Abstract
Congenital valve disease, predominantly in the pulmonary position, requires multiple replacements as current bioprosthetic valves degrade and calcify over time. An allogenic, decellularized bioengineered tissue with minimum immune response and calcification would provide an ideal solution for young patients and could substantially improve the standard of care. In this study, a transcatheter pulmonary valve with bioengineered tissue leaflets on a self-expanding nitinol stent was implanted for up to 18 months in five juvenile sheep using an 18Fr transapical delivery system. Serial echo monitoring showed an increase in cardiac output as the juvenile sheep grew without any decrease in effective orifice area (>2 cm2) or increase in pressure gradients (<10 mmHg for both peak and mean). No valvular regurgitation, leaflet thickening, or tissue abnormality was reported at any time points. Micro-CT and histological assessment showed mild calcification and mild thrombosis. The 18-month implants in the highly calcific juvenile sheep model showed no loss of function and no deterioration of the bioengineered tissue, suggesting a suitable alternative leaflet material for younger patients where functional longevity is critical.
This is a visual representation of the abstract.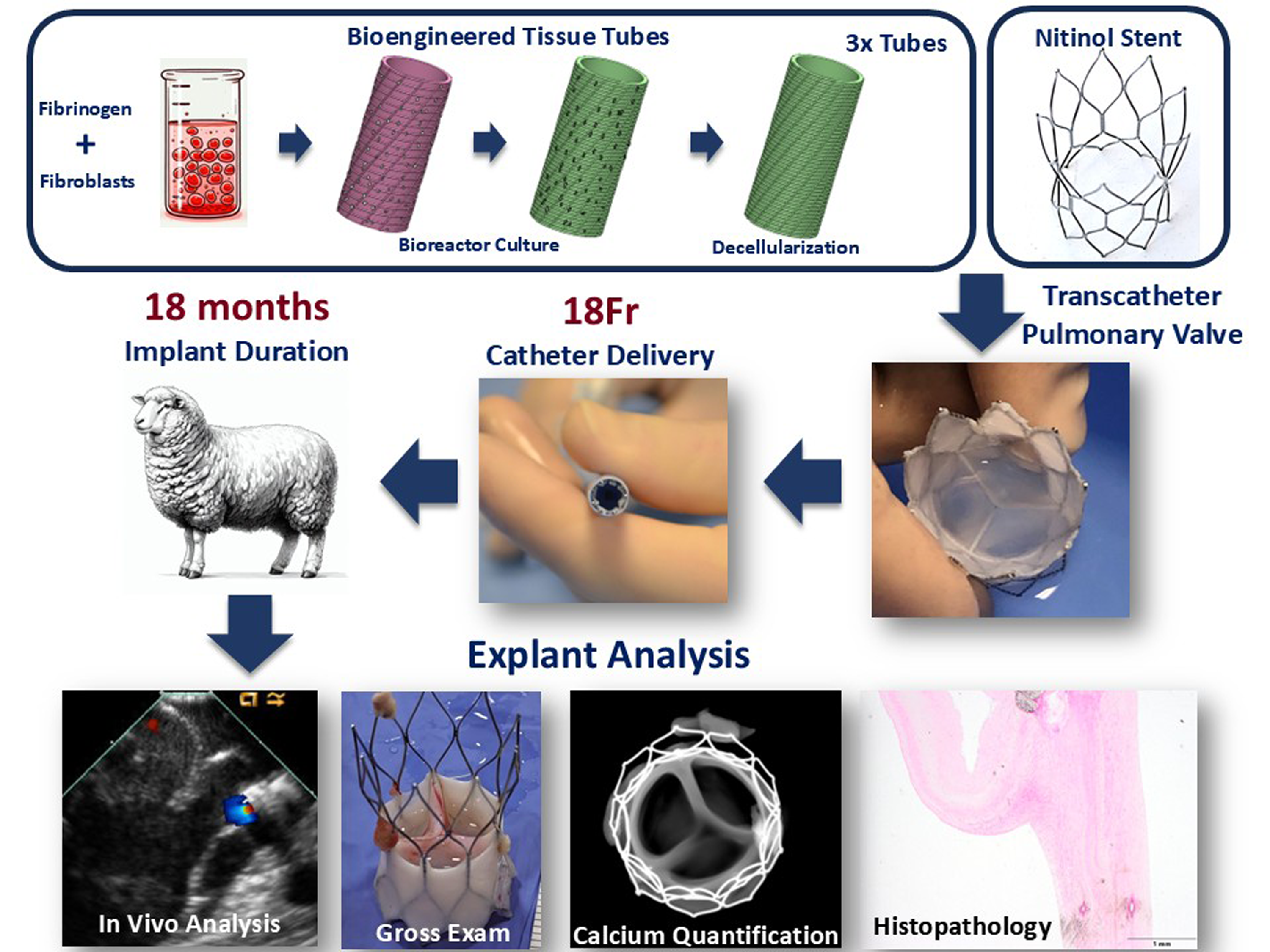
Keywords
Introduction
Heart valve diseases are a significant cause of morbidity and mortality worldwide, necessitating surgical interventions that often involve valve replacement.1,2 Traditional heart valve replacement options include mechanical valves and bioprosthetic valves, each with its own set of advantages and limitations. While mechanical valves offer durability, they require lifelong anticoagulation therapy and often fail early in the low-pressure right heart circulation. In contrast, bioprosthetic valves, typically made from bovine or porcine tissues, are favored for their biocompatibility and lack of need for long-term anticoagulation, making them particularly appealing for specific locations such as the pulmonary valve and in specific patient populations, including children and young adults.3,4
However, the performance of bioprosthetic heart valves in young adults has been suboptimal compared to in older patients.5,6 Historic outcomes reveal that while bioprosthetic valves initially provide satisfactory hemodynamic performance and improved quality of life, their durability is significantly compromised in younger patients.3,6 Accelerated degeneration and calcification of the valve tissue is a predominant failure mode in younger patients, resulting in progressive stenosis, regurgitation, and ultimately requiring reoperation. 7 Studies indicate that the median lifespan of bioprosthetic valves in young adults is markedly shorter, often necessitating repeat interventions within 5–10 years post-implantation.
Tissue calcification is a critical challenge in bioprosthetic heart valve durability. 3 The pathological deposition of calcium salts within the valve leaflets leads to stiffening and loss of function. Biological and mechanical stresses imposed on the valve exacerbate this process, particularly in the younger, more active population. Several factors such as the patient's metabolic profile, valve tissue properties, and implantation technique influence the propensity for calcification. Despite advances in anti-calcification treatments and valve design, current bioprosthetic valves still fall short of the ideal durability needed for younger patients.5,8
The urgent need for improved bioprosthetic heart valves has spurred the development of tissue-engineered heart valves (TEHVs).9–11 These novel valves aim to overcome the limitations of traditional bioprosthetic materials by utilizing bioengineered tissues that can potentially grow, repair, and remodel in response to the dynamic environment of the cardiovascular system.11–14 Innovations in biomaterials, scaffold design, and cellular engineering are pivotal in this endeavor. The ideal leaflet material for TEHVs would resist calcification, integrate seamlessly with the host tissue, and maintain structural integrity under physiological conditions.
In this study, bioengineered tissue, having already demonstrated the capacity for recellularization and biocompatibility in a growing sheep model as vascular grafts, 14 pulmonary valved conduits, 11 and in human clinical trials for dialysis access, 15 was evaluated for long-term hydrodynamic performance and calcification resistance in the transcatheter pulmonary valve (TPV) application in the juvenile sheep model. By addressing the shortcomings of current bioprosthetic valves, this study aims to pave the way for durable and functional heart valve replacements that can meet the long-term needs of younger patients.
Materials and Methods
Valve Fabrication
In-house expertise was leveraged to design and fabricate the valve stent frame using self-expanding nitinol material. The design was chosen to provide stable placement in the cylindrical pulmonary artery without paravalvular leak in multiple diameters, which is the anticipated need in congenital repaired pulmonary outflow tracts. 15 The valve material was comprised of bioengineered tissue tubes 16 mm in diameter manufactured as described previously. 11 Briefly, neonatal human dermal fibroblast cells at 1million/ml concentration were mixed in fibrin gel at 4 mg/ml in a 16 mm tubular mold. The formed gel was cultured in a bioreactor in DMEM media with 10% FBS for 9 weeks. After maturation, the tissue tubes were decellularized using 1% SDS, and 1% Triton-X, then extensively washed in phosphate-buffered saline. Aseptically, each decellularized engineered tissue tube was then cut to create a single leaflet where three tubes were manually stitched using Teleflex 7-0 suture. Additional bioengineered tissue was used as a wrap on nitinol frame (Figure 1a, b). Each leaflet had a height of 11 mm and a width of 25 mm (commissure to commissure). For porcine pericardium, the same dimensions of 11 mm × 25 mm were used for the leaflets and stitched using same pattern as the bioengineered tissue (Figure 1c, d). The nitinol frame was designed with an hour-glass shape 3 cm in height. The diameter at the two ends is 30 mm and the middle is 24 mm. The nitinol frame was manufactured by laser cutting a 6 mm Inner Diameter × 0.5 mm Thick SE508 nitinol tube (purchased from Confluent Medical, CA USA) with a nine cell design. The laser cutting, expansion, shape setting, and electropolishing processes were performed by Northern Nitinol LLC (MN, USA). The porcine pericardium tissue used in the control valve was cleaned and fixed in 2% glutaraldehyde and then cut into the exact same dimensions as bioengineered tissue.

Images of assembled transcatheter pulmonary valve with bioengineered tissue. (a) End-on view, (b) side-view, and with fixed porcine pericardium, (c) end-on view, (d) side-view. End-on images during hydrodynamic testing in (e) systolic and (f) diastolic phase. (g) Flow pressure profile during hydrodynamic testing, accelerated wear testing with. (h) Image of valve prior to start durability testing. (i, j) End-on views of valve after 200 million cycles and (k) pressure wave applied for durability testing.
Accelerator Wear Testing
A stentless valve (n = 1) was mounted on a custom silicone fixture using stitching consistent with those of the transcatheter valve design and placed in the VDT-3600i Heart Valve Durability System (BDC Labs, CO USA) with two chambers. The system maintains 37 °C temperature and the pressure gradient is monitored using two pressure transducers (Deltran Transducer, Utah Medical, UT USA). Valves were tested at 12 Hz frequency with an end-diastolic pressure of ∼60-80 mmHg (transducer accuracy ±0.1% max scale), which exceeds the pulmonary diastolic pressure gradients, including for hypertensive clinical cases. One valve manufactured using bioengineered tissue was run for 200 million cycles with concurrent visual inspections. Post durability testing, valve stitch lines were visually inspected for failures, then hydrodynamic properties were measured to evaluate any changes associated with accelerated wear testing.
Pulse Duplicator Operation
A customized pulse duplicator system previously described 11 was used. It consists of a commercial wave generator and pump (ViVitro Lab, BC Canada), reservoir, valve mounting chamber, variable compliance chamber, and mechanical bi-leaflet valve to ensure one-directional fluid movement. The system had pressure transducers (Deltran ±0.1% max scale, Utah Medical, UT USA) immediately upstream and downstream of the valve to allow for accurate pressure measurements. Additionally, the valve had an electromagnetic flow meter model FM501, reading accuracy ±1% (Carolina Medical, NC USA), upstream to measure the flow rate in both directions. A custom LabVIEW® program was used to record flow rates and pressures. The valves (n = 2 bioengineered tissue and n = 2 fixed porcine pericardium) were mounted inside a custom silicone sleeve to be secured inside the system. The pulse duplicator loop was run with phosphate-buffered saline as the test fluid at room temperature. Each valve was tested with end-diastolic pressure gradient (range 40−80 mmHg) to mimic pulmonary pressure conditions at a prescribed flow rate (set by pump stroke volume and frequency). While normal pulmonary pressure gradients are 20mmHg or less, higher pressures are reported for patients with right ventricular outflow tract dysfunction, 16 hence the range used here is appropriate. An average flow rate of 5 liters per minute (LPM) was used to mimic pediatric cardiac output. 17 Pressure was controlled by changing the downstream flow resistance, stroke volume, and upstream hydraulic pressure head. During valve testing, end-on-camera images were obtained. Valve properties, including systolic pressure gradient, effective orifice area (EOA), and regurgitation fraction were calculated using pressure and flow traces.
Tensile Strain-to-Failure
Test strips with dimensions ∼3 mm × ∼20 mm were cut from the bioengineered tube, the fixed porcine pericardium, and the explanted tri-tube valve. Each strip was tested in tensile strain-to-failure as previously described.13,18 For pre-implant tissue, strips in both circumferential and axial directions were cut corresponding to circumferential being in the commissure to commissure direction of the leaflets and axial being the height of the leaflets. Sample dimensions were measured before testing using a digital caliper. The strips were mounted in custom grips attached to the actuator arms of an Instron tensile testing system (accuracy ±0.5% max scale, Instron Systems, MA USA) and straightened with a 0.05 N tensile load. Strain was calculated by taking the natural logarithm of the sample's deformed length over its initial length. Stress was defined as the force divided by the undeformed, cross-sectional area of the strip. Modulus and ultimate tensile strength were taken as the slope of the linear region of the stress-strain curve and the maximum stress, respectively.
Valve Implantation
A juvenile sheep model (age <6-month-old) was selected for this study based on prior literature showing rapid calcification. 19 Animals were selected based on pulmonary valve annulus via echocardiogram measurement. An echocardiogram-guided transapical delivery method was utilized to ease implant and animal recovery. Juvenile Dorset breed sheep of ∼5 months old were used for the implant. All animal procedures and housing were performed at American Preclinical Service (APS) (now NAMSA). The studies were conducted using an approved Institutional Animal Care and Use Committee (IACUC) protocol. Five sheep were implanted with the bioengineered tissue valves.
Under general anesthesia, a mini thoracotomy was performed to expose the apex of the heart. The valve was crimped and inserted into an 18Fr delivery catheter. Using epicardial echocardiography imaging, the delivery catheter was passed from the apex to the pulmonary artery and deployed over the native pulmonary valve, ensuring the stent does not protrude into the ventricle. The implanted valve function was evaluated with echocardiography, which included leaflet motion, systolic gradient, paravalvular leak, and regurgitation across the valve. Incision sites were repaired, and the animal was recovered per protocol. Animals were kept on the anticoagulant enoxaparin (1 mg/kg BID) for 4 weeks following implant. An echocardiogram was performed every 2 months to evaluate the valve performance. Planned explants were performed at 6 months (n = 1), 12 months (n = 1) and 18 months (n = 2).
Explant Examination and Histology
The heart was grossly examined from the exterior and interior with the valve exposed at term. The explanted valve was imaged in Faxitron®. Valve leaflets and root sections were cut for tensile testing and histology. Histology samples were fixed in 10% formalin and sent to CVPath Institute for further processing. At CVPath Institute, samples were imaged with Micro-CT, then processed for wax embedding, sectioning, and staining with Masson's Trichrome, Pentachrome, Von Kossa and Hematoxylin and Eosin (H&E) stains. Stained slides were digitally scanned and examined.
Statistics
Bioengineered tissue thickness and tensile properties for implants and explants were evaluated using one-way ANOVA with Tukey post hoc test for differences at P < 0.05 using GraphPad Prism software. Paired symbols are used to show statistical differences between groups. Linear regression analysis was performed on serial echocardiogram measurements to evaluate any trends over time.
Results
In Vitro Hydrodynamics
Figure 1e, f shows the 24 mm prototype valves manufactured using bioengineered tissue depicting the full opening during the systolic phase and the complete closure during the diastolic phase. The flow and pressure profile during a single cycle is shown in Figure 1g. The hydrodynamic properties outlined in ISO 5840 Cardiovascular Implant Guidelines specify an EOA requirement of >1 cm2 and a regurgitation fraction <10%. The engineered tissue valve demonstrated a systolic pressure drop of 5.5 ± 0.1 mmHg, leading to an EOA of 2.3 ± 2 cm2 and a regurgitation fraction of 12.4 ± 1.0%. The measured hydrodynamic properties of bioengineered tissue valve and fixed pericardium sheet valve based on identical design are reported in Table 1. The bioengineered tissue valve had a 17% greater EOA given an equivalent valve diameter and leaflet dimensions. The tensile properties of bioengineered tissue showed anisotropy with modulus being 5.7-fold higher in the circumferential direction compared to axial direction (Table 2), while fixed porcine pericardium showed isotropic modulus with similar values in two directions.
Hydrodynamic Properties of Bioengineered TPV and Fixed Porcine Pericardium TPV with Same Design.

TPV, transcatheter pulmonary valve; EOA, effective orifice area; AWT, accelerated wear testing; ΔP, change in pressure.
Tensile Mechanical Properties of Bioengineered and Fixed Porcine Pericardium Tissue.

Paired symbols are used to show statistical differences between groups.
In Vitro Durability Testing
A bioengineered valve mounted in an accelerated wear tester (Figure 1f) was evaluated over a 5-month period during which the valve cycled at 12 Hz to 200 million cycles (Figure 1g). At termination, the valve was visually inspected (Figure 1h-j), showing no damage at stitch line or propagation of stitch holes. Furthermore, the evaluation of the valve hydrodynamic properties showed no change in EOA, degree of regurgitation, or pressure drops (Table 1).
Valve Crimp Testing and in Vivo Delivery
During in vitro testing following baseline hydrodynamic testing, the valves were crimped to clinical standard 18Fr diameter (Figure 2a, b) with no qualitative visual damage observed. Hydrodynamic testing after crimping showed no change in the performance metrics of the valves (Table 1). Based on in vitro confirmation, implants were performed using an 18Fr catheter with echocardiography-guided transapical delivery (Figure 2c).

Images of the implantation procedure, (a) transcatheter valve crimped into delivery system (18F), (b) end-on image of the valve in the delivery catheter, (c) valve deployment into lamb heart using echocardiogram-guided transapical approach.
In-Life Echocardiographic Evaluation of Valve Performance
All implanted valves had normal hemodynamic function in the juvenile sheep model. The healthy compliance of the native pulmonary artery (as opposed to the repaired post-surgical outflow tract) and softness of the nitinol stent led to stent migration downwards into the right ventricle in three cases. In one implant, the stent migrated upwards into the pulmonary artery leading to a non-functional valve, which was taken out at 30 days as native valve in series did not allow for implant valve to function. Explants were performed at 6 months (n = 1), 12 months (n = 1) and 18 months (n = 2). The anatomical growth (weight gain, Table 3) did not show any adverse effect on the valve function as followed over 18 months. The long-term performance showed no change in leaflet thickness (visually), pressure drop, or hemodynamic performance (Figure 3, Table 3). Linear regression analysis of systolic pressure drops and EOA showed no significant changes. Before explant at 18 months, valves were visually imaged using intravascular echocardiography which showed no flow restriction (Figure 3a, Suppl Video 1), fully coapting leaflets (Figure 3b, Suppl Video 1), and end-on view with leaflet coaptation and no central regurgitation (Figure 3c, Suppl Video 1). There was a paravalvular leak in the two 18-month implants due to valve migration downwards.

Echocardiogram imaging after 18 month implant, (a) color Doppler in forward flow, (b) color Doppler in diastolic flow with no central regurgitation and uniform leaflet thickness, (c) end-on view of leaflets, (d) mean and maximum systolic pressure drop, (e) cardiac output and effective orifice area (EOA), and (f) regurgitation with EOA. N = 3.
Hemodynamic Function Measured during the Course of Implant using Echocardiography.

EOA, effective orifice area.
Gross Explant Pathology
At all-time points, the explanted heart had normal anatomy with no evidence of ventricular wall thickening. Grossly, lungs were healthy with no time points showing evidence of embolism. Once the myocardium was dissected, the valves showed no major pannus growth around the implant site (Figure 4a, b) and the frame was removed easily from the implant site (Figure 4c, d, 18 months explants).
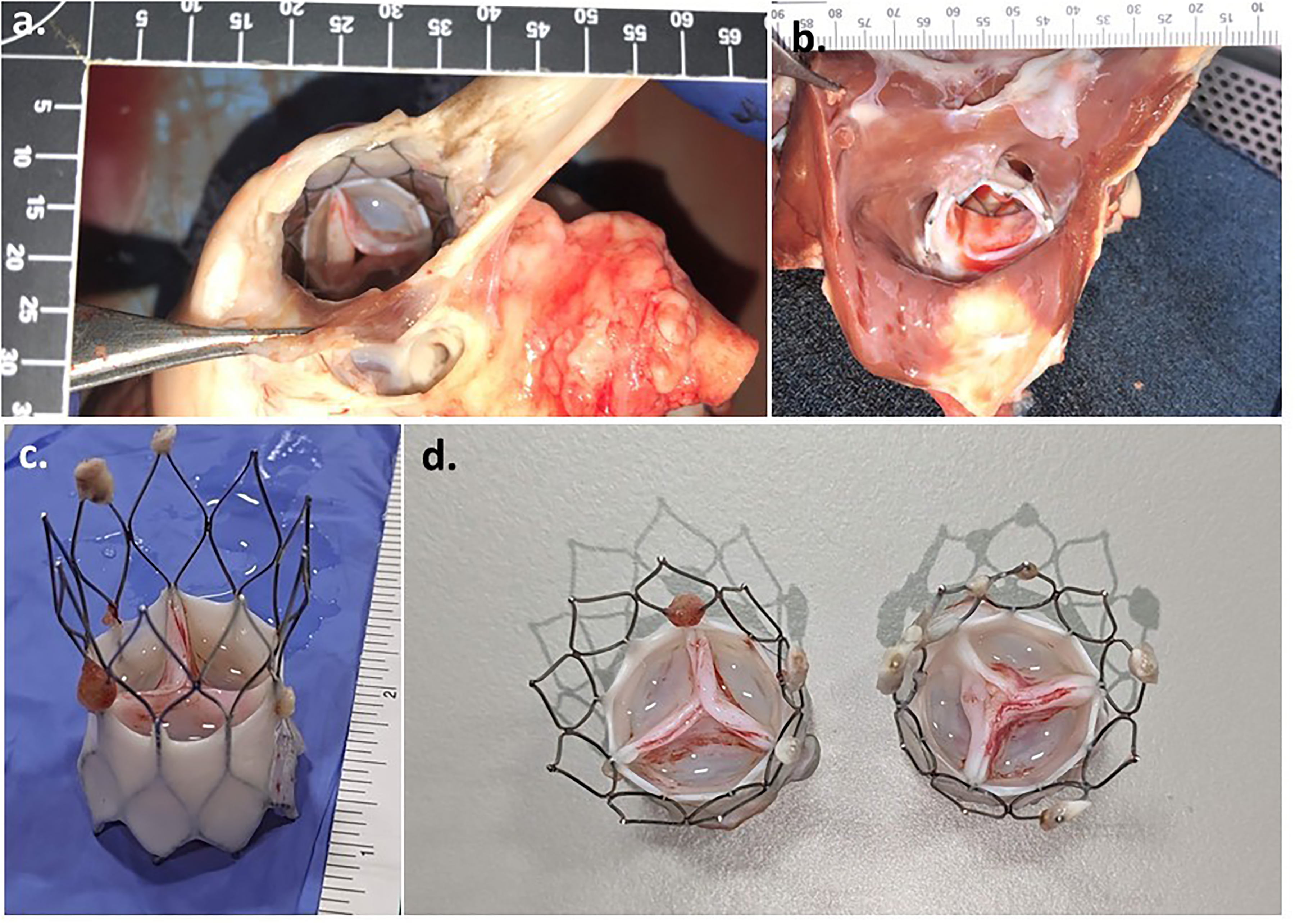
Representative Images of 18mo explant, (a) valve view from pulmonary artery side, (b) valve view from the ventricular side, (c) explant valve side-view, and (d) explant valves end-on view.
The comparison between implanted tissue and explanted valves after 6 (Figure 5a), 12, (Figure 5b) and 18 (Figure 5 c, d) months showed a comparable tissue thickness with no deterioration. A thrombogenic nodule was visible on a single leaflet at 6mo and 18mo explants.
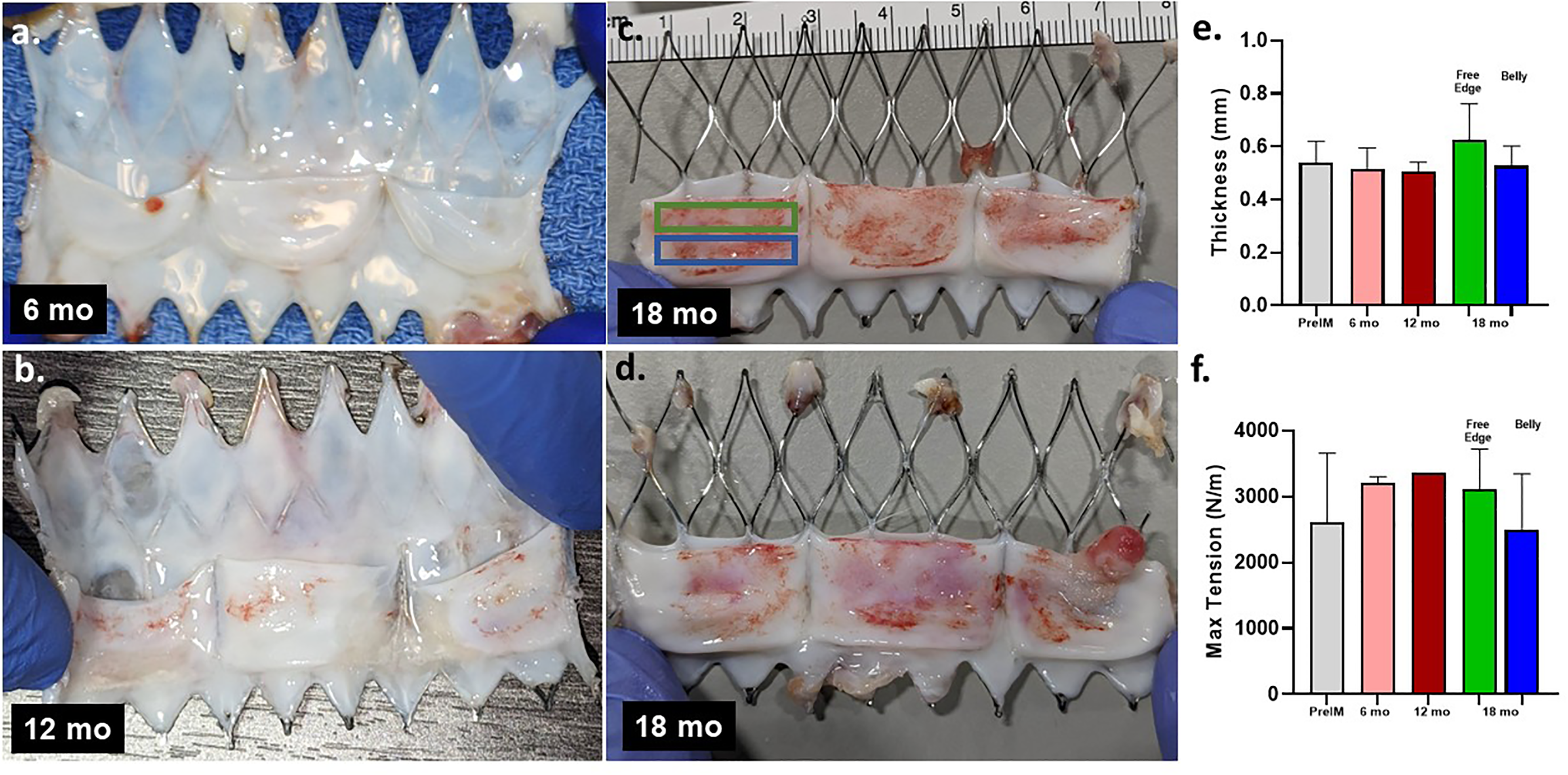
Image of bioengineered valve frame cut open to show flattened leaflet (a) 6 months, (b) 12 months, (c) 18 months #1, (d) 18 months #2, (e) thickness and (f) tensile strength of leaflet before implant and after 6-, 12- and 18-month explant. The thickness measurements in belly and free edge measured were averaged for explanted valve at 6 and 12 months, and shown separately for 18 months (the tissue location with matching color marked on 18month explant in (c)).
Explant Tissue Mechanical Properties
Bioengineered tissue explanted valve leaflets strips for tensile testing were cut from commissure to commissure such that one side was the free edge of the leaflet. A secondary strip was cut below the first strip, capturing the middle belly region of the leaflet. The thickness and maximum tension showed no change over implant material in 18-month implants (Figure 5e, f). As seen in histology, the tissue is sparsely populated with cells, indicating original ECM is durable to withstand 75 million cycles (100bpm average heart rate for sheep) without any tissue deterioration, including on the free edge. Even with some paravalvular leakage, the echocardiogram confirmed end-diastolic pressure gradient indicating leaflets cycled with cyclic pressure load for the entire implant duration.
Calcification Quantitation
Following the explant and gross examination (Figure 6a, b), each valve was imaged using Faxitron® at APS (Figure 6c, f) and dissected for further mechanical and histological analysis with a portion of each leaflet saved for calcium analysis. NAMSA Labs performed mass spectrometry quantification analysis on the pre-implant and explanted samples. The calcium content was measured and reported on a per dry-weight basis. Pre-implant tissue had a value of 0.181 mg/mg dry-weight. Two explanted valves at 18 months had average values of 0.141 mg/mg dry-weight and 0.158 mg/mg dry-weight (Figure 6g) indicating no significant increase in calcium content. The background of the instrument was 0.1 mg/mg dry-weight.

(a, b) End-on images of explanted valves at 18 months, (c-f) Faxitron® images of 18-month explanted valves showing no evidence of leaflet calcification, (g) quantified calcium content of pre-implant and 18-month explanted valves, (h) micro-CT of 12 month explant, (i, j) micro-CT of 18-month explants.
Histopathology Analysis
Portions of the valve leaflets explanted at 6 months (n = 1),12 months (n = 1) and 18 months (n = 2) were sent for histology analysis at CVPath Institute (Figure 6a-g) and imaged using micro-CT (Figure 6h-j). Overall, the leaflet thickness of the bioengineered tissue explants at 6 (Suppl Figure 1), 12 (Figure 7), and 18 (Figure 8, Suppl Figure 2) months were thin and uniform throughout the leaflets. Mild calcification was seen predominantly near the base of leaflets with micro-CT (Figure 6) and Von Kossa stain (Suppl Fig. 3). The valve explanted at 12 months showed the most significant calcification in the root of the leaflet.
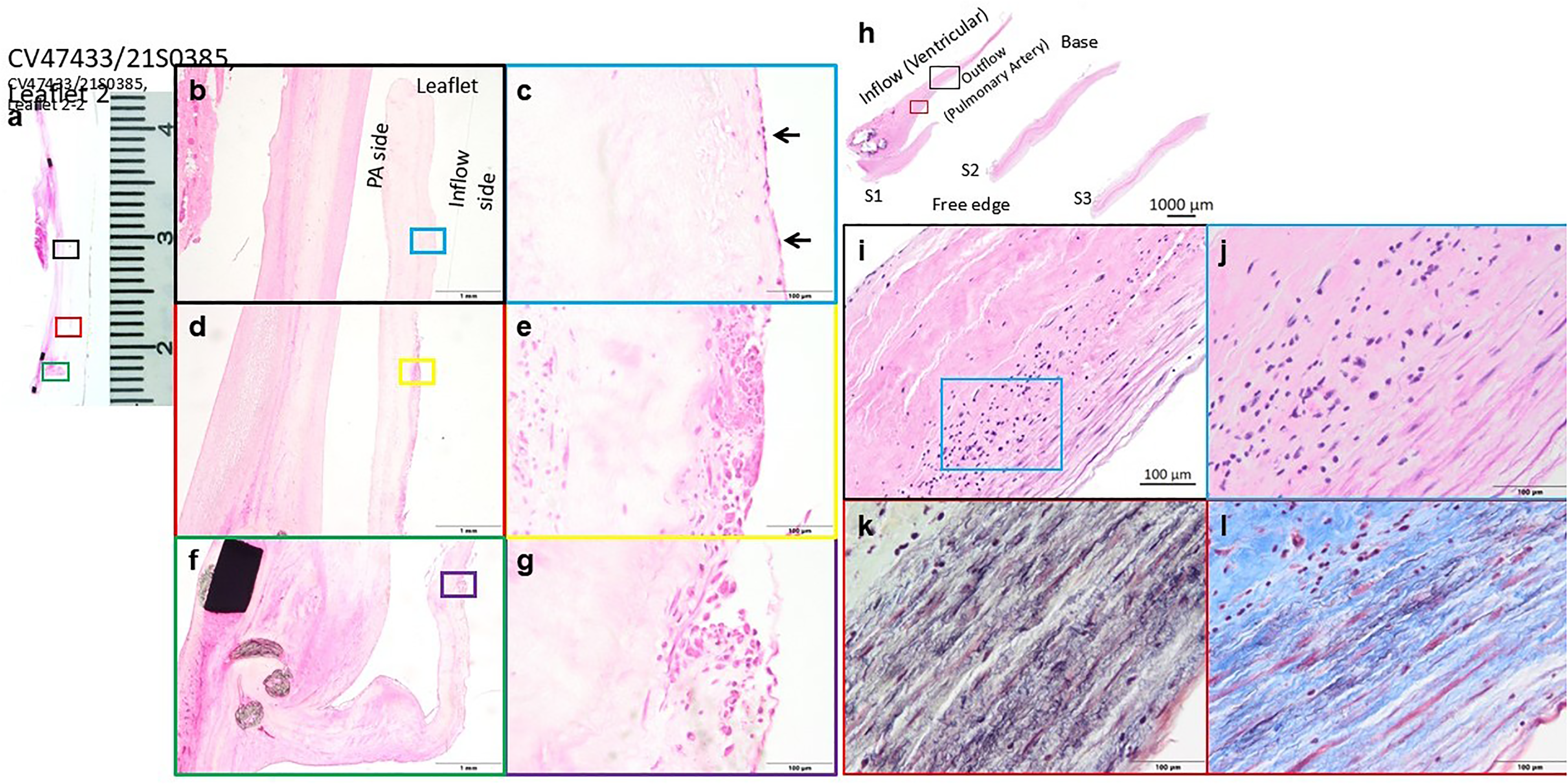
(a) Histopathology H&E images of 1 year explant, (b-g) explant shows mild immune response with leaflet remodeling including endothelium on lumen surface, (h-j) interstitial cells, and (k-l) evidence of elastin.

18-month explant H&E staining with (a) side-view of leaflet and frame, showing uniform thickness, with recellularization near. (b-e) Base of leaflet by interstitial cells and sparse immune cells. Colored boxes show magnified sections in adjacent panel.
After 18 months, evidence of regeneration in terms of endothelial coverage was observed in significant portions of the conduit with lack of endothelialization present only in areas of localized inflammation or thrombus-attached areas (Figure 8). Analysis showed interstitial cell migration from the root into leaflets and full cellularization by interstitial cells in the tissue within the frame. Additionally, there was some evidence of elastin deposition near the root of the leaflet (Figure 7).
One 6-month and one 18-month explant leaflet showed a nodule on the free surface of the leaflet with organized thrombus on the free edge of the inflow side (Suppl Fig. 3). Scattered inflammatory cells were observed mainly on the surface of the leaflets. Some areas demonstrated aggregation of inflammatory cells on the surface of the leaflets.
To summarize, histological findings demonstrate bioengineered tissue maintain thin leaflets over the course of the implant without pannus growth or an overt immune response. Endothelial and interstitial cells were present in all explants.
Discussion
While all four heart valves (aortic, mitral, pulmonary, and tricuspid) are impacted by aging or congenital defects, aortic valve replacements represent the majority of heart valve disease interventions globally. Pulmonary valve replacements, while not as numerous as aortic valve replacements, are primarily used as an intervention to address congenital defects. Over the last decade, substantial progress has been made in the treatment of heart valve disease, with the majority of advancements being made on delivery systems or frame design with very little focus on leaflet material. New bioprosthetic valves with a propriety treatment reduced pericardium calcification and deterioration in a juvenile sheep study 19 and demonstrated good performance in a 5-year preclinical study. 20 However, even with the improved material, limitations were evident in the early pulmonary valve implant data 6 signifying the need for an innovative material for the leaflets.
Bioengineered tissue derived from human cells has been shown to be both durable and uniform.11,13,21–23 Decellularized human tissue further reduces the potential for an immunological response that can lead to deterioration and calcification. The bioengineered tissue in the valve leaflets demonstrated better in vitro hydrodynamic performance than porcine pericardium control. In practice, this allows for better performance in vivo (smaller pressure drop and lower flow resistance) without oversizing the implanted valve. This improved outcome is likely due to the compliance of the bioengineered tissue being similar to the properties of the native leaflet tissue including anisotropic attributes. In contrast, the fixed pericardium was measured to be isotropic unlike the native valve leaflets.24,25
In this study, a total of five valves were implanted. To leverage previous experience in long-term performance in the pulmonary position, 11 and ease of implant, the current study was conducted on the right heart circulation of a juvenile sheep model. The pulmonary valve in the juvenile sheep model also provides an ideal environment to evaluate calcification, especially with the 18-month implant duration. All bioengineered tissue valves showed normal leaflet function with no remarkable changes over the course of the study.
To our knowledge, no bioprosthetic tissue valves or engineered tissue valves have been assessed for such a long duration in juvenile sheep. While ISO 5840 standards recommend 5-month in vivo implants, in our pre-submission discussion with the FDA, it was recommended for the heart valves with remodeling capabilities to be evaluated for up to 18 months. Hence, as more bioengineered tissue and biodegradable polymers are developed, these long-term studies would be warranted to ensure safety prior to clinical translation.
Benninnk et al evaluated a novel valve design based on bioabsorbable polymer in the pulmonary position out to 12 months. 26 The study showed significant thickening of leaflets along with cellularization and remodeling. The study did not report the impact of thickened leaflets on hemodynamic parameters such as pressure drop. In this study, no such thickening was observed with serial hemodynamic measurements showing no change in pressure gradient or leaflet function over the course of the study. For fixed bioprosthetic tissue valves, Flameng et al reported an 8-month study in juvenile model. 19 The bioprosthetic valves were implanted in the mitral position and after explant showed average calcification ranging from 1.9 to 6.8 mg/mg. In comparison, the two leaflets evaluated at 18 months in this study measured calcium content of 0.141 mg/mg and 0.158 mg/mg.
Over the course of implant, even with some anatomical growth (weight gain), there was no change in systolic pressure gradient thus no change in EOA. Paravalvular regurgitation was evident in two valves where the stent migrated within the first 30 days after which no further migration was observed. The migration was likely due to limitations of frame design not being flared enough to catch the healthy native pulmonary artery. There was no central regurgitation at any time point, indicating good leaflet coaptation with no evidence of changes for the duration of the study.
Two explanted bioengineered valves at 18 months showed excellent leaflet morphology with no evidence of tears, thickening, or other abnormal growth. After imaging, leaflets were evaluated for tensile mechanical properties, calcium content, and histology which confirm leaflet durability, remodeling with new cells, and mild calcification. In histology, it was noteworthy to observe new elastin deposition and endothelium along the leaflet surface. Evidence of elastin potentially indicates healthy normal regeneration rather than fibrotic pannus tissue formation, as generally seen with fixed pericardium leaflets.
Limitations
Some limitations of the current study include the use of transapical delivery instead of transfemoral delivery, which would be required for clinical translation. The primary goal of this study was to evaluate whether bioengineered tissue can be crimped into an 18F delivery system, deployed, and assessed for in vivo performance over 18 months. As such, a clinical delivery system was not developed and will likely require additional acute in vivo studies to ensure that the delivery system can successfully deploy the valve at the desired location. Additional work is also required to ensure the animal model mimics a stenosed pulmonary artery as the TPV migrated in the healthy PA upon implant.
Furthermore, the study was conducted under non-GLP (Good Laboratory Practice) conditions, and additional GLP studies would likely be required to comply with FDA regulations for clinical trials. The fixed pericardium used in the study was produced in-house to reduce costs, rather than using commercially available TPVs. However, GLP studies may require the use of currently approved TPVs as comparators to support clinical trials.
All limitations can be addressed in the IND/IDE (Investigational New Drug/Investigational Device Exemption) enabling GLP studies in the next phase.
Conclusion
Here, we demonstrated the long-term in vivo performance of bioengineered tissue valves in the juvenile model. All valves maintained normal leaflet motion and function throughout the duration with histology showing mild calcification and mild thrombus. The excellent leaflet function with bioengineered tissue in a highly calcific animal shows significant promise as a candidate for future valve leaflet development. Additionally, there were no changes in mechanical properties after 75 million cycles (18 months at 100bpm) which is, to our knowledge, the first study to demonstrate robustness using bioengineered tissue.
Supplemental Material
Supplemental Material
sj-docx-1-hvs-10.1177_30494826251325474 - Supplemental material for 18-Month Implant of a Transcatheter Pulmonary Valve Using Bioengineered Tissue Leaflets in Juvenile Sheep
Supplemental material, sj-docx-1-hvs-10.1177_30494826251325474 for 18-Month Implant of a Transcatheter Pulmonary Valve Using Bioengineered Tissue Leaflets in Juvenile Sheep by Mariah McMahon, Abrielle Prunty, Jacob Benkofske, James Berry, Rika Kawakami, Rick Murphy, Renu Virmani, Gurumurthy Hiremath and Zeeshan Syedain in Journal of the Heart Valve Society
Footnotes
Abbreviations
Acknowledgement
Authors would like to acknowledge Eric Solein and the animal care staff for providing support. The authors would also like to acknowledge the staff of CVPath Institute for processing and imaging of the explanted valves.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Regenerative Medicine Minnesota, (grant number RMM TR014).
Declaration of Conflicting Interests
Mariah McMahon, Abrielle Prunty, Jacob Benkofske, Richard Murphy, and Zeeshan Syedain have stocks options in Vascudyne Inc. All other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
