Abstract
Objective
Transcatheter aortic valve replacement (TAVR) with self-expanding valves (SEVs) may have different outcomes with supra-annular valves (SAVs) or intra-annular valves (IAVs) in patients with a small aortic annulus (SAA). We aimed to evaluate different SEVs, namely SAVs (CoreValve/Evolut R/PRO/PRO+/FX) versus IAVs (Portico/Navitor).
Methods
Systematic review and meta-analysis of studies (published until September 2025) comparing SEV-SAV versus SEV-IAV in patients with SAA. Meta-analyses were conducted to assess early outcomes and mortality in the follow-up.
Results
Five observational studies met our eligibility criteria, including 3303 patients (SAV: 2295; IAV: 1008). We did not find statistically significant differences for 30-day mortality, stroke, acute kidney injury, coronary occlusion, major bleeding, and paravalvular leak; however, we found lower rates of permanent pacemaker implantation in the SAV group (RR 0.75; 95%CI 0.60-0.94; P = .013). Despite slightly larger effective orifice area (mean difference +0.05 cm2/m2; 95%CI 0.01-0.10; P = .038), higher indexed effective orifice area (mean difference +0.07 cm2/m2; 95%CI 0.01-0.12; P = .012) and slightly lower mean gradients (−0.91 mm Hg; 95%CI −1.77 to −0.05; P = .038) with SAVs, we did not find statistically significant differences in terms of residual mean gradients ≥20 mm Hg (RR 1.65; 95%CI 0.73-3.74; P = .232); however, SAV showed a lower risk of prosthesis-patient mismatch (RR 0.64; 95%CI 0.42-0.98; P = .042). We observed a statistically significant difference in survival at 5 years in favor of the SEV-SAV group (HR 0.770; 95%CI 0.600-0.980; P = .034).
Conclusions
IAVs and SAVs may have a different performance in terms of hemodynamic structural and nonstructural dysfunction in patients with SAA, which should be confirmed by randomized studies.
This is a visual representation of the abstract.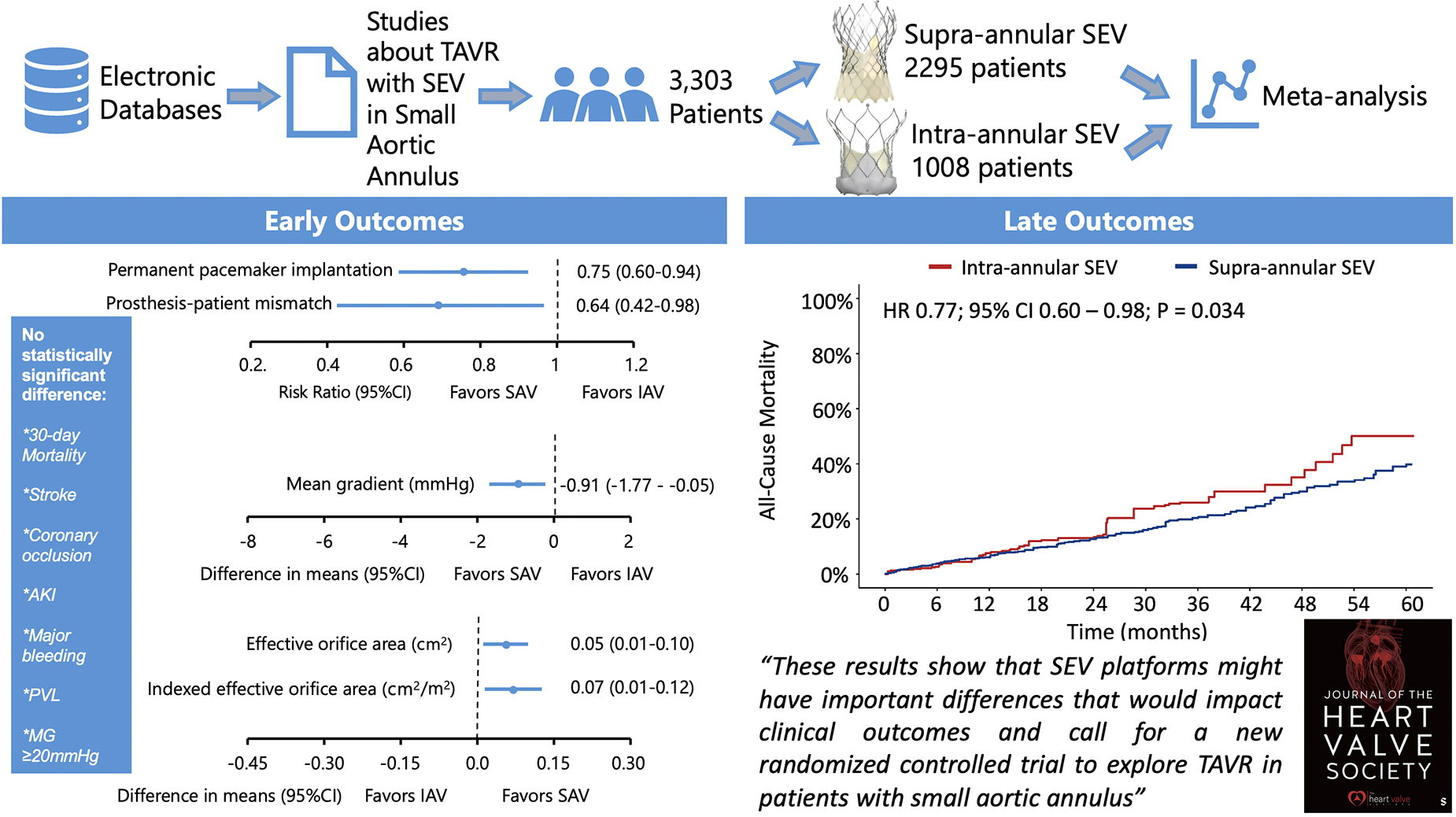
Keywords
Key Points
This is the first pooled meta-analysis of early and late outcomes of transcatheter aortic valve replacement with 2 different self-expanding platforms (supra-annular vs intra-annular) in patients with small aortic annulus.
We did not find statistically significant differences for 30-day mortality, stroke, acute kidney injury, coronary occlusion, major bleeding, and paravalvular leak, but we found lower rates of permanent pacemaker implantation in the group of patients who received supra-annular valves.
Despite slightly larger effective orifice area, higher indexed effective orifice area, and slightly lower mean gradients with supra-annular valves, we did not find statistically significant differences in terms of residual mean gradients ≥20 mm Hg.
Supra-annular valves showed a lower risk of prosthesis-patient mismatch.
We observed a statistically significant difference in survival at 5 years favoring the supra-annular valves.
Introduction
Transcatheter aortic valve replacement (TAVR) has become the most groundbreaking technology in the management of severe aortic stenosis (AS) over the last two decades and came to be the gold-standard treatment for patients at high, intermediate, and even low risk for surgical aortic valve replacement (SAVR). Although the CoreValve/Evolut (Medtronic) and SAPIEN (Edwards Lifesciences) TAVR platforms are the most used commercial devices with their performance constantly under scrutiny,1–4 new valves such as Portico/Navitor (Abbott Vascular) arose as a new option on the market. 5
The presence of a small aortic annulus (SAA) poses a considerable challenge in the management of patients with severe AS, especially in elderly women, in whom it is a very common finding and associated with increased risk of prosthesis–patient mismatch (PPM), 6 which in turn is associated with an increased risk of overall mortality proportionally to its severity, suboptimal valve hemodynamics, and less left ventricular mass regression after both TAVR 7 and SAVR.8,9
The hemodynamic performance of transcatheter heart valves (THV) has been considered a reliable predictor for long-term clinical outcomes.7,10 Within the SAA subgroup, evidence from observational registries indicates better hemodynamic performance for TAVR with self-expanding valves (SEV) outperforming balloon-expandable valves (BEV).3,11–13 Noteworthy, these studies3,11–13 compare SEVs with a supra-annular design against BEVs which have an intra-annular design. Therefore, behind the SEV-versus-BEV comparison lies a comparison between supra-annular valves (SAV) with intra-annular valves (IAV). This is important because IAVs are associated with lower effective orifice area (EOA) and, thus, tend to present higher post-TAVR gradients and higher rates of PPM. 6 In this scenario, a question remains underexplored: are there significant differences in the performance of SAVs and IAVs if both platforms are SEVs?
In this context, we aimed to compare the hemodynamic and clinical performance of 2 SEV platforms in contemporary TAVR practice: the Medtronic SAV (CoreValve, Evolut R, Evolut PRO/PRO+ and Evolut FX) versus the Abbott Vascular IAV (Portico/Navitor) in patients with SAA.
Methods
Eligibility Criteria, Databases, and Search Strategy
This study followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) reporting guideline.
14
Using the PICOS strategy (Population, Interventions, Comparison, Outcome, and Study design), studies were included if the following criteria were fulfilled:
the population comprised patients who underwent TAVR for severe AS and had SAA; there was an intervention group undergoing TAVR with a SEV-SAV; there was a second intervention group undergoing SEV-IAV; outcomes studied included any of the following outcomes: 30-day mortality, survival and/or mortality in the follow-up (with Kaplan-Meier curves), bleeding, acute kidney injury (AKI), permanent pacemaker implantation (PPI), post-procedural mean gradients, effective orifice area (EOA), indexed EOA, PPM, paravalvular leak (PVL); the study design was retrospective/prospective, randomized/non-randomized, mono/multicentric, with matched/unmatched populations.
The following sources were searched for articles meeting our inclusion criteria and published by September, 2025: PubMed/MEDLINE, EMBASE, SciELO, LILACS, CENTRAL/CCTR (Cochrane Controlled Trials Register), Google Scholar, and the reference lists of relevant articles. We searched for the following terms: (“self-expanding” OR “self-expandable” OR “Navitor” OR “Portico OR “CoreValve” OR “Evolut” OR “Evolut R”OR “Evolut PRO” OR “Evolut PRO+” OR “Evolut FX”) AND (“transcatheter aortic valve replacement” OR “TAVR” OR “transcatheter aortic valve implantation” OR “TAVI”) AND (“small aortic annulus” OR “small aortic annuli” OR “small annulus”). The following steps were taken for study selection: (1) identification of titles of records through database search; (2) removal of duplicates; (3) screening and selection of abstracts; (4) assessment for eligibility through full-text articles; and (5) final inclusion in study. Studies were selected by 2 independent reviewers. When there was disagreement, a third reviewer made the decision to include or exclude the study. Ethical approval was not applicable for this study, as it consisted of a systematic review and meta-analysis. There were no language restrictions.
Assessment of Risk of Bias
The Risk of Bias in Non-Randomized Studies of Interventions tool (ROBINS-I) was systematically used to assess included studies for risk of bias. 15 The studies and their characteristics were classified into low, moderate and serious risk of bias. Two independent reviewers assessed risk for bias. When there was a disagreement, a third reviewer checked the data and made the final decision.
Statistical Analysis
The early outcomes were investigated using traditional meta-analysis. Risk ratios (RR) with 95% confidence intervals (CI) and P-values were combined across studies using a random-effects model. 16 Forest plots were created to represent the individual study and combined ORs with 95% CI. Chi-squared and I2 tests were performed for assessment of statistical heterogeneity. 17 To assess publication bias (Supplemental materials), funnel plots were generated and Begg and Mazumdar's test 18 and Egger's test were performed. 19
For the late outcomes (mortality in the follow-up), the analysis was performed using the “curve approach,” 20 which reconstructs individual patient data (IPD) based on the published Kaplan-Meier graphs from the included studies. In a first stage, Kaplan-Meier plots were digitized to raw data coordinates (time and survival probability). In a subsequent step, IPD was reconstructed from the raw data coordinates obtained from the digitized Kaplan-Meier graphs and the respective numbers at risk at given timepoints using the R package “IPDfromKM” (version 0.1.10). 21 The reconstructed IPD from each study was merged to create the final study dataset. Kaplan-Meier analyses and Cox proportional hazard (PH) models were fitted using the reconstructed IPD using the R packages “survival” (version 3.2-13), “survminer” (version 0.4.9) and “coxph” (version 4.0.2). HRs with 95% confidence intervals (CIs) were calculated from reconstructed IPD using a Cox frailty model. Study groups (SAV-SEV vs IAV-SEV) were included in the model as a fixed effect. Between-study heterogeneity was assessed by the inclusion of a γ frailty term, where individual studies modelled as a random effect using random intercepts. A likelihood ratio test was used to test the significance of this γ frailty term. The proportionality of the hazards of each Cox model was assessed with the Grambsch-Therneau test and diagnostic plots based on Schoenfeld residuals. 22 Our protocol stated that flexible parametric survival models with B-splines and landmark analysis would be performed in case the proportional hazards assumption was violated as apparent either from these tests or from visual inspection of the Kaplan-Meier curves.23–25 This approach allowed us to apply conventional Cox regression models to each of these regions separately, and to estimate the overall HR with 95% CI with each of these regions while respecting the proportionality assumption.
To ensure that our Kaplan-Meier-derived dataset was accurate enough to reproduce the original Kaplan-Meier curves, the reconstructed Kaplan-Meier curves were individually reviewed for similarity with the original versions. We observed that all reconstructed curves remained within a 99% confidence interval when compared with the original curves published. These were also evaluated by using the root mean square error (RMSE) and the mean and max absolute error. A RMSE ≤0.05, mean absolute error ≤0.02, and max absolute error ≤0.05 indicate that the extracted data points are sufficiently well-captured for subsequent analyses, which was achieved in our analyses.
All analyses were completed with R Statistical Software (version 4.1.1, Foundation for Statistical Computing, Vienna, Austria).
Results
Study Selection and Characteristics
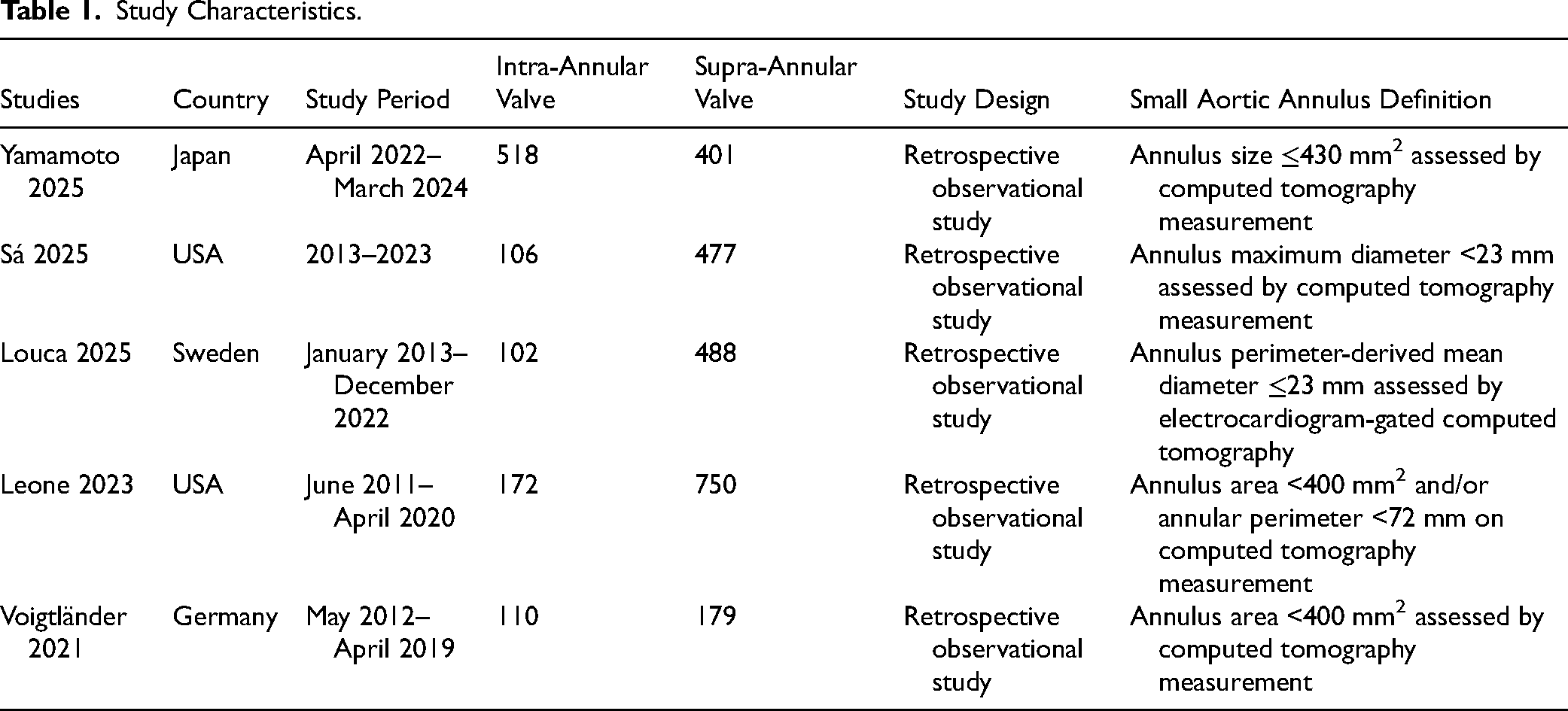
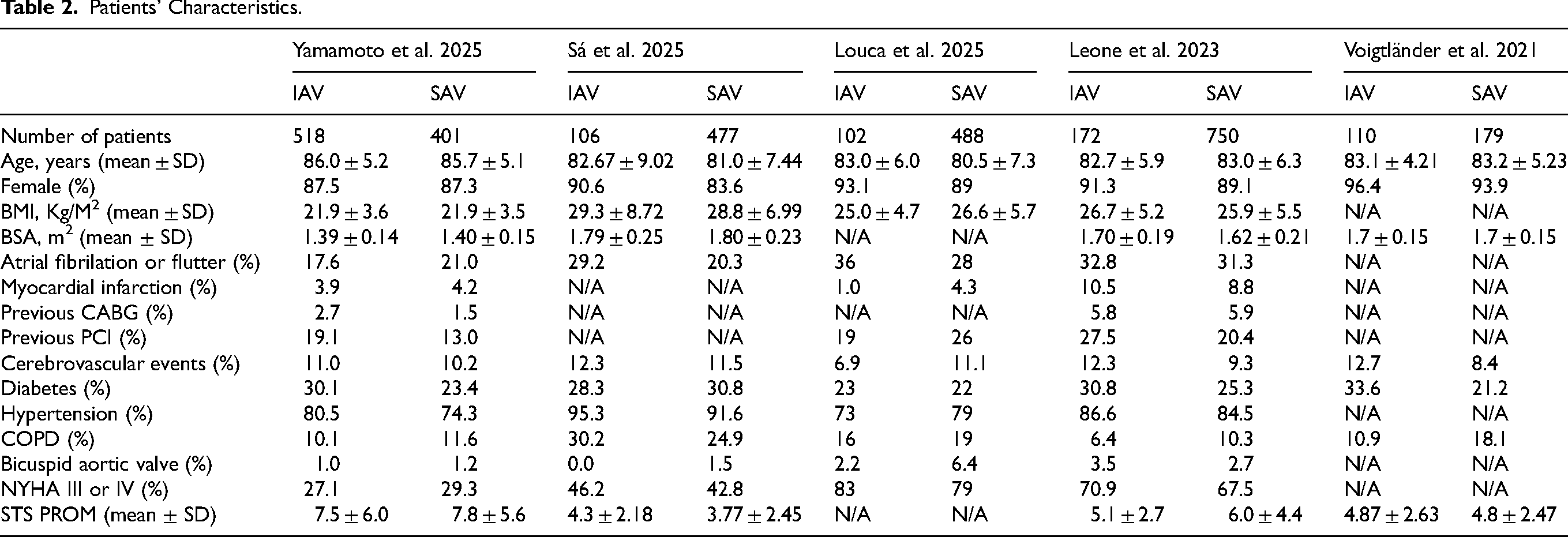
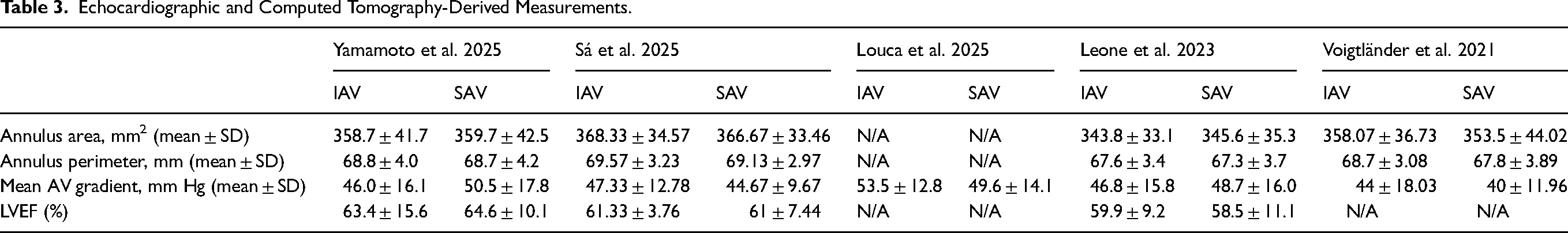
After excluding duplicates and noneligible studies, 5 studies13,26–29 met our eligibility criteria (Figure 1) and included a total of 3303 patients (SEV-SAV: 2295; SEV-IAV: 1008). All the studies were nonrandomized, observational, and retrospective (Table 1). Patients’ characteristics are shown in Table 2. Mean age was over 80 years in all groups and there was a high prevalence of female patients ranging from 83.6% to 96.4% in the studies. Sizes of the aortic annulus, aortic valve gradients and left ventricle ejection fraction are shown in Table 3. The mean aortic annulus area ranged from 343.8 mm2 to 368.3 mm2 and the mean aortic annulus perimeter ranged from 67.3 mm to 69.5 mm in the studies. Figure 2 shows the qualitative assessment of the studies with the ROBINS-I tool. In our qualitative analysis, we observed a moderate-to-high risk of bias.
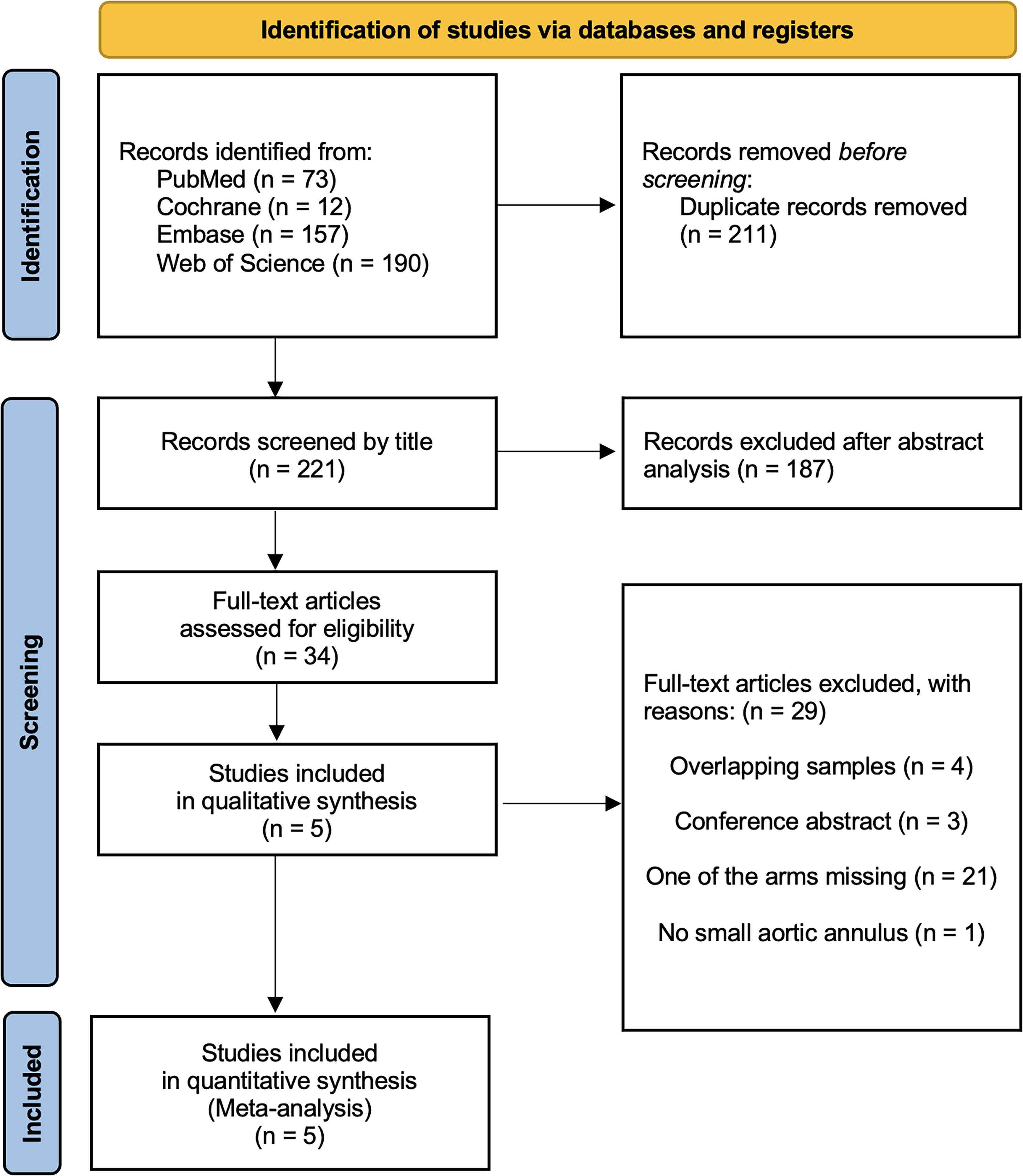
Flow diagram of studies included in data search.
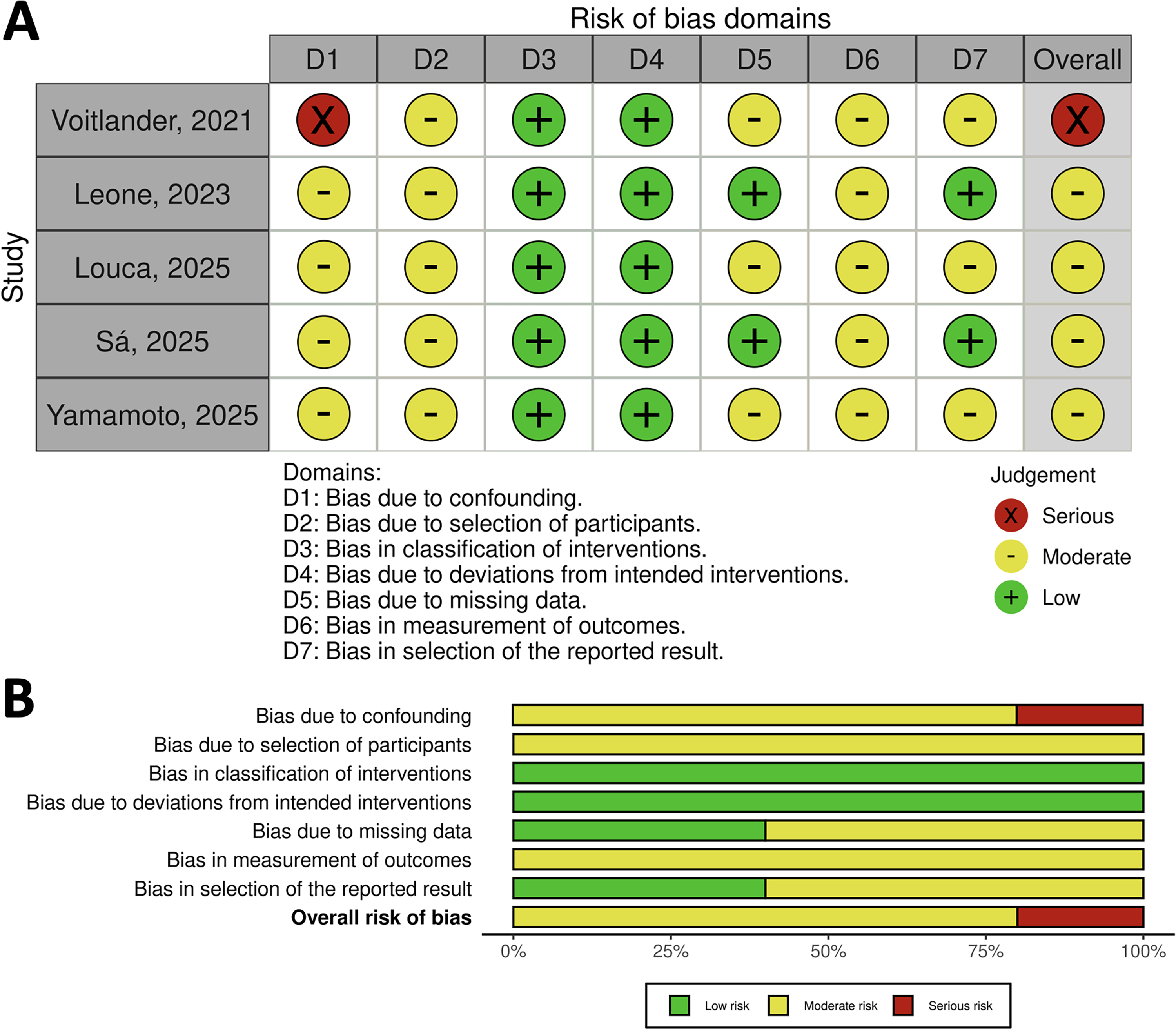
Risk of bias summary—ROBINS-I tool with traffic lights and summary plot.
Study Characteristics.
Patients’ Characteristics.
Echocardiographic and Computed Tomography-Derived Measurements.
Early Outcomes
Figure 3 shows early clinical outcomes of TAVR with SEV (SAV vs IAV) in patients with small aortic annulus. After TAVR, we did not find statistically significant differences for the following outcomes: 30-day mortality, stroke, AKI, coronary occlusion and major bleeding; however, we found lower rates of PPI in the SAV group (RR 0.75; 95%CI 0.60-0.94; P = .013).
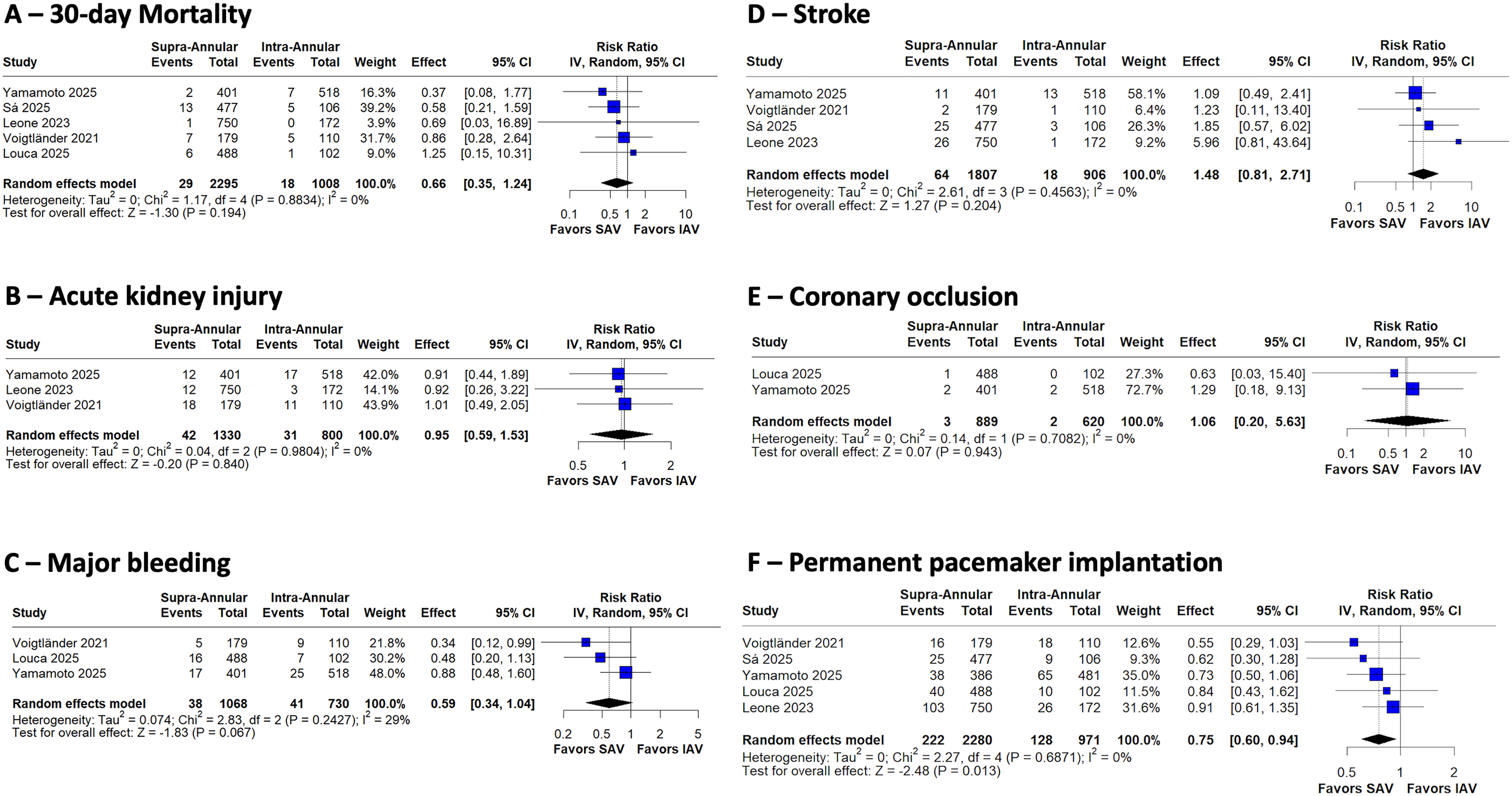
Forest plots of early clinical outcomes.
Figure 4 shows early echocardiographic outcomes of TAVR with SEV (SAV vs IAV) in patients with small aortic annulus. Despite the slightly larger effective orifice area (mean difference +0.05 cm2/m2; 95%CI 0.01-0.10; P = .038), higher indexed effective orifice area (mean difference +0.07 cm2/m2; 95%CI 0.01-0.12; P = .012) and slightly lower mean gradients (−0.91 mm Hg; 95%CI −1.77 to −0.05; P = .038) with SAVs, we did not find statistically significant differences between the groups in terms of residual mean gradients ≥20 mm Hg (RR 1.65; 95%CI 0.73-3.74; P = .232); however, SAV showed a lower risk of PPM (RR 0.64; 95%CI 0.42-0.98; P = .042). We did not find statistically significant differences for PVL.
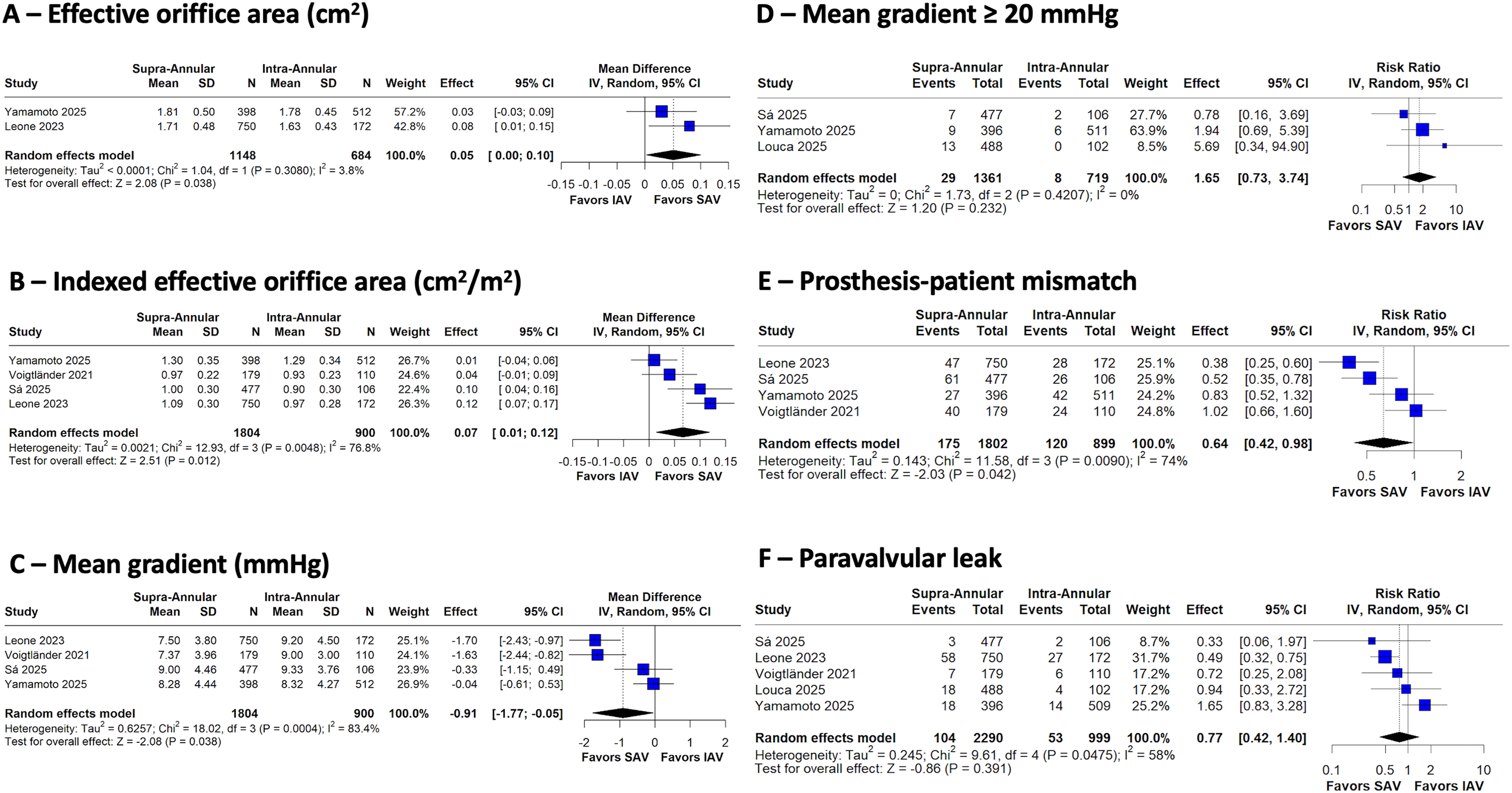
Forest plots of early echocardiographic outcomes.
Late Outcomes
We observed a statistically significance in survival at 5 years in favor of the SEV-SAV group (HR 0.770; 95%CI 0.600-0.980; P = .034) (Figure 5).
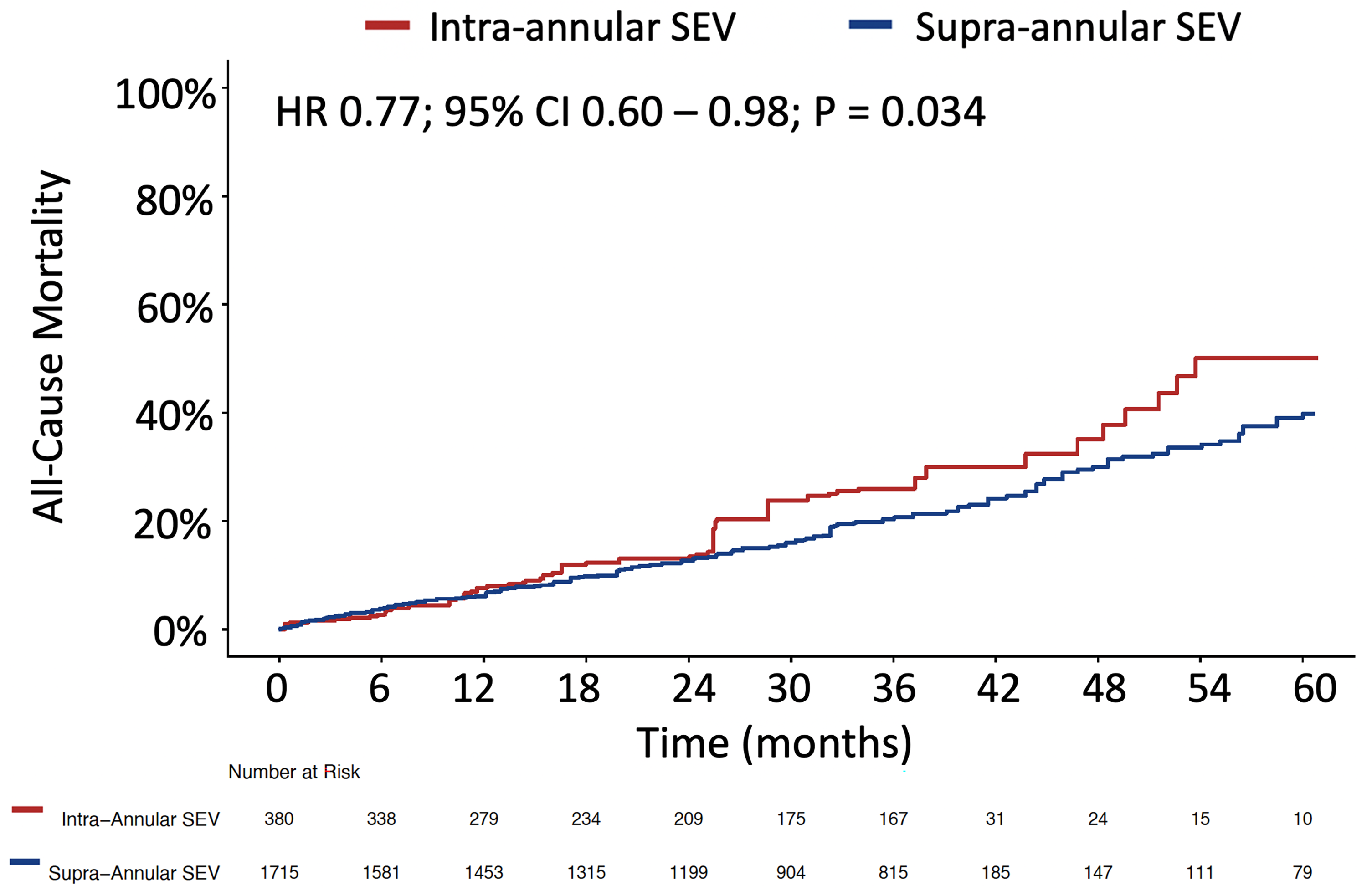
Pooled Kaplan-Meier curve for all-cause mortality in the follow-up.
Discussion
Summary of Evidence
To the best of our knowledge, this is the first pooled meta-analysis of early and late outcomes of TAVR with 2 different SEV platforms (SAV vs IAV) in patients with SAA. The main findings are as follows (Figure 6—Graphical Abstract):
TAVR performed with SAV and IAV was found to have no statistically significant differences in 30-day mortality, stroke, AKI, coronary occlusion, and major bleeding. Despite slightly larger EOA, higher indexed EOA and lower mean gradients in the SAV group, we did not find a statistically significant difference in the rates of residual mean pressure gradient was ≥20mm Hg, thus, no statistically significant difference in terms of hemodynamic structural valve dysfunction (HSVD) as defined by the Valve Academic Research Consortium-3 (VARC-3) definitions.
30
Although there was no statistically significant difference in the rates of PVL between the groups, SAV was associated with lower rates of PPM, thus, a statistically significant difference in terms of nonstructural valve dysfunction (NSVD) as per VARC-3 definitions. SAV was associated with lower rates of PPI in comparison with IAV. At 5 years, TAVR with SEV-SAV was associated with lower rates of mortality.
Comments
Comparative follow-up data, including TAVR platforms in the scenario of SAA, is scarcely represented in randomized trials. The sole trial comparing 2 TAVR platforms in the setting of SAA was the recently published SMART trial, 31 which randomly assigned a total of 716 patients with symptomatic severe AS and an AV annulus area of 430 mm2 or less (mean age 80 years; 87% women; mean STS-PROM score 3.3%) to receive either an Evolut PRO/PRO+/FX (SEV-SAV) or SAPIEN 3/3 Ultra (BEV-IAV). This trial showed that patients with severe AS and SAA who underwent TAVR with a SEV-SAV were noninferior to a BEV-IAV with respect to clinical outcomes and were superior regarding bioprosthetic-valve dysfunction and hemodynamic performance through 12 months. It is unclear if this is related only to the supra-annular design of these THVs or if this is also related to the continued expansion of the nitinol frame in SEV-SAVs. If we consider the latter, the fact the Portico/Navitor THV is mounted on a self-expanding nitinol frame might help with hemodynamics despite its intra-annular design, and, in our study, we aimed to investigate the comparison between CoreValve/Evolut iterations versus Portico/Navitor iterations in SAA.
On the one hand, despite EOA and indexed EOA slightly lower than that of a SEV-SAV and mean gradients slightly higher than that of a SEV-SAV, this difference did not translate into higher rates of HSVD (residual mean pressure gradient was ≥20 mm Hg). Therefore, one might consider the Portico/Navitor platform as an SEV with “intra-annular design and supra-annular hemodynamics.” On the other hand, we should bear in mind that we observed higher rates of PPM in the IAV-SEV group, which has been associated with higher rates of mortality over time7,32 and may have contributed to the higher 5-year mortality observed in the IAV-SEV group in our study.
Another important observation in our study was the higher risk of PPI with IAV-SEV in comparison with SAV-SEV. This difference might be explained by the initial acquisition of experience (learning curve) with the SAV-SEV platform which happened earlier in the TAVR era and later with IAV-SEV. The consistent adoption of cusp-overlap view for optimal implantation depth of both TAVR-SEV platforms would surely reduce the rates of PPI in both arms.33,34 Furthermore, pre-TAVR measurement of the membranous septum length may be helpful in the optimal implantation of any SEV device and, consequently, reduce the PPI rates after TAVR with SEVs. 35 Be that as it may, we must strive to decrease the rates of PPI after TAVR with any platform because PPI is associated with a higher risk of all-cause mortality and rehospitalization for heart failure.36,37 Indeed, the higher rates of PPI in the IAV-SEV may have compounded with the higher rates of PPM and contributed to a higher risk of 5-year mortality.
Study Limitations
The findings of this study may be under the effect of unmeasured confounders due to the pooling of studies of observational nature. Another key limitation of this study is the uncertainty of how patients were directed to one therapy or the other in each study. THVs may have been chosen based on the Structural Heart Team's preference. When it comes to SEVs for SAA, the members of the team could select an SAV or IAV based on the preference of the operators (interventional cardiologist and cardiac surgeon) in each individual case, and this can lead to biases since the selection of the THV was not randomized.
In addition, the definition of SAA used for inclusion in each study was not the same (see Table 1).
Moreover, instead of using the same concept of PPM (such as projected/predicted PPM), some studies explored PPM measured by Doppler echocardiography. While the former can mitigate the effect of low-flow states on the measurements, 38 it has the downside of including the extrapolation of the average values of a group (from other studies) to an individual patient (from each study).32,38
Finally, our findings apply only to the THVs studied and should not be extrapolated to other TAVR platforms.
Conclusions
This study provides important insights into the comparative clinical and hemodynamic performance of iterations of the Corevalve/Evolut and Portico/Navitor TAVR platforms in patients with SAA. The results should be seen as a motive for a more comprehensive understanding of TAVR in patients with SAA, guiding future research and patient management strategies. There is a clear need for further investigation through randomized trials.
Supplemental Material
sj-docx-1-hvs-10.1177_30494826261424814 - Supplemental material for Supra-Annular Versus Intra-Annular Self-Expanding Transcatheter Heart Valves in Patients With Small Aortic Annulus: A Meta-Analysis
Supplemental material, sj-docx-1-hvs-10.1177_30494826261424814 for Supra-Annular Versus Intra-Annular Self-Expanding Transcatheter Heart Valves in Patients With Small Aortic Annulus: A Meta-Analysis by Michel Pompeu Sá, Gabriel Neves, Leo Consoli, Asad Iqbal, Leonardo Dexheimer, David Abraham Batista da Hora, Thiago Camarotti, Xander Jacquemyn, Federico Napoli, Antonio Polanco, Nicolas A. Brozzi and Jose L. Navia in Journal of the Heart Valve Society
Footnotes
Acknowledgments
None.
Funding
Te authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
