Abstract
Tricuspid regurgitation (TR) has shifted from being considered a benign condition to a clinically relevant entity with a potentially poor prognosis, leading to increasing diagnostic and therapeutic interest, particularly following the development of new transcatheter intervention techniques. Imaging is fundamental in the evaluation of valvular heart disease, and in the case of TR, cardiac magnetic resonance (CMR) and cardiac computed tomography (CCT) have emerged as essential and complementary tools to echocardiography. Although specific thresholds for intervention have not yet been clearly defined, integrating functional and anatomical parameters through CMR and CCT may help determine the optimal timing for treating patients with severe TR, thereby avoiding progression to irreversible right ventricular remodeling and the onset of heart failure, which significantly impacts prognosis. CMR enables accurate assessment of right ventricular volumes and function, as well as quantification of regurgitation severity. Moreover, advanced CMR techniques such as 4D-flow and myocardial strain analysis using feature tracking provide additional prognostic information. On the other hand, CCT, thanks to its high spatial and temporal resolution, allows detailed visualization of the tricuspid valve anatomy, playing a crucial role in patient selection and procedural planning for percutaneous interventions, as well as in follow-up evaluation.
This is a visual representation of the abstract.
Keywords
Key Points
Cardiac magnetic resonance (CMR) and cardiac computed tomography (CCT) play complementary roles in the evaluation of patients with tricuspid regurgitation undergoing transcatheter valve interventions.
CMR is the reference standard for right ventricular volume and function assessment, which are critical parameters in determining timing and prognosis of tricuspid valve intervention.
CCT offers superior spatial resolution and is the preferred modality for anatomical assessment, device sizing, and procedural planning in transcatheter therapies.
The integration of multimodality imaging enhances patient selection, procedural success, and early detection of complications after transcatheter tricuspid interventions.
Further studies, including randomized controlled trials, are needed to establish evidence-based imaging thresholds and define the prognostic impact of CMR and CCT in this clinical setting.
Introduction
Despite being considered a benign disease for many years, tricuspid regurgitation (TR) has been gaining interest in recent years due to the progressive recognition of its association with poor outcomes and the increasing availability of transcatheter therapies that can safely treat TR, particularly interesting considering that TR patients are usually elderly, frail, and carry significant comorbidities. Imaging techniques are essential in diagnosing and guiding treatment in all valvular heart disease, and particularly in the field of transcatheter therapies, have become crucial to appropriately select candidates and to ensure adequate guiding of procedures.
Although echocardiography is currently the first-line imaging modality for detecting tricuspid valve (TV) disease and estimating its severity and hemodynamic impact, other imaging modalities such as cardiac computed tomography (CCT) and cardiac magnetic resonance (CMR) can provide complementary information on the detailed anatomy and the functional impact of TV disease. Specifically, CMR is useful for evaluating the severity of TR and for assessing right ventricular (RV) function and remodeling. Conversely, CCT plays a key role in procedural planning, as well as in follow-up imaging, and is gaining importance in the assessment of RV function. Both imaging modalities are essential in understanding and characterizing the precise anatomy and 3-dimensional (3D) geometry of the TV, which plays a key role in planning and selecting candidates for percutaneous transcatheter interventions. Potentially, these techniques could also provide improved quantification of TV dysfunction with further technical developments and clinical evidence.
This review aims to provide an overview of the current role of CMR and CCT in the assessment of patients with TR eligible for valve interventions.
Quantification of TR With CMR and CCT
CMR is the gold standard noninvasive imaging technique for cardiac volumetric assessment, and particularly for the right cavities of the heart, due to its ability to accurately evaluate both the morphological and the functional changes of the heart.1,2 Moreover, CMR has emerged as a good imaging technique for the diagnosis and evaluation of TR and its impact on the right heart performance, and consequently helping in the decision-making regarding TR intervention.1,2
Quantification of TR by CMR is typically indirectly assessed with the RV stroke volume (calculated in the short-axis cine) and the forward pulmonary flow volume (assessed by phase-contrast imaging), values that, in the absence of valvular heart disease, should be similar. However, when TR is present, the regurgitant volume can be estimated as the difference between RV stroke volume and pulmonary forward stroke volume. The TR regurgitant fraction, therefore, can be calculated as (TR volume/RV stroke volume) × 100%. It is important to emphasize that this approach is inherently indirect and may be influenced by several technical and physiological factors. Specifically, CMR-based TR quantification may be affected by arrhythmias, the presence of pacing leads, and challenges in accurately delineating RV borders. Even if TR regurgitant fraction ≥ 40% is considered to be hemodynamically significant, 3 cutoffs to determine the TR severity using CMR have not yet been established. In addition to phase-contrast imaging, advanced 4D-flow MRI sequences have shown promise for the direct quantification of regurgitant volume (Figure 1). However, recent studies have demonstrated discrepancies between transthoracic echocardiography and 4D-flow in the grading of TR. 4 These inconsistencies underscore the need for further technical refinements and validation studies to improve reliability and support broader clinical adoption.
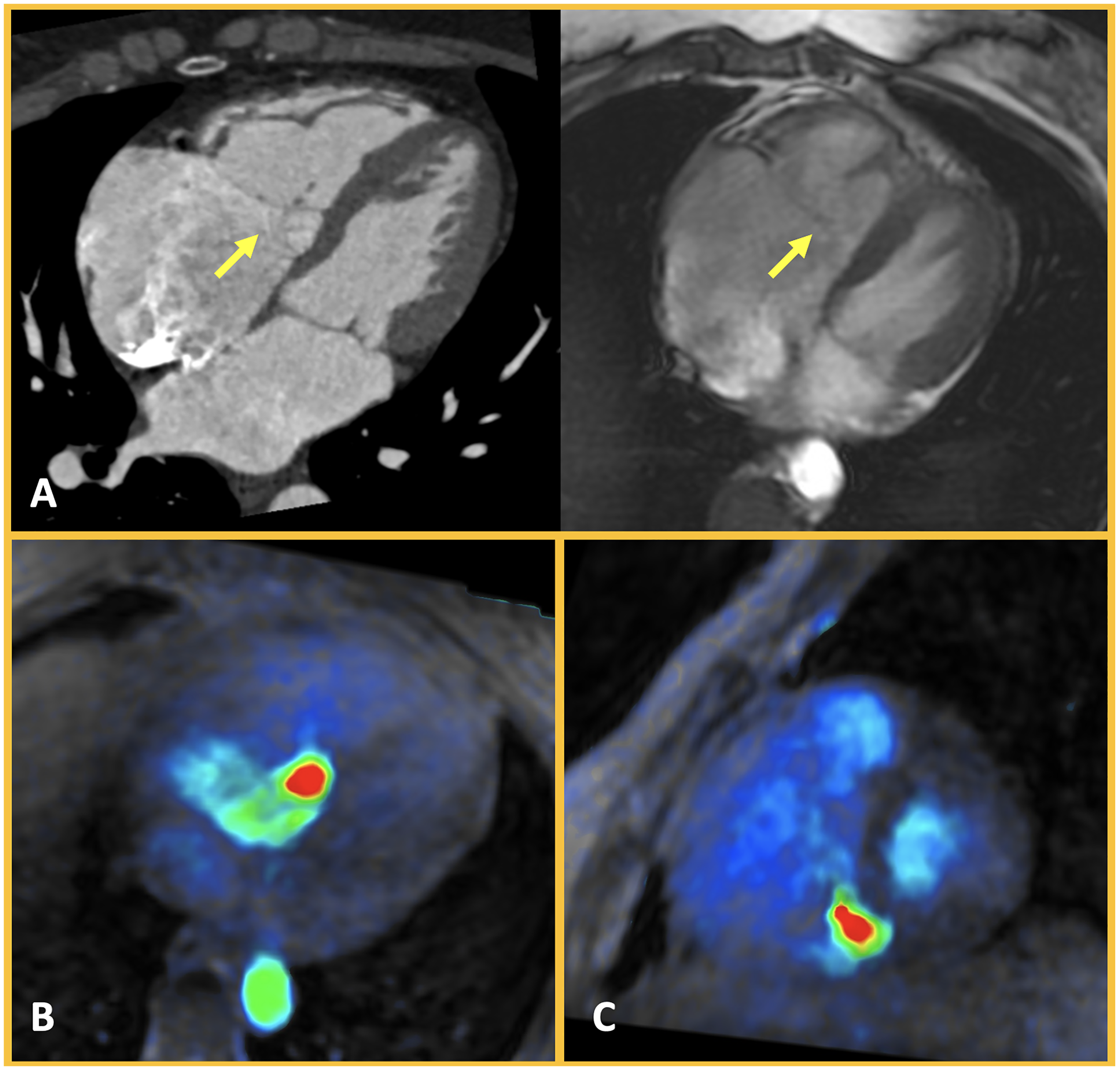
Analysis of Tricuspid Regurgitation Severity. Patient with Ebstein anomaly leading to severe tricuspid regurgitation. Cardiac CT, with its excellent temporal and spatial resolution, allows accurate assessment of the anatomy of the various components of the valvular apparatus, as well as the mobility of the valvular leaflets (panel A, arrow). Cardiac MRI also enables anatomical and functional evaluation of the tricuspid valve (panel B), additionally allowing for the analysis of the severity of the valvulopathy using 4D flow sequence (panels B and C).
CCT has higher spatial resolution than CMR, and it is playing an increasingly important role in the assessment of the TV morphology and becoming essential for the planning of transcatheter procedures. TR quantification by CCT has not been fully validated, but it might be useful in those patients with old defibrillators or pacemakers that are not CMR compatible. CCT cannot directly quantify TR, but it can indirectly assess TV regurgitant volume and TR severity by calculating the difference between the right and left stroke volume, in the absence of other significant valvular heart disease or associated shunts. Moreover, CCT has proven useful for directly measuring the regurgitant orifice in mid-end-systole (defined as the area of the smallest orifice at the level of the tip of the leaflets), and this might work as an additional tool to assess TR severity. 3
Currently, there is a lack of prospective data defining imaging thresholds for TR severity assessed by CMR or CCT. This limitation poses challenges in guiding patient selection for intervention. Acknowledging this gap highlights the need for future studies to establish standardized criteria.
Evaluation of RV and RA Remodeling: Indication for Intervention
The natural history and underlying pathophysiology of TR and its relationship to morphological and functional changes in the right heart cavities is less understood as compared to mitral or aortic valve disease. The degree of RV and the right atrium (RA) remodeling secondary to the volume overload induced by TR might lead to worsening TR, resulting in a vicious cycle. Despite specific cut-offs are not well established for patients with TR, RA, and particularly RV remodeling and function are currently the main imaging biomarkers. These parameters can help in making decisions about whether an intervention is necessary in patients with severe TR in whom symptoms are usually mild until very advanced stages of the disease, where multiorgan damage has already been established and interventions might have less benefit or even might be futile.5,6
The RV morphology and function can be assessed by CMR with conventional balanced steady-state free-precession cine sequences in a short-axis view that covers both ventricles. Biventricular function analysis involves semi-automatic contouring of the inner and outer boundaries (epicardial and endocardial) of the left ventricle and the endocardial boundaries of the RV, at both end-diastole and end-systole. This enables the calculation of the RV end-diastolic volume (EDV), end-systolic volume (ESV), and ejection fraction (EF). Additionally, conventional balanced steady-state free-precession cine sequences in a short-axis view covering both atria or in a 4-chamber view allows assessment of the RA volume and RA area, respectively. 7
Overt RV dysfunction in patients with TR is associated with poor prognosis and increased operative risk.6,8 Indeed, a study by Park et al 9 demonstrated that impaired RVEF and increased indexed RV-ESV were associated with more postoperative events and cardiovascular death in patients with severe TR undergoing TV surgery. In another recent study that included a cohort of 184 patients with severe TR, RV-EDV was shown to be a better predictor of death or surgery at 2-year follow-up for a threshold of 100 mL/m2 of body size area, as compared to RVEF for a threshold of 46%. 10 However, since the analysis of RV function is a time-consuming procedure, techniques based on artificial intelligence for assessing ventricular function using both CT and MRI are currently under development. Kirchner et al 11 recently demonstrated in a retrospective study involving 100 patients who were candidates for percutaneous valvular intervention on the TV that RV function analysis using CT (based on the measurement of RV EDV and EF) was a predictor of risk of death or hospitalization following such procedures. Similarly, Gonzales et al 12 showed that the assessment of RV function using tricuspid annular plane systolic excursion (TAPSE) and peak systolic velocity in CMR (in 2- and 4-chamber views), based on residual neural networks, was reproducible compared to manual measurement, with reduced analysis time.
Regional contractility might also be assessed by CMR with feature tracking (FT), which measures myocardial deformation, thus recognizing and characterizing subclinical and earliest evidence of ventricular systolic dysfunction. FT can be performed offline using the long-axis cine 4-chamber view, where RV longitudinal strain can be assessed regionally or globally as an average of the 3 RV-free wall segments. When considering RV-FT values, caution should be taken if compared with strain values assessed by echocardiography, since the agreement for the RV between these 2 techniques has been found to be weak. 13 However, an RV-free wall longitudinal strain ≥ −16% by FT has been recently proved in a multicentric study as an independent predictor of mortality in patients with severe functional TR, incremental to common clinical and other imaging risk factors. 14 Another recent study by Kresoja et al 15 has shown that survival after transcatheter TV repair (edge-to-edge) could be predicted by a worse RV contraction pattern, with RVEF < 45% and diminished global longitudinal and circumferential RV strain.
CMR is a unique, noninvasive technique that enables tissue characterization of the RV with late gadolinium enhancement (LGE) sequences, which can be challenging due to the small thickness of the RV wall. Myocardial fibrosis may develop in patients with TR as a result of volume overload from regurgitation, often appearing as focal enhancement at the RV anterior or inferior insertion points, similar to findings in pulmonary hypertension. 16 However, its prognostic significance in this specific patient group remains unclear. Native T1 mapping and extracellular volume (ECV) calculation 17 at the RV insertion points might also provide valuable insights into the presence of interstitial fibrosis in these areas. Indeed, while diffuse interstitial fibrosis has been well correlated with adverse RV remodeling in 2 pulmonary hypertension cohorts,18,19 data on its role in TR patients remain limited.
One of the primary challenges for patients with TR is the delayed decision to treat, often occurring too late, which leads to suboptimal or even futile interventional outcomes (either with surgery or transcatheter therapies). However, integrating comprehensive CMR data with the patient's symptoms can help identify the optimal timing for intervention. This approach is crucial in preventing irreversible RV remodeling or cardiac failure, which can significantly increase surgical risk.20,21 Current clinical practice guidelines recommend TV intervention at the time of left-sided valve surgery in patients with severe TR or tricuspid annular dilation. Isolated intervention is recommended in patients with severe TR when accompanied by symptoms or RV dilation, and only after ruling out severe pulmonary hypertension or significant RV dysfunction in cases of secondary TR. 22 While the utility of CMR is unquestioned, the exact thresholds for intervention have yet to be clearly defined.
Assessment of the Anatomy of the TV
The TV is a complex apparatus consisting of leaflets (classically identified as 3: anterior, posterior, and septal), chordae tendinae, 3 discrete papillary muscles (septal, posterior, and anterior), and the fibrous tricuspid annulus, being its valve orifice and annulus larger than the mitral ones. The TV and tricuspid annulus are dynamic structures, with conformational changes observed throughout the cardiac cycle that adapt to changes in loading conditions. 23
CMR might be of help to analyze the anatomy of TV, although it can be challenging given the thin nature of the leaflets and through-plane motion from the tricuspid annulus. Instead, retrospective ECG-gated CCT allows for a better understanding of the 3D morphology of the annulus and its dynamics during the cardiac cycle, the anatomy of the other components of the TV apparatus, and the mechanism causing the valve dysfunction, due to its excellent spatial and temporal resolution. Retrospective acquisition is of great importance since it enables subsequent multiplanar reconstructions at an adequate time point of the cardiac cycle.
In patients with primary TR, CCT might be particularly of help to analyze the anatomy and motility of the leaflets as well as the involvement of the subvalvular apparatus. In patients with secondary TR, CCT may be useful to assess the annular dimensions and valvular tenting (Figure 2). In those patients in whom TR may be related to the presence of leads from implantable cardiac devices such as pacemakers or defibrillators, CCT is useful to assess whether there is an interaction between the lead and the leaflets and its location (Figure 3). This is particularly interesting as some older devices are not CMR conditional, and therefore not safe for scanning even at lower magnetic field strength (1.5 T),
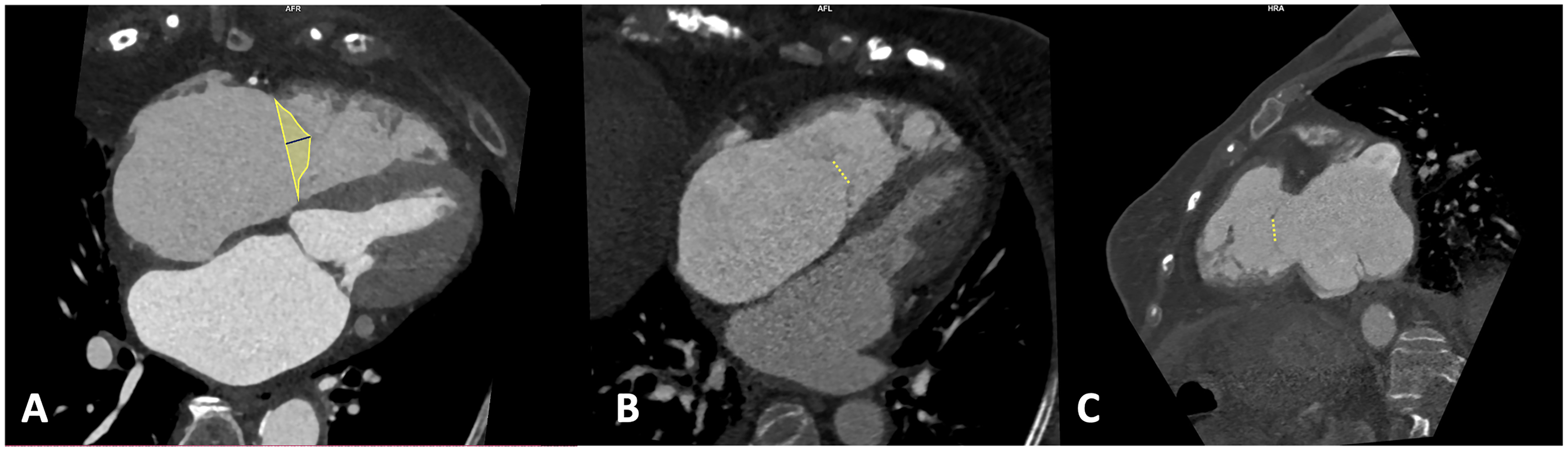
Tenting Height, Area, and Coaptation Defect Measurements in Tricuspid Regurgitation. The tenting height and area, defined as the space between the valvular leaflets and the annular plane in a 4-chamber mesosystolic view (panel A), provide additional insights into the severity and mechanism of tricuspid regurgitation (TR). Additionally, the analysis of the morphology and dimensions of the coaptation defect during meso-telesystole offers further information regarding the mechanism and severity of TR. A patient with severe tricuspid regurgitation presents a large coaptation defect in both the 4-chamber and 2-chamber views of the right ventricle (panels B and C).
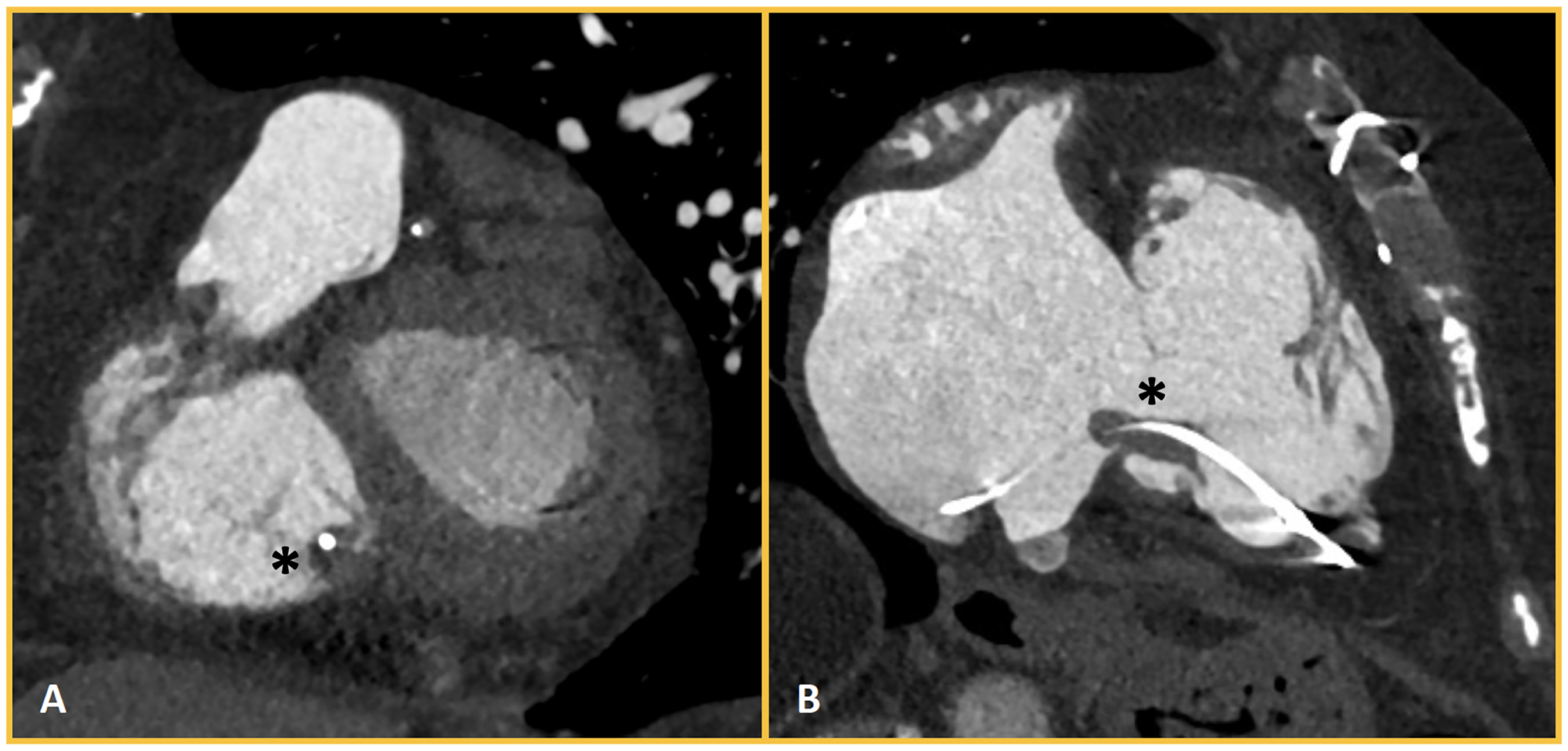
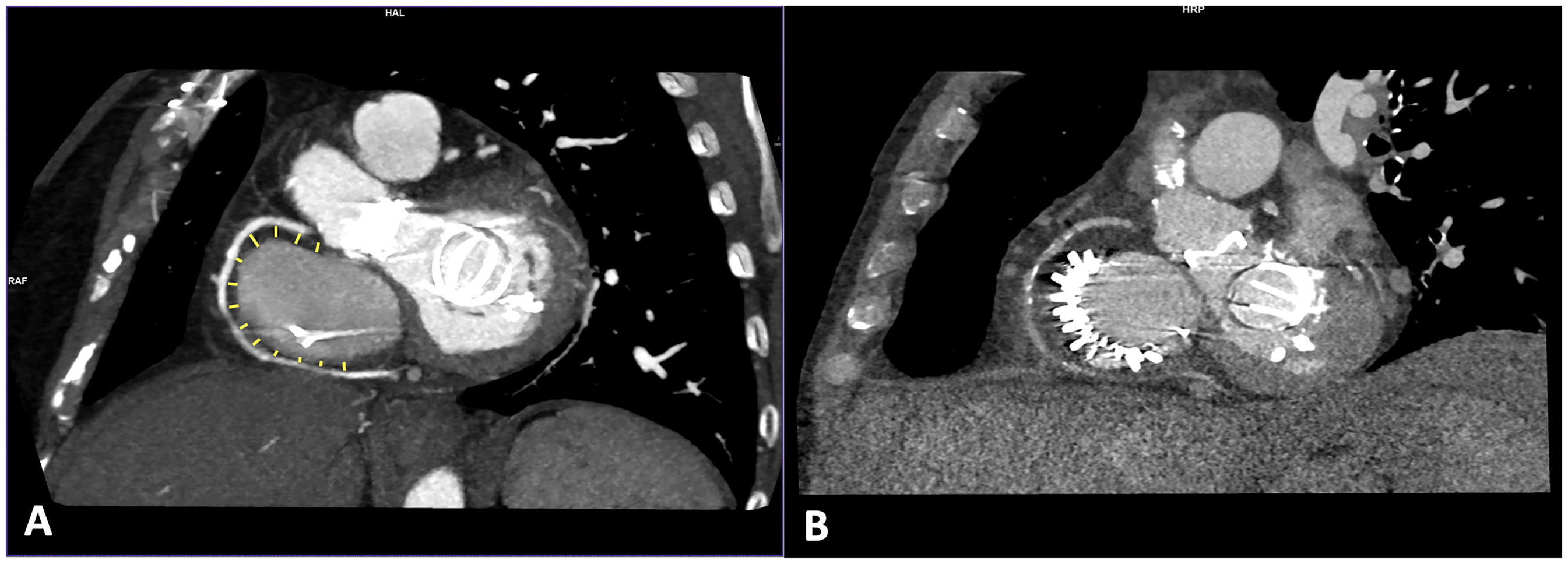
Lead-Related Tricuspid Regurgitation. Patient with permanent atrial fibrillation and a pacemaker implanted 12 years prior, presenting with severe tricuspid regurgitation. The short-axis (panel A) and 2-chamber (panel B) views of the right ventricle show the interaction between the septal leaflet and the pacemaker lead. The lead appears thickened as a result of fibrous encapsulation and fibrin deposits (*), which restrict the mobility of the septal leaflet.
Multiplanar 4-chamber and 2-chamber view reconstructions from CCT acquisitions offer a short-axis plane through the tricuspid annulus. In this plane, the annulus dimensions (major and minor diameters, area, and perimeter) can be determined with excellent inter- and intra-observer reproducibility 24 (Figure 4). Due to its dynamic nature, shape and size changes of the TV annulus during the cardiac cycle occur up to 30%, thus annular measurements of both systole and diastole are recommended to plan any TV procedure.
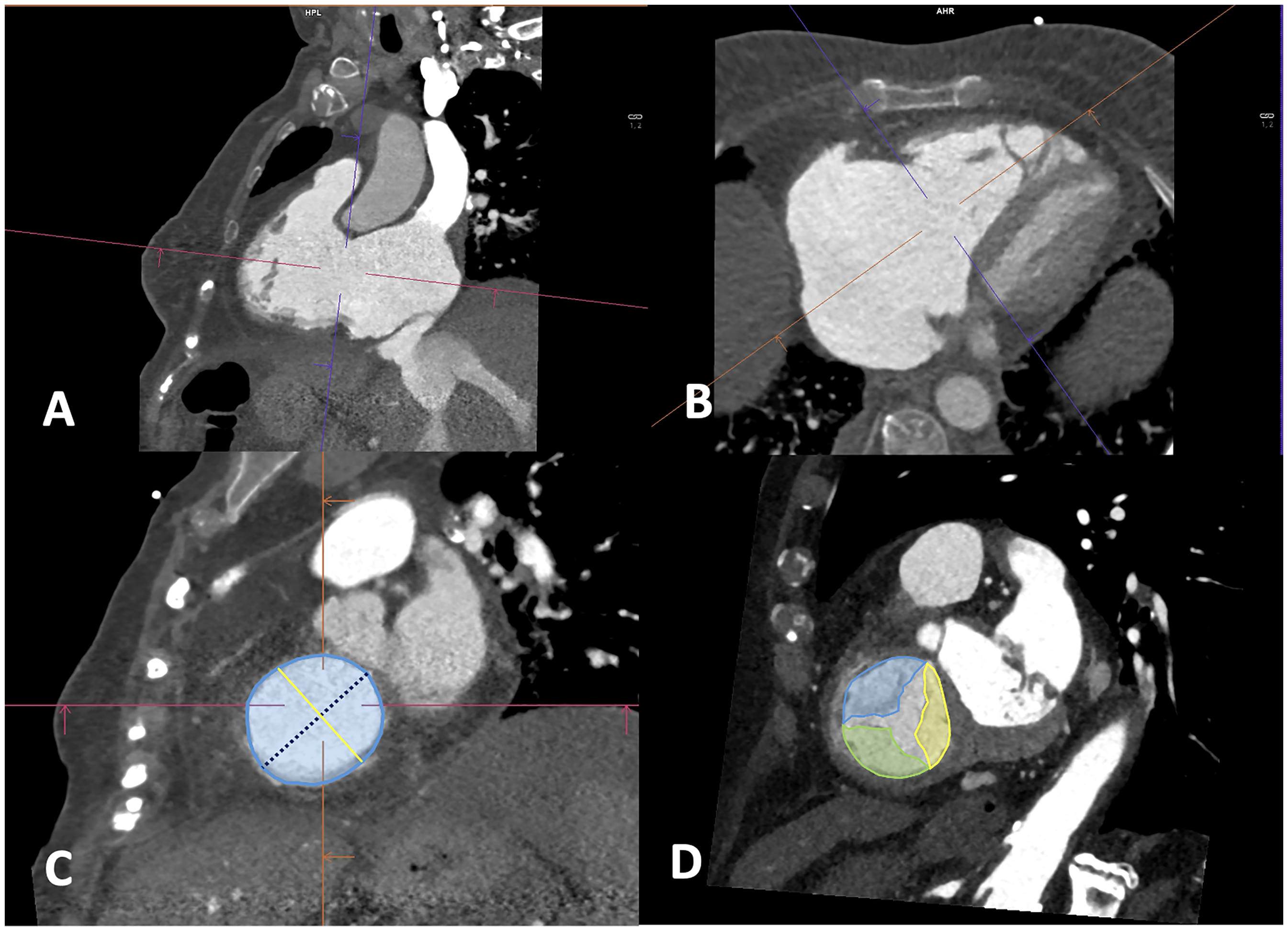
Anatomical Assessment of the Tricuspid Ring and Leaflets. Cardiac computed tomography (CT) allows for an accurate assessment of valvular anatomy. Through multiplanar reconstructions from the 4-chamber and 2-chamber views (panels A and B), the valvular ring plane can be reconstructed. Measuring various parameters such as perimeter, area, and both major and minor diameters can be useful for the appropriate selection of the device (panel C). Additionally, from the short-axis view of the valvular ring with apical displacement, simultaneous visualization of all 3 valvular leaflets is possible (panel D).
Planning for Transcatheter Interventions: Repair and Replacement Techniques
Current clinical practice guidelines recommend TV surgery in various clinical scenarios, depending on whether it is an isolated valve disease or associated with left valve disease. 22 However, due to the aging population and the increased surgical risk that this entails,25,26 there is a significant number of patients who are not candidates for valve surgery, leading to a growing development of percutaneous valve intervention techniques in recent years. 27
Types of Devices
In the field of percutaneous tricuspid intervention, there are currently various devices, both in clinical use and still limited to research purposes. Depending on the mechanism by which these devices correct the valve disease, they can be classified as follows:
Edge-to-edge repair devices (Triclip© Abbott, Pascal© Edwards Lifesciences, DragonFly© Valgen Medical): These are the most commonly used in clinical practice. They reduce the regurgitant volume by increasing the coaptation surface of the valve leaflets.
28
Direct annuloplasty devices (Cardioband© Edwards Lifesciences): They reduce the dimensions of the annulus by anchoring directly to it, and thus improving the coaptation of the leaflets.
29
Percutaneously implanted valve bioprostheses: Depending on the potential implantation site, they are further classified into orthotopic and heterotopic prostheses. Orthotopic prostheses: prostheses implanted in the tricuspid position. There is currently a commercially available prosthesis
30
(Evoque© Edwards Lifesciences) and various models in advanced research phases
31
(such as Cardiovalve© Cardiovalve or LuX-Valve© Jenscare). Also, transcatheter bioprosthesis can be implanted in a bioprosthesis (valve-in-valve). Valve-in-ring in the tricuspid position is rarely performed, as most surgical TV annuloplasties are performed with an open ring, which does not provide enough support to the transcatheter prosthesis. Heterotopic prostheses: Prostheses that are implanted in a nontricuspid position, generally in the inferior vena cava or both caval veins,32,33 reducing systemic congestion despite not reducing the regurgitant volume, on the RA (TricValve© Products + Features).
Given the anatomical complexity of the TV 34 and the procedures of percutaneous intervention, imaging techniques, primarily echocardiography 35 and CCT, are both complementary and essential imaging modalities for therapeutic strategy selection, and for assessment of complications during the follow-up.4,36
Preprocedure Study
Anatomy of the TV and Sizing
The main role of CCT in planning or evaluating a potential candidate for a TV procedure is the evaluation of the size of the annulus, which is essential for the sizing of transcatheter devices (either annuloplasty or bioprostheses, including sizing of the caval veins for heterotopic transcatheter prosthesis).37,38
Relation to Nearby Structures
Due to the specific double chamber geometry of the RV with inlet and outlet in 2 different directions, RV outflow obstruction is not a typical complication limiting transcatheter prosthesis implant, as it is often the case for the mitral valve.
In the context of transcatheter procedures on the TV, the most important anatomical relationship is the right atrioventricular groove and the right coronary artery, since, given its proximity, the coronary artery could be damaged by the implantation of orthotopic prostheses and by direct annuloplasty devices. The distance between the annulus and the right coronary artery, both in systole and diastole, can be precisely measured by CCT.
Procedure Planning: Fluoroscopy Angles
Fluoroscopy angles can be estimated prior to the procedure, which offers several advantages: reducing the duration of the procedure, minimizing the volume of iodinated contrast used, lowering the risk of contrast-induced nephropathy, decreasing the total radiation dose for both the patient and healthcare personnel, and potentially minimizing the risk of device malposition.
Similar to percutaneous intervention for left heart valves, a nomenclature system in fluoroscopy has been developed that directly correlates with echocardiographic planes and can initially be inferred from CCT. 39 Thus, short-axis, 2-chamber, 4-chamber, and bi-caval views can be obtained, all based on the combination of right versus left oblique angulation and cranial versus caudal angulation. This has allowed for the development of a common language between both modalities. In addition, it is also possible to simulate the intracardiac trajectory of the catheter as well as estimate the angle between the inferior vena cava and the apex of the right ventricle.
Specific Parameters Based on Specific Procedures and Devices
In addition to the general assessment of valvular anatomy, mechanism, and severity of valve dysfunction, CCT is useful to evaluate the anatomical suitability of a given therapeutic strategy and the optimal transcatheter technique/device according to the particular anatomy of each patient.40,41 Depending on the therapeutic strategy, there are mandatory measurements and others that, while not compulsory, provide useful information.
Edge-to-edge repair devices: These devices are currently the most commonly used transcatheter devices, and CCT or CMR are often not performed as the main anatomical limitation for this therapy is leaflet length and mobility, which are usually well visualized by transesophageal echocardiography.
42
CCT can be particularly useful to evaluate the relationship of the leaflets with leads and to evaluate their interference. More rarely, CCT can be used to depict the extent of calcification into the leaflet in some very unusual cases of tricuspid calcification. Annuloplasty devices: These devices are useful for patients with annular dilation, functioning similarly to surgical annuloplasty by reducing the dimensions of the annulus and thereby increasing leaflet coaptation. The Cardioband© device is a partial annuloplasty device consisting of multiple anchors to the native valve annulus, requiring an analysis of annular morphology and its anatomical relationships.
43
Both the distance between the annulus and the right coronary artery and the quality of tissue present between both structures are important to reduce the risk of coronary artery injury, with a gap of < 2 mm and the presence of annular calcification being significant risk factors for the procedure
40
(Figure 5). Orthotopic prostheses on native or on prior ring/bioprosthetic TV: CCT is an essential tool for the sizing of the new prosthesis by measuring the annulus and estimating the trajectory from the origin of the caval veins.
44
Heterotopic prostheses: These prostheses are implanted in both caval veins, and CCT becomes essential to size the device by measuring the size of both caval veins, typically at different levels (Figure 6) in order to select the most appropriate size and also to avoid obstruction of the suprahepatic veins, the appearance of periprosthetic leak, or device migration.
45
Cardiac CT in Planning Direct Anuloplasty Procedures. In the preprocedural evaluation of patients who are candidates for percutaneous annuloplasty, it is crucial to measure the distance between the RCA and the tricuspid annulus at every anchoring point of the cardioband device (panel A). After implanting the device, a reduction in the size of the annulus is achieved (panel B), which improves the coaptation area of the leaflets and subsequently reduces the regurgitant volume.
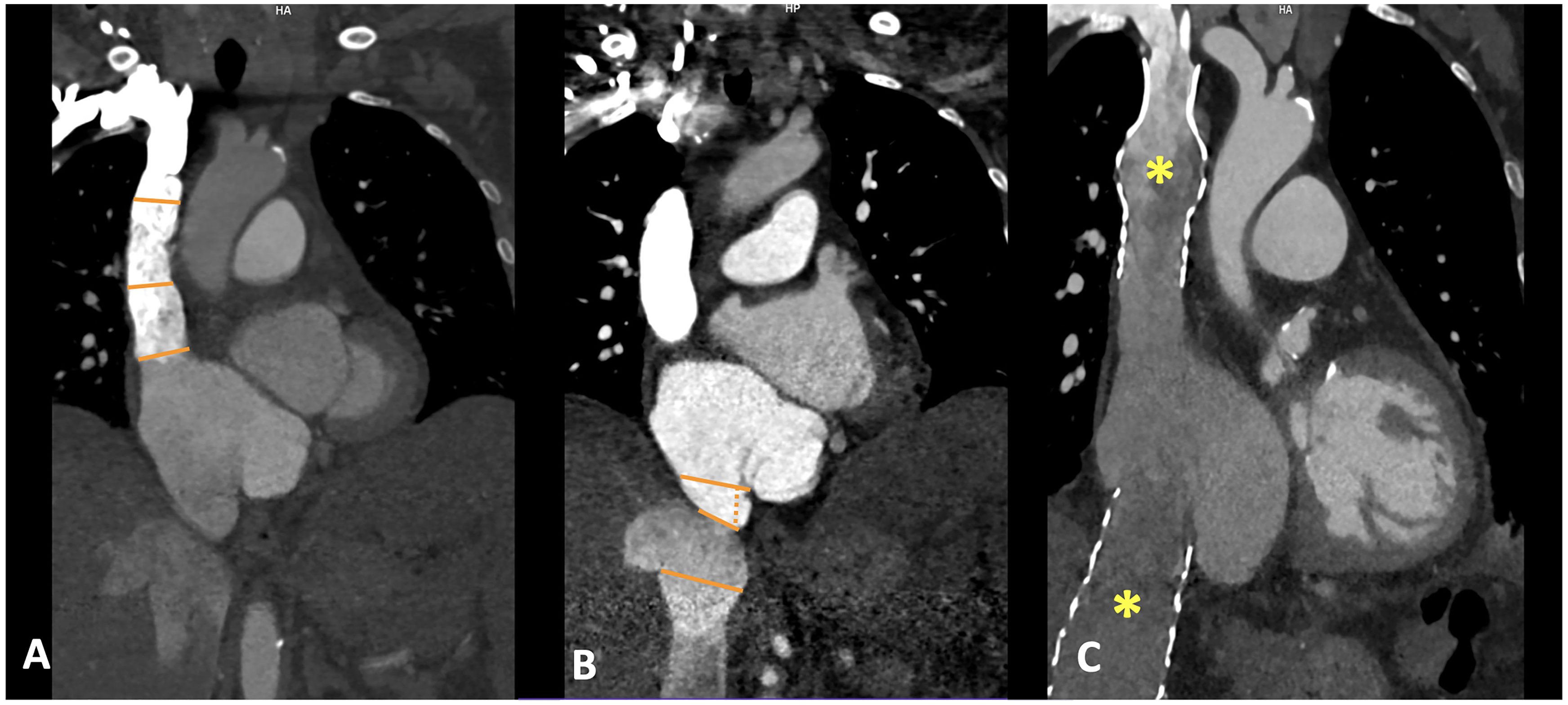
Cardiac CT in the Implantation of Heterotopic Prostheses. Accurate selection of the prosthetic size is essential to reduce the risk of complications, requiring the measurement of the diameters of the SVC (panel A) and IVC (panel B) at various levels. Postimplantation, the bicaval prosthesis can be seen in a properly aligned position (*), extending longitudinally along the distal segments of these vessels (panel C).
Utility of CCT and CMR in Follow-Up of Patients Undergoing TV Transcatheter Procedures
While echocardiography remains the first-line imaging technique for follow-up, the presence of prosthetic material that generates acoustic shadowing can complicate the assessment of procedural success. Therefore, other imaging techniques, primarily CCT, can be useful for detecting possible complications. Thus, in cases where there are elevated prosthetic gradients or abnormal flows on echocardiography without being able to determine the cause, CCT can provide adequate diagnosis of complications such as thrombosis, pannus formation, prosthetic endocarditis, and perivalvular leak.
Although the evidence available in the field of tricuspid intervention is limited, it is possible to extrapolate data from percutaneous prostheses in the aortic position. Therefore, in cases of suspected prosthetic stenosis, CCT allows for to study of the presence of low attenuation thickening of the leaflets (hypoattenuated leaflet thickening, HALT, defined as an increase in leaflet thickness presenting a typical meniscus appearance) and restrictive leaflet motion (RLM) by analyzing leaflet mobility throughout the cardiac cycle. 46 Additionally, the presence of prosthetic dysfunction due to pannus formation can also be adequately assessed by cardiac CT, with studies showing that pannus has higher attenuation values (with a cutoff > 145 HU) as compared to thrombus, 47 potentially allowing differentiation from prosthetic thrombosis and thus adding diagnostic value and treatment guidance.
Conclusion
In conclusion, CMR and CCT have become essential imaging modalities in the evaluation of patients with TR eligible for valve intervention, each providing greater utility at different steps of the diagnostic and therapeutic process (Table 1 and Figure 7). While CMR provides an accurate assessment of RV volumes and function, CCT depicts a detailed assessment of the TV anatomy. Randomized controlled trials are needed to better define the prognostic impact of integrating CMR and CCT into the management of patients with TR, particularly regarding the optimal timing of intervention based on RV assessment and TR quantification.
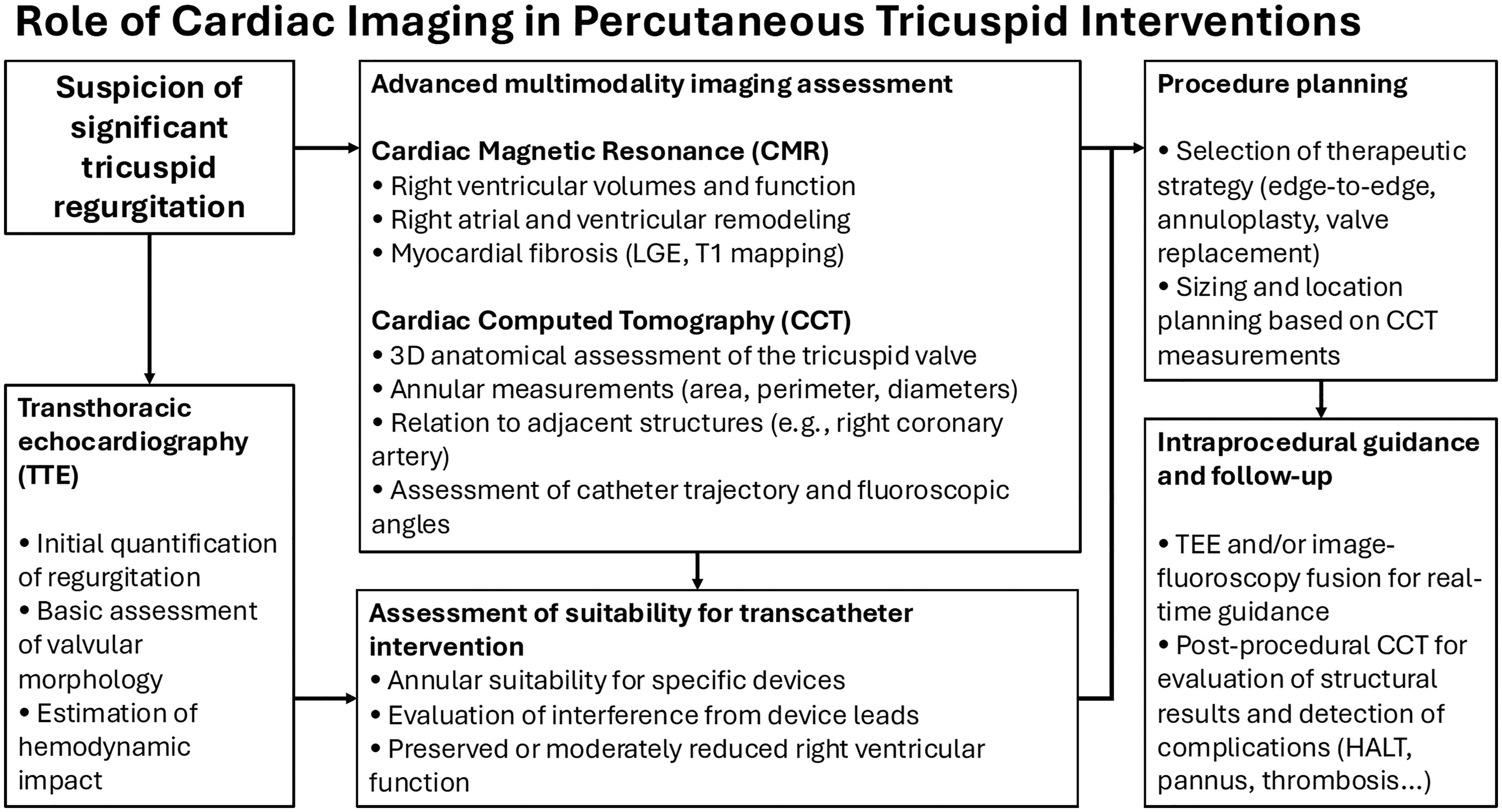
Stepwise Integration of TTE, CMR, and CCT Enables Diagnostic Assessment, Procedural Planning, and Post–Intervention Follow–Up in Patients Undergoing Transcatheter Treatment for Tricuspid Regurgitation.
Comparative Overview of CCT and CMR in the Evaluation of TR.

Abbreviations: CCT, cardiac computed tomography; CMR, cardiac magnetic resonance; TR, tricuspid regurgitation; 4D, 4-dimensional; 3D, 3-dimensional; RV-LV, right ventricular and left ventricular; EDV, end-diastolic volume; ESV, end-systolic volume; EF, ejection fraction; ICDs, implantable cardioverter-defibrillators; MRI, magnetic resonance imaging; LGE, late gadolinium enhancement; ECV, extracellular volume; HALT, hypoattenuated leaflet thickening.
Footnotes
Authors’ Contributions
BDX and MS: guarantors of the study integrity. RJA, BDX, and MS: study conception. RJA, BDX, CMvdH, and MS: study design. NA: data acquisition, data analysis and interpretation, and statistical analysis. RJA and BDX: literature review and manuscript drafting. BDX, CMvdH, and MS: critical revision of the manuscript for important intellectual content. RJA, BDX, CMvdH, and MS: approval of the final version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
