Abstract
Biological heart valves (BHVs) are currently the main prosthetic substitute used to treat patients with severe valvular heart diseases. Unlike mechanical valves, BHVs provide better hemodynamic profile and lower thrombogenicity. However, their durability remains one of the main concerns in the field. Structural bioprosthetic valve degeneration (SVD) is a progressive phenomenon occurring early postimplantation and characterized by the fibro-calcification of the BHV leaflets, which leads to BHV failure over ∼10 years. SVD is currently an unpreventable and untreatable condition occurring in all BHV. The pathophysiological mechanisms leading to the development and progression of SVD remain largely unknown. However, a better understanding of these mechanisms will provide valuable data to improve BHV durability via the development of preventive treatments and/or newly engineered biological tissues. In this review, we provide a contemporary overview of the complex and interrelated pathophysiological mechanisms leading to SVD. We discussed the relevant studies in the fields, addressing the role of (i) the biomaterial structure and manufacturing processes, (ii) the active cell-mediated fibro-calcific processes, (iii) the residual immunogenicity of the currently animal-derived biomaterial, and (iv) the subclinical thrombosis, in the development and progression of SVD.
This is a visual representation of the abstract.
Keywords
Key points
Introduction
The burden of valvular heart diseases has constantly grown over the past decades, especially because of the aging of the population, which leads to an exponentially increased number of diagnosed patients.1–4 The most recent epidemiological data reported an incidence of valvular heart diseases reaching up to 10% in patients >70 years old, a rate that is expected to double by 2050.1–5 Without medical treatment for these life-threatening conditions, the only option offered to the patients with symptomatic severe valve diseases is to replace the diseased native valves with prosthetic valves, either with mechanical or biological heart valves (BHVs).2,6,7 Even if this invasive approach is not curative, valve replacement is associated with a significant reduction in morbidity and mortality.4,8–11 Over the last 2 decades, the implantation of BHV, via conventional surgery and transcatheter implantation, has overpassed the use of mechanical prosthetic valves: indeed, the recent advances and developments of the transcatheter aortic valve implantation have revolutionized the field with a constant increase in the use of BHV instead of mechanical valves.12–15
BHVs present significant benefits as opposed to mechanical valves: they provide better hemodynamic profiles and lower risk of bleeding for the patients due to the absence of a life-long anticoagulation regimen, coupled with better patient quality of life.6,7 However, the main limitation of the BHV is their durability, which arises as one of the main concerns in the field and a cornerstone for an even larger expansion of their use. The constant increased use of BHV associated with the implantation in younger patients with longer life expectancy will lead to a considerable rise in the burden of this prosthesis-related disease in the coming years.
Nowadays, the mechanisms leading to BHV degeneration remain largely unknown, but likely involved complex and multiple pathophysiological processes which start early in the prosthetic's life. In this state-of-the-art review, we provide a contemporaneous overview of BHV degeneration, covering its definition, its clinical presentation and associated risk factors, as well as a discussion of the currently described underlying pathophysiological mechanisms.
BHV dysfunction: Nonstructural and structural valve degeneration
BHV dysfunctions are classified as nonstructural and structural valve degeneration (SVD). Nonstructural degeneration is defined as a non-intrinsic deterioration of bioprosthetic leaflets, such as patient–prosthesis mismatch and paravalvular regurgitation, as well as a preventable and reversible phenomenon with pharmacological treatment, such as prosthetic valve thrombosis and endocarditis.16–19 SVD is characterized by acquired leaflet abnormalities resulting from a progressive fibro-calcific remodeling, thickening, and stiffening of the leaflets or matrix disruption leading to tear or perforation of the leaflet, resulting in BHV stenosis and/or regurgitation.16–20
Until recently, the durability of BHV related to SVD was defined by the reoperation rate. 21 However, this metric solely encompassed end-stage and advanced SVD, without any consideration regarding significant but nonsevere stages of SVD that do not require redo surgery, as well as patients no longer eligible for surgery.16–18 This definition thus underestimated the incidence and clinical impact of SVD and provided overoptimistic data regarding the durability of BHV. In 2017, the European Association of Percutaneous Cardiovascular Interventions (EAPCI) has provided the first standardized definitions of SVD based on morphological and functional abnormalities of the leaflets, 16 which has been updated in 2018 by the “Valve in Valve International Data” (VIVID) investigators, 17 and finally the “Valve Academic Research Consortium 3” (VARC-3) in 2021 (Table 1). 18 Based on these definitions, which encompassed morphological and hemodynamic alterations of the BHV, a significantly higher incidence of SVD has been recently reported, reaching up to ∼30% in surgically implanted BHV at 10 years post-surgery. 22 Importantly, in this study, SVD was also associated with more than a 2-fold higher risk of mortality. 22 These findings, concordant with other reports on the early development of SVD in BHV,20,23,24 highlight an unrecognized phenomenon occurring earlier than expected, with significant impacts on the management and outcomes of the patients.
Standardized definitions of structural valve degeneration based on EAPCI, VIVID, and VARC-3 consensus statements.

Concerning transcatheter BHV, available data on their mid- and long-term durability remain sparse. 25 A recent sub-analysis of a randomized clinical trial reported that SVD develops earlier and more frequently with the 2nd generation of balloon-expandable transcatheter BHV as compared to surgical BHV. 26 This finding was not observed with the 3rd generation of the transcatheter BHV, suggesting better durability with newer transcatheter BHV that could finally raise similar durability than surgical BHV. 26 The most recent follow-up report from the NOTION trial showed a higher rate of SVD in surgically implanted BHV as compared to transcatheter valves, while the overall rate of BHV dysfunctions was similar. 27 Importantly, in this trial, one-third of the surgical BHV was externally mounted, a prosthetic design associated with early development of SVD. 27 Questions regarding the potential impact of the remaining native diseased valve after transcatheter valve implantation have also emerged in the literature, but our current understanding remains limited, and further studies are needed to determine its potential adverse effects on the future development of SVD. 28
To limit redo open-heart surgery and its associated risks, one of the options is to perform a valve-in-valve intervention, consisting of the deployment of a transcatheter BHV within the initial surgically implanted or transcatheter-delivered BHV.29–34 Even if this approach is technically feasible and provides short-term benefits, multiple implantations for the same patient remain limited, and important drawbacks remain present, whether in terms of device/interventional costs than health-related adverse events at mid- and long-term perspectives.35–39 The concern related to the durability of BHV, which will be more and more prominent in the coming years with the opening of BHV implantation toward the younger population and the constant developments and evolutions in the field of transcatheter BHV, highlights the need to prevent and/or treat SVD. To achieve these objectives, a comprehensive description of the clinical presentation and risk factors associated with SVD, coupled with a better understanding of the underlying pathophysiological mechanisms, is mandatory.
Clinical presentation and risk factors for SVD
The analyses of large cohorts of patients who received BHV coupled with the studies of explanted BHV at the time of redo surgery have provided insightful clinical data to identify risk factors associated with SVD and describe its morphological presentation.
Clinical risk factors associated with SVD can be classified into 4 main categories: (i) the traditional cardiovascular risk factors, usually referred to the atherosclerotic-like factors, (ii) the factors related to phospho-calcic metabolism and renal insufficiency, (iii) the stress-related factors, and (iv) the procedural- and valve-related factors. Recently, the role of subclinical leaflet thrombosis (SLT) in the development of SVD, a more and more frequent finding post-BHV implantation, has been hypothesized.40–43
Age and sex are known as the main cardiovascular risk factors in multiple cardiovascular diseases. In the context of SVD, few studies reported a significant association between younger age and early development of SVD, even if conflicting clinical data have also been reported depending on the definition used for SVD (ie, hemodynamic- or redo intervention-based definitions).24,44,45 Interestingly, a recent study provides mechanistic insights supporting the clinical association observed between age and SVD. 46 Data on sex-specific mechanisms related to the development of native valve diseases are currently emerging,47–57 but the available data regarding differential incidence and development of SVD in men and women remain controversial.58–61
The traditional metabolic risk factors, such as diabetes mellitus, metabolic syndrome, and/or hypercholesterolemia, were first identified in large cohorts of patients who received BHV.22,58,59,62–66 More recently, increased plasma apolipoprotein B/apolipoprotein A1 ratio (ie, a marker of a pro-atherogenic profile), small and dense low-density lipoprotein (LDL) particle size (ie, LDL particles with increased abilities to infiltrate BHV and with a greater rate of oxidation), the plasma level of lipoprotein-associated phospholipase A2, and the proprotein convertase subtilisin/kexin type 9 serum level were associated with SVD.22,67,68 Controversial data have also been published regarding the association of lipoprotein (a) [Lp(a)] with SVD,69,70 and further studies are needed to confirm these findings. Inflammation and related monocyte/macrophage activation and recruitment have also been associated with a higher incidence of SVD. 71 Even if these findings come from clinical association studies, the effect of these well-known atherosclerotic-like factors on SVD supports the idea that inflammatory and lipid-mediated mechanisms would be among the main triggers for the development of SVD.
Dysregulation of the phospho-calcic metabolism, renal insufficiency, and hyperparathyroidism are associated with SVD.59,65,66,72,73 These conditions favor the establishment of a pro-calcifying environment, with an increase in circulating calcium and inorganic phosphate favoring ectopic calcification in tissue such as BHV. 74
BHV faces multiple mechanical and shear stresses.17,18 These successive, cyclic, and diverse stresses imposed on BHV leaflets over the prosthesis's life have been identified as potential contributors of structural biomaterial degradation and further implementation of degenerative processes (mechanisms discussed in following sections).75–78 As well described in native valves, the disturbed or turbulent flow post-BHV implantation could also participate in these processes, via the exacerbation of cell recruitment and activation into the biomaterial. 79 –82 These complex stresses can be further intensified by patient-related factors (ie, hypertension and small annulus size), procedural-related factors (ie, small prosthetic size, suboptimal BHV positioning and/or expansion, patient–prosthesis mismatch, and subsequent turbulent flow postimplantation), and BHV-related factors (ie, valve types, sizes, and designs), all described as promoting factors for early development of SVD.17,18,59,66,76,77,82–90 On this purpose, some studies have underlined differences between surgically implanted and transcatheter-delivered BHV that could mitigate the development of SVD: (i) the preparation protocols of the transcatheter valves (ie, valve crimping, loading, balloon deployment, and post-dilatation) can damage the leaflet structure; 91 (ii) the deployment of these BHV is done in a turbulent aortic flow context, as opposed to surgically implanted BHV on non-beating heart;92,93 and (iii) the expansion of the transcatheter BHV can lead to asymmetric and/or elliptical shape of the implanted BHV, which in turn impairs normal function and hemodynamics.92,94 Overall, these data highlight the interrelated involvement of patient-, procedural-, and BHV-related factors on SVD, at least in part, via their impact on the mechanical and shear stresses imposed to BHV leaflets, as well as the resulting structural modification of the biomaterial.
At the tissue level, histological analysis of explanted BHV reveals a (pseudo)endothelization of the BHV, leaflet thrombosis, lipid and protein accumulation into the leaflets, presence of multiple and abundant subpopulations of macrophages, and red blood cells, fibroblast-like cells, and foam cells at the end-stage disease.95–99 A recent analysis of the ultrastructure of explanted BHV confirmed these findings. 99 As described in native aortic valve calcification,79,80,100 we can postulate that these biological and cellular factors promote extracellular matrix (ECM) degradation and the subsequent calcification/mineralization of BHV.
Altogether, these data identify clinical risk factors and potential underlying mechanisms associated with the development and progression of SVD, which should certainly be factored into the management of patients undergoing valve replacement. However, the understanding of the pathophysiological mechanisms leading to SVD, and their kinetics and regulations, appears as the cornerstone to significantly modify the clinical course of BHV and enhance BHV durability.
Pathophysiological mechanisms leading to SVD
Most of the currently implanted BHVs are manufactured with xenograft substitutes, coming from bovine or porcine pericardium, or “intact” porcine aortic valves.101–103 As opposed to the native valves in which endothelial and interstitial valvular cells control tissue homeostasis, these xenografts are decellularized and usually fixed with glutaraldehyde, a process conferring adequate viscoelasticity and biomechanical properties to the BHV leaflets and allowing suitable BHV hemodynamic function.101–103 However, this manufacturing process also predisposes the tissue to early and accelerated degenerative processes, via both passive dystrophic calcification and cell-mediated mineralization.102–107
Four main hypotheses are currently investigated with regard to the development and progression of SVD, covering multiple interrelated pathophysiological mechanisms: (i) the intrinsic structure and design of the biological leaflets prone to dystrophic calcification, (ii) the implementation of cell-mediated active fibro-calcifying mechanisms, (iii) the contribution of the residual immunogenicity of the implanted xenograft biomaterial, and (iv) the potential impact of SLT.
Biological leaflet structure as a favorable substrate for SVD
The manufacturing process of animal-derived biomaterials, such as the one used for aortic graft and BHV leaflet preparation, is based on the neutralization and stabilization of a “decellularized” Type-1 collagen-rich matrix using glutaraldehyde cross-linking procedure (Figure 1).102,103 Some studies highlighted that this specific biomaterial preparation can promote calcification,108–113 and interestingly, this process has been recently applied in the field of bone-designed scaffolds, with the objective to develop new biomaterials with enhanced calcification properties. 114 One of the mechanisms intrinsically related to the calcification of implanted biomaterials is called dystrophic calcification: this passive phenomenon is described as hydroxyapatite crystal precipitation in “damaged” tissue, mainly due to the local imbalance of calcium and inorganic phosphate.106,107
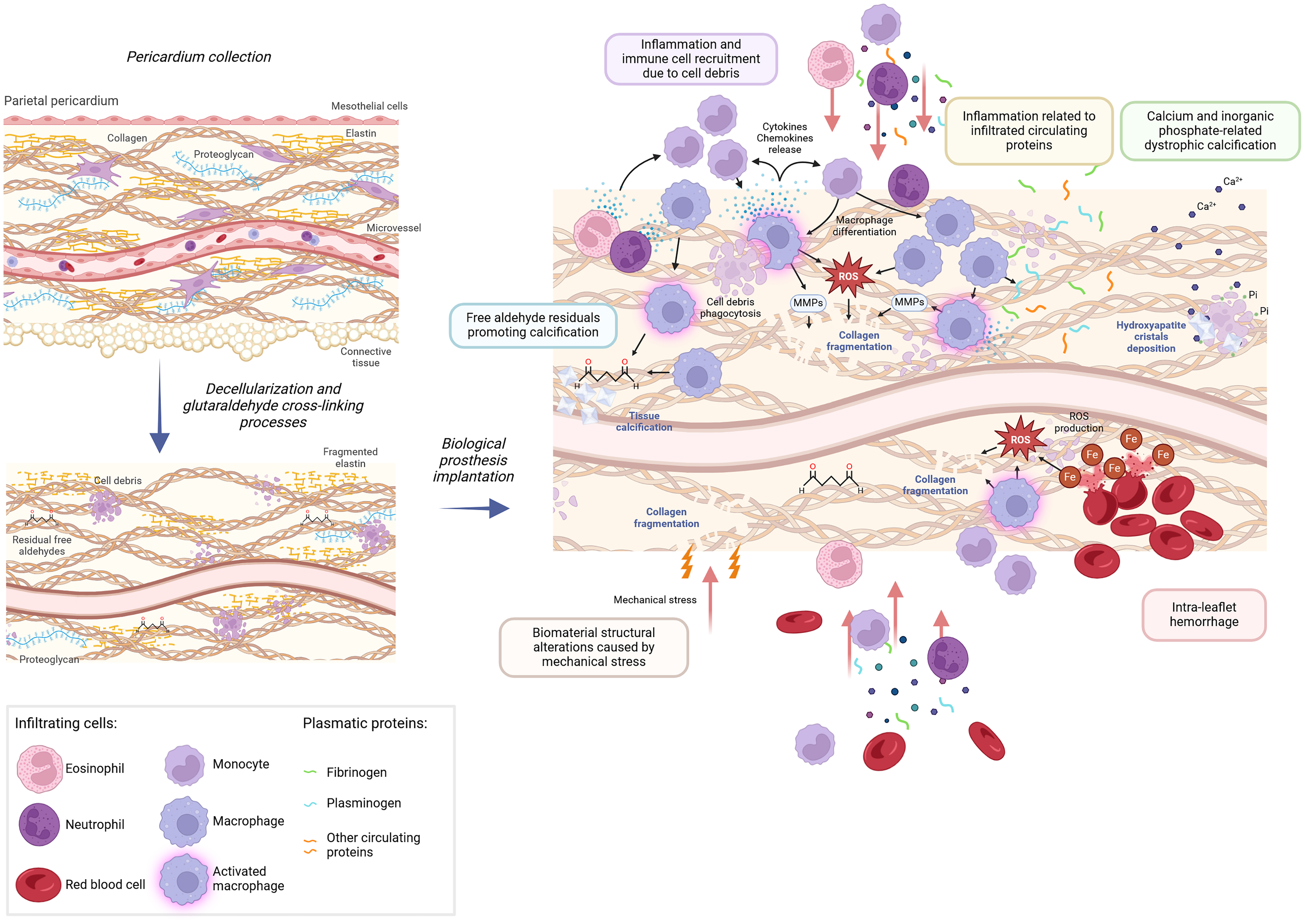
Biomaterial-derived pathophysiological mechanisms associated with SVD. Decellularization and fixation via glutaraldehyde are applied to animal-derived tissue to generate a cross-linked Type-1 collagen-rich biomaterial. These manufacturing processes are associated with significant changes in the structure and composition of the newly generated biomaterial: free aldehydes and cell debris are still present, proteoglycan content is reduced, and elastin fibers are degraded. Following BHV implantation, interrelated pathophysiological mechanisms related to the development of structural bioprosthetic degeneration have been described: (i) the increased local bioavailability of calcium (Ca2+) and inorganic phosphate (Pi) induce dystrophic calcification; (ii) the cytotoxicity and pro-calcifying properties of the free aldehydes potentiate dystrophic calcification; (iii) the modified structure and composition of the biomaterial (ie, cross-linked collagen fibers coupled with reduced and/or degraded proteoglycans and elastin fibers) reduce its abilities to face the mechanical stresses imposed on BHV leaflets, which in turn contribute to collagen degradation and subsequent dystrophic calcification; (iv) the absorption of proteins from the circulation, especially fibrinogen and plasminogen proteins, promotes dystrophic calcification; (v) the cell adhesion and infiltration, particularly macrophages, lead to the development of a pro-calcifying microenvironment, which further enhance calcification processes; and (vi) the intra-leaflet hemorrhage contributes to the local inflammation and enhanced oxidative stress via the local iron release. Created in BioRender. Capoulade, R. (2025) https://BioRender.com/r61e021.
In the context of BHV, the glutaraldehyde cross-linking procedure has been widely applied to balance the benefits and disadvantages in terms of structural/functional properties, hemocompatibility, and immunogenicity of the biomaterial used for BHV and has been more recently coupled with anti-calcification pretreatment. 103 However, pro-calcifying structural characteristics of this animal-derived biomaterial could participate in the development of SVD.
First of all, the decellularization process applied to the biomaterial eliminates resident living cells.101–103 As well described in native valves, resident valvular cells continuously remodel the leaflet's ECM to mitigate the impact of diverse stresses and stimuli imposed to the valves and maintain their structure and function over time. 80 The lack of ECM turnover and remodeling in the context of BHV leaflets, due to the absence of tissue-resident cells, could then favor ECM degradation and further calcification. Conjointly to this phenomenon, the decellularization process does not completely eliminate all resident cell components: indeed, residual cell debris can still be observed in non-implanted pericardial tissue (Figure 1). 115 The incomplete clearing of cellular elements (including inorganic phosphate-rich cell membranes) coupled with the bioavailability of calcium (from both the circulation and the remaining cell debris into the biomaterial) is associated with a local increase in calcium and inorganic phosphate, a microenvironment favoring dystrophic calcification.115,116 In addition, the glutaraldehyde cross-linking procedure is inevitably associated with the presence of free aldehydes in the biomaterial: the well-described cytotoxicity and pro-calcifying properties of these free aldehydes could then contribute to (and/or exacerbate) the process of dystrophic calcification post-BHV implantation, as well as limit its neo-endothelization (Figure 1).112,117–119
The manufacturing process also significantly modifies the structure (ie, composition, stiffness, and elasticity) of the biomaterial, which further impacts its mechanical properties (Figure 1).102,103 Indeed, as opposed to native valves, BHV leaflets are mainly composed of cross-linked Type-1 collagen fibers, with relatively low amounts of proteoglycans and elastin fibers, that are not efficiently stabilized by glutaraldehyde treatment and are then prone to degradation and fragmentation over time.102,103 The ability of this collagen-rich biomaterial to face the repetitive and cyclic diverse mechanical stresses imposed on valve leaflets is then restricted and leads to a progressive disruption and degradation of collagen fibers.102,120 These structural damages coupled with the unmasking collagen “hole” zones related to the degradation/absence of proteoglycans have been associated with the development of BHV dystrophic calcification. 116 These findings are also supported by the consistently reported clinical associations between patient-, procedural-, and BHV-related factors, such as hypertension, patient–prosthesis mismatch, small annulus size, and early development of SVD.121–124 Implementation of the vicious circle related to the implantation of BHV with “nonoptimal” biomaterial structure facing important mechanical stresses, early dystrophic calcification, and progressive biomaterial ECM degradation leading to its structural and mechanical modifications supports the development and progression of dystrophic calcification causing SVD.
Finally, the absence of endothelium on top of the collagen-rich biomaterial will favor protein absorption into the BHV, a phenomenon recently described as promoting dystrophic calcification (Figure 1).113,125–130 In addition, cell adhesion and infiltration are also potentiated in this context and could contribute to the pathological processes. Macrophage invasion into BHV is consistently described in SVD and can promote pro-calcifying phenomena, both following foam cell apoptosis/necrosis and active pro-inflammatory- and ECM degradation-related mechanisms (mechanisms discussed in the next section; Figures 1 and 2).97,130–132 Evidence of BHV intra-leaflet hemorrhage, related to the accumulation of red blood cells into the biomaterial, is often reported in histological studies. Interestingly, studies performed in the context of native valve calcification highlight the role of red blood cells in the initiation and progression of native valve calcification, with insights into the underlying mechanisms promoting active fibro-calcific processes.133–135 We can then postulate that red blood cell infiltration into the BHV could also participate in the implementation and/or progression of SVD over time (Figure 1).

Host-related pathophysiological mechanisms associated with SVD: the interrelated roles of inflammation, lipid accumulation and oxidation, residual immunogenicity and subclinical leaflet thrombosis. Interrelated host-mediated processes have been associated with SVD: (i) the active fibro-calcific mechanisms mainly driven by immune cell recruitment (particularly macrophages and giant cells) and the associated inflammation and oxidative stress, and lipoprotein retention and oxidation; (ii) the residual immunogenicity of the animal-derived biomaterial with subsequent activation of the complement cascade due to the binding of human natural antibodies against the xenogeneic αGal and Neu5Gc carbohydrates; and (iii) the role of subclinical thrombosis favoring cell adhesion and activation of macrophages and platelets. Created in BioRender. Capoulade, R. (2025) https://BioRender.com/d44q524.
Importantly, all these findings highlight that the manufacturing processes and the resulting intrinsic structure of the biomaterial provide a favorable substrate for the development of SVD, especially by triggering dystrophic calcification in BHV leaflets. Advances in tissue engineering, and their industrial implementation as a routine workflow, will certainly provide, in the future, opportunities to mitigate these detrimental biomaterial-related properties and develop new BHV substitutes, presenting more favorable structural properties to counterbalance calcification propension and then significantly enhance their durability.
Active fibro-calcific mechanisms
Several clinical association studies and histological analyses of explanted BHV supported the implementation of active fibro-calcifying mechanisms in BHV tissue, mainly driven by (i) the recruitment of immune cells and associated inflammation and oxidative stress and (ii) the lipoprotein retention and oxidation (Figure 2).95–97,127,130,136–138 These processes have been well described in the context of native aortic valve calcification,79,80,100 supporting the hypothesis of the partial recapitulation of the fibro-calcifying processes observed in native aortic valves in BHV, in a more rapid fashion. Importantly, the faster development of this active fibro-calcific remodeling in BHV leaflets as compared to the native valve calcification could be explained, at least in part, by the potentiation of immune cell and lipoprotein accumulation in BHV, related to the intrinsic biophysical and biomechanical properties of the fixed collagen-rich tissue used to manufacture BHV leaflets (discussed in the previous section).
Relying on the (immuno)histological analysis of 18 explanted BHVs, Shetty et al reported a dense inflammatory infiltrate in BHV explanted for SVD, mostly composed by CD68+ macrophages which colocalized with oxidized LDL. 136 Recent data confirmed the accumulation of CD68+ macrophages in explanted BHV for SVD and their colocalization with apolipoprotein E in the calcified area.98,99,130,139 Neutrophils have also been observed in explanted BHV for SVD, an immune cell type involved in the innate immune response and favoring local inflammatory environment. 99 Interestingly, these data support the hypothesis of the implementation of a chain reaction process leading to SVD: infiltration of lipoproteins and their oxidation coupled with recruitment and activation of immune cells lead to the development of an inflammatory microenvironment via the release of pro-inflammatory cytokines/chemokines and reactive oxygen species which will further exacerbate immune cell recruitment (Figure 2). This vicious circle will potentiate the local accumulation of highly inflammatory cells (ie, canonical macrophages, foam cells, mononucleated macrophages with multiple cytoplasmic inclusions, and multinucleated giant cells),98,99,139 participating in the fibro-calcification of BHV (Figure 2).130,136 In the context of biomaterial implantation, the “foreign body reaction” process, 140 mediated by macrophages and giant cells, is typically described and could also be part of the process leading to SVD.141,142 Ultimately, apoptosis or necrosis of foam cells will further promote the recruitment and activation of immune cells, as well as the mechanisms of dystrophic calcification related to cell debris and apoptotic bodies, and then maintain (or exacerbate) the calcification of BHV (Figure 2). 130
Macrophages and foam cells are also known to release matrix metalloproteinases (MMPs), particularly MMP-9, involved in ECM degradation with preferential substrates described as Type-1 collagen, fibronectin, and elastin. Higher MMP activity has been reported in explanted BHV for SVD.136,139 This local release of MMPs by activated macrophages, coupled with MMPs from the circulation, contributes to the ECM degradation and calcification of BHV leaflets (Figure 2). These data highlight the central and interrelated roles of lipoprotein oxidation, leukocyte recruitment, ECM degradation, and calcification in SVD. 116
Analysis of explanted BHV by electron microscopy highlights the presence of active fibroblast-like cells at the end-stage SVD. 99 Pro-inflammatory microenvironment related to immune cell recruitment and activation has been described as a trigger for myofibroblast- and osteoblast-like cell differentiation in the native valve. 99 We can thus postulate that circulating progenitor cells and fibrocytes could infiltrate BHV biomaterial, as described for other cell types, and then undergo activation and differentiation into myofibroblast- and osteoblast-like cells, favoring the implementation of active fibrosis and calcification processes on BHV (Figure 2). To support this hypothesis, a recent study reports a significant association between the circulating progenitor cells expressing the osteocalcin pro-calcifying marker and early development of SVD. 143 Macrophages also secrete pro-osteogenic molecules, such as osteocalcin and osteonectin.144–146 These osteogenic factors will potentiate the osteoblastic differentiation of fibroblast-like cells and participate in the calcification of BHV biomaterial (Figure 2).
Immune cell mechano-transduction is an emerging field, with multiple pieces of evidence suggesting the role of the biophysical environment in immune cell recruitment and activation.131,147–149 Interestingly, macrophages express mechanosensitive ion channels, such as Piezo-type mechanosensitive ion channel component 1 (Piezo1) and transient receptor potential vanilloid 4 (TRPV4), as well as integrins, selectins, and cadherins, all modulating in vivo macrophages polarization and activation.131,147–149 As previously discussed, BHV leaflets face important and repetitive mechanical stresses, a phenomenon modified and exacerbated by the progression of SVD and ECM remodeling, as well as concomitant host comorbidities such as hypertension. The constant biomechanical stresses and their evolution over time postimplantation could then contribute to immune cell recruitment and activation in BHV, which will potentiate the activation of fibro-calcific processes in BHV.
Altogether, these findings highlight the central role of leukocytes, particularly macrophages, in the development and progression of SVD, via a feedforward loop between immune cell recruitment/activation and lipoprotein accumulation/oxidation, which will contribute to calcification of BHV. Interestingly, the subcutaneous implantation of BHV-derived biomaterial in an animal model depleted for macrophages has confirmed the reduction of BHV tissue calcification related to macrophage elimination, 150 thus reinforcing the central role of these cells in the pathophysiology of SVD.
Residual immunogenicity
The implantation of animal-derived biological tissue in humans suffers from important challenges, mainly related to cross-species immunologic barriers, such as the production of natural antibodies by humans targeting antigens specifically expressed by nonhuman mammals.151,152 In the context of the hyperacute rejection following xenotransplantation, two main xenogeneic antigens play a pivotal role: the carbohydrate a-galactose 1,3-galactose (αGal) and the N-glycolylneuraminic acid (Neu5Gc).151,152 Interestingly, the anti-αGal and anti-Neu5Gc antibodies are the most abundant circulating immunoglobulins in humans. 151
Despite continuous efforts to reduce the immunogenicity of the animal-derived biological tissue used to manufacture BHV, these tissues still display residual expression of αGal and Neu5Gc carbohydrates.111,153–158 High levels of anti-αGal and anti-Neu5Gc antibodies were also observed in explanted BHV for SVD, coupled with the activation of the complement cascade, suggesting active immune response following BHV implantation mediated by xenogeneic antigens.111,154,158,159 Several animal studies using α-Gal knock-out porcine pericardium have reinforced these findings: the use of this α-Gal knock-out tissue was associated with a lower propension for calcification over time.160–163 These experimental data support that xenogeneic antigens present in BHV are recognized by the human immune system and could then act as a trigger for active pathophysiological mechanisms leading to SVD (Figure 2). It is also important to note that other potentially unexplored xenogeneic antigens could also participate in SVD, and further studies are needed to comprehensively characterize BHV immunogenicity. 164
From a clinical standpoint, Senage et al have provided the first clinical evidence of this xenogeneic antigen-mediated response post-BHV implantation: in this prospective and multicenter study, patients were enrolled at the time of surgery and those who received BHV, as compared to controls who underwent cardiac surgery without implantation of biological material, had a significant increase in circulating levels of anti-αGal and anti-Neu5Gc antibodies over 6 months post-intervention. 158 This acute elevation of xenogeneic antibodies was also observed in a relatively small cohort of patients who underwent transcatheter BHV implantation. 165 Interestingly, some studies looking at the αGal syndrome, a rare allergic disorder to αGal due to tick bites or red meat consumption, reinforced these findings: hyperimmunized patients diagnosed with αGal syndrome who underwent BHV implantation presented an early and acute development of severe SVD.166–170 Overall, these findings suggest that immunological response to the animal-derived biomaterial and its residual expression of xenogeneic antigens is a central contributor of SVD (Figure 2).
The underlying pathophysiological mechanisms related to the residual immunogenicity observed in BHV involve the activation of the complement cascade, which in turn results in the activation of the innate immune cells, and particularly macrophage chemotaxis, polarization, and phagocytosis (Figure 2).171,172 This recruitment and activation of immune cells, including macrophages and foreign giant cells, will in turn potentiate the inflammatory microenvironment of BHV and actively participate in the ECM remodeling (as previously discussed). Interestingly, the complement activation has also been linked to pro-thrombotic events mediated by platelets and coagulation cascade activation, initiating a vicious circle between complement cascade, immune cell recruitment, and (subclinical) thrombotic events (Figure 2).172–174
Subclinical leaflet thrombosis
SLT, reported in up to 30% of the patients 1-year post-surgery, is characterized as hypo-attenuated leaflet thickening (HALT), reduced leaflet motion, and hypo-attenuation affecting motion, without significant hemodynamic changes or associated symptoms, and ultimately may spontaneously resolve without treatment.18,40,42,98,175,176 However, the SLT has recently been associated with a higher incidence of SVD, supporting a potential mechanistic link between fibrin deposit and the implementation of pathophysiological mechanisms associated with SVD.41,175
Morphological analysis of explanted transcatheter BHV using histology, immunohistology, and electron microscopy provided insights into the thrombogenic responses postimplantation of BHV: by analyzing BHV explanted over a large period of time (ie, from few days postintervention up to 7 years), Sellers et al firstly reported the presence of leaflet thrombus, described as the formation of fibrine deposit, in all explanted BHVs, suggesting that this phenomenon could have much more importance post-BHV implantation with regards to the implementation of pathophysiological mechanisms leading SVD (Figure 2). 98 This study also underlines, even if the direct link cannot be formally proven, the sequential implementation of events leading to SVD, including thrombus formation, fibrosis development, and then calcification of the BHV leaflets. 98 The role of platelet activation, which is potentiated by the thrombus formation at the surface of the BHV leaflets and also mediated by the von Willebrand factor, has also been reported. 176
Interestingly, fibrin deposit has been described to promote monocyte adhesion and macrophage activation, which could potentiate the pathophysiological processes leading to SVD.177,178 A recent mechanistic study by Sakaue et al highlights the role of fibrinogen as a key mediator of macrophage activation in the context of SVD. 130 These findings have been reinforced by the recently published animal study demonstrating a link between fibrin deposits on top of bovine pericardial tissue and calcification of the BHV tissue (Figure 2). 179
Overall, these findings point out a role of biological leaflet properties, and especially the impact of bioprosthetic matrix remodeling and calcification coupled with subclinical thrombosis, on SVD, that could be, at least in part, mediated by monocyte/macrophage adhesion and activation. A recent clinical study has highlighted an association between Lp(a) plasma levels (combined with high C-reactive protein levels) and the risk of developing HALT in a cohort of patients receiving transcatheter BHV. 180 Even if this study will need further validation, these data support a strong interrelationship between all pathophysiological mechanisms involved in the development and progression of SVD and the need for a better understanding of their interactions and the kinetic of their implementation.
Conclusion
With the exponential rise in the use of BHV, the occurrence of SVD and its socioeconomic burden will constantly increase in the coming years, highlighting the urgent need to improve BHV durability. The understanding of the pathophysiological mechanisms involved in this process is the cornerstone to achieve this objective. Recent advances in the field have provided first insights regarding the complex and multifactorial mechanisms involved in the development and progression of SVD, with a strong interrelationship between the manufacturing process of the biomaterial and the host-related factors. A continuous effort should be made to better dissect the interaction between all these mechanisms and the kinetic of their implementation: these valuable and necessary data will provide the mechanistic basis to develop innovative approaches in terms of tissue engineering and/or identify preventive treatments to mitigate SVD development and progression and, in turn, significantly improve BHV durability.
Footnotes
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MS holds a PhD scholarship grant from the ‘Fondation pour la Recherche Médicale’ (PBR program—Prix Beziat-Rouanet; PBR202206016091). RC is supported by a “Connect Talent” research chair from Région Pays de la Loire and Nantes Métropole, and by research grants from the “Agence Nationale de la Recherche” (ANR-23-CE17-0007-01) and from the “Fondation de France” (grant no. 00133548 / WB-2022-45051).
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
