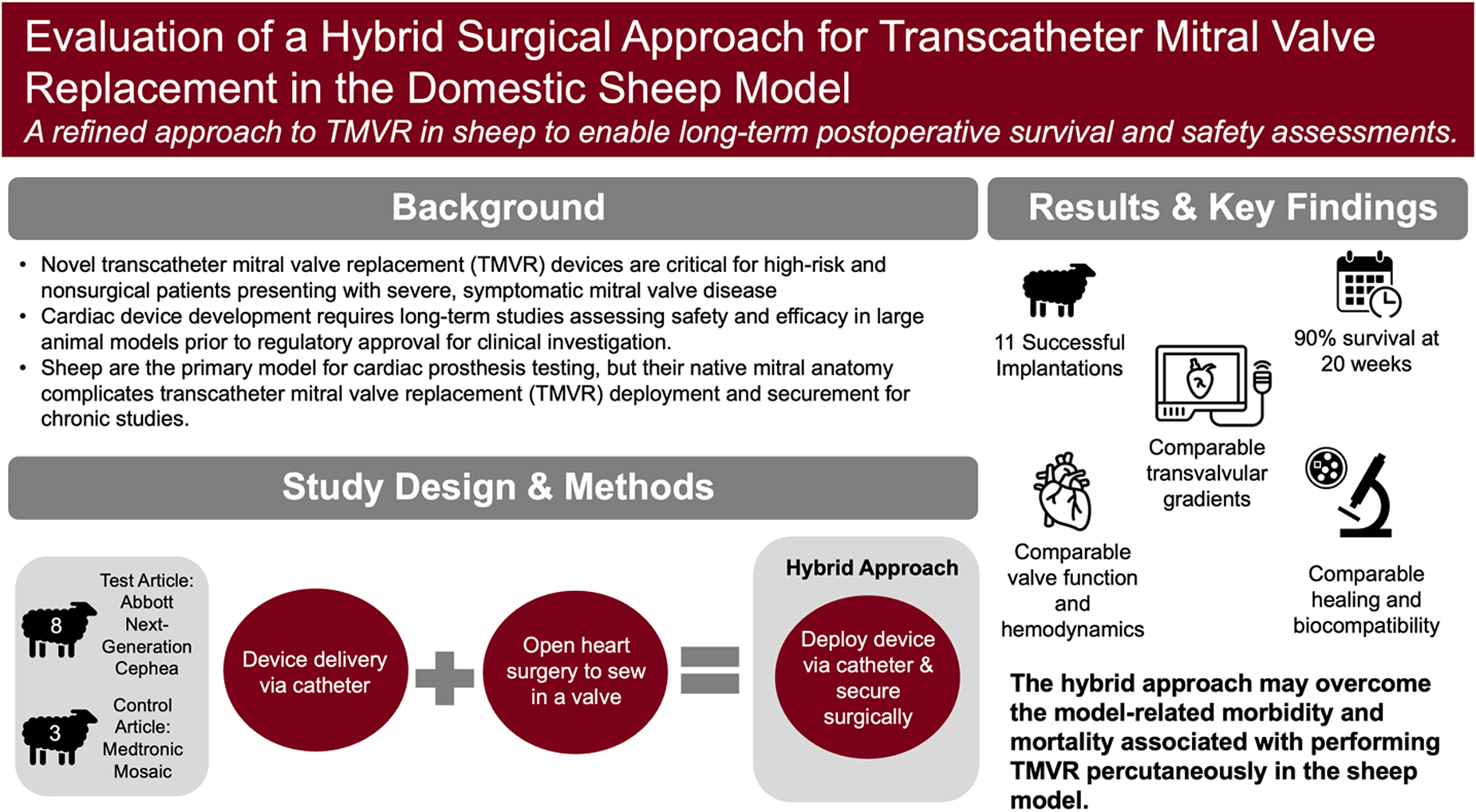
Keywords
Key Points
Sheep are the primary in vivo model for testing novel cardiac devices, though their mitral anatomy complicates transcatheter mitral valve replacement (TMVR) device deployment and securement for chronic studies.
We developed a hybrid surgical approach for TMVR in sheep to enable long-term evaluation prior to clinical investigation.
This refined approach may overcome the model-related morbidity and mortality associated with performing TMVR percutaneously in the sheep model, and facilitate long-term device evaluation required for regulatory approval.
Introduction
Mitral valve disease (MVD) is one of the most common cardiac valve diseases in the United States. 1 While a surgical valve repair or replacement is the first-line treatment option in most cases, minimally invasive, catheter-delivered replacement valves are crucial for treating patients for which surgery is contraindicated because of disease etiology or comorbidities. 2 In another common cardiac valve disease, aortic stenosis, transcatheter aortic valve replacement (TAVR) has already shown success as an alternative to surgical valve replacement. The growing use of TAVR has led to marked advancements in catheter and material technology, as well as increased comfort and experience of physicians utilizing catheter-based devices. Given a similar need for non-surgical alternatives in treating severe, symptomatic MVD, transcatheter mitral valve replacement (TMVR) has become an emerging area of cardiac device development. 3 There are several early clinical studies of novel TMVR devices currently ongoing in patients in the United States and Europe with encouraging initial results, which will likely lead to additional clinical trials and increased access to these novel devices by physicians and their patients.3–5
Prior to clinical use, regulatory agencies require comprehensive preclinical feasibility and chronic safety evaluations of novel cardiac devices in large animal models. 6 One of the most often cited animal models for such preclinical studies is the domestic sheep (Ovis aries). 7 Use of the sheep model in evaluating novel cardiac devices and their translation to the clinical setting has been widely reported in published literature, especially in evaluating long-term hemodynamic performance and durability, thromboembolic complications, and biocompatibility.8,9 As a result, the sheep has become the standard large animal model for in vivo testing and evaluation of novel cardiac valve replacement devices prior to human use. However, there are few resources describing the utility of the sheep model for long-term evaluation of novel catheter-based cardiac valves. Previous studies discovered chronic evaluation of TAVR in animals is challenging given the presence of a healthy, nondiseased heart in this model. In human clinical patients, the diseased heart valve provides a natural mechanism for securing transcatheter replacement devices. Absence of a diseased valve and associated lack of transcatheter valve securement in the aortic position can lead to a variety of complications unrelated to the performance of the device including improper positioning, annular rupture, severe paravalvular leak, and device migration. 10
We have experienced similar challenges in the mitral position during our preliminary unpublished feasibility studies for early-stage TMVR devices. We attributed our adverse events to the innate challenges of using a native, nondiseased sheep model as opposed to the clinical performance of the TMVR prostheses in the diseased valve of the intended human patient population. Thus, we sought to leverage established methods of surgical mitral valve replacement to facilitate insertion and securement of a novel TMVR device for long-term evaluation in the sheep model. We aimed to implement a novel hybridized surgical approach to TMVR in the sheep model in which we combined open heart surgical access and securement with interventional transcatheter valve deployment. We hypothesized this method would mitigate the largely model-related adverse events observed in our initial studies and establish a suitable method to create successful chronic animal models in which to study novel TMVR devices prior to clinical use.
Materials and Methods
Approval was obtained by the Institutional Animal Care and Use Committee prior to the start of the study.
Experimental Design
The experimental design is provided in Table 1. Animals receiving the test article (n = 8), a next generation Abbott Cephea Valve (Cephea), underwent TMVR utilizing a hybrid surgical approach and were recovered. Commercially available Medtronic Mosaic bioprostheses were used as control articles in this study, implanted in sheep (n = 3) with standard open-heart surgical techniques. Animals were carefully observed postoperatively, with follow-up transthoracic echocardiography (TTE), clinical pathology, and veterinary observation performed monthly to monitor animal health. At the study endpoint, twenty weeks following valve implant, animals were anesthetized for a terminal intracardiac catheterization to collect cardiac hemodynamic data, followed by humane euthanasia and a comprehensive gross necropsy by a board-certified veterinary pathologist.
Study Design and Data Collection: Summary of Animal Numbers, Implant Device Model, and Postimplant Investigations for Test and Control Groups.
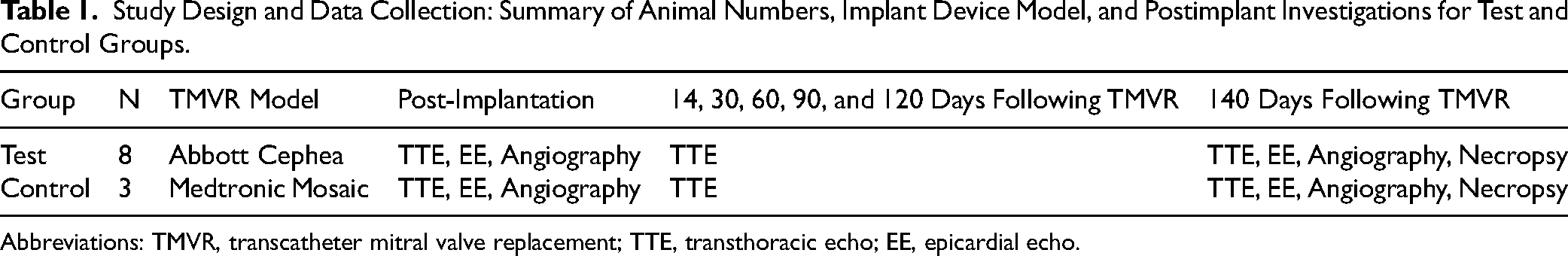
Abbreviations: TMVR, transcatheter mitral valve replacement; TTE, transthoracic echo; EE, epicardial echo.
Animals
Eleven (n = 11) domestic sheep were selected for this study, consisting of 11 castrated males, 16.0 ± 2.5 months old (mean ± SD); weighing 78.9 ± 9.1 kg. Upon arrival to the laboratory, a veterinarian performed a physical examination for heath assessment and vaccinated as well as dewormed the animals as appropriate. Animals were housed in AAALAC accredited pens in large animal housing.
Fasting/Preoperative Preparation
Animals were fasted for 12–24 h prior to anesthetic events with water provided ad libitum. Sustained release (SR) Buprenorphine (SQ) was used for pre- and postoperative analgesia at a dose of 0.12–0.27 mg/kg, given in the 24 h period prior to surgical induction.
Hybrid Surgical Approach for TMVR
Animals were sedated with 0.04 mg/kg atropine IM, 10 mg/kg ketamine IM, and 2–6 mg/kg propofol IV. Sheep were intubated, maintained on isoflurane at 2%–4% for the duration of surgery, and monitored for heart rate, mean blood pressure, fixed pupil location, palpebral reflex absence, and oxygen saturation to ensure proper anesthesia. Surgery was performed through a left fourth intercostal space thoracotomy to expose the heart and the aorta, as well as a small cutdown incision in the left neck for carotid arterial access to complete postimplant angiograms. The animals were anticoagulated and placed on cardiopulmonary bypass (CPB) using standard techniques. 11 A left atriotomy was made to expose the native mitral valve. Due to the expansive size of the atrial cuff of the Cephea valve, the sutures intended for securing the valve in place were placed before valve deployment. Pledgetted 3-0 braided polyester sutures were placed trans-annularly, using interrupted mattress stitches, around the circumference of the native mitral valve. Once all sutures were placed, the Cephea valve, loaded into its delivery system, was deployed under direct visualization. Annular stitches were placed through the atrial cuff of the Cephea valve and tied to secure the prosthesis within the annulus. The atriotomy was closed in two layers with 4-0 polypropylene sutures, while also de-airing the heart. The heart was then defibrillated with internal paddles. Once the heart converted to normal sinus rhythm, animals were weaned from CPB and cannulas were removed. Angiograms were collected by introducing a 5 Fr pigtail catheter into the left ventricle from the carotid access site. Angiograms were used to confirm prosthesis leaflet function and identify any regurgitant flow around the prosthesis or through the native aortic valve. An epicardial echocardiography exam was completed for collection of two-dimensional views of the implanted prosthesis and Doppler-derived hemodynamics. The thoracotomy and neck incisions were closed in a standard manner. Postoperatively, the animals recovered under the care of a veterinarian and received carprofen 2–4 mg/kg IM and buprenorphine 0.001–0.01 mg/kg SQ as needed for pain management.
Surgical Mitral Valve Replacement for Controls
For controls, surgical mitral valve replacement was performed using the same approach as above. Like for the hybrid approach, the heart was accessed through a thoracotomy and after commencement of CPB, the mitral valve exposed through a left atriotomy. Instead of the transcatheter Cephea device, a standard surgical valve prosthesis (Medtronic Mosaic) was sutured into the mitral annulus. Subsequently, the animal was closed and recovered as described above.
Postoperative Monitoring
Animals were observed post operatively for normal recovery from surgery including appetite, fluid intake, voiding, ambulation, respiratory rate, respiratory effort, heart rate and rhythm, willingness to stand when approached, willingness to ambulate, development of ascites, and survival. An independent veterinary assessment was performed in the presence of any atypical clinical appearance. After discharge from postoperative care, animals were housed long-term in a natural environment with pasture and appropriate shelter with onsite veterinary technical support.
Echocardiography
Transthoracic Echocardiography (TTE) exams were performed postoperatively at 14, 30, 60, 90, 120, and 140 days following TMVR. A comprehensive exam was performed following established guidelines for assessing prosthetic valves. 12
Endpoint Data Collection
On the day of the study endpoint, animals underwent an intracardiac catheterization procedure for collection of cardiac hemodynamic data, in accordance with ISO 5840:1-3 guidance. To test the device function over a range of cardiac outputs, dobutamine was administered IV (0.25 mg/h-4 mg/h) and titrated to response (increase in cardiac output from baseline: low challenge = 10%–30%, high challenge = 40%–50%). Animals subsequently underwent humane euthanasia with intravenous administration of Beuthanasia-D IV at 87 mg/kg. After death, a comprehensive gross necropsy and tissue collection was performed by a board-certified veterinary pathologist, including macroscopic examination of the implanted device and photographic documentation. Downstream tissues and gross lesions identified at necropsy or during trimming were examined microscopically for evidence of infarction, thromboembolism, inflammation, foreign material, and treatment-related morphological changes. Semiquantitative scoring of these findings was performed. All sections containing valve leaflets were examined for the presence of thrombus, pannus, endothelialization, inflammation, calcification, necrosis, vegetations, tears, and neointimal formation.
Statistics
All data were collected using Microsoft Excel 2016. Statistical analyses were performed using GraphPad PRISM software (GraphPad Software Inc). Two-sample t-tests were used to test for significance between test and control groups included in the study. Statistical significance was defined as p ≤ .05. All numerical data is reported as median and interquartile range.
Results
All 11 animals underwent successful implantation of either test or control article, were weaned from CPB, and transferred to postoperative housing. Left ventricular angiograms and epicardial echocardiograms collected following implantation of the Cephea valve showed only trace amounts of regurgitant blood flow across the mitral annulus and prosthesis (Figure 1, Figure 2A, and Table 2). No animal showed signs of moderate or severe regurgitation on postimplant angiograms. Cardiac function was measured following implantation and monitored throughout the postoperative period by TTE (Supplemental Table 1). Longitudinal postoperative echo-derived hemodynamic data showed overall comparable peak and mean mitral valve gradients in both groups at 14 and 60 days post-TMVR, apart from a significantly lower mean gradient at 14 days postprocedure in the Cephea group (6.5 [5.0, 8.5] vs 14.0 [13.0, 23.0] mm/Hg, p = .001). Group comparisons of serial echocardiography are summarized in Table 3.
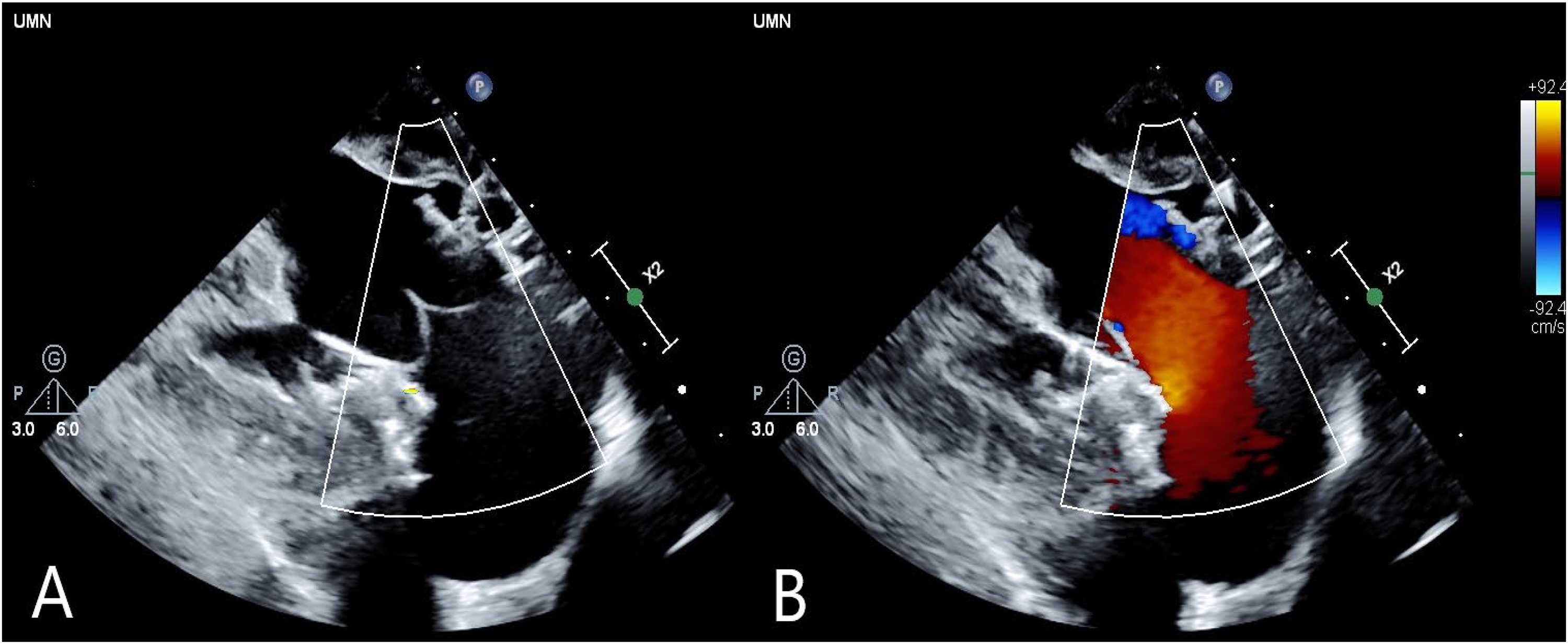
Echo imaging of Cephea valve postimplantation: (A) 2D long axis and (B) with flow Doppler applied.
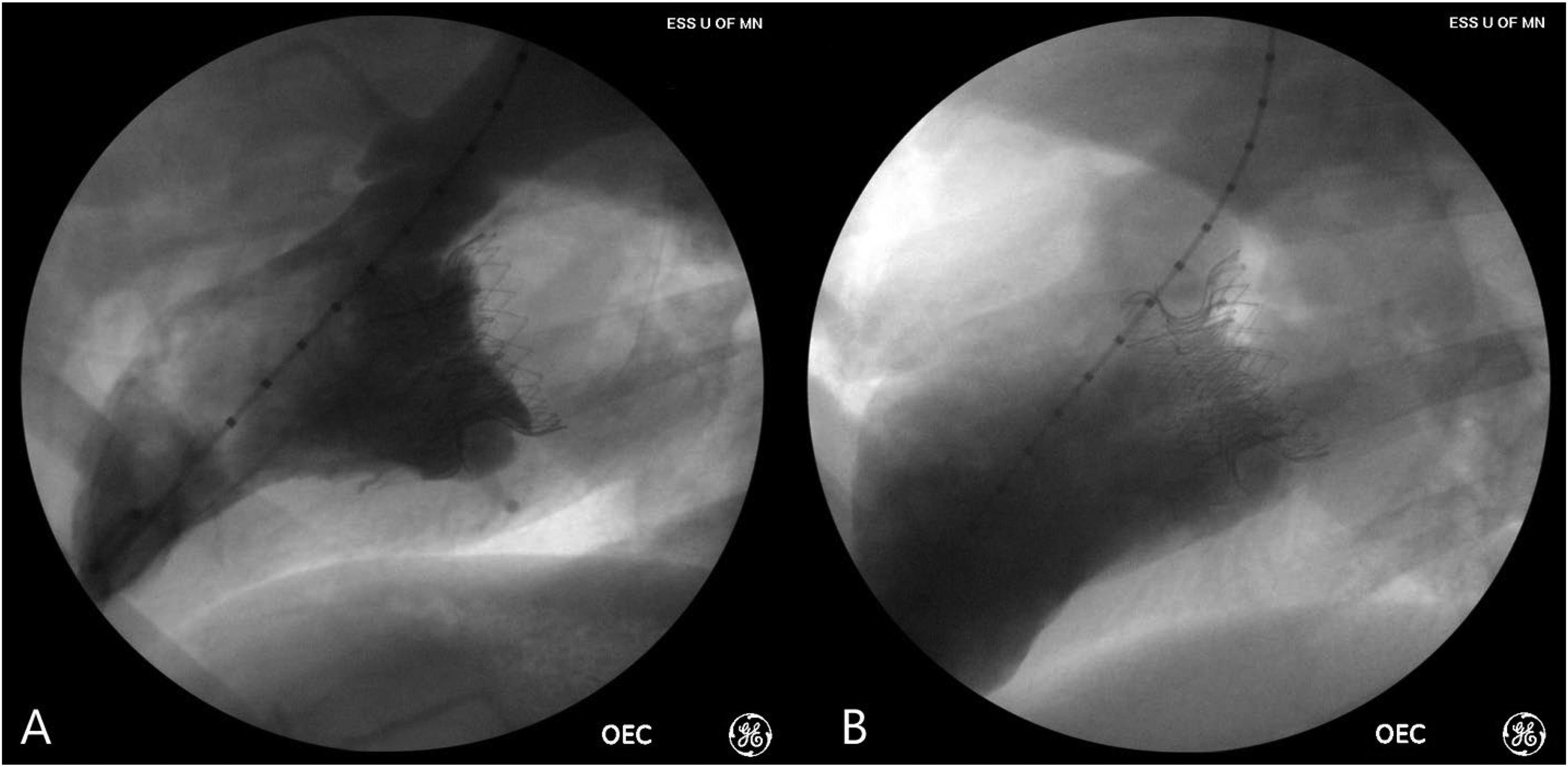
Angiography image of Cephea valve: (A) perioperatively following weaning from cardiopulmonary bypass and (B) at 20-week study endpoint.
Angiography Assessment of Mitral Regurgitation: Assessment of Mitral Regurgitation Severity Postimplantation and at 140-Day Study Endpoint for Test and Control Groups.

*0 = none, 1 = mild, 2 = moderate, 3 = marked, 4 = severe; median [IQR].
Transthoracic Echocardiography Data: Serial Measurements of Mitral Regurgitation, Aortic Peak Velocity, Left Ventricular Ejection Fraction, Peak and Mean Mitral Valve Gradient, Effective Orifice Area, Cardiac Output and Stroke Volume from 14 Days Post-TMVR Until the Study Endpoint.
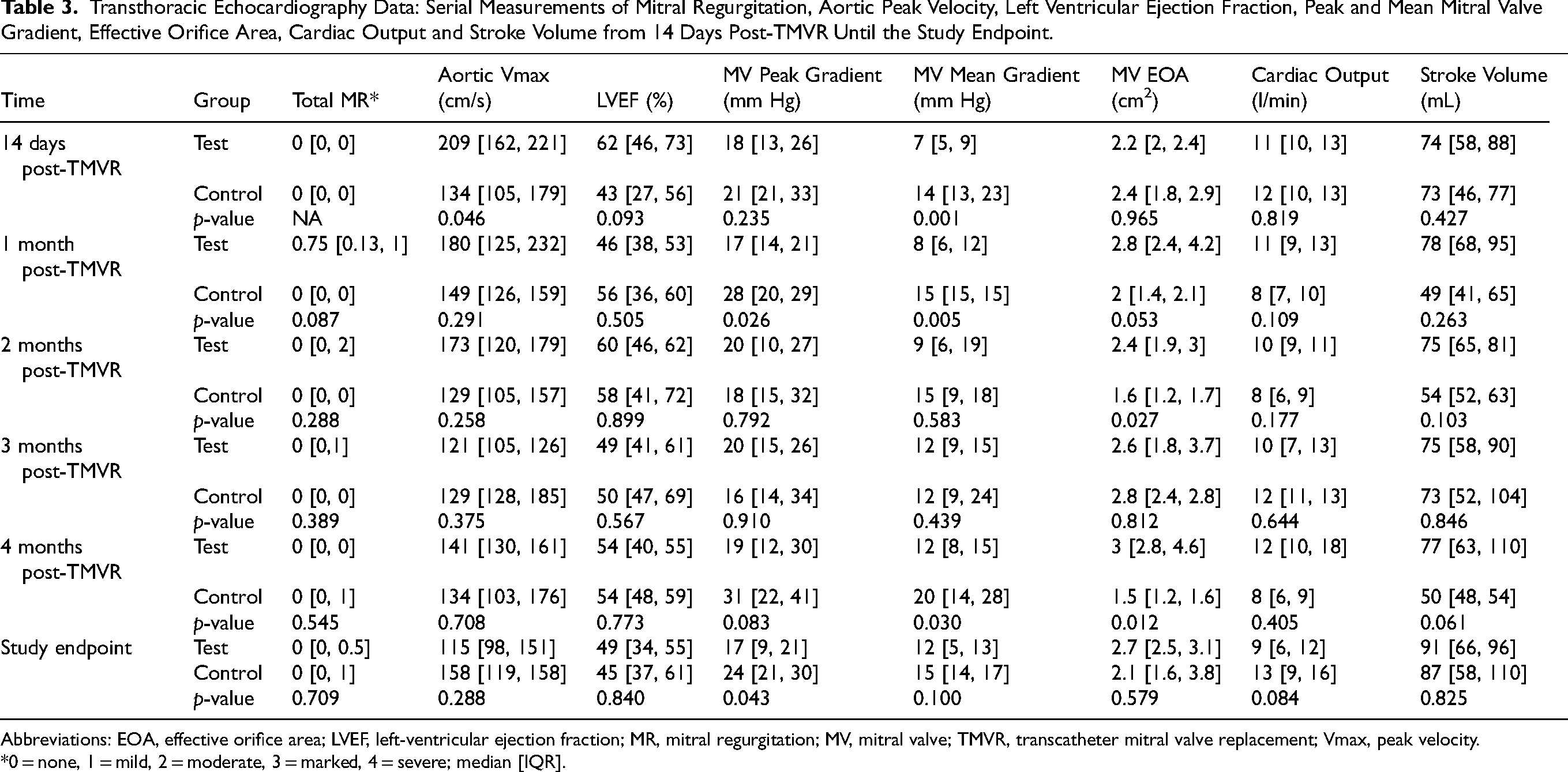
Abbreviations: EOA, effective orifice area; LVEF, left-ventricular ejection fraction; MR, mitral regurgitation; MV, mitral valve; TMVR, transcatheter mitral valve replacement; Vmax, peak velocity.
*0 = none, 1 = mild, 2 = moderate, 3 = marked, 4 = severe; median [IQR].
Ten out of eleven animals enrolled survived to their predetermined study endpoint of 140 days postimplant. Seven out of eight animals implanted with the Cephea valve lived to the 140-day study endpoint, compared to three out of three animals implanted with the Mosaic control valves. One animal implanted with the Cephea valve exhibited signs of declining health and was euthanized on postoperative day 58. A comprehensive necropsy was performed by a board-certified veterinary pathologist. Postmortem analyses concluded the cause of animal's decline to be due to systemic complications of a surgical wound infection and thus unrelated to the implanted TMVR device.
At the 20-week endpoint, animals underwent collection of hemodynamic data by intracardiac catheterization (Supplemental Table 2) which revealed mild regurgitation in both the Cephea and control groups (0.5 [0.0, 2.0] vs 0.0 [0.0, 2.0], p = .68, Table 2). One control and three test animals had moderate mitral regurgitation on angiogram. Cardiac output was comparable between both groups (4.1 [3.5, 4.8] vs 5.2 [4.6, 5.6] L/min, p = .057, Table 4), while mitral transvalvular gradients were significantly higher in controls compared to test animals both at rest (2.0 [1.0, 4.0] vs 10.0 [4.0, 21.0] mm/Hg, p = .033, Table 4) and under dobutamine-induced stress challenge (6.0 [5.0, 10.0] vs 17.0 [9.0, 28.0] mm/Hg, p = .029, Table 4). On TTE, peak transvalvular gradients were lower in the Cephea group compared to controls (17.0 [9.0, 21.0] vs 24.0 [21.0, 30.0] mm/Hg, p = .043), however there was no significant difference in mean gradients (12.0 [5.0, 13.0] vs 15.0 [14.0, 17.0] mm/Hg, p = .1, Table 3).
Intracardiac Catheterization Data: Hemodynamic Measurements at 140-Day Study Endpoint of Cardiac Output and Mitral Gradient Measured at Baseline and Under Low and High Dobutamine-Induced Stress.

Abbreviations: TMVR, transcatheter mitral valve replacement; median [IQR].
At necropsy, gross pathological assessment revealed the Cephea valves to be well seated and in proper position comparable to controls (Figure 3). The valves appeared biocompatible with minimal evidence of structural valve degeneration. There were no gross macroscopic findings in any downstream sentinel organs which were related to the presence of either valve. Histopathologic evaluation identified chronic to acute-subacute myocardial infarction in three test group animals and low-severity distal renal ischemia in two test group animals. Detailed histopathology results are provided in Supplemental Table 3. Histopathological assessment of the valve leaflets showed the leaflet-associated tissue response across study animals to be characterized by partial biointegration, with incomplete endothelialization, immature neointimal formation, and residual fibrin deposition, including evidence of persistence in some sections. No pathologically significant inflammatory response or cellular infiltration was identified within the leaflet tissue. Inflammatory findings associated with the leaflets were graded as absent to minimal and were consistent with a limited chronic tissue response (Supplemental Table 4 and Supplemental Figure 1).
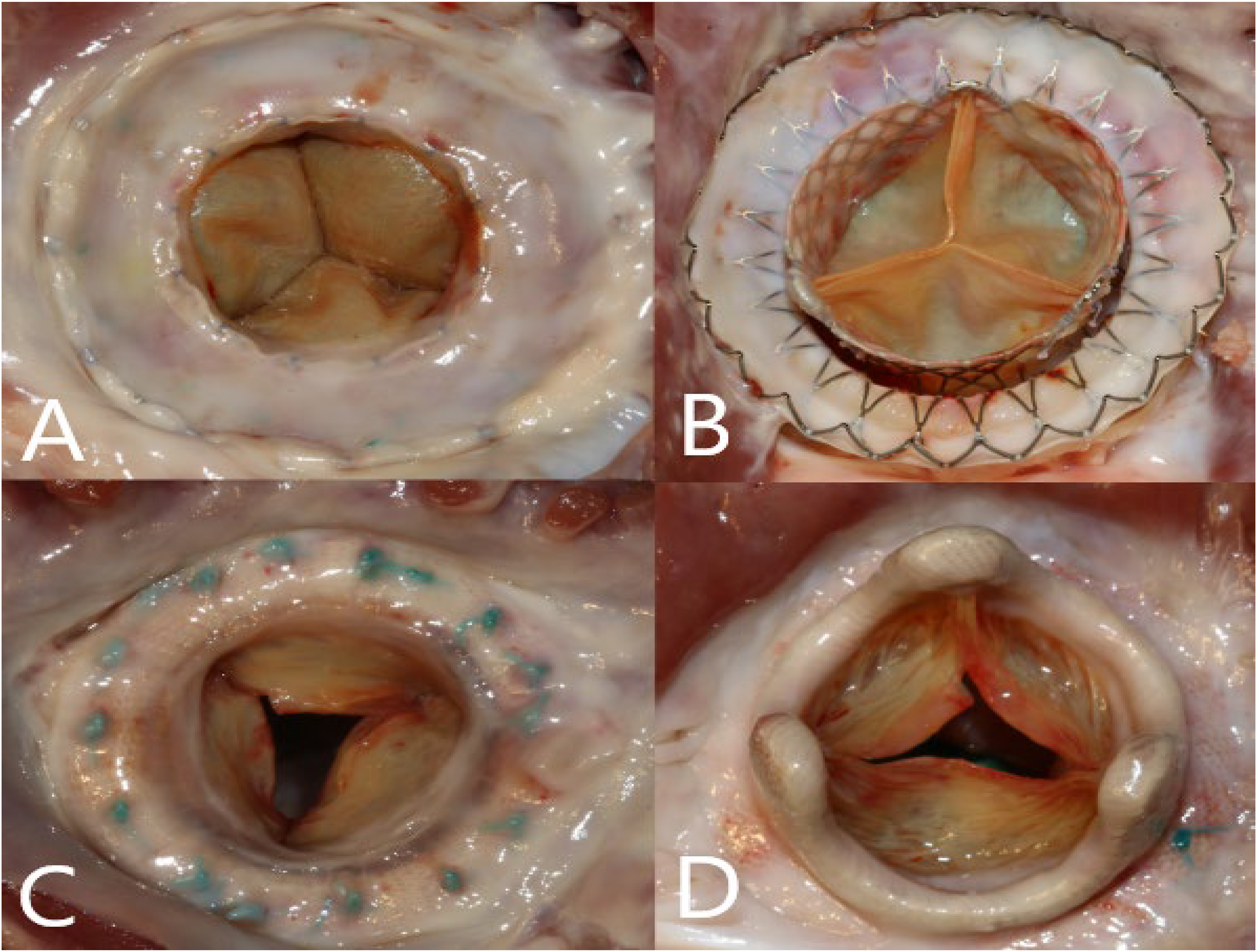
Cephea and Mosaic valves at the 20-week study endpoint: (A) inflow aspect of the Cephea valve (B) outflow aspect of the Cephea valve (C) inflow aspect of the Mosaic valve (D) outflow aspect of the Mosaic valve.
Discussion
There is a clinical need for nonsurgical approaches to mitral valve replacement in high-risk patients suffering from severe symptomatic MVD. A promising alternative are TMVR devices, however, device development requires long-term studies assessing safety and efficacy in animal models prior to clinical use. In this study, we evaluated the feasibility of using a hybrid surgical approach in sheep to evaluate the chronic in vivo performance of a novel TMVR device intended for clinical investigation approval. We aimed to show how this approach can successfully overcome the challenges of percutaneous TMVR delivery in the sheep model to ensure excellent survival and long-term data acquisition. Using CPB, we gained access to the mitral valve through a thoracotomy and left atriotomy which allowed excellent exposure and secure placement of the TMVR device. Intraoperative angiography and echocardiography confirmed the correct positioning and function of the device prior to chest closure. All eleven animals tolerated the procedure, were successfully weaned off bypass and experienced no device-related complications. Thorough postoperative monitoring included regular veterinary assessments and TTE imaging. Excellent overall survival (100% at 30 days and 88% at the 20-week endpoint) allowed collection of longitudinal, multimodal data on device feasibility, biocompatibility, and in vivo performance.
To our knowledge, this is the first report of successful long-term evaluation of a TMVR device in a chronic sheep model. Presently, there is a paucity of information available in the scientific literature regarding the preclinical study findings for TMVR devices currently in early clinical usage. The few existing publications of preclinical studies assessing transcatheter mitral valve approaches in large animal models are limited by the use of low animal numbers with only short- to medium-term follow-up. 13 Although sheep are well-established preclinical models for surgical valves, the evaluation of transcatheter devices in sheep poses unique challenges. The structure of a healthy sheep heart precludes the ability to position and secure TMVR devices in a similar fashion to interventional methods used in the human clinical population. This is associated with early postoperative mortality in sheep unrelated to the safety and efficacy of the device. The lack of information regarding chronic TMVR evaluation in animals presents a critical obstacle for investigators looking to advance novel TMVR devices to clinical use.
The novel TMVR device tested in this study was an Abbott Next-Generation Cephea. Control animals underwent standard open-heart surgical implantation of a Medtronic Mosaic bioprosthesis. Intra- and postoperative imaging and hemodynamic measurements ensured comprehensive evaluation of device function throughout the study period. Regular TTE follow-up showed comparable mitral valve gradients in test and control animals and at the 20-week study endpoint only mild regurgitation was seen in both groups. In comparison to a surgical prosthesis, the Cephea valve was not designed to have a sewing cuff seal the annulus, which may have contributed to the mild regurgitation. Intracardiac catheterization at term measured lower mitral transvalvular gradients in the Cephea group both at rest and under dobutamine-induced stress. Dobutamine challenge provided functional data and showed good performance across a broad spectrum of cardiac outputs. After sacrifice, assessments of the device and surrounding tissue allowed for further qualitative and quantitative analysis of device function and biocompatibility. This confirmed good deployment of both the test and control prosthesis as well as excellent biocompatibility.
Using this hybrid surgical approach, we were able to securely implant a novel TMVR device into an established large animal model with minimal postprocedural morbidity and mortality. This allowed us to acquire long-term data to assess device safety and performance and shows how hybrid approaches can be used in preclinical studies to safely and effectively test novel percutaneous valve prostheses in large animal models. This technique eliminates the variables associated with improper valve positioning in the model due to anatomical differences between sheep and humans, ensuring optimal preclinical evaluation of devices to provide the robust data required for regulatory approval of human studies. Furthermore, this study shows how CPB and securing sutures can facilitate valve deployment in an animal model, highlighting how a hybrid surgical approach can be a feasible and safe option in situations where interventional device placements encounter difficulties or intraprocedural complications.
We chose to utilize classically implanted surgical valve prostheses as controls for this study, since they represent the gold standard with regards to reproducibility and device function. Furthermore, in a clinical setting, TMVR is considered as an alternative to open-heart surgical approaches and hence a direct comparison in preclinical models is warranted to inform clinical decision making. The same nominal prosthesis size was used for both test and control devices. However, structural differences between surgical and transcatheter prostheses can lead to discrepancies in prosthesis sizing, especially between nominal and orifice sizes, which may have contributed to the slight differences seen in device function. Discrepancies in gradients between different imaging modalities can be explained by the fact that animals were not sedated and only physically restrained during follow-up TTE measurements. At the study end point, animals were fully anesthetized and intracardiac hemodynamic data collected according to ISO 5840:1-3 standards. This approach provides reliable functional measurements at baseline as well as across a spectrum of cardiac output indices during dobutamine challenge. Since intracardiac catheterization is considered the gold-standard modality, no TTE was performed during dobutamine stress test.
A limitation of this model is the technical expertise required to perform open-heart valve implantation using CPB. This study was performed at a high-volume center with extensive experience with both sheep model and open-heart surgical approaches. Initial survival after CPB for this center is >99% across all animal studies, which facilitated the animal survival seen in this study. Proficient use of CPB is required in order to reproduce this animal model with similar survival rates. Similarly, secure placement of the valve prosthesis requires a skilled operator capable of precise and careful suturing. Therefore, this approach can only be performed in research environments with the required equipment and expertise. Furthermore, as with all preclinical animal models, the sheep model does not completely capture the pathophysiological conditions found in human patients requiring mitral valve replacement and the device implantation technique differs from valve deployment in humans. While pigs have a similar cardiac anatomy to humans and can be used for valve replacement studies, their continuous growth is an important confounding factor, which is not translatable into humans. Hence, sheep are the most appropriate model for long-term safety study endpoints, once challenges of device securement are overcome. The relevance of the histopathological findings seen in healthy test and control animals, including myocardial infarction and distal organ ischemia, to human clinical use remains uncertain. Differences in anatomy, tissue characteristics, and mechanical environment between the ovine model and human patients with annular pathology may influence device-tissue interaction. Accordingly, the clinical significance of these observations cannot be determined based on the current model. Despite these challenges, building upon the extensive use of sheep models for the long-term study of valvular prostheses, the data acquired from this model provides important insights into device performance and safety, serving as the evidence base for future clinical application.
In conclusion, we were able to safely test a novel TMVR device using a hybrid surgical approach in an established large animal model. Excellent long-term survival allowed for acquisition of comprehensive qualitative and quantitative data demonstrating the valve's safety and biocompatibility in an established in vivo model. This study shows how a hybrid surgical approach can be effectively used in a domestic sheep model to assess long term performance of new TMVR devices and provides valuable insights into the application of preclinical animal models in the field of percutaneous valve technologies.
Supplemental Material
sj-docx-1-hvs-10.1177_30494826261424812 - Supplemental material for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model
Supplemental material, sj-docx-1-hvs-10.1177_30494826261424812 for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model by J. P. Carney, K. N. Potel, W. Tollison, E. Liljequist, L. Ogle, M. T. Lahti, A. M. Kaparos, Z. Vidlund, M. Evans, R. F. Kelly, J. T. Schappa Faustich and R. W. Bianco in Journal of the Heart Valve Society
Supplemental Material
sj-docx-2-hvs-10.1177_30494826261424812 - Supplemental material for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model
Supplemental material, sj-docx-2-hvs-10.1177_30494826261424812 for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model by J. P. Carney, K. N. Potel, W. Tollison, E. Liljequist, L. Ogle, M. T. Lahti, A. M. Kaparos, Z. Vidlund, M. Evans, R. F. Kelly, J. T. Schappa Faustich and R. W. Bianco in Journal of the Heart Valve Society
Supplemental Material
sj-docx-3-hvs-10.1177_30494826261424812 - Supplemental material for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model
Supplemental material, sj-docx-3-hvs-10.1177_30494826261424812 for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model by J. P. Carney, K. N. Potel, W. Tollison, E. Liljequist, L. Ogle, M. T. Lahti, A. M. Kaparos, Z. Vidlund, M. Evans, R. F. Kelly, J. T. Schappa Faustich and R. W. Bianco in Journal of the Heart Valve Society
Supplemental Material
sj-docx-4-hvs-10.1177_30494826261424812 - Supplemental material for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model
Supplemental material, sj-docx-4-hvs-10.1177_30494826261424812 for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model by J. P. Carney, K. N. Potel, W. Tollison, E. Liljequist, L. Ogle, M. T. Lahti, A. M. Kaparos, Z. Vidlund, M. Evans, R. F. Kelly, J. T. Schappa Faustich and R. W. Bianco in Journal of the Heart Valve Society
Supplemental Material
sj-docx-5-hvs-10.1177_30494826261424812 - Supplemental material for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model
Supplemental material, sj-docx-5-hvs-10.1177_30494826261424812 for Evaluation of a Hybrid Surgical Approach for Transcatheter Mitral Valve Replacement in the Domestic Sheep Model by J. P. Carney, K. N. Potel, W. Tollison, E. Liljequist, L. Ogle, M. T. Lahti, A. M. Kaparos, Z. Vidlund, M. Evans, R. F. Kelly, J. T. Schappa Faustich and R. W. Bianco in Journal of the Heart Valve Society
Footnotes
Acknowledgments
The authors thank the staff and students of the University of Minnesota's Experimental Surgical Services Laboratory and Research Animal Resources for their care and husbandry of animals in this study.
Author Contributions
J. P. Carney conceptualized the hybrid surgical approach, performed the sheep surgeries, analyzed the data, and wrote the manuscript. K. N. Potel collected and analyzed the data and contributed to writing, editing, and preparation of the manuscript. W. Tollison, E. Liljequist, L. Ogle, and M. T. Lahti contributed to performing surgeries, data collection, and data analysis. A. M. Kaparos, Z. Vidlund, and M. Evans created the experimental design and edited the manuscript. R. F. Kelly edited the manuscript and provided clinical guidance. J. T. Schappa Faustich performed data collection, data analysis, and edited the manuscript. R. W. Bianco developed the project concept and supervised the overall project.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Abbott Laboratories,
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Michael Evans, Zach Vidlund, and Anna-Maria Kaparos are full-time employees of Abbott Laboratories. No other authors have conflicts to disclose.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
