Abstract

Keywords
Bioprostheses (BPs), used for their low thrombogenicity, have gained ground over mechanical valves in the past 2 decades. Their use is expected to rise for both aortic and mitral valve replacements as a result of population aging, patient preference, and the possibility of transcatheter options for degenerated BPs. The types of surgical mitral valves currently implanted are stented valves, either porcine—made from native pig aortic valve leaflets—or bovine—with leaflets constructed from cow pericardium. Since the introduction of the first model in the late 1960s, over 10 different prosthesis models have been commercialized. It is essential to examine their distinctions and understand their expected individual performance.
Paper Aim and Conclusion
In the present study, Delanoë et al compared the hemodynamic performance of 2 recent models of surgical mitral BPs: the porcine Epic Plus (Abbott) size 29 mm and the bovine pericardial Mitris Resilia (Edwards Lifesciences) size 27 mm in an in vitro pulse cardiac simulator. 1 The tested valves were submitted to various physiological and pathological flow conditions. Both valves exhibited excellent hemodynamic performance. The pericardial type Mitris demonstrated a modestly larger geometric orifice area (GOA) based on camera measurements, a lower mean transvalvular pressure gradient (mTPG), and a greater effective orifice area (EOA) as determined by combined Doppler echocardiography and flowmeter analysis compared to the porcine Epic Plus.
The authors should be congratulated for providing a valuable in vitro head-to-head, hemodynamic comparison of these 2 BP models, tested in their respective label sizes that are theoretically suitable for the same mitral annulus. They also proposed an interesting prediction of indexed EOA across a range of plausible body surface areas and anticipated higher risk of patient-prosthesis mismatch (PPM) with Epic Plus implantation compared to Mitris.
Editorial Question
Does this mean that pericardial mitral BPs are superior to porcine ones overall? A simple “yes” would assume that tissue type alone determines hemodynamic performance, and that this performance is the sole measure of a BP's effectiveness in improving patient quality of life. However, the reality is more complex.
Zoom-in: Hemodynamic Performance
Although the Mitris and Epic Plus valves differ in tissue type, they both feature internally mounted leaflets and exhibit intra-annular function—traits characteristic of mitral BPs—and share similar stent internal diameters (27 mm vs 26.3 mm), as reported by the manufacturers. Nevertheless, the 2 BPs exhibited notable hemodynamic differences.
Specific Design Features
Despite close industrial specifications of these 2 studied valves, they exhibit distinct profiles. The porcine Epic Plus valve features sloped posts and a smaller outflow-side diameter compared to the inflow, resulting in a less favorable funnel-shaped profile. In contrast, the pericardial Mitris valve shows minimal diameter reduction from inflow to outflow, leading to a more hydrodynamic, tubular profile. This may influence the valve's maximal opening and help explain the slightly higher GOAs observed for the Mitris compared to the Epic Plus. 2 It may also affect flow patterns across each valve model—particularly the contraction coefficient (CC)—resulting in even more pronounced differences in EOA and mTPG. The CC defined as the ratio of EOA to GOA—also referred to in clinical practice as the performance index—reflects how efficiently the flow utilizes the valve orifice. In most tested conditions, it favored the Mitris valve. Moreover, the Epic Plus has a relatively bulky leaflet insertion. One might question whether this bulkier annulus could negatively affect inflow geometry and, in turn, contribute to altering the CC.
Design Effects Are Flow-Dependent
Surprisingly, the overall hemodynamic advantage of the Mitris valve was lost under low-flow, normal heart rate conditions. This may reflect both potentially suboptimal opening of the pericardial valve and increased flow contraction (ie, a lower CC) at reduced flow rates. These complex fluid-structure interactions could be influenced by prosthesis design, including leaflet tissue characteristics, such as thickness (slightly greater for pericardial bovine leaflets), and elasticity. Conversely, pressure gradients appeared to be more affected by high flow in the porcine valve than in the pericardial one. So, the valve performance and the influence of design (including tissue type) are modulated by flow conditions, which are patient-specific.
Valve selection requires consideration of each patient's characteristics, such as flow state. The possibility of incomplete valve opening in low-flow situations should be balanced with the risk of higher gradients at elevated flow rates. Because of these complex mechanisms, the hemodynamic performance of a BP cannot be determined solely by valve type or design; instead, model-specific empirical data that include flow dynamics are necessary, emphasizing the value of comprehensive reference datasets.
Zoom-out: Other Valve Features
Mitral bioprosthetic valves feature several other important characteristics, some of which are related to hemodynamics, while others are not.
Durability
Durability remains one of the biggest challenges in the era of BPs. Regardless of whether the valve is porcine or bovine pericardial, the risk of reoperation increases sharply after the first decade following implantation. 3 Several recent studies have suggested that porcine mitral BPs may offer greater durability than bovine pericardial ones, particularly in younger patients (eg, under 65 years).4–8 This finding contrasts with the general trend, in which pericardial BPs typically offer superior antegrade hemodynamics. This suggests that durability is not solely associated with hemodynamic performance, but also depends on other factors potentially related to leaflet-structure interaction and the material itself—including tissue properties such as flexibility, thickness, source (native valve vs pericardial sheet), and treatments used for tissue fixation, anti-calcification, and storage.
Porcine BPs tend to fail due to leaflet tears leading to regurgitation, whereas pericardial BPs are more prone to calcification and thickening, resulting in mitral stenosis.5,9 Although porcine valves are generally associated with smaller EOAs and thus a higher risk of PPM, their mode of failure may confer greater tolerance to the high shear stress conditions imposed by PPM compared to pericardial tissue. This could help explain the lower rates of reintervention or valve explant reported for porcine valves in some series.
However, these interpretations remain to be approached with caution, as some bias cannot be excluded, eg, related to valve selection and to non-standardized structural valve deterioration definition, which is often based on valve explant rather than echo follow-up findings. 10 Furthermore, patient's response to BP dysfunction and its therapeutic management might differ depending on the BP mode of failure. Pericardial BPs may also be preferentially chosen in anatomically challenging or higher surgical risk cases based on the assumption that they offer superior hemodynamic performance.
The mechanisms of valve failure likely involve complex interactions between tissue properties, valve design, and flow dynamics. Comprehensive, well-structured in vivo studies with extended follow-up periods of over 10 to 15 years, tailored to each BP model and, optimally, each size, utilizing standardized echocardiography-based definitions of structural valve deterioration, are essential to accurately assess whether new devices meet their intended outcomes and to facilitate meaningful comparisons among them.
New Material Technologies
Emerging technologies have the potential to significantly enhance durability. For instance, the innovative Resilia tissue used in the Mitris valve—featuring enhanced anti-calcification properties such as dry storage—has shown promising results in the aortic position with the Inspiris valve. 11
Furthermore, the development of new synthetic, non-animal materials—such as polymers or bioengineered tissues12–14—holds great potential to overcome the main barrier to patient's global access to these BPs: their cost. These materials could facilitate the manufacture of these devices and reduce reliance on animal-derived tissues.
Implantability—Anatomical Compatibility
BPs can also be distinguished by their implantability. Engineers have worked on design features such as larger, more flexible, and anatomically tailored sewing skirts (as seen in the Epic Plus and Mitris valves) to improve conformability and sealing, while reducing stress on leaflets; low-profile frames to prevent left ventricular outflow tract obstruction 15 ; and even color-coded markers to aid orientation during implantation.
Transcatheter Valve-in-Valve
Finally, since the perfect valvular device does not yet exist, surgeons should anticipate potential valve degeneration at the time of BP selection and optimize the feasibility of valve-in-valve procedures.15,16 For example, selecting a valve with radio-opaque stent at the annulus level can facilitate future interventions.
Conclusion
Hemodynamic performance is crucial for preventing PPM, improving patient outcomes, and ensuring valve durability. It depends on both tissue type and device design, as well as flow dynamics. Consequently, there is a need for up-to-date independent and unbiased hemodynamic reference data for each BP model in use—especially the latest generations. In this regard, in vitro studies are particularly valuable, as they allow for objective characterization across a wide range of conditions, eliminating confounding factors such as patient anatomy, flow variability, and measurement errors. However, it is important to remember that hemodynamic performance is just one of several key determinants when selecting a mitral BP. Achieving optimal performance demands a careful balance among hemodynamics, durability, ease of implantation, and anatomical compatibility, through the integrated consideration of design features, tissue type, geometry, anticipated mode of failure, and patient-specific factors such as anatomy and flow state. Attention should also be given to the risk of reintervention, and the potential need for future strategies such as valve-in-valve procedures (Figure 1).
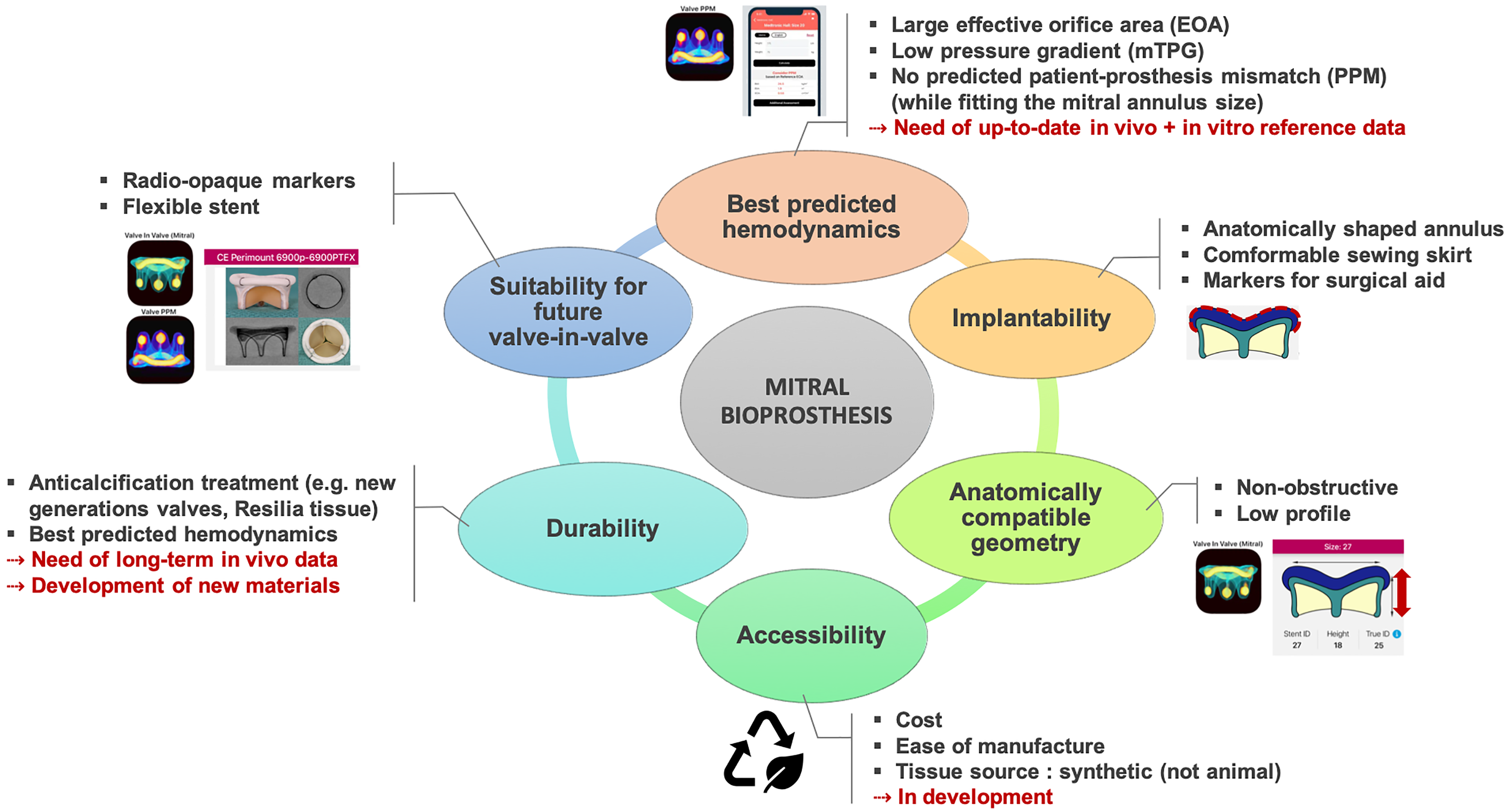
Key Features of a Mitral Bioprosthesis. The squared logos refer to the mobile applications—Valve PPM and Valve in Valve (Mitral)—which centralize key features and reference data for most bioprostheses currently in use. Valve-related images are adjusted from these apps.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
