Abstract
Background
Prosthesis-patient mismatch (PPM) has been shown to be related to worse survival and hemodynamic outcomes following valve replacement. Women with aortic stenosis (AS) have a smaller annulus than their male counterpart, making them more susceptible to PPM. The objective of this study was to compare the incidence of PPM in a female population with small aortic annulus (SAA) undergoing transcatheter aortic valve replacement (TAVR) versus surgical aortic valve replacement (SAVR) using the Carpentier Edwards Magna Ease bioprosthetic valves (19-21 mm) and the Edwards Sapien 3 and Sapien 3 Ultra transcatheter valves (20-23 mm).
Methods
This retrospective study was conducted using harmonized databases from two high-volume cardiac surgery centers. Female patients with SAA (≤23 mm) measured by echocardiography who underwent SAVR or TAVR between 2011 and 2022 were included. Patients with pure aortic regurgitation were excluded. The primary endpoint was to compare the incidence of moderate to severe PPM, both measured and predicted, in women receiving TAVR versus SAVR. Secondary endpoints included various clinical outcomes.
Results
A total of 426 women were included. 64.8% underwent SAVR (mean age: 75) and 35.2% underwent TAVR (mean age: 81). SAVR patients received 19- or 21-mm prostheses (32% and 68%, respectively). Almost all TAVR patients (94%) were assigned a 23 mm prosthesis, with the others receiving a 20 mm prosthesis. While 53% of SAVR patients were predicted to have a moderate to severe PPM (P-PPM), 39% of TAVR patients were predicted to do so (P = .005). When it came to measured PPM (M-PPM), 73% of SAVR versus 32% of TAVR patients had moderate to severe PPM (P < .001). Proportions were lower when adjusting for BMI > 30 (P-PPM - SAVR: 25%; TAVR: 19%; P = .148; M-PPM – SAVR: 64%; TAVR: 23%; P < .001).
Conclusion
In women with SAA, a larger proportion of SAVR patients had moderate to severe P-PPM or M-PPM compared to TAVR patients. Adjusting for BMI > 30 showed similar trends, although overall proportions were lower.
Keywords
Background
Aortic stenosis (AS) is the most common valvular heart disease affecting Western countries. Severe AS is an indication for treatment and its prevalence increases with age. It is estimated to be 3.4% in those aged >75 years 1 and is higher in women compared to men in those aged >80 years.2,3 Definitive treatment options in the current era are transcatheter versus surgical aortic valve replacement (TAVR vs SAVR). There is an equipoise in terms of durability, reoperation, structural valve deterioration (SVD) and survival between TAVR versus SAVR on the mid- to long-term in high- and intermediate-risk patients. However, for specific populations of patients such as those that are low-risk, younger, have a bicuspid valve, a small aortic annulus (SAA), or women, more studies are required to draw definitive conclusions. 4 There are signals in the literature that have showed disproportionate benefits of TAVR over SAVR in women in regards to long-term survival. In fact, female patients appear to have a better long-term survival post-TAVR compared to their male counterparts. 5

Incidence of P-PPM and M-PPM in females with SAA undergoing SAVR versus TAVR. SAVR: surgical aortic valve replacement; TAVR: transcatheter aortic valve replacement; PPM: prosthesis-patient mismatch; M-PPM : measured prosthesis-patient mismatch; P-PPM: predicted prosthesis-patient mismatch.
Now that we have access to multiple novel types of technologies, valve options, and surgical techniques such as the enlargement of the aortic annulus, the refinement of our understanding in these specific population groups would help shape treatment decisions based on more personalized patient characteristics.
Although current treatment guidelines do not differentiate between sexes but suggest indexing echocardiographic values, 3 studies have shown that there are clinical and pathophysiological differences in AS between women and men. Women have higher incidence of AS later in life, are often diagnosed at later ages, are more symptomatic and have fewer comorbidities than men.2,6,7 In terms of their pathophysiology, they tend to have less aortic valve calcification, more fibrotic deposits on their aortic valve, and reach hemodynamically more severe AS for similar amounts of aortic valve calcification compared to men.6–8 Moreover, women are more likely to harbor a SAA, 9 increasing the risk of prosthesis-patient mismatch (PPM).
PPM occurs when a prosthetic valve has an effective orifice area (EOA) less than that of a native valve, which may influence postoperative outcomes. 10 A PARTNER I trial substudy showed that patients with SAA undergoing TAVR had larger aortic valve annulus than patients undergoing SAVR which translated into a lower rate of PPM. 11
Meanwhile, the VIVA trial highlighted that among patients with severe AS and SAA where women comprised the majority, there was no evidence of superiority of TAVR versus SAVR in valve hemodynamic outcomes. 4
The objective of this study was to compare the incidence of measured and predicted PPM in women with SAA undergoing TAVR versus SAVR using intra-annular Carpentier Edwards Magna Ease bioprosthetic valve and the balloon-expandable Edwards Sapien 3 and Sapien 3 Ultra transcatheter aortic valves.
Methods
Study Population
This study included women ≥18 years with SAA who underwent SAVR (Carpentier Edwards Magna Ease 19-21 mm) or TAVR (Edwards TAVI Sapien 3 & Sapien 3 ultra 20 and 23 mm) between 2011 and 2022. Inclusion criteria comprised having a baseline echocardiography, an echocardiography within 30 days post-intervention, and at least one longer-term follow-up echocardiography in the year following the intervention. Patients with pure aortic regurgitation (AR) were excluded. Patients who underwent concomitant procedures such as percutaneous coronary intervention (PCI) in the month prior to TAVR, coronary artery bypass grafts (CABG), left atrial appendage closure or replacement of the ascending aorta for SAVR patients were included. These criteria were fulfilled by 426 patients. Retrospective data collection was conducted using harmonized databases from two high-volume cardiac surgery centers in the province of Quebec. These databases centrally collect all preoperative, perioperative, and postoperative data including the echocardiographic data in a prospective fashion. Mortality was compiled using provincial vital statistics. Approval for the study was obtained from the local ethics committee on November 23rd, 2020 (IRB 20.261).
Study Endpoints
The primary endpoint was the incidence of moderate or severe PPM. Both measured PPM (M-PPM) and predicted PPM (P-PPM) were assessed.
Secondary endpoints included hospital length of stay (LOS) and short-term outcomes such as procedural, 30-day and 1-year mortality, reintervention for bleeding, peri-operative stroke, transient ischemic attack (TIA), acute renal failure, permanent pacemaker (PMP) implantation, bundle branch block (BBB), endocarditis, valvular thrombosis, SVD, and 1-year readmission. Endpoints also included hemodynamic data such as measured EOA, indexed EOA (iEOA), mean aortic gradient (MAG), and left ventricular ejection fraction (LVEF).
Measurement of PPM and other hemodynamic data involved echocardiographic assessment within the initial 30 days and up to 1 year post-intervention. Echocardiographies were conducted as routine follow-up or when mandated by a change in the patient's clinical status.
Definitions
SAA was defined as an aortic annulus measuring less than 23 mm at the preoperative echocardiography. 9
P-PPM was determined based on predicted EOA values derived from charts published by Lancellotti, Pibarot, et al for SAVR 12 and Hahn, Pibarot, et al for TAVR. 13 Regarding SAVR, the predicted EOA was 1.3 ± 0.3 cm2 and 1.5 ± 0.3 cm2 for the Edwards Magna Ease 19 mm and 21 mm valves, respectively. Regarding TAVR, the predicted EOA was 1.22 ± 0.22 cm2 and 1.45 ± 0.26 cm2 for the Edwards SAPIEN 3, 3 ultra 20 mm and 23 mm valves, respectively (Table 1).
Predicted Effective Orifice Area (EOA) by Valve Size and Type.

EOA: effective orifice area; SAVR: surgical aortic valve replacement; TAVR: transcatheter aortic valve replacement.
M-PPM was determined based on measured EOA derived from the continuity equation: EOA = CSALVOT × (LVOTVTI ÷ AoVTI), where EOA is in cm2, CSA is the cross-sectional area of the left ventricular outflow tract (LVOT), LVOTVTI is the subaortic velocity–time integral and AoVTI is the aortic velocity–time integral, both in cm.
The severity of PPM was determined by specific ranges of iEOA obtained by dividing a patient's EOA by their body surface area (BSA). Moderate PPM was defined by an iEOA of 0.65 to 0.85 cm2/m2 and severe PPM by an iEOA < 0.65 cm2/m2. Different thresholds were applied in obese patients with a body mass index (BMI) exceeding 30 kg/m2 according to the VARC-3 definition, with iEOA values between 0.56 and 0.70 cm²/m² classified as moderate and those <0.55 cm²/m² as severe.13,14 Herein, PPM defined according to the VARC-3 definition was referred to as PPM adjusted for BMI > 30.
The Kidney Disease: Improving Global Outcomes (KDIGO) definition was used to define acute renal failure which translates to an absolute increase in the serum creatinine level of 26.5 μmol/L (0.3 mg/dL) or more or an increase in the serum creatinine level of more than 150% to 200%. 15
The definitions of hemodynamically significant SVD, valve thrombosis and endocarditis were based on the VARC-3 criteria, 16 and categorized dichotomically (presence/absence).
Statistical Analysis
Women receiving TAVR versus SAVR were compared. Continuous variables were expressed as mean ± standard deviation or median [interquartile range] and were compared using ANOVA or Kruskal–Wallis tests, as appropriate. Categorical variables were expressed as frequencies (%) and were compared using Pearson's χ2 test or Fisher's exact test, as appropriate. For each variable, only patients with complete data were reported, with a proportion of 100% referring to the total number of patients with complete data for that specific variable. Analyses were conducted using SAS version 9.4. A P < .05 was considered significant.
Approval for the study was obtained from the local ethics committee. When necessary, patients were contacted, and permission to obtain echocardiography reports from other centers was secured.
Results
Baseline Characteristics
Out of 426 patients, 64.8% (n = 276) underwent SAVR and 35.2% (n = 150) underwent TAVR. TAVR patients were older than SAVR patients (SAVR: 75 ± 8; TAVR: 81 ± 7 years old; P < .001). 36.2% (n = 100) of SAVR and 30.7% (n = 46) of TAVR patients had a BMI > 30 kg/m2. Baseline characteristics are described in Table 2.
Baseline Characteristics of the Study Population (n = 426).

SAVR: surgical aortic valve replacement; TAVR: transcatheter aortic valve replacement; NYHA: New-York Heart Association; PCI: percutaneous coronary intervention; LVEF: left ventricle ejection fraction; MAG: mean aortic gradient.
While a larger proportion of SAVR patients had a bicuspid valve (SAVR: 18% (n = 49); TAVR: 4.7% (n = 7); P < .001), TAVR patients were more likely to have had previous atrial fibrillation or flutter (SAVR: 19% (n = 51); TAVR: 32% (n = 47); P = .003) and manifested more advanced signs of heart failure (NYHA class III or IV: SAVR: 54.2% (n = 148); TAVR: 69.4% (n = 103); P = .002). There was no significant difference in coronary artery disease prevalence between SAVR and TAVR patients (SAVR: 42% (n = 114); TAVR: 48% (n = 72); P = .205). However, TAVR patients were more likely to have had previous PCI (SAVR: 11% (n = 31); TAVR: 27% (n = 41); P < .001) or cardiac surgery (SAVR: 6.2% (n = 17); TAVR: 15% (n = 22); P = .004). Although both SAVR and TAVR patients had similar preoperative LVEF (SAVR: 60 ± 9; TAVR: 60 ± 9; P = .960), TAVR had higher MAG (SAVR: 45 ± 16; TAVR: 51 ± 15; P < .001).
Operative Characteristics
Operative details are summarized in Table 3. Regarding SAVR, median cardiopulmonary bypass and cross-clamp time were 88 [73, 118] and 69 [56, 92] minutes, respectively. Four patients (1.4%) had a concomitant ascending aorta replacement and 92 (33%) had a concomitant coronary artery bypass graft (CABG) operation.
Operative Details.
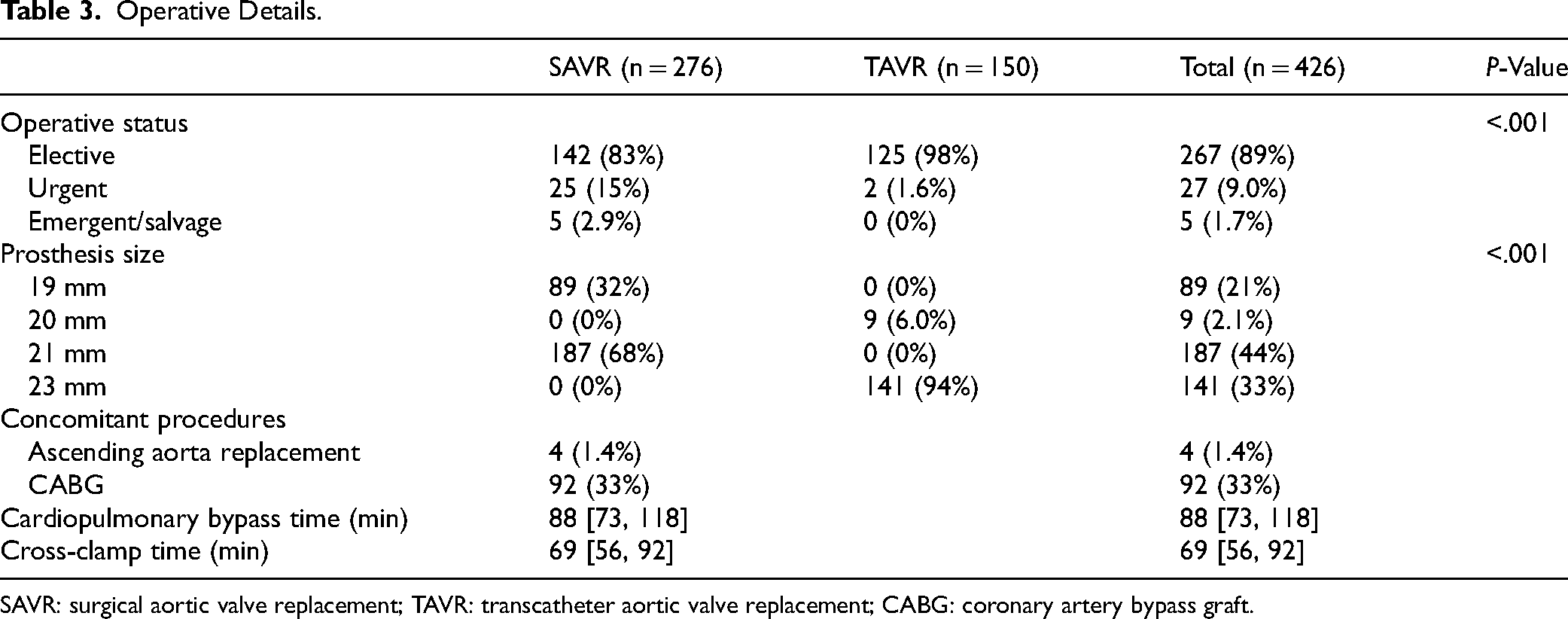
SAVR: surgical aortic valve replacement; TAVR: transcatheter aortic valve replacement; CABG: coronary artery bypass graft.
In terms of prosthesis size, most SAVR patients (68%, n = 187) were assigned a 21 mm prosthesis, while the others (32%, n = 89) received a 19 mm prosthesis. Almost all TAVR patients (94%, n = 141) were assigned a 23 mm prosthesis, with the others (6%, n = 9) receiving a 20 mm prosthesis.
PPM Incidence and Postoperative Hemodynamic Status
SAVR patients were significantly more likely to have moderate to severe P-PPM compared to TAVR patients (SAVR: 53%; TAVR: 39%; P = .005). When adjusting for BMI, proportions were lower and the difference between SAVR and TAVR was no longer significant (SAVR: 25%; TAVR: 19%; P = .148). Similarly, SAVR patients were significantly more likely to have M-PPM compared to TAVR patients (SAVR: 73%; TAVR: 32%; P < .001). Again, proportions were lower when adjusting M-PPM for BMI although the difference between SAVR and TAVR remained significant (SAVR: 64%; TAVR: 23%; P < .001). Interestingly, the proportion of SAVR and TAVR patients with M-PPM was generally higher than those with P-PPM both adjusted and unadjusted for BMI > 30, except for unadjusted PPM in TAVR patients where a smaller proportion had M-PPM compared to P-PPM (P-PPM: 39%; M-PPM: 32%). Other hemodynamic data can be found in Table 4 and Figure 1.
PPM and Hemodynamic Data.

SAVR: surgical aortic valve replacement; TAVR: transcatheter aortic valve replacement; PPM: prosthesis-patient mismatch; P-PPM: predicted prosthesis-patient mismatch; M-PPM: measured prosthesis-patient mismatch; BMI: body mass index; EOA: effective orifice area; iEOA: indexed effective orifice area; MAG: mean aortic gradient; LVEF: left ventricle ejection fraction.
Short-Term Clinical Outcomes
Regarding the postoperative course, the median hospital LOS was five days longer for SAVR compared to TAVR patients (SAVR: 8 [6, 11]; TAVR: 3 [1, 6] days; P < .001). One-year mortality was 4.3% (n = 10) for SAVR compared to 6.5% (n = 7) for TAVR, although this difference was not significant (P = .377). Patients undergoing TAVR had more new LBBB/RBBB compared to SAVR patients (SAVR: 4.0%; TAVR: 16%; P < .001). There were significantly more readmissions following SAVR compared to TAVR (SAVR: 8.2%; TAVR: 3.0%; P = .044). There were significantly less TIA following SAVR compared to TAVR (SAVR: 5.5%; TAVR: 13%; P = .009). There was no significant difference in acute renal failure (SAVR: 15%; TAVR: 12%; P = .441). Other short-term clinical outcomes are described in Table 5.
Short-term Clinical Outcomes.
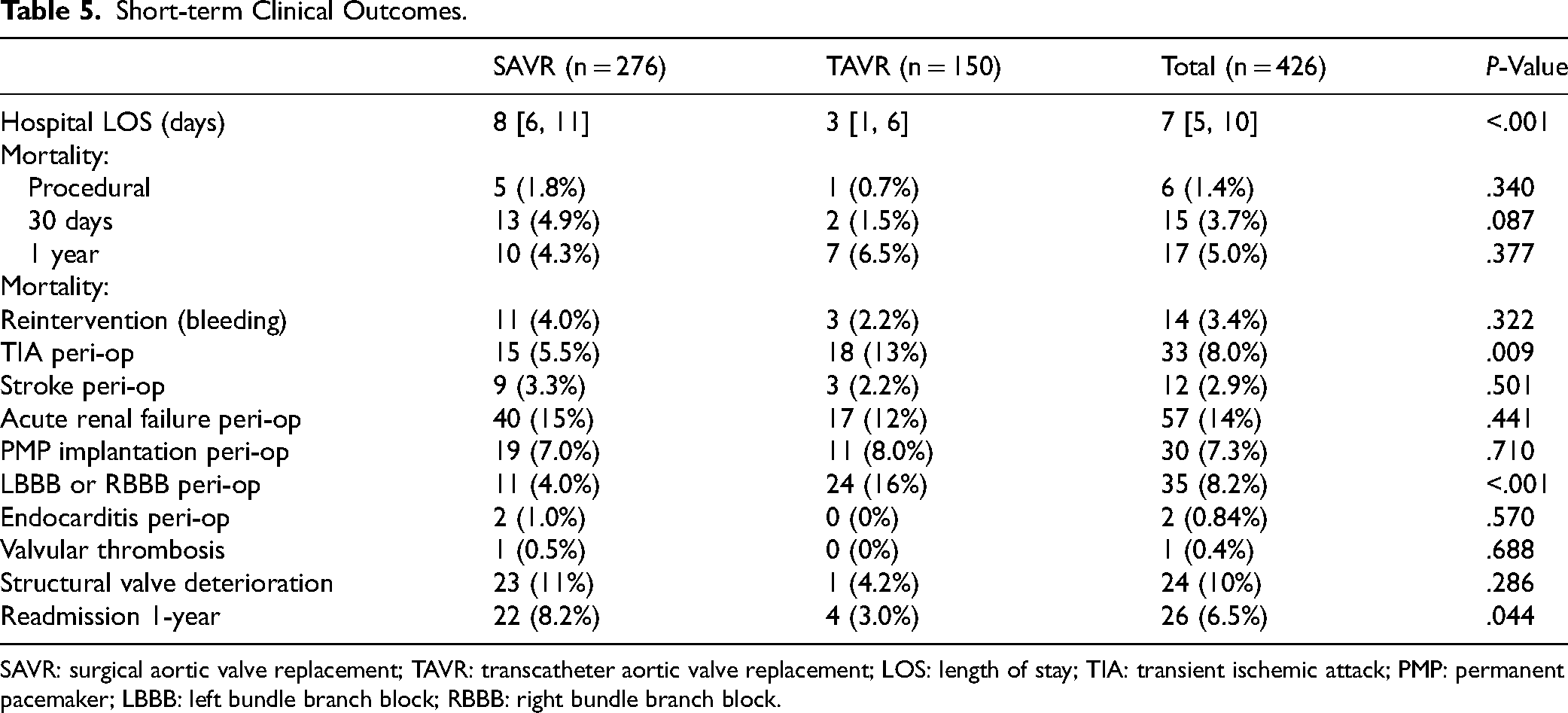
SAVR: surgical aortic valve replacement; TAVR: transcatheter aortic valve replacement; LOS: length of stay; TIA: transient ischemic attack; PMP: permanent pacemaker; LBBB: left bundle branch block; RBBB: right bundle branch block.
Discussion
To our knowledge, this is the first study assessing the incidence of P-PPM and M-PPM in women with SAA undergoing SAVR versus TAVR using intra-annular bovine pericardial prostheses. The main findings of the study are the following: (a) there were significantly more moderate to severe P-PPM and M-PPM in SAVR compared to TAVR; (b) the proportion of PPM in SAVR and TAVR was lower when adjusting PPM for BMI > 30; (c) the proportion of M-PPM was higher than P-PPM in SAVR and TAVR patients except for unadjusted for BMI PPM in TAVR where a smaller proportion had M-PPM compared to P-PPM.
Aortic Stenosis in Women
Although women have been historically underrepresented in many landmark studies forming the basis of guideline recommendations and risk prediction tools, it is now increasingly acknowledged that there are sex-based differences in AS pathophysiology and clinical presentation. Women with severe AS have less overall aortic valve calcification than men, although they develop similar AS severity as men for lower aortic valve calcification loads, possibly due to valvular fibrosis.3,17 They tend to have concentric LV hypertrophy in AS with higher relative wall thickness and smaller end-diastolic diameter while men often present an eccentric LV hypertrophy picture. 18 Moreover, women have a smaller AV annulus and shorter distance from annulus to coronary ostia, corresponding to smaller BSA. They tend to have more paradoxical low-flow/low-gradient AS and are less likely to have a congenital bicuspid aortic valve.7,19,20 In this study, 13% of women had a bicuspid valve. In regards to their clinical presentation, women with severe AS are often older and more symptomatic at presentation, although they tend to have fewer comorbidities. 21 They are less seen by specialists and undergo fewer diagnostic tests.5,22 They also experience later referral for interventions.3,23 At the time of referral, women have lower prevalence of concomitant coronary artery disease which decreases the rate of concomitant CABG.24–26 In this study, 44% of women had coronary artery disease and 33% had a concomitant CABG. Finally, AS interventions in women are more technically demanding. Indeed, there is a strong female sex preponderance for SAA which increases surgical risk, 9 and women are at increased need for aortic annulus enlargement. 5
Short-Term Clinical Outcomes
Regarding short-term clinical outcomes, no significant differences were found in terms of mortality and stroke between SAVR and TAVR. However, hospital LOS and 1-year readmission rates were significantly higher for SAVR. Similarly, in the VIVA trial, a prospective multicenter randomized controlled trial looking at patients with SAA and severe AS undergoing TAVR versus SAVR where women represented 93% of the study population, no differences were found in terms of mortality and stroke between both interventions. The trial also showed longer median hospitalization time in SAVR patients 4 A PARTNER-1 sub-study also showed no difference in mortality in patients with SAA although there was a higher incidence of stroke and major vascular complications in the TAVR group. 11
Prosthesis-Patient Mismatch
Although common, PPM is less frequent after TAVR versus SAVR as transcatheter valves have larger EOAs and lower gradients.27–29 One explanation for this may be related to the absence of a sewing ring in TAVR valves. The presence of SAA increases the risk of PPM, 30 which is itself associated with higher mortality, morbidity, rehospitalization rate, left ventricular workload, and reduced aortic prosthesis longevity in surgical prostheses.31–33 In this study, PPM rates were lower in TAVR versus SAVR regardless of whether PPM was measured or predicted and adjusted or unadjusted for BMI > 30, although this difference was not significant for P-PPM adjusted for BMI > 30. Similarly, in a substudy of the PARTNER I trial, patients with SAA undergoing TAVR had larger aortic valve areas than those undergoing SAVR, which translated into a lower rate of PPM. 11 However, in the VIVA trial comprised of patients with severe AS and SAA where women were the majority, there was no evidence of superiority of TAVR versus SAVR in valve hemodynamic results, suggesting that the two therapies represent a valid alternative for treating patients with severe AS and SAA. 4
PPM incidence after SAVR is high but varies widely in the literature. A meta-analysis by Head et al comprised of 34 observational studies and 27,186 patients found an incidence of 44% (34% moderate PPM and 10% severe PPM). 33 This study found an incidence of 73% moderate to severe M-PPM after SAVR, and 64% when adjusting for BMI > 30. These proportions are higher than the results from the meta-analysis where only 8 of the 34 studies used EOAs measured by echocardiography. However, when using predicted EOA, the results of this study are closer to the ones mentioned in the meta-analysis : 53% had moderate to severe P-PPM after SAVR and 25% when adjusted for BMI > 30. In regards to TAVR, the WIN-TAVI registry, comprised exclusively of female patients with severe AS, reported a PPM rate of 32.8%. 34 Similarly, in this study, P-PPM and M-PPM rates after TAVR were 39% and 32%, respectively. However, these proportions were reduced to 19% and 23%, respectively, after adjusting for BMI. The reason why adjusting for BMI reduced the incidence of PPM in this study may lie in the motive behind the VARC 3 definition itself. Indeed, the definition adjustment for BMI > 30 was introduced as a high BSA can lead to the over-indexing of the iEOA and the overestimation of PPM. 35
There remains a debate on whether measured or predicted EOA is preferred to assess PPM following SAVR and TAVR. Measured EOA of a given prosthetic valve type and size depends on the echocardiographic quality and correctness. The measurement varies from individual to individual and within serial measurements in one same patient. There may be sex variations as well given different aortic root anatomies at baseline and women being more likely to have a smaller left ventricular cavity with concentric hypertrophy for example. Moreover, pressure recovery, which impacts Doppler derived gradients across the prosthetic valve, may explain why echocardiography-measured EOA might underestimate the real EOA, falsely classifying patients’ PPM outcome.36,37 Meanwhile, predicted EOA is based on various sources, such as mean echo data from various patient cohorts or the reported size of manufacturers, and depends on the quality of the data. TAVR studies tend to use more M-PPM while SAVR studies commonly use P-PPM, which may cause discrepancies between series. As such, a PARTNER-2 study by Ternacle et al aimed to compare the incidence of P-PPM and M-PPM after SAVR and TAVR and found that the incidence of P-PPM was much lower than M-PPM in both SAVR and TAVR. 38 Similarly, in this study, the proportion of SAVR and TAVR patients with M-PPM was generally higher than those with P-PPM both adjusted and unadjusted for BMI, except for unadjusted PPM in TAVR patients where a smaller proportion had M-PPM compared to P-PPM. This is similar to a previous study by Natalis et al which found a lower rate of M-PPM versus P-PPM in women undergoing TAVR. Low-flow state was found in 16% of patients in the Natalis et al study. 39 Meanwhile, in the Ternacle et al study, low-flow states were quite frequent (50%) and may have been responsible for the more frequent occurrence of M-PPM, as low-flow states can lead to underestimation of EOA and diagnosis of pseudosevere PPM.
Aortic root enlargement was the first surgical alternative proposed to treat SAA and reduce PPM. A posterior enlargement of the aortic root by the Rittenhouse-Manouguian or Nicks-Nuñez techniques is the most used method, however an anterior enlargement is also possible with the Konno-Rastan technique.9,40 A more contemporary technique is the Y-incision technique described by Bo Yang from Michigan. 41 Aortic root enlargement poses no significant increase in surgical risk according to recent studies and may explain a recent decline in the prevalence of PPM after SAVR.39,42,43 Although we did not collect data regarding aortic annulus enlargement, we suspect that 10% of the SAVR cohort had an annulus enlargement.
Limitations
In this retrospective study, we conducted comparative analysis between SAVR and TAVR, focusing solely on routinely used pericardial valves including the Carpentier Edwards Magna Ease bioprosthetic valve as well as the balloon-expandable Edwards Sapien 3 and Sapien 3 Ultra transcatheter aortic valves. These results cannot be extrapolated to other valves.
Although the small patient sample size may have impacted the statistical power of this study, this patient population depicts a real life portrait of a female patient population assessed by our cardiac heart team. Moreover, statistical power may have been influenced by missing data in some patients which limited the total number of patients being compared. It is important to note that PPM was measured at different timepoints perioperatively, would it be prior to discharge, in the weeks or in the months up to one year following intervention, depending on the patient. Mortality was assessed at the time of the procedure as well as at 30-day and 1-year postoperatively. It would have been interesting to follow patients for a longer postoperative period, as some differences in clinical outcomes may only become evident at a later time.
Stroke volume index was not collected in this database. This data would have been interesting as low-flow states can lead to underestimation of EOA and diagnosis of pseudosevere PPM.
Nonetheless, the results of this study which looked at the incidence of P-PPM and M-PPM after SAVR versus TAVR using pericardial prostheses in a specific population of women with SAA may help in shaping more personalized diagnostic and treatment options for this specific population in the near future.
Conclusion
Women with SAA have significantly more moderate to severe P-PPM and M-PPM after SAVR versus TAVR. The proportion of PPM diminishes when adjusting for BMI > 30. This is the first study assessing the incidence of predicted and measured PPM in women with SAA undergoing SAVR versus TAVR using intra-annular bovine pericardial prostheses. More research is needed to assess the sex-based predictive value of P-PPM and M-PPM in SAVR versus TAVR, in a larger study population and with longer postoperative follow-up.
Footnotes
Abbreviations
Ethics committee approval
IRB 20.261, approved on November 23rd, 2020.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Edwards Lifesciences (grant number 60000 CAD).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Kalavrouziotis is a proctor for Edwards Lifesciences. Dr Forcillo is a proctor, consultant, speaker and part of the advisory board for Edwards Lifesciences, Medtronic and Terumo. All the remaining authors do not have any disclosures.
