Abstract
Hemodynamics and biomechanics are essential for the accurate and early diagnosis of patients with aortic valve diseases and those undergoing transcatheter aortic valve replacement (TAVR). Despite astonishing advancements in medical imaging, current imaging modalities cannot quantify hemodynamics and biomechanics adequately. This review explores the impact of personalized simulations and artificial intelligence (AI)-based methods in clinical practice and how they could transform cardiology and influencing cardiovascular care. It outlines future directions while addressing current challenges and opportunities related to translating these technologies into clinical settings. The chapter highlights the contributions of various computational methods in quantifying hemodynamics and biomechanics specifically for patients with aortic valve diseases undergoing TAVR. It identifies barriers to adopting these methods in clinical practice and emphasizes the critical need for interdisciplinary collaboration to bridge this gap. By leveraging computational insights to inform clinical decisions, there is potential to revolutionize patient outcomes through personalized diagnostics and treatment strategies. Integrating advanced computational models into clinical practice requires concerted efforts from both computational engineers and clinical cardiologists. Together, they can develop user-friendly, clinically relevant computational tools that elevate the standard of cardiovascular care.
This is a visual representation of the abstract.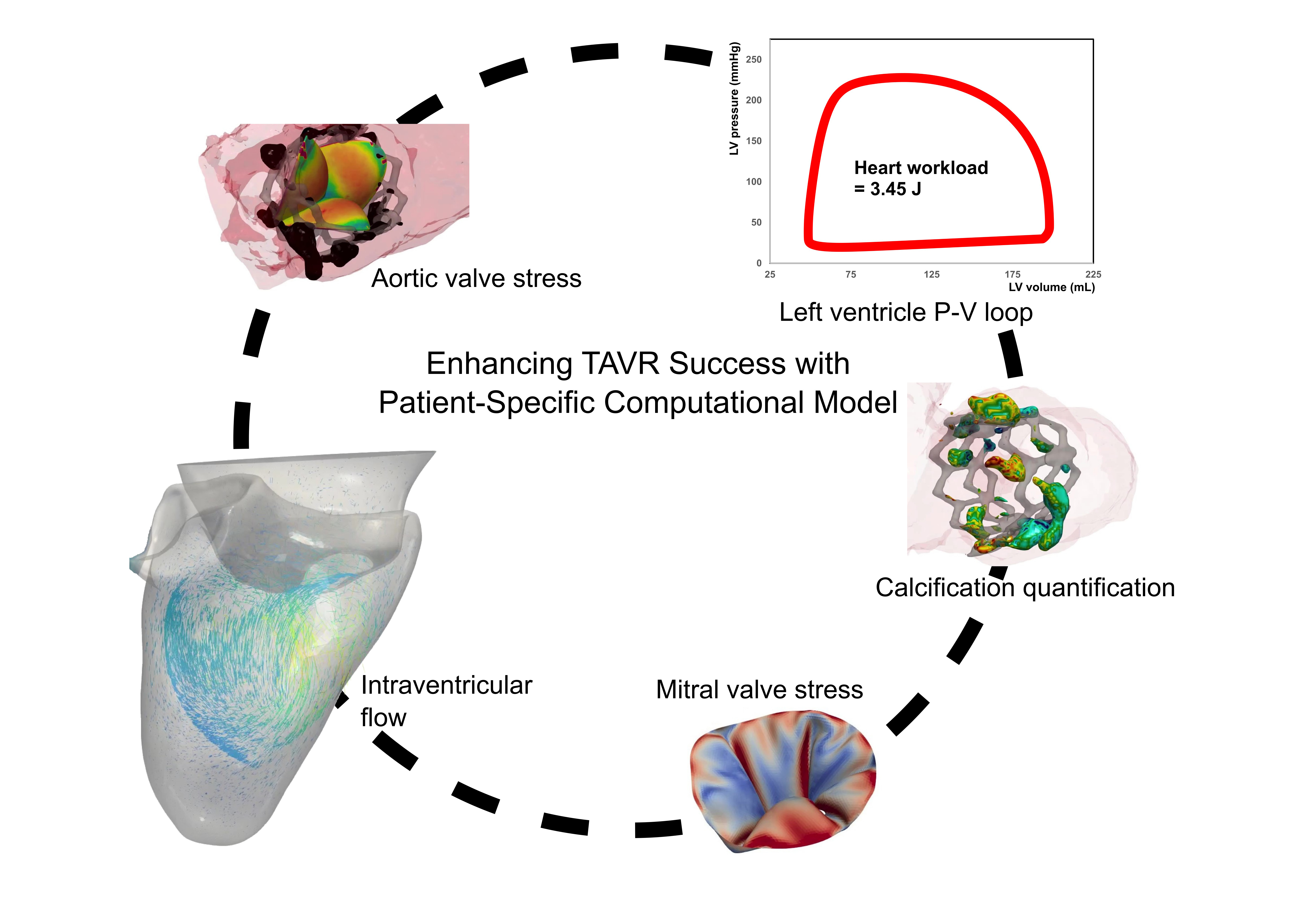
Keywords
Key points
Hemodynamics and biomechanics play a pivotal role in the precise and early diagnosis of aortic valve diseases, particularly for patients undergoing transcatheter aortic valve replacement (TAVR).
While medical imaging has seen remarkable advancements, current technologies remain inadequate in accurately quantifying hemodynamics and biomechanics, highlighting the pressing need for innovative approaches.
Personalized simulations hold the potential to transform cardiology by enabling precise diagnostics and customized treatment strategies, ultimately improving patient outcomes.
Bridging the gap between computational insights and clinical practice requires close collaboration between engineers and clinicians. Such partnerships are vital to developing intuitive, clinically relevant tools that seamlessly integrate into patient care.
Introduction
Aortic valve stenosis (AS) is one of the most acute and chronic cardiovascular disease (CD) where the aortic valve becomes damaged and fails to function properly. 1 This disease can quickly impair the heart's pumping action, potentially leading to heart failure. Historically, the only option for high-risk AS patients was surgical valve replacement, a procedure associated with a high mortality rate. However, transcatheter aortic valve replacement (TAVR) has emerged as a minimally invasive alternative. TAVR is increasingly being used not only for high-risk patients but also for those with moderate valvular disease or younger individuals, offering a broader spectrum of patients a safer treatment option.2,3
Over the past decade, computational breakthroughs have significantly advanced both technology and medicine. 4 With an increasing focus on personalized healthcare and the significant impact of CDs, interest in cardiovascular computational modeling has grown. These in-silico models have allowed researchers to achieve a more thorough understanding of these diseases and are starting to aid clinicians in creating personalized and optimal treatment plans.5,6 Latest advancements in medical imaging and computational modeling have significantly propelled this field forward. As technology progresses, researchers, and physicians are working towards the ultimate goal of developing a personalized whole-heart computational model. This comprehensive model would combine personalized genomics, cellular behavior, tissue structure, heart mechanics, and fluid dynamics.7–9 Although significant progress has been made, each of these fields still faces numerous challenges. Overcoming these obstacles is essential to fully unlock the potential of personalized cardiovascular care.
Hemodynamics and biomechanics are crucial for the accurate and early diagnosis of AS patients and those undergoing TAVR.10–13 However, current diagnostic methods are lacking because fluid dynamics methods, which could power new diagnostic tools, are not yet fully developed. Additionally, most interventions aim to restore healthy conditions, making the ability to predict hemodynamics and biomechanics following specific interventions highly impactful for saving lives. Unfortunately, predictive tools for these patients are scarce.6,14,15 Such tools are essential for evaluating the effectiveness of interventions and systematically testing clinical solutions.
While TAVR has yielded positive outcomes and significantly reduced mortality rates, the procedure is not without risks. While many patients experience substantial improvement following TAVR, some see their condition worsen, or their pre-existing valvular disease may evolve into other forms of cardiac diseases.11,16–25 In addition, following TAVR, abnormal hemodynamics and biomechanics are still common, for example, disturbed wall shear stress, high workload, disturbed vortical structure, and turbulence are present.10–12,22,23,26,27 The complexity of fluid dynamics and biomechanics in AS and TAVR limits the effectiveness of TAVR.26,28,29 Moreover, prediction of the outcome of TVR in each individual, requires development of novel advanced tools to predict changes in hemodynamics and biomechanics in response to TAVR. To effectively and safely utilize TAVR in patients, it is crucial to address several key questions: Who stands to benefit the most from this costly procedure? What will be the impacts on heart function post-TAVR? What is the optimal timing for the intervention? How can we predict which patients are likely to have better or worse outcomes? A tool that can provide answers to these questions, tailored to each patient's specific conditions, is urgently needed.
This article explores how the personalization of computational modeling to quantify hemodynamics and biomechanics is revolutionizing patient-specific care for patients with AS and TAVR. This article delves into the synergies between these technologies in developing innovative diagnostic and predictive tools. The article also addresses future directions, current challenges, and opportunities for integrating these advancements into clinical practice.
Synergizing Patient-Specific Simulations and Imaging: A New Era in Personalized Cardiology in Patients with Aortic Valve Diseases Undergoing TAVR
Recent research has highlighted that valvular disease is a multifaceted condition shaped by the interactions between the valve, ventricle, and vascular system.1,10,11,21,30–35 For patients with aortic valve disease undergoing TAVR, it is essential to consider the interplay among these three components: valves, ventricle, and the vascular system in personalized simulations of hemodynamics and biomechanics.1,10,11,21,30–35 In these patients, AS often exists alongside other complex valvular, ventricular, and vascular diseases (referred to as C3VD in this article). In C3VD, multiple pathologies in the valve, ventricle, and vascular system interact mechanically, with the physical phenomena of each pathology amplifying the effects of the others on the cardiovascular system.16–19,36–38
Comprehensive Fluid Dynamics in Intraventricular and Coronary Systems
Current diagnostic methods: Intraventricular flow and vortex analysis
With every cardiac beat, the left ventricle (LV) and its valves experience a complex flow, which becomes even more intricate in C3VD. The heart's chirality results in this flow being strongly three dimensional38,39 and unsteady. 39 During the normal heart filling phase, blood entering the LV through the mitral valve forms a vortex, minimizing energy dissipation, preventing blood stagnation, and optimizing pumping efficiency. 39 In the presence of C3VD and TAVR, however, the LV's vortical structure is altered. 11 : the vortex dynamics become less synchronized with heart contractions, and non-native vortices may emerge and interact, reducing efficiency.
Accurately quantifying the complex flow in C3VD is crucial for early and precise diagnosis.40,41 Phase-contrast magnetic resonance imaging (MRI) can provide a 3-D velocity field but suffers from poor temporal resolution,42,43 is time-consuming, and is unsuitable for patients with implanted devices. Recently, Doppler echocardiography (DE) has been able to provide 2-D velocity measurements.44–46 However, since 2-D velocity does not represent 3-D velocity, some 3-D DE techniques have been developed, but these techniques suffer from low temporal resolution and cannot precisely quantify the velocity field. Recent advances in Doppler velocity measurements include: (1) Echo-PIV, an adaptation of particle image velocimetry (PIV), which computes flow velocity by tracking speckles, often enhanced with contrast agents like microbubbles.44,45 While promising, Echo-PIV may underestimate high velocities depending on the acquisition frame rate, impacting diagnosis. 47 Additionally, the contrast agent must homogeneously and consistently fill the field to avoid saturated and dark areas. 48 (2) Color-Doppler vector flow mapping (VFM), which estimates the velocity field without contrast agents via color Doppler. 46 VFM enhances the 1-D velocity measurement of color Doppler by estimating the velocity component perpendicular to the beam in the LV using the continuity equation and LV-wall speckle tracking. 46
Current diagnostic methods: Coronary flow analysis
Quantifying the complex flow in C3VD is essential for early and accurate diagnosis.40,41 This includes impacts on coronary perfusion and myocardial function, making the management of concurrent coronary artery disease (CAD) post-TAVR a crucial aspect of follow-up care.27,49 Various imaging techniques are available for coronary arteries, such as angiography, computed tomography coronary angiography, MRI, echocardiography, ultrafast ultrasound, intravascular ultrasound, and optical coherence tomography. 27 However, except for MRI, all modalities cannot noninvasively quantify hemodynamics. Although MRI can quantify local hemodynamics, it is limited to patients without pacemakers, except those with MRI-compatible pacemakers. Additionally, MRI is not routinely used for coronary imaging in clinical practice due to its limited spatial resolution.
Clinical implementation of patient-specific flow dynamics simulations: Challenges and opportunities
Simulations of patient-specific flow dynamics start by reconstructing the 3-D geometry of the ventricle, valves, and vascular region from medical images. Next, the geometry is divided into smaller subvolumes (meshing), boundary conditions are applied, and the governing equations are solved (Figure 1).50,51 Patient-specific flow dynamics are incredibly valuable and crucial for the following reasons:
Enhanced understanding of hemodynamics and biomechanics: To provide detailed insights into the blood flow patterns and hemodynamic forces inside the heart and at the vicinity of heart valves to provide critical flow parameters such as vortex rings inside the ventricles,52–54 kinetic energy and energy loss during cardiac motion,
55
and/or pressure gradient, shear stress and flow patterns across mitral and aortic valve.56,57 Post-TAVR, patients might face thromboembolic events such as strokes, along with valve-related complications like thrombosis, hemolysis, and prosthesis degeneration,
58
all of which are linked to abnormal blood flow. Additionally, the heart's recovery and adaptation post-TAVR present unique challenges. This includes effects on coronary perfusion and myocardial function, emphasizing the importance of managing concurrent CAD as a critical aspect of follow-up care.
49
Personalized medicine: To predict how a patient's cardiac system will respond to the interventions. Optimization of medical devices: To design and optimize cardiac devices such as heart valves and ventricular assist devices which has significant impact on accelerating the development process and reducing the need for extensive in vitro and animal testing while also significantly de-risking emerging technologies. Preoperative planning: To aid in preoperative planning for complex interventions to minimize surgical risks and to improve procedural outcomes.
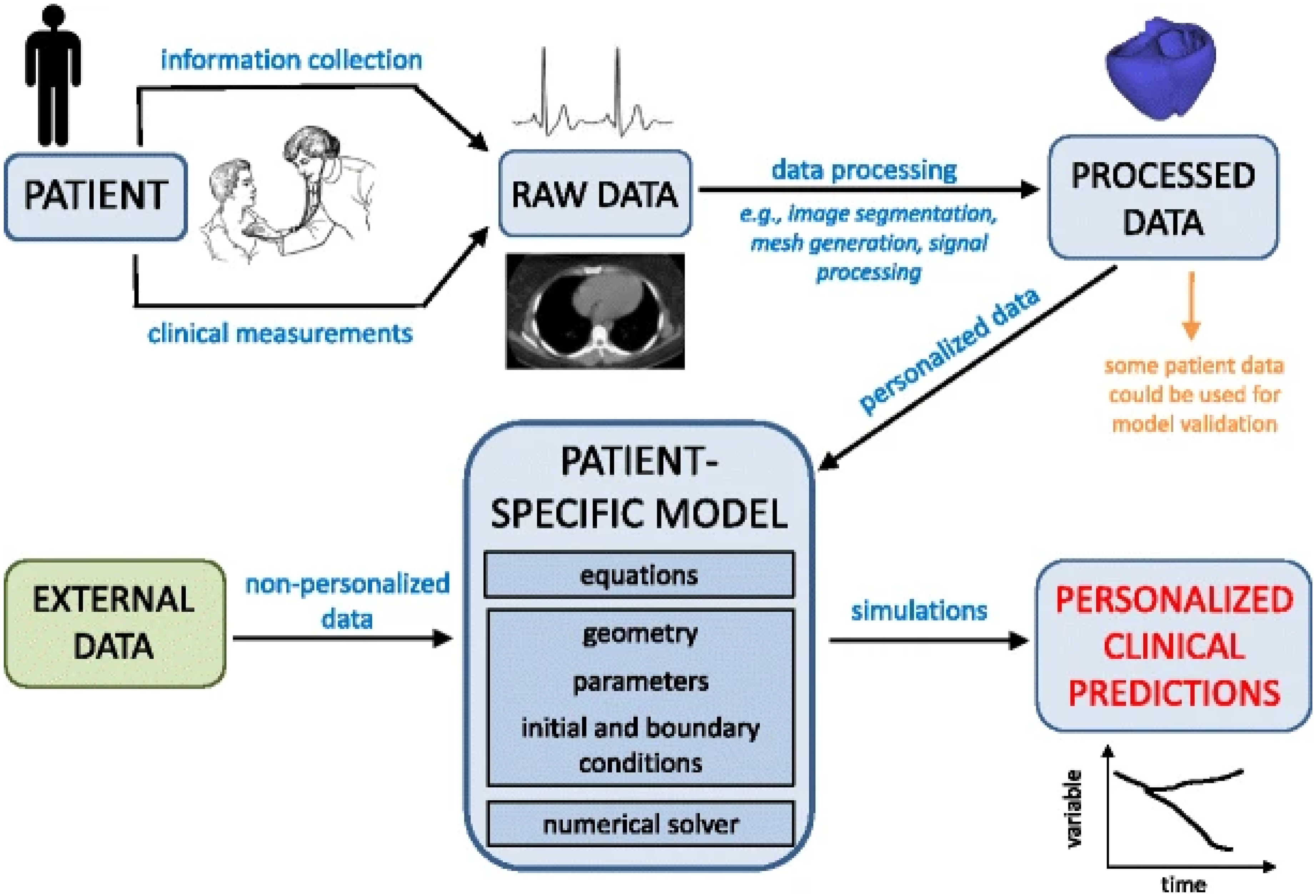
The Patient-Specific Computational Modelling Workflow (from reference 50 ).
The complexity of patient-specific flow dynamics simulations stems from the following factors:
Geometrical complexity: Accurate 3-D reconstruction of heart components is essential for reliable personalized computational fluid dynamics simulations. However, current methods, whether manual or semi-automatic, often compromise geometric accuracy.59,60 Manual methods can be time-consuming and prone to human error, while semi-automatic methods may lack the precision needed for detailed heart geometry. This compromises the fidelity of the simulations, which depend on precise anatomical details to accurately model the flow dynamics and interactions within the heart. Consequently, advancements in imaging techniques and automated reconstruction algorithms are crucial to enhance the accuracy and efficiency of these simulations, ultimately improving their reliability and clinical applicability. Mathematical formulations: The simulations rely on solving at least the Navier-stokes equations, which describe the motion of fluid substances (and sometimes involve coupling fluid and solid dynamics simulations). These equations are nonlinear partial differential equations (PDEs) and can be challenging to solve, especially for complex geometries and flow conditions.23,26,61,62 Boundary conditions: Defining appropriate boundary conditions is crucial for accurate simulations, as they significantly influence the outcomes. The challenge lies in the scarcity of precise cardiovascular flow and pressure measurements in clinical settings, making it difficult to establish accurate boundary conditions for simulations.15,22 Clinical data often lack the detailed and specific measurements needed to precisely define these conditions, which can lead to inaccuracies in the simulations. Moreover, the variability among patients further complicates the process, requiring a tailored approach to each individual case. This complexity underscores the need for advanced techniques and tools to capture the necessary data, ensuring the boundary conditions are reflective of real physiological conditions. Mesh generation: Creating a computational mesh that accurately captures the geometry and flow characteristics is a complex task. The mesh needs to be fine enough to capture critical details, yet not so fine that it becomes computationally impractical. Flow dynamics algorithms require advanced mesh generation methods for the interface and very small time steps for stability and convergence.
63
The significant deformation of cardiovascular tissues, such as heart valves and ventricles, makes this type of modeling one of the most challenging Multiphysics problems.
63
Material properties: The accuracy of models heavily relies on the precise characterization of the material properties of biological tissues, such as heart valves and ventricles, which vary significantly between individuals and are challenging to measure in vivo. For instance, the left ventricle's tissue exhibits a complex multilayered anisotropic architecture.
64
Various empirical frameworks have been developed to describe its dynamics, based on ex vivo experiments conducted on animal hearts, which differ structurally from human hearts.
65
Given the complicated nature of directly measuring cardiac tissue behavior in a living organism, many research efforts have focused on simulating blood flow by treating the tissue as a dynamic boundary, with motions derived from medical images such as MRI.
66
These approaches essentially bypass the direct impact of cardiac tissue, considering it instead as a moving wall. Alternatively, some researchers have pursued more personalized simulations of tissue by adjusting material characteristics to match displacement fields captured from patient MRI scans.
67
Beyond conventional 3-D MRI, the use of tensor diffusion MRI is crucial for a precise assessment of myocardial fibers and the anisotropic features of the left ventricle's tissue.
68
Despite these challenges, the recent work of Khodaei et al22,26,56 stands out as a significant advancement (Figure 2). They have developed a framework that fine-tunes and replicates the material properties of the left ventricle, leveraging a sophisticated Doppler-based, patient-specific algorithm.15,69 Computational resources: Computational resources: High-fidelity fluid dynamics simulations demand substantial computational resources and considerable time to accurately solve fluid dynamics equations or the coupled dynamics of fluid flow and structural deformation. This computational time can range from a few hours to a few days or even weeks, depending on the complexity of the model and the available computing power. Such demands can be restrictive in clinical settings, where quick decision making is often essential. Validation and verification: Validating computational models with clinical data poses significant challenges and often leads to inconsistencies between the model's predictions and actual clinical outcomes.6,70,71 These challenges arise primarily from the difficulty in obtaining and collecting clinical data that accurately aligns with computational requirements in terms of both time and spatial resolution.
72
For instance, data collection must be timed accurately to capture dynamic physiological changes, and the spatial resolution must be fine enough to reflect the detailed structure and function of the cardiovascular system. Ensuring this level of detail and accuracy in clinical data collection can be logistically challenging. To bridge these discrepancies, it is crucial to perform a sensitivity analysis. Sensitivity analysis evaluates how variations in the specific data and numerical parameters impact the outcomes of the computational model. By systematically assessing these effects, researchers can better understand the robustness of their models and identify areas that require further refinement.
72
Adoption of patient-specific flow dynamics in healthcare: Beyond the inherent complexities of these methodologies and their inputs, healthcare professionals generally lack familiarity with the simulation processes of patient-specific flow dynamics modeling. This unfamiliarity often results in hesitance to adopt these methods.
73
Additionally, there is an issue with effectively conveying the benefits of patient-specific flow dynamics simulations by researchers and cardiologists, which worsens the situation. Communication and collaboration between computational researchers and clinicians are frequently limited. Researchers may not fully understand the practical challenges within clinical environments, and clinicians may not be up-to-date with the latest advancements in patient-specific computational models and their applications in research. This disconnect creates a significant obstacle in translating research findings into practical and clinically applicable solutions.
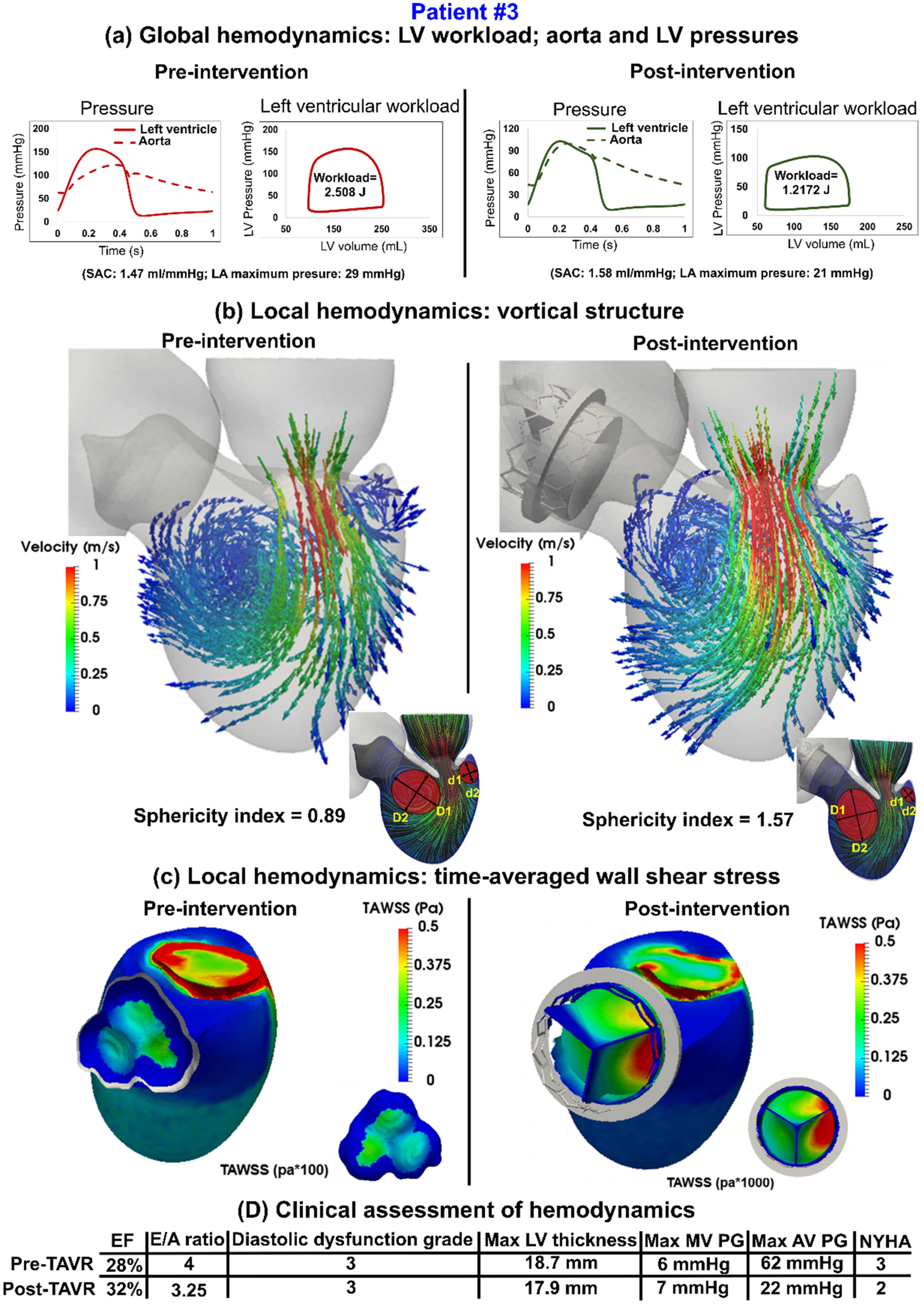
Sample Results of Intraventricular Flow and Cardiac Function Analyses in an AS Patient Pre- and Post-TAVR (from reference 56 ). Abbreviations: AS, aortic valve stenosis; TAVR, transcatheter aortic valve replacement.
Building on the foundational understanding of the necessity for comprehensive fluid dynamics modeling in coronary arteries following TAVR, recent studies have employed innovative approaches to address this gap. The research conducted by Khodaei et al stands out, utilizing a multiscale, patient-specific computational framework to assess the effects of TAVR on coronary as well as on cardiac hemodynamics.10,11,22,23,26,27,74 This study highlights the complexity of CAD in TAVR patients, demonstrating how computational simulations can be a powerful tool for the early diagnosis of long-term hemodynamic complications that are often challenging to detect clinically.23,74,75 The model revealed significant variability in coronary blood flow dynamics postintervention, illustrating the personalized nature of TAVR's impact and underscoring the potential of computational models in enhancing clinical decision making for cardiovascular interventions. These studies represent a paradigm shift in CAD research within the TAVR context, emphasizing the critical role of patient-specific computational models in understanding and mitigating complex cardiovascular interactions post-TAVR. They pave the way for more personalized and effective treatment strategies by considering the unique hemodynamic characteristics of each patient.
Left Ventricle Pressure-Volume Loop Analysis and Cardiac Hemodynamics
Aortic valvular disease leads to an increase in LV pressure, LV end-diastolic pressure, LV workload, systemic arterial stiffness, and LV afterload, all contributing to both systolic and diastolic dysfunction, 78 a significant cause of heart failure in affected patients. Although TAVR has shown positive outcomes and significantly reduced mortality rates, it fails in approximately 25% to 35% of cases, with patients either not surviving or not regaining a reasonable quality of life postprocedure. 79 The immediate and long-term hemodynamic changes impacting the aortic-LV system post-TAVR are poorly understood. While TAVR consistently reduces the transvalvular pressure gradient, it is expected to enhance LV systolic and diastolic function in the long run. Despite the benefits, invasive LV P-V loop analysis revealed impaired LV systolic and diastolic function in the early phase following TAVR.77–79 LV P-V loop analysis also revealed exacerbated heart failure despite successful TAVR procedures in many patients,21,57,80 indicating that reductions in transvalvular pressure gradient do not always correlate with improvements in LV workload. In many cases, TAVR has no effect on LV workload, and in some instances, it even increases the workload postprocedure.21,57,75
Clinical Implementation of Left ventricle P-V Loop Analysis and Cardiac Hemodynamics Algorithm: Challenges and Opportunities
In the field of circulatory mechanics, only patient-specific lumped parameter modeling has the distinct capability to assess both cardiac function and global flow dynamics using one single model.33,57,71,81,82 This dual assessment is vital for accurately predicting and diagnosing cardiovascular diseases, given the cardiovascular system's complex interconnections. The integration of patient-specific lumped parameter modeling with advanced imaging techniques enables the creation of detailed patient-specific physiological models.21,33,83–85 The flexibility of such algorithms to adapt to patient-specific data (in comparison to other complex computational methods) enhances the accuracy of these simulations, making them invaluable in clinical decision making, from diagnosing cardiovascular conditions to optimizing treatment plans.86,87
The validation of algorithms designed to quantify cardiac function and hemodynamics through rigorous clinical trials is crucial for establishing their efficacy and reliability across diverse conditions and patient populations. As the evidence base for such models continues to grow, thorough validation remains a significant barrier to their widespread adoption.6,88 Until these models are substantiated through comprehensive research and clinical trials, their integration into routine clinical practice may be limited.71,89
A notable example of successful implementation of patient-specific lumped parameter modeling in translational medicine is the first and the only Doppler-based noninvasive patient-specific diagnostic, monitoring, and predictive tool that can investigate and quantify effects of interventions, medications, and C3VD constituents on the function of the heart and circulatory system at no risk to the patient.13,15,90 This pioneering method12,27,28,69,74 offers several capabilities (Figure 3, sample results): (a) It quantifies details of physiological pulsatile flow and pressures throughout the heart and circulatory system in patients with C3VD and TAVR. (b) It tracks cardiac and vascular states using accurate time-varying models that replicate physiological responses. (c) It performs LV P-V loop analysis to quantify heart function metrics, particularly in terms of heart workload. (d) It provides a detailed breakdown of how disease constituents affect the global function of the heart (eg, contributions and impacts of each disease on the heart workload), aiding in predicting intervention effects and planning the sequence of interventions for C3VD. (e) It quantifies other heart function metrics, such as LV end-diastolic pressure, instantaneous LV pressure, and contractility. None of these metrics can be obtained noninvasively in patients, and when invasive cardiac catheterization is performed, the collected metrics are not as comprehensive as those provided by this novel method. Such information is crucial for the effective use of advanced therapies to improve clinical outcomes and guide interventions in patients. However, these capabilities are currently not accessible in clinical settings.
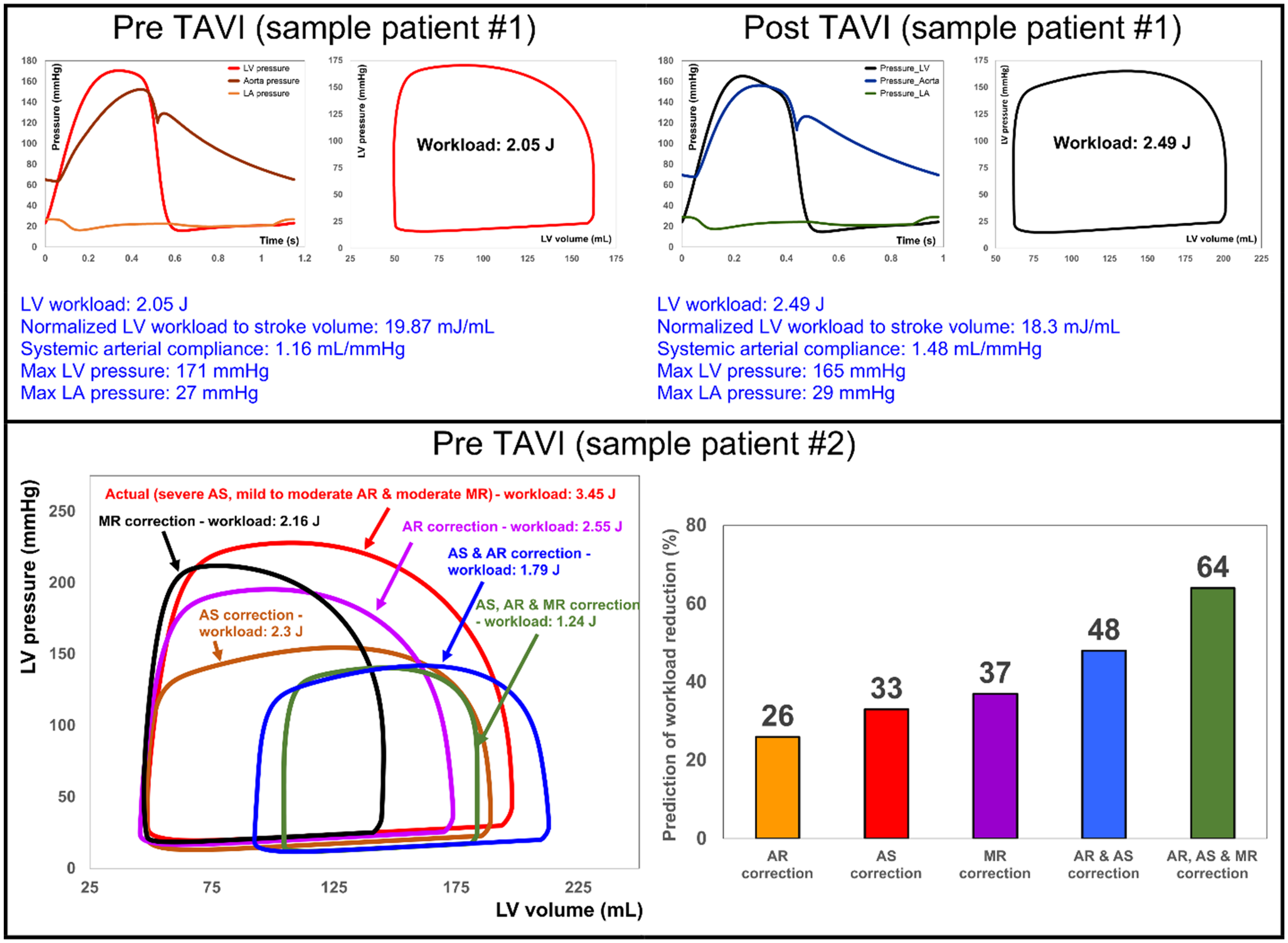
Samples Results of LV P-Vanalyses in AS Patients in Both Pre- and Post-TAVR (from reference 13 ). First row: Diagnosis and monitoring of a sample patient #1 from baseline to 90 days: This patient didn’t fully benefit from the TAVI intervention. Instead of improving the heart condition by reducing the LV workload, TAVI caused an increase in the LV workload. Second row: Example of workload breakdown analysis and prediction for the effects of interventions in a sample patient #2: Right: P-V loops of the actual diseased condition and predictions of several valve interventions. Left: Predicted percent decrease in the LV workload following valve interventions. Both mitral valve regurgitation (38% increase) and aortic valve stenosis and regurgitation (48% increase) had substantial contributions to increasing the workload. In reality, this patient only underwent TAVI. However, considering this calculation, the decision of whether to also perform a mitral intervention at the time of the aortic valve intervention should have been evaluated and considered. Abbreviations: AS, aortic stenosis; AR, aortic regurgitation; MR, mitral regurgitation; LV, left ventricle; LA, left atrium; P-V, pressure-volume; TAVI, transcatheter aortic valve implantation; TAVR, transcatheter aortic valve replacement.
Aortic Valve Dynamics and Stress Analysis
Current diagnostic methods
Understanding valve dynamics through stress analysis and motion assessments, if available, offers critical insights into the patient's cardiac deterioration and recovery. This information can profoundly impact patient care, intervention planning, and making crucial clinical decisions that carry life-threatening risks.91,92 It is important to recognize that calcification patterns often vary on leaflets, with each exhibiting a distinct level of calcification.24,25,29 Mechanical strain and stress on the aortic valve promote and progress calcification, significantly affecting treatment outcomes.12,28,93
AS often results in, or arises from, abnormal stress and strain distributions on aortic valve leaflets, both before and after interventions. 94 Additionally, mechanical stresses play a crucial role in the degeneration and eventual failure of prosthetic heart valves. 95 Developing effective treatment strategies heavily relies on a comprehensive understanding of valve dynamics. 96 Currently, there are no clinical tools available to invasively or noninvasively measure valve dynamics. Despite significant advances in medical imaging, imaging alone cannot provide the detailed valve dynamics essential for the long-term health of the heart and the durability of valve leaflets.
Clinical implementation of patient-specific valve dynamics simulations: Challenges and opportunities
The condition of the heart valves not only relies on blood flow, but the geometry and material properties of the leaflets as well as the interaction between the flow and the valve.
97
Patient-specific valve dynamics are valuable for the following reasons:
Personalized valve repair and replacement: Understanding the specific dynamics and stress profiles of a patient's aortic valve allows for more precise and personalized surgical interventions. Surgeons can plan repairs or replacements that are tailored to the patient's unique anatomy, potentially improving outcomes and reducing complications. Estimation of valve durability: Analyzing stress distributions on the valve helps predict areas of potential wear and tear. This information is crucial for predicting the longevity of both natural and prosthetic valves, enabling proactive management and timely interventions. Monitoring postintervention outcomes: Postoperative valve stress analysis helps in monitoring the success of interventions. It can reveal how well a repair or replacement is functioning and whether any adjustments are needed to maintain optimal valve performance.
The complexity of patient-specific valve dynamics simulations arises from several factors:
Mathematical formulations: It relies on solving at the dynamic equation of motion in which motion equations are solved by a nonlinear finite element method (FEM). Anisotropic tissues of aortic valve leaflets experience extremely large deformations in interaction with transvalvular blood flow in each cardiac cycle so any FEM numerical solver should accurately address these complexities.
98
Boundary conditions: Similar to what was mentioned in the “Comprehensive fluid dynamics in intraventricular and coronary systems” section (intraventricular flow dynamics and vortex analysis). Mesh generation: Similar to what was mentioned in the “Comprehensive fluid dynamics in intraventricular and coronary systems” section (intraventricular flow dynamics and vortex analysis). Material properties: The accuracy of models heavily relies on the precise characterization of material properties of heart valves. Material properties of each patient should be calibrated to mimic each patient's tissue behavior. Several experimental tests were performed on normal aortic valve tissues; however, for calcified aortic valve leaflets, there should be a calibration step for material properties of each individual patient.99,100 Furthermore, because calcification patterns on aortic leaflets differ, each leaflet's dynamic behavior should be evaluated on a leaflet-by-leaflet basis24,25,29 which is crucial for patients with valvular disease who undergo TAVR. Despite all these challenges, recent advancements have made significant strides in addressing them. Bahadormanesh et al12,28 developed highly innovative Doppler-exclusive computational frameworks that combine our Doppler-based lumped-parameter model and 3-D nonlinear finite element method to quantify aortic valves’ dynamics and material properties (Figure 4) at no risk to the patients in both pre and post-TAVR conditions (eg, transient 3-D distribution of stress and displacement, leaflet-based material properties, 3-D deformed shape of leaflets).12,28 This approach represents an example for potentially bridging the gap between computational modeling and practical clinical application, offering a pathway to overcome existing challenges by providing a more accurate representation of cardiac tissue behavior in FEM models. Validation and verification: Similar to what was mentioned in the “Comprehensive fluid dynamics in intraventricular and coronary systems” section (intraventricular flow dynamics and vortex analysis). Methodological and clinical barriers to computational model integration: High-fidelity models can be computationally expensive and time-consuming, making them impractical for real-time clinical use. Engineers need to find ways to optimize these models to make them faster and more efficient. In addition to the methodological constraints and limitations, there are several obstacles from clinical point of view. Clinicians need to interpret and utilize the results of these models, which requires an understanding of both the modeling techniques and the underlying cardiovascular physiology. While computational engineers are compelled to acquire a foundational understanding of cardiology to inform their models, clinicians, as end users, may not feel an equivalent imperative to delve into the complexities of FEM models, especially given their extensive clinical commitments.
101
Therefore, due to the limited time and prioritization of their clinical responsibilities, cardiologists often do not feel comfortable integrating computational models into their workflow, as they lack the opportunity to fully understand and engage with these complex models. In addition, engineers should develop user-friendly interfaces that allow clinicians to interact with and interpret the models without needing extensive training in computational techniques. All these require significant collaborations between engineers and clinicians.
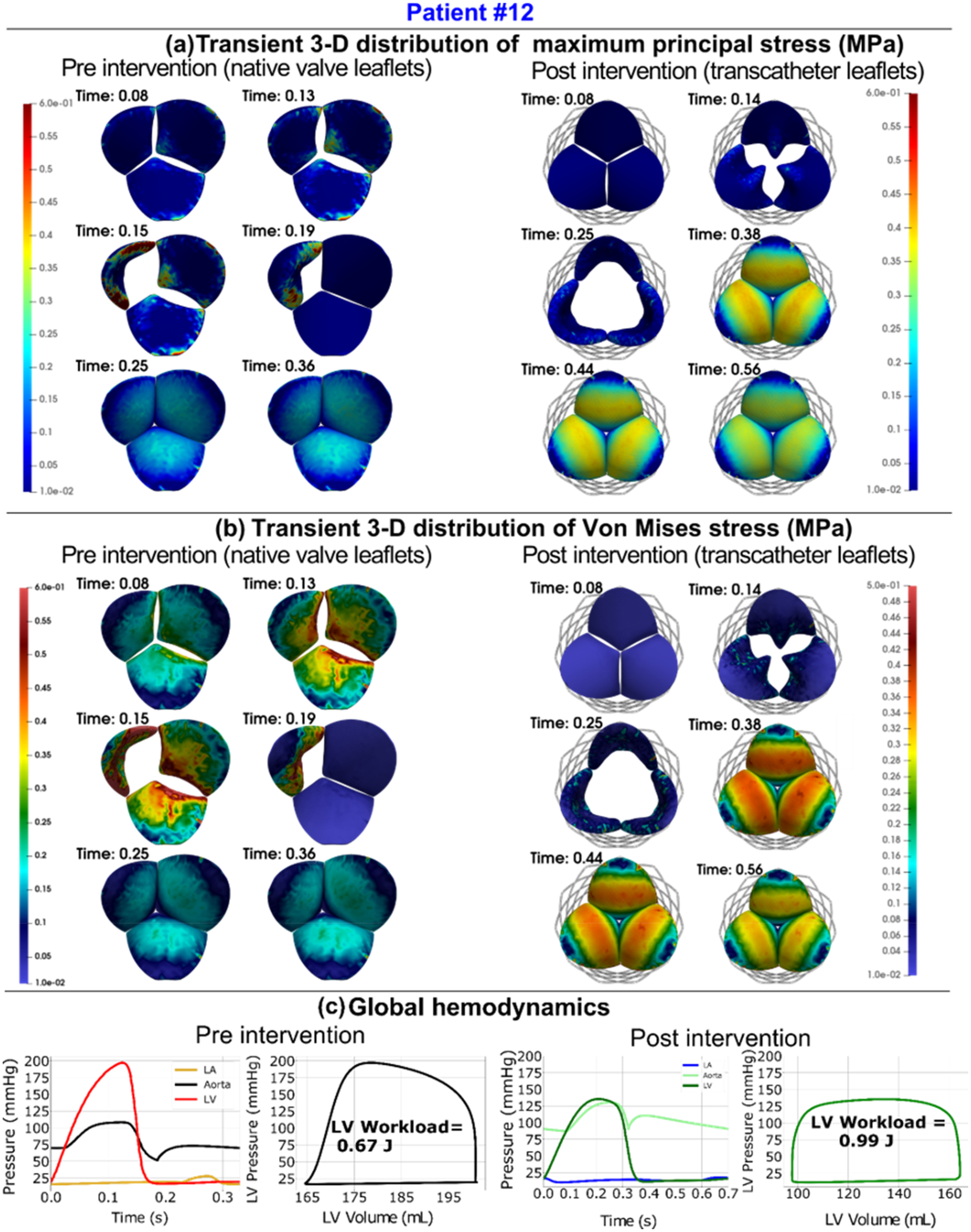
Doppler-Based Patient-Specific Simulations to Quantify Valve Dynamics and Stress Analyses in AS Patients in Both Pre- and Post-TAVR (from reference 28 ). Abbreviations: AS, aortic valve stenosis; TAVR, transcatheter aortic valve replacement.
AI-Driven Insights: Modeling Aortic Valve Diseases and TAVR
Recent advancements in artificial intelligence (AI), particularly machine learning (ML) methods, have opened new pathways for predicting CDs and their interventions.6,102
Implementing AI-Based Flow Dynamics Methods In Clinical Practice Versus Patient-Specific Algorithms: Challenges and Opportunities
Geometrical complexity: One key aspect of preprocessing computational models is the precise geometry reconstruction of the model. Traditionally, computational methods for cardiovascular applications have been labor-intensive, involving the manual extraction of blood vessel structures, heart valves, the ventricle, and other features.56,103 However, recent advancements in AI and ML technologies offer a transformative shift, enabling automatic and rapid extraction of these geometric features for computational modeling.104–107 AI techniques have utilized nearest distances in grid points and deep learning to predict flow resistance 108 and segment medical images, streamlining the process for simple and complex geometries alike. 109 ML models are transforming patient-specific biomechanical modeling by automatically segmenting and reconstructing 3-D geometry from medical images, accommodating the diverse morphologies of various cardiovascular diseases. Although these advancements are significant, fully aligning with anatomical structures and expert assessments remains a challenge. This highlights the complexity of patient-specific shapes and the continuous need to enhance ML models for broader generalization and greater precision in capturing the subtle details of cardiovascular morphology. 110
Mathematical formulations: AI and ML are enhancing the efficiency and effectiveness of solving partial differential equations (PDEs). 111 By leveraging data-driven and physics-driven models, AI methodologies enhance PDE resolution without relying on traditional empirical or model equations.112,113 Data-driven approaches use high-fidelity data from existing simulations or experiments to train neural networks, generating new solutions that replace conventional PDEs.114,115 This method enables quick and efficient numerical solutions for flow fields, increasing data accuracy by integrating computational and experimental hemodynamics data. Physics-driven models, on the other hand, formulate specific input-output relationships through ML, based on physical principles, bypassing traditional model equations entirely. 116 These AI-driven solutions have demonstrated promising results in predicting hemodynamic parameters and stress distributions in cardiovascular tissues, illustrating AI's potential to streamline computational analyses in biomechanics.112,117,118
Despite these advancements, using AI and ML to solve PDEs instead of patient-specific algorithms faces several challenges and limitations. Data-driven neural networks, for instance, need extensive high-fidelity data, which may not always be available or sufficient for intricate cardiovascular models. 119 These AI models are often validated on simplified or canonical problems, while more complex cardiovascular disease modeling still requires exploration. 120 Additionally, the creation of physics-informed neural networks (PINNs), which embed physical equations into neural frameworks, offers a novel approach to solving PDEs. However, these models encounter difficulties in ensuring generalizability across various cardiovascular conditions and accurately incorporating boundary conditions without labeled data. 111 The success of AI in cardiovascular modeling relies on overcoming these challenges, necessitating further research to develop more versatile and broadly applicable ML models that can effectively represent the complex nature of cardiovascular diseases.
Conclusions and Future Works
Closing the Research-Clinical Gap
The gap between computational cardiovascular biomechanics and clinical application is more than just an academic issue; it signifies a missed opportunity in patient care. Bridging this gap is crucial, not only for translating theoretical models into practical diagnostic and therapeutic tools but also for the transformative potential it holds in patient care. The integration of patient-specific computational models—such as computational fluid dynamics, FEM, fluid-structure interaction models, and lumped parameter algorithms—combined with the capabilities of ML and AI, into clinical practice, promises to enhance diagnostic precision, tailor treatments to individual patient needs, and greatly improve the understanding and management of cardiovascular diseases.
Enhancing Patient Care: Effects on Clinical Outcomes
Bridging the research-clinical gap has profound implications for patient care and clinical outcomes. By integrating advanced computational models into routine practice, clinicians can gain detailed insights into cardiovascular mechanics, enabling earlier diagnoses, more accurate prognoses, and personalized treatment plans. This shift not only aims to enhance the quality of patient care but also seeks to reduce the overall burden of cardiovascular diseases through improved management strategies and outcomes. The ability to directly influence patient survival rates, quality of life, and treatment efficacy highlights the critical importance of addressing this gap.
Pioneering Innovation: Advancing Collaboration Between Engineers and Cardiologists
Successfully translating research into clinical practice requires a collaborative effort between engineers and cardiologists. This call to action highlights the importance of forming multidisciplinary teams that work together to address the challenges of incorporating computational models into healthcare settings. The innovations resulting from such collaborations can lead to the development of user-friendly, clinically relevant tools that seamlessly integrate into the diagnostic and treatment frameworks of cardiovascular medicine.
Building common ground for communication, sharing knowledge, and focusing on the ultimate goal of enhancing patient care are essential steps toward fostering an environment where technological advancements and clinical needs intersect. This convergence has the potential to redefine the future of cardiovascular healthcare. By addressing these aspects, we can envision a future where the gap between cardiovascular biomechanics research and clinical application is not only narrowed but ultimately bridged, ushering in a new era of patient-centered, technology-driven cardiac care.
Footnotes
Abbreviations
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Conflicts of interest
Dr Zahra Keshavarz-Motamed serves as the Founder and Chief Executive Officer of Diagnics Inc.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
