Abstract
The ideal aortic valve substitute in adults remains a matter of intense debate. With the advent of transcatheter procedures, the concept of lifetime management of aortic valve disease has gained further momentum. The Ross procedure represents the only replacement option that ensures long-term viability of the aortic valve substitute. This provides unique biological and hemodynamic benefits, which have translated into clinically relevant benefits for patients. In recent years, better understanding of the technical nuances of the operation and recognition of the importance of surgical expertise have further improved outcomes. The current review provides a comprehensive examination of the current role of the Ross procedure in adults in contemporary practice, including indications, technical elements and special considerations.
Central illustration
Title: The Ross procedure: Current clinical evidence and future directions
Key point box
Conventional aortic valve replacement in young and middle-aged adults is associated with a loss in life-expectancy, likely due to valve-related complications, imperfect hemodynamics and non-living substitutes. The Ross procedure provides a unique opportunity to patients, as it preserves a living valve in the most critical position. This combination of preserved biology and hemodynamics has repetitively been shown to translate into superior clinically relevant outcomes. The operation is now standardized, patient-tailored and performed with excellent durability of both valves in patients with preoperative aortic stenosis and/or aortic insufficiency in reference centers. Responsibilities and challenges for the future include determining the best technique for individual patients, the dissemination of evidence to the community and patients, better defining centers of excellence, and fair translation of this evidence to clinical practice recommendations.
Background
The choice of prosthetic heart valves in adults needing aortic valve replacement (AVR) has long been a decisional dilemma.1–3 Non-elderly patients with aortic valve disease face a complex decision, and the conversation regarding valve selection usually centers around substitute-specific durability and avoidance of lifelong anticoagulation.4–6 Beyond considering durability and need for anticoagulation, life expectancy and quality of life should be important components of the valve selection process in non-elderly, active individuals with an anticipated long life expectancy.
The optimal valve substitute in young adults continues to be a matter of debate. In recent years, there has been a reexamination of the role of the Ross procedure (pulmonary autograft translocation) in adults. 7 It is the only surgical procedure that sustains long-term viability of the aortic valve tissue. 8 Current evidence strongly implies that the Ross procedure is the most appropriate option for non-elderly patients with aortic stenosis (AS) and/or aortic insufficiency (AI) needing AVR.4,9–13 Despite recent findings favoring the Ross procedure, there remains misalignment between evidence and American and European guideline recommendations for AVR.5,6 And in times where reportedly ≥50% of all valve replacements in patients ≤60 years in the United States consists of transcatheter aortic valve implantation (TAVI), 14 a trend unsupported by current guidelines, it is increasingly important to reexamine the role of the Ross procedure.
Since the publication of a prior review on the Ross procedure in 2018, 15 a wealth of scientific evidence has emerged. The aims of the current review are to provide a comprehensive examination of current literature involving the Ross procedure and to pinpoint the current position of the Ross procedure in the treatment of aortic valve disease.
History of the Ross procedure
The first pulmonary autograft in a human was implanted as a subcoronary valve in 1967. 16 The finding that early autograft insufficiency, which posed a significant problem in the early experience, improved when using a freestanding root technique, led many to abandon the subcoronary technique. 17 A similar freestanding root technique was simultaneously adopted with aortic homografts, and was also associated with better late patient survival and valve durability compared to the subcoronary technique. 18 It is proposed that constant oscillation of different component parts of the aortic root after the subcoronary technique may cause structural valve deterioration.19,20
The hypothesis that a living valve was fundamental for long-term durable patient outcomes, correctly postulated by Donald Ross in his 1967 Lancet paper, 16 was evaluated over the span of decades.8,12 This hypothesis was based on reproduction of the sophisticated functional characteristics of the aortic valve complex; later stressed in numerous studies focusing on mechanobiology and fluid dynamics.21–23 After the Ross procedure, there is likely preserved ventriculo-arterial coupling, in contrast to prosthetic AVR.
Widely accepted and practiced in the 1990s, the procedure fell out of favor in the early 2000s due to a higher incidence of complications than expected, namely operative morbidity, autograft failure and early reoperations.24,25 Following the publication of favorable long-term results in adults from dedicated centers,8,13,26 there has been revived interest for the Ross procedure. Use of the Ross procedure across the Society of Thoracic Surgeons (STS) database has risen from a nadir of <1% in 2017 to 7.2% in 2023 in patients <60 years 27 due to a better appreciation of the clinical benefits of the operation and clearer understanding of the surgical technique.
Young and middle-aged adults with aortic valve disease represent a challenging population to treat and require a personalized lifelong management strategy. The current generation of prosthetic valves is not ideal as they are associated with long-term excess morbidity and loss of life expectancy.1,4,28,29 In 2006 it was suggested for the first time, using microsimulation models, that the Ross procedure could lead to restored survival that was comparable to expected survival in a matched general population (Figure 1). 30 It took more than half a century to support the hypothesis that a living aortic valve indeed translates to such clinical benefits, which is now evidenced by a wealth of data 15 that continues to accumulate. The Ross procedure offers the only long-lasting option that does not require anticoagulation and is today widely accepted as the replacement of choice for children and most adults with a reasonable life expectancy. 15
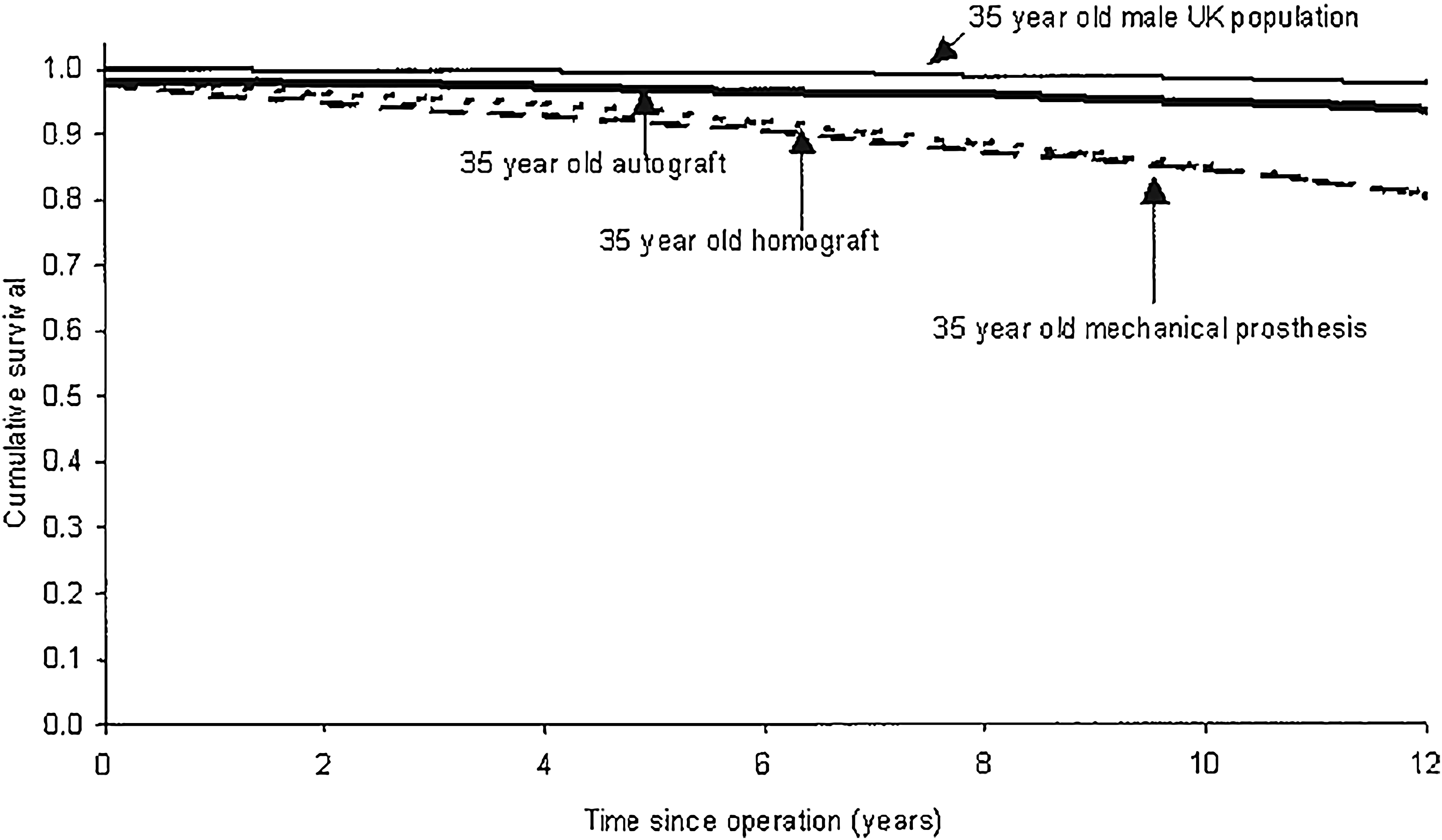
Microsimulation-based survival after the ross procedure (autograft), mechanical aortic valve replacement and homograft aortic valve replacement in a 35-year-old patient compared to a 35-year-old patient from the general population. This was the first signal of restored survival after the Ross procedure.
Underlying mechanobiology and hemodynamics
The aortic root is a living, functional unit
The aortic root is a structural and functional unit that consists of the aortic annulus, the aortic cusps, the sinuses of Valsalva, and the sinotubular junction. It has been shown that homeostasis of the native aortic valve complex is tightly regulated by valvular endothelium, interstitial cells, and extracellular matrix components,22,31 and is highly adaptive to shear stress and humoral changes. 32 The aortic root's expansion and contraction throughout the cardiac cycle reduces cusp stress, minimizes flow turbulence in systole, and enhances coronary flow reserve.21,33,34 The deformations in root geometry precede changes in transvalvular pressure, confirming the dynamic and living nature of the aortic valve and its intimate coupling with ventricular activity. 21 This dynamic function of the aortic root is the premise behind the Ross procedure. The pulmonary autograft is the only aortic valve substitute that preserves the aortic root as a functional unit with long-term viability, and the ability to maintain this hemodynamic pattern throughout the cardiac cycle is believed to contribute to better clinically meaningful long-term outcomes.8,21,35
The pulmonary autograft: similarities and differences
The pulmonary and aortic root share their embryological origin, the superior end of the bulbus cordis, and therefore their basic histological composition and anatomy. The pulmonary valve is a mirror image of a normal aortic valve: it is composed of three semilunar cusps with respective sinuses. As suggested by Anderson, the three cusps are referred to by their relationship to the aortic valve: the left-facing, the right-facing, and the non-facing cusp. 36 Several differences from the aortic root include the absence of coronary ostia in the pulmonary root, thinner leaflets, and direct attachment of the leaflets to infundibular muscle which pushes the valve above the fibrous skeleton of the heart, unlike the three other valves. As such, the pulmonary valve has no fibrous annulus, making the cusps solely supported by the subpulmonary infundibulum (Figure 2). A lack of fibrous continuity between the pulmonary and tricuspid valve base allows for harvesting of the pulmonary valve with no repercussion on the tricuspid apparatus. In addition to these differences, the anatomical relationship between the aortic and pulmonary roots and the interplay with different coronary artery anatomies, must be understood by surgeons performing the Ross procedure.

Longitudinal coupe of aortic and pulmonary sinus tissue (including cusp) demonstrating insertion of the aortic cusp to the fibrous skeleton, opposed to insertion of the pulmonary cusp directly to the muscular rim below the valvar plane (infundibulum). Reprinted with permission from Yacoub et al (2023). 20
Adaptive autograft remodeling
The pulmonary autograft is viable and, therefore, adaptive to the left-sided environment after translocation to the aortic position. When exposed to systemic pressures, the transplanted pulmonary root can remodel and ultimately grow (in children) through cell proliferation, matrix synthesis, and the transformation of valvular interstitial cells into myofibroblasts- developing into a normal aortic valve. Analyses performed on explanted pig autografts showed evidence suggestive of growth activity (presence of TGF-B, bFGF, and Collagen III) and revascularization without degeneration of the autograft wall. 37 Additionally, in a sheep model, Vanderveken et al found that the autograft had mechanical behavior mimicking an aorta, when responding to biaxial tensile stress testing. 38 Another example of its adaptive capabilities is the late expression of EphrinB2, which is a marker of left-sided (systemic) arterial but not right-sided endothelium, in over 85% of pulmonary autograft endothelial cells. 39 The exact timeframe during which pulmonary autograft remodeling takes place is undefined. But adaptations in pulmonary autograft wall and cusp tissue have been demonstrated to start as soon as 2 weeks after the procedure, suggesting that precautions to promote adaptive remodeling are critical at least in the first months. This active process reinforces the autograft wall against excessive tensile stress from higher systemic pressures. These examples illustrate how adaptive remodeling of the pulmonary autograft replicates both the microarchitecture and function of a native aortic root.
Hemodynamic performance
From a functional viewpoint, the pulmonary autograft executes the sophisticated three-dimensional deformations of the aortic root during the cardiac cycle, contrary to any prosthetic valve. The pulmonary autograft preserves the mobility of every component part of the aortic root as opposed to prostheses that rigidly fix the aortic annulus. This explains why the Ross procedure has consistently shown excellent hemodynamic results. At discharge, autograft valves invariably exhibit mean transaortic gradients <5 mm Hg.8,40 In magnetic resonance flow studies comparing pulmonary autografts with mechanical or biological prostheses (stentless or stented), only the autograft group exhibited similar eccentricity, helicity, and vorticity flux when compared to healthy subjects, indicating a physiological central flow jet. 41 Furthermore, wall shear stress at the sinotubular junction and distal ascending aorta was even lower in the autograft group compared to healthy controls. On the contrary, mechanical and biological prostheses exhibited markedly higher flow velocity and jet eccentricity than both autografts and healthy controls. 42 Figure 3 shows an example of improved, near-physiological helical and vortical flow patterns after the Ross procedure, as reported by Stellon et al 43 Such flow studies in patients post-TAVI showed more energy loss and jet eccentricity compared to healthy subjects, 44 in some instances showing the same level of eccentricity as untreated severe aortic stenosis.

Flow characterization by 4D flow magnetic resonance imaging before (top row) and after the ross procedure (bottom row), compared to controls (far right column), demonstrating major improvements to near-normalization of flow after the ross procedure. Asterisks (*) indicate abnormal helical flow, whereas arrows (→) indicate abnormal vortical flow.
Compared to other root replacement procedures such as aortic homografts and xenografts, the pulmonary autograft has also demonstrated better late hemodynamics and flow characteristics, as well as the lowest peak transvalvular jet velocity mimicking that of a native valve.41,45 This is almost certainly the result of a larger effective orifice area (EOA) of the autograft. In a randomized clinical trial comparing autograft with homograft root replacement, there were clinically relevant differences in mean gradients at 13 years postoperatively favoring the Ross procedure. 8 This may explain the survival advantage reported in this trial and other studies. It has been suggested that the benefits of lower time-related gradients in such a young population should not be overlooked, as small reductions in mean gradients in patients undergoing AVR are associated with a lower heart failure incidence afterwards. 46
Patient-prosthesis mismatch (PPM), when the EOA of an inserted prosthesis is too small in relation to patient body size, is well-documented in tissue AVR and is associated with increased mortality, negative remodeling, and recurrence of symptoms.47,48 The Ross procedure is particularly advantageous for patients in whom an increased risk of PPM is anticipated, given the hemodynamic advantages and absence of PPM. 8 For an annulus of equal size, a pulmonary autograft always depicts lower transvalvular gradients and higher indexed EOA (EOAi) than other substitutes. 8 Indeed, short-term autograft hemodynamics are comparable to xenografts or homografts, but autograft hemodynamics and EOAi remain stable and endure over time. 49
Exercise capacity
The hemodynamics of the autograft are near-physiological at high exercise state, with peak jet velocities, mean gradients and EOAi akin to healthy controls. 50 In contrast, patients after mechanical AVR experience significantly higher peak jet velocities and mean gradients at exercise when compared to healthy controls. 51 When observed during maximal exertion, gradients across the autograft do not increase which poses another major benefit compared to valve prostheses in a population of highly active patients. 52
Particularly beneficial for individuals with high activity levels and women planning to conceive, these superior hemodynamic outcomes result in reduced oxygen consumption and postoperative left ventricular remodeling- all of which translate into enhanced exercise capacity and quality of life improvement.53,54 Compared with the age- and sex-matched population, exercise tolerance after the Ross procedure is within the normal range and does not decline over time. 53 Takajo and colleagues showed that Ross patients had preserved exercise capacity (METs) and improved VE/VCO2, which has been traditionally used as a predictor of heart failure prognosis. 54 On the contrary, patients who underwent prosthetic AVR (88% mechanical valve) saw their exercise capacity and predicted VO2 decrease at median follow-up, with further deterioration over time. 54
Surgical techniques
Since its first description, the technique for pulmonary autograft implantation principally evolved from a subcoronary to a total root replacement with coronary reimplantation (Figure 4). Several technical alterations of the Ross procedure have additionally been published over the years (Figure 5), mostly to account for late neo-aortic root dilatation causing insufficiency. Failure mechanisms that cause decreased leaflet coaptation are i) neo-aortic sinus dilatation, ii) annular dilatation and iii) distal native aortic dilatation and ensuing autograft sinotubular junction (STJ) stretching. 55 These mechanisms all predispose to late autograft insufficiency and/or need for reintervention. Moreover, pulmonary homograft stenosis/regurgitation is another mechanism of failure. Although a particular technique might be better in one surgeon's hands than in the other's, we believe that a tailored total root technique offers the most physiological, durable, reproducible and teachable technique, besides offering all benefits of a living root. We will nevertheless examine the main technical approaches to the Ross procedure.

Surgical techniques for the ross procedure; subcoronary technique (A) and full root replacement technique (B).

Technical modifications to the full root replacement ross procedure to mitigate late dilatation and insufficiency; autologous inclusion technique (A), dacron wrapping technique (B), and tailored approach (C).
Subcoronary technique
Implanted as a scalloped autograft at the level of the aortic annulus below the coronaries, the subcoronary Ross procedure (Figure 4A) has shown excellent long-term results.17,56,57 The main benefit of the subcoronary technique is that it precludes late failure by neo-aortic wall dilatation. However, mismatches of the autograft and aortic annulus can produce early insufficiency and failure, as well as dilatation of the aortic annulus over time. Aortic insufficiency by leaflet degeneration or prolapse is the most common mechanism of failure, as there is no autograft wall to dilate. 57 Furthermore, this technique is not applicable to all aortic root phenotypes or coronary origin patterns. Dr. Sievers has published excellent outcomes with 90% freedom from autograft reintervention at 20 years in 630 patients using the subcoronary technique. 57 Similarly, Dr. David applied this technique to a substantial number of patients with very low reintervention rates; his subgroup of subcoronary patients exhibits a comparable incidence of reintervention at 20 years compared to total root or inclusion techniques. 56
Total root technique
The total root technique for pulmonary autograft implantation was pioneered and perfected by Sir Magdi Yacoub and subsequently widely adopted 58 (Figure 4B). Early experience with this technique has demonstrated early and late pulmonary autograft dilatation,59,60 which is now mitigated by technical modifications for autograft implantation. These include i) proximal and distal scalloping of the pulmonary autograft to exclude proximal devascularized muscle and distal pulmonary artery wall at risk of dilatation, ii) implantation inside the left ventricular outflow tract (LVOT) for external support by the native annulus, and iii) blood pressure control to allow for adaptive remodeling.15,55 The technique is now standardized and teachable and allows for multiple tailored modifications to provide essential support. Step-by-step details are now clearly articulated.55,61 Radial and height symmetry during implantation of the autograft are crucial to take into account, especially in unicuspid and bicuspid valves. 62 To promote adaptive pulmonary autograft remodeling and prevent early dilatation (which begets late dilatation), strict systolic blood pressure control should be the aim, starting immediately when the cross-clamp is taken off. Echocardiographic studies have shown that in patients who present with late autograft dilatation, most of it has already occurred by hospital discharge, both at the level of the sinuses of Valsalva (neo-aortic wall) and aortic annulus. 59 The target systolic pressure should be ≤110 mm Hg for 6–12 months to achieve gradual exposure of the autograft to higher blood pressure, preferably with beta-blockers conjointly with an ACE-inhibitor or an angiotensin receptor blocker. In experienced hands, the total root technique has been shown to yield a 20-year cumulative incidence of reintervention of 14%. 12 In a recent update of the German Ross registry, there was no difference in the 25-year durability between subcoronary and total root techniques. 26 Most importantly, these series demonstrate a signal of restored survival, 15 up to 27.5 years in the Harefield series. 12
Risk factors for pulmonary autograft dilatation and subsequent reoperation have been widely reported15,60,63,64 and include: pre-operative isolated aortic insufficiency (AI), aortic annulus >27 mm, size mismatch between the aortic and autograft annuli, and preoperative aortic dilatation. The most common pathophysiology for autograft failure is progressive annular or STJ dilatation and subsequent leaflet malcoaptation. Deep implantation of the autograft in the LVOT and trimming of the distal pulmonary artery to 2 mm above the commissures are important steps to prevent further dilatation of the autograft. Recent modifications have been shown to further improve outcomes for patients with AI/dilated aortic annuli and are believed to significantly reduce the hazard of autograft failure (Figure 5C).65,66 Extra-aortic ring annuloplasty using a Dacron ring is an effective technique to mitigate proximal autograft dilatation by preventing native annular dilatation, hence preventing AI.67,68 The size of the ring is tailored to the autograft annular diameter. 55 Mismatch between the aortic annulus and the autograft ≥2 mm is another instance where extra-aortic ring annuloplasty prevents premature failure. In situations of mismatch between autograft and aortic dimensions, a short interposition graft prevents STJ dilatation. Moreover, the native aortic remnants can be tacked to above the distal autograft anastomosis to provide further nonrestrictive external support. 55 In our experience of 455 patients, this tailored approach has shown very low hazards of both aortic and pulmonary valve dysfunction up to 12 years (<5%) and, importantly, comparable outcomes in patients with AI and AS.
Root inclusion
Inclusion of the pulmonary autograft in the native aortic root (Figure 5A) has been championed by Dr Skillington 13 and modified by others.69,70 In the autologous inclusion technique, the autograft is sutured into the aortic annulus, and the native aortic wall is then tailored to the autograft size. This technique was devised in response to observed autograft dilatation after a freestanding root technique, while avoiding the use of prosthetic material. It provides autologous nonrestrictive support to the pulmonary autograft to withstand left-sided pressure. However, this technique may not be applicable to all aortic roots, such as those with familial aortopathy or certain anatomical features. From a hemodynamic standpoint, this technique preserves the integrity and function of the living root as a unit. The root inclusion is associated with a very low incidence of significant root dilatation and reoperation. In a study involving 333 patients, the 15-year freedom from redo AVR was 96% and survival was comparable to the Australian general population. 13 Importantly, freedom from reintervention in patients with a bicuspid aortic valve (BAV) and pure AI was 85% at 20 years. 71
Dacron wrapping
Autograft wrapping by means of an external Dacron tube (Figure 5B) has been proposed by several groups.72,73 Similar to the root inclusion technique, this modification aims to eliminate any possibility of future root dilatation by stabilization of the autograft wall. The autograft is included and sewn in a Dacron or Valsalva graft before implantation into the aortic annulus. While seemingly straightforward and reproducible, it risks distorting the autograft's natural shape and may lead to immediate/early failure. Additionally, it may predispose to serious complications with coronary reimplantation.74,75 Complications after external aortic wrapping such as vascular erosion and migration have been reported as early as 1986.76–78 This raises concerns when wrapping is applied to the autograft, which is a living and dynamic structure. Animal studies have demonstrated that autograft wrapping is associated with smooth muscle cell disarray and wall thinning in the long term. 79 The longest follow-up series in human wrapped Ross recipients reported mid-term results (median follow-up: ± 4 years) in a population with BAV, showing a low incidence of reintervention (4%) at 10 years. 73 Of note, all reinterventions were within the first postoperative year, suggesting an association with surgical technique. The exact role of this technique will require better definition in upcoming decades. However, we believe that the combination of blunting root dynamics, increasing stresses of the cusps, and impedance on the ventricle may negate the very long-term benefits of the Ross procedure.
Ross-PEARS
Analogous to Dacron inclusion, personalized external aortic root support (PEARS) has been successfully applied to the pulmonary autograft, preventing neo-aortic dilatation and insufficiency for up to 3 years. 80 This technique uses a bespoke, soft and pliable wrap for the aortic root and was developed in 2004 to treat patients with Marfan's syndrome and native aortic root aneurysms. 81 The material is a polyethylene terephthalate macroporous mesh, produced from an individual's pulmonary root measurements obtained by computed tomography. It has a strong proximal edge which is anchored to the pulmonary autograft annulus and prevents its dilatation. 82 However, sheep models have demonstrated thinning of the medial wall due to smooth muscle cell atrophy after wrapping it with a macroporous mesh. Long-term follow-up studies are needed to establish its role in supporting the autograft in the Ross procedure.
Right ventricular outflow tract replacement
Options for right ventricular outflow tract (RVOT) reconstruction include pulmonary homografts, xenograft roots or valved conduits.
Use of pulmonary homografts is the most common and favored option in the Ross procedure. In most long-term series, RVOT reconstruction was performed using cryopreserved pulmonary homografts.12,26,83,84 The homograft, with its muscle skirt, facilitates easy anastomosis to the infundibulum. It is also the replacement option with the best hemodynamic profile and longevity. 85 Systematic oversizing of pulmonary homografts is advisable for optimal hemodynamics as well as to mitigate the hemodynamic impact of early pulmonary homograft stenosis, which can occur in a subset of patients.86–89
Cumulative pulmonary homograft reintervention incidence ranges between 3.4% 90 and 16.0% 91 at 20 years. In a multicenter evaluation of homograft function after the Ross procedure using decellularized pulmonary homografts (n = 466), the only independent risk factor associated with mid-term homograft failure was younger patient age (<45 years; Hazard Ratio (HR): 3.1; p = .03). 89 The hazard of reintervention was highest in the first year after implantation (3.5% in first year) and decreased to less than 1% per year afterwards. 89 In cases of homograft failure, a transcatheter technique has become the approach of choice, with wide applicability and excellent results. The tradeoff between decellularized and standard cryopreserved homografts is currently based on the availability of decellularized homografts as well as surgeon and center preference.
In the absence of pulmonary homografts, use of stentless xenografts has been proposed (eg, Medtronic Freestyle). In a French series of 61 patients undergoing a Ross procedure with a stentless xenograft (mean age: 38 years), there was a 100% freedom from valve stenosis at 5 years. At latest echocardiographic follow-up, the mean RVOT gradient was 11.7 ± 6 mm Hg and no regurgitation was reported. The mean transpulmonary gradient increased faster over time in individuals with a stentless xenograft diameter of <29 millimeters. 92 Very recently, a German series comparing homografts with bovine jugular veins and stentless xenografts in the Ross procedure demonstrated that 10-year freedom from reintervention was lowest with bovine jugular veins (89%) compared to homografts (99%) and xenografts (100%; p = 0.156). Additionally, the progression of mean pulmonary conduit gradients was comparable between homografts (2.6 mm Hg/year) and xenografts (2.9 mm Hg/year). 93
As such, xenografts represent a reasonable alternative to pulmonary homografts. Continued research in the field of tissue engineering is anticipated to provide viable and scalable long-term solutions.
Outcomes of the Ross procedure
Single arm studies
There are now multiple expert centers reporting outcomes after the Ross procedure well into the second or third postoperative decades. A summary of recent evidence for the Ross procedure is provided in Table 1. The most recent update of the Toronto experience with the Ross procedure in 212 patients (mean age: 34 years) demonstrated 89% patient survival at 20 years, not different from the matched Ontario population, accompanied by a low incidence of reintervention on both valves (17% at 20 years). 56 The largest published analysis of patients with a root inclusion technique (n = 322; mean age: 40 years) showed excellent aortic valve durability, with freedom from autograft reintervention of 96% at 18 years. 94 Such excellent outcomes were confirmed even in 129 patients with pure bicuspid AI up to 20 years. 71 The most recent update of the German Ross cohort included 2444 patients (mean age: 44 years) with a median follow-up of 9.2 years. 26 At 20 years, survival mirrored that of the German population matched for age and sex with 78% freedom from any Ross-related reintervention. Importantly, occurrence rates of all other valve-related complications were extremely low. 26 The original subcoronary Ross is associated with excellent durability outcomes in expert hands, with a similar signal of restored survival; in 630 subcoronary Ross recipients (mean age: 45 years), a low incidence of reintervention (14% at 20 years) was observed. 57 With a median follow-up of 24.1 years (completeness of follow-up: 98%), the Harefield series represents the longest reported follow-up after the Ross procedure. 12 All 108 patients (mean age: 38 years) had a freestanding root technique for autograft implantation and systematic homograft oversizing. Survival mimicked that of the general population up to 27.5 years after surgery (Figure 6). Although there was a progressive rate of recurrent AI over time, 71% of patients were free of any reintervention at 25 years. 12 Mastrobuoni and colleagues reported high long-term survival and only 3% valve-related deaths at 16 years in 306 consecutive subjects undergoing the Ross procedure (mean age: 42 years) using various techniques. 10 Finally, the group from Leuven recently examined the outcomes of the Ross procedure at their institution in 173 patients (median age: 32 years), showing excellent 20-year patient survival of 89% and autograft reintervention-free survival of 75%. 95 Coupled with long-term stability of autograft and homograft function for all techniques, this body of global evidence demonstrates a consistent signal of restored survival after the Ross procedure across various settings.
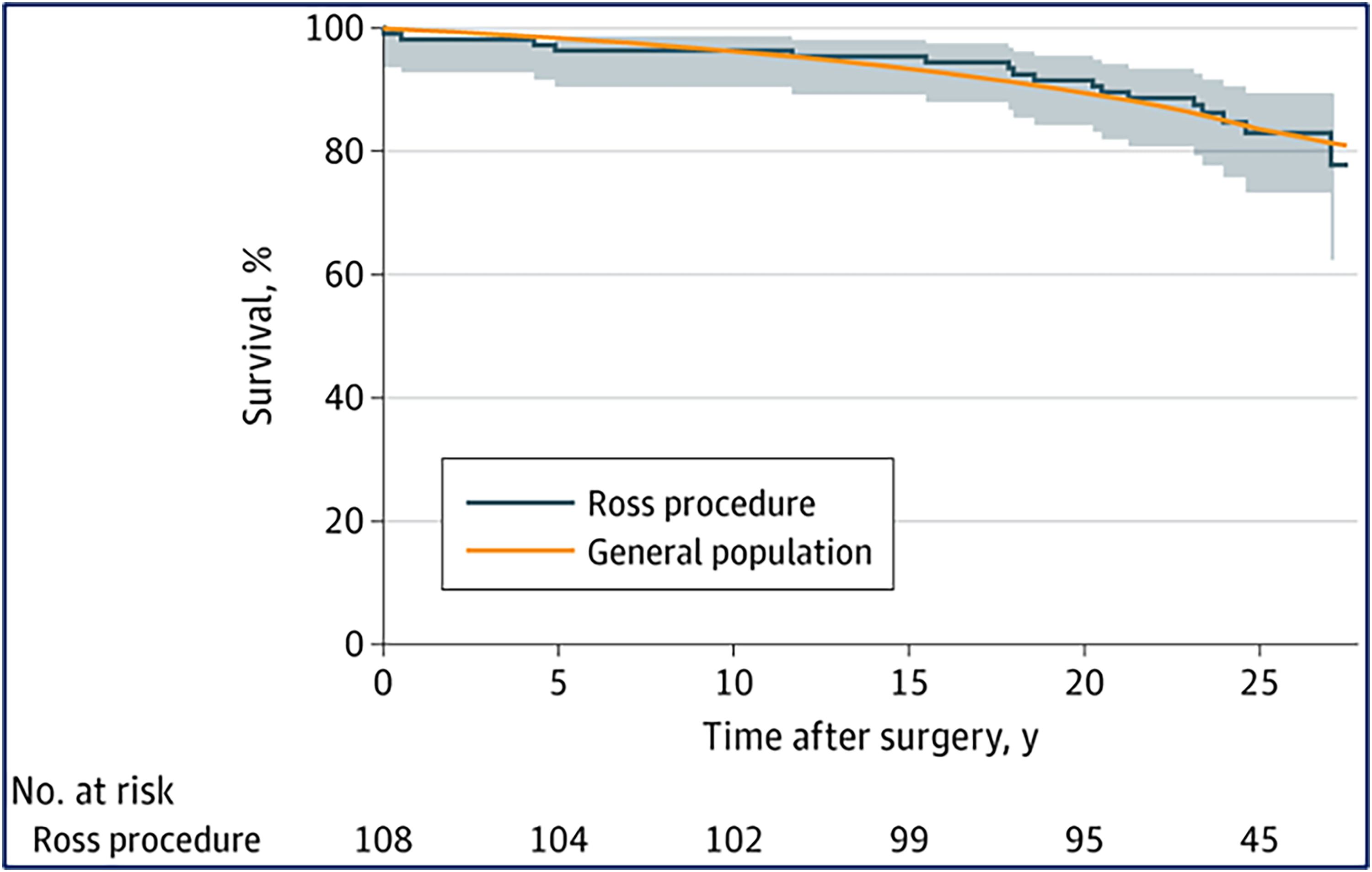
Postoperative survival after the ross procedure mimics survival in the matched-general-population up to 27.5 years after the operation (median follow-up: 24.1 years; completeness 98%).
Summary of contemporary evidence for the Ross procedure (2018-2024).

Abbreviations: MP, mechanical prosthesis; BP, biological prosthesis; SD, standard deviation; IQR, interquartile range; AI, aortic insufficiency; AS, aortic stenosis; SC, subcoronary; RR, root replacement; RR + I, root replacement with inclusion (autologous inclusion or Dacron wrapping). ^ = depending on subgroup.
Comparative studies
A number of studies aim to compare the Ross procedure to other substitutes (Table 1). A randomized clinical trial including 216 consecutive patients needing AVR (mean age: 38 years) showed superior survival following the Ross procedure (95% at 13 years) compared to homograft aortic root replacement (78% at 13 years). Freedom from any reintervention was 94% at 13 years after the Ross procedure and clearly superior to the 51% at 13 years in homograft recipients. 8 Other recent nonrandomized studies have attempted to demonstrate differences in outcomes between substitutes, most using propensity-score matching (PSM) to reduce the effect of baseline differences on outcomes. Mazine and associates in 2016 conducted a propensity-matched analysis comparing the Ross procedure to mechanical AVR (n = 208 pairs), showing less cardiac- and valve-related deaths (HR: 0.22) after the Ross procedure and comparable durability to after mechanical AVR at 20 years. 11 Similarly, an Australian study utilizing PSM demonstrated better survival and lower valve-related complication occurrence after the Ross procedure compared to mechanical AVR. 9 A propensity-matched analysis of 108 Ross (mean age: 41 years) and 108 biological AVR recipients showed a significant survival benefit and a lower need for reoperation and pacemaker implantation in favor of the Ross procedure. 96 Recently, a comparison of valve substitute outcomes with mandatory statewide databases was performed, aimed at mitigating limitations related to single-center biases and patient selection. Using California and New York state data, outcomes of matched pairs of 434 patients undergoing the Ross procedure, mechanical or bioprosthetic AVR (mean age: 36 years) were analyzed, showing superior survival after the Ross procedure compared to all prostheses types 4 (Figure 7). Compared to bioprosthetic AVR, but not mechanical AVR, the Ross procedure was associated with a lower hazard of any reintervention. Late bleeding (HR: 0.32) and stroke (HR: 0.37) were lower among Ross versus mechanical AVR patients, whereas late endocarditis (HR: 0.37) was lower among Ross versus bioprosthetic AVR patients. Of the valve-related complications after all modes of AVR, the one associated with the lowest 30-day mortality was reoperation. 4 Although interest in biological AVR has grown due to future valve-in-valve possibilities, it should be cautiously considered in operable younger adults as it may predispose to a loss in life expectancy – as previously demonstrated. 97 Several large-scale meta-analyses of original studies have confirmed the aforementioned excellent outcomes after the Ross procedure compared to other substitutes.98,99
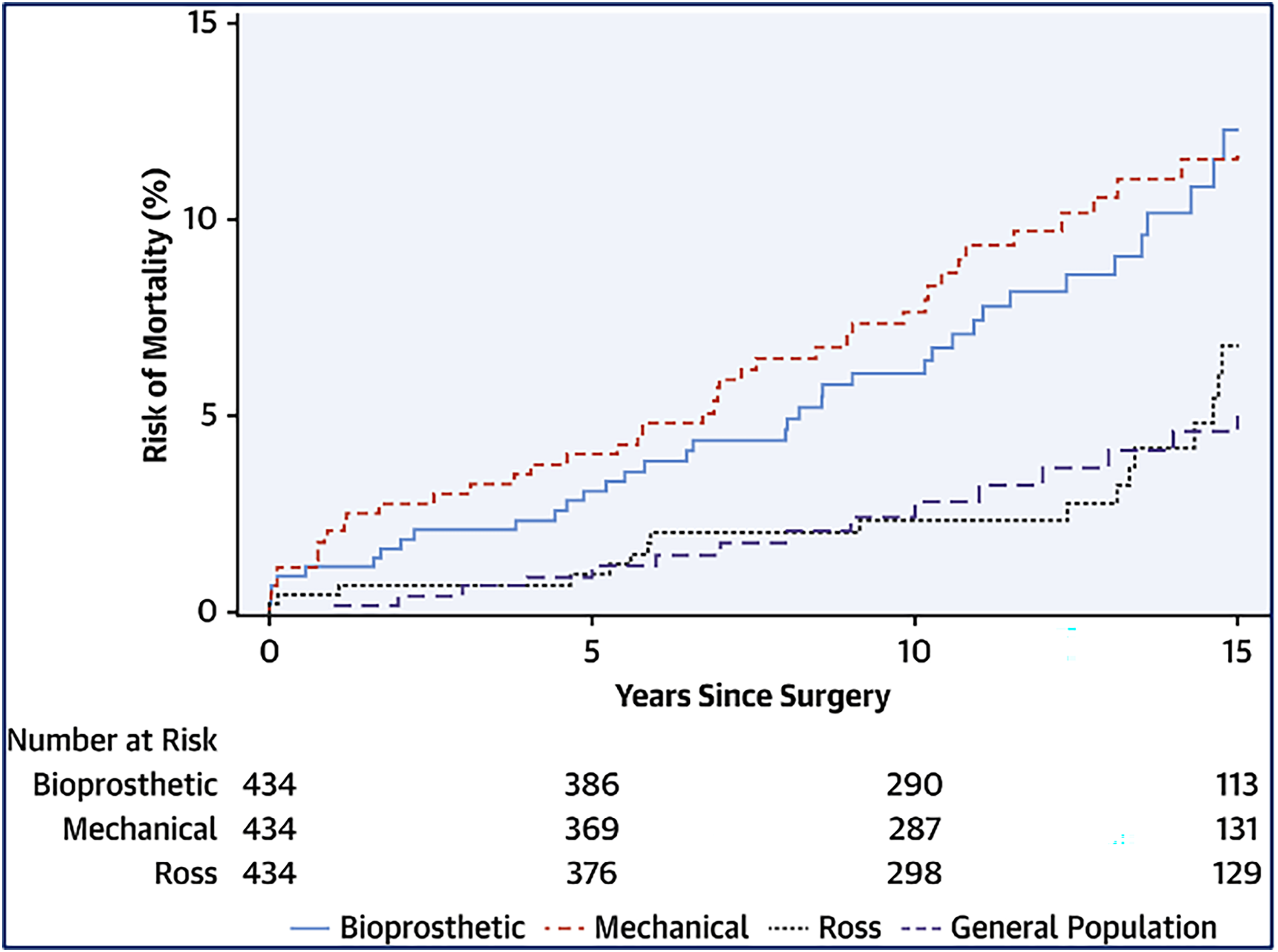
Long-term postoperative survival after the ross procedure is superior to other available substitutes and comparable to the matched-general-population, unlike mechanical or biological aortic valve replacement.
Observational studies have additionally suggested that quality of life after a Ross procedure may be superior to quality of life after a mechanical AVR,100,101 especially in terms of vitality and mental health – important aspects in non-elderly adults. 100 The previously described randomized trial also demonstrated higher self-assessed physical functioning and general health domain scores in Ross compared to homograft patients, 8 supporting the notion that a living aortic valve can lead to improved quality of life.
The findings from these comparative studies are consistent and support the preferred use of the Ross procedure over other alternatives in non-elderly adults.
Important considerations
Reference centers
The relationship between volume and outcomes in cardiac surgery in general, and in aortic root surgery specifically, is well-established. 102 In addition to case volume, late outcomes after the Ross procedure are influenced by the surgical technique and patient selection. 103 A recent study examined Ross procedure volumes and outcomes using the STS database from 2008–2023. 27 It showed that the nadir of the Ross procedure's popularity was in 2015–2017, where it constituted <1% of all AVRs in adults <60 years, whereas in 2023 this had risen to 7.2%. Besides this Ross renaissance, the main take-home message of this study was the relationship between center volume and early mortality. Compared with centers that performed 1–2 annual Ross procedures, in-hospital mortality was lower in centers that performed 3–10 (OR: 0.56 [95%CI: 0.24-0.89]) and lowest at centers performing ≥11 Ross operations/year (OR:0.18 [0.06-0.52], p = .013). Indeed, the observed/expected ratio for mortality was 1.0 in the latter group. 27 This stresses that the Ross procedure should only be performed in high-volume centers, where the perioperative mortality is not different from conventional AVR. 104 With the benefit of hindsight and the accumulation of new studies confirming the superiority of the Ross procedure at more experienced institutions, these findings place earlier findings 24 into context. They underscore our current understanding of the operation's successes and failures.
The time has thus come for Ross reference centers. These centers should be defined by criteria such as case volume, operative mortality rates, echocardiographic outcomes, and the availability of long-term follow-up to demonstrate efficacy.
103
Aortic insufficiency
Although an increased hazard of autograft failure and reintervention has been reported in patients with preoperative AI or a dilated aortic annulus,60,63 these patients always showed a similar survival benefit.12,64,71 Of note, all series in Table 1 included a substantial proportion of patients with preoperative AI (median proportion of pure AI across series: 34.5% (range: 13.8-45.4%)). Furthermore, the increased hazard of functional pulmonary autograft decline can now be mitigated by a tailored surgical approach. The main principles of the tailored approach are based on the principal failure mechanisms of the pulmonary autograft in AI patients (Figure 8).
55

Tailored approach for the Ross procedure. A total root replacement is performed, and annular and/or sinotubular junction support provided on an individual basis (if indicated). The native aortic remnants of the non-coronary sinus and left-right commissure are tacked to above the distal autograft anastomosis to provide non-restrictive autologous external support.
In 2011 Ryan and colleagues demonstrated that patients with preoperative AI were at significantly greater risk of requiring late reintervention after the Ross procedure, but their survival was still excellent and indistinguishable from AS patients. 64 Groups that used other techniques for the Ross procedure in patients with pure AI recently published longer follow-up series. Poh et al examined late outcomes following the Ross procedure in 129 patients with AI and BAV. Attributed to proper external pulmonary autograft root reinforcement, they reported an 85% freedom from redo AVR or AI >2 at 20 years. Similarly, there was excellent survival. A larger STJ diameter was a predictor for late reoperation or AI development, suggesting that radical addressment of autograft-to-STJ mismatch may further reduce the need for reintervention. 71 We recently reported a 25-year freedom from autograft reintervention of 81.5% in 44 subjects with preoperative severe AI (45.4% of total cohort) and a freestanding total root replacement (median follow-up: 22.5 years). The relative survival (observed/expected) was still optimal in the third decade after the Ross procedure, which confirms a consistent signal of excellent survival also in these patients with AI. 12
Considering these findings –coupled with suboptimal outcomes of prosthetic AVR in these patients– the Ross procedure is an excellent option in unrepairable AI.
Small aortic annulus Reinterventions after a Ross procedure
The advantages of the Ross procedure become increasingly apparent in the presence of a small aortic annulus – often in patients with AS. There are two reasons for these advantages. First, long-term durability of the Ross procedure in AS patients with a small aortic annulus is thought to be superior compared to those in AI patients with or without a dilated aortic annulus.56,63,91 This is principally attributed to a lower hazard of pulmonary autograft dilatation and insufficiency in AS patients compared to AI patients. Second, the incidence of PPM is higher after conventional AVR with a small aortic annulus, and it has been clearly shown that the negative impact of PPM is disproportionally higher in younger, more active patients than their elderly counterparts.
105
In contrast, PPM is not observed after a Ross procedure.
The pulmonary autograft fails late, often in the second or third decade, with a characteristic pattern of autograft insufficiency with or without dilatation.106,107 Pulmonary autograft reinterventions are safe and often need to address AI and root dilatation12,108 - rendering them suitable for valve-sparing root replacement. Patients with progressive pulmonary autograft insufficiency should be considered for a reintervention. However, the timing of autograft reintervention for isolated root dilatation is not well-defined. Though autograft root dilation rarely causes acute dissection,109110111112–113 it may more likely induce chronic cusp stretching causing large stress fenestrations – damage which may preclude a successful valve-sparing operation.
114
Reoperation should be considered when the root diameter reaches 55 mm if there is a high likelihood of sparing the autograft valve. Alternatively, it is likely safe to wait until it reaches 60 mm in the absence of significant insufficiency. Several groups have shown that a sizeable proportion of autografts can be spared during reoperation if performed timely and in reference centers.115,116 Alternatively, TAVI has sporadically been performed in failing autografts but is not established as a suitable mode of reintervention and deserves further investigation.
117
Pulmonary homografts fail in a bimodal fashion.
118
Early on, an increased hazard phase is observed in the first 12–18 months, which is followed by a constant low hazard of dysfunction. This is the case in standard cryopreserved
118
as well as decellularized homografts.
89
Homograft reinterventions are indicated for valvular dysfunction – often stenosis – in the presence of symptoms, right ventricular systolic dysfunction, or right ventricular dilatation. This can now be routinely addressed with transcatheter techniques in the majority of cases,89,119 with durability comparable to surgical pulmonary valve replacement (PVR).
120
Multiple transcatheter options are available for reinterventions, such as: a Melody valve, a Harmony valve or a TAVI valve deployed inside an RVOT stent. Use of the Melody valve in this context has been largely abandoned because of size limitations and the inherent risks of stent fracture or prosthesis endocarditis. Surgical PVR is preferred only when there is a need for concurrent autograft reoperation or when the left coronary system is closely situated (<2-3 mm) behind the homograft. In cases of uncertainty, the feasibility of transcatheter solutions can be assessed by inflating a balloon in the RVOT before performing a left coronary angiography to ensure there is no obstruction to coronary flow.121,122
Expanding indications
Most of the contraindications to the Ross procedure are considered to be relative contraindications. The only absolute contraindications are when large pulmonary valve fenestrations (Figure 9) are discovered, patients who perform high-intensity isometric exercise (eg, competitive weightlifting) and rare coronary anomalies hampering autograft harvesting (RCA from LAD).

Examples of large pulmonary valve cusp fenestrations precluding a ross procedure.
Rheumatic valve disease continues to be the predominant cause of aortic valve disease worldwide, affecting approximately 40 million people in total, predominantly children and young adults. 123 There have been ongoing concerns regarding the durability of autografts in this patient population. Both Pergola as well as Da Costa et al have shown that patients with rheumatic aortic valve disease may benefit from a Ross procedure given favorable outcomes in terms of survival and reintervention, and rare recurrence of rheumatic pulmonary autograft disease.124,125 In this setting, to reduce recurrence hazards, medical prophylaxis with penicillin benzathine is advisable. Additionally, surgery should be avoided during the active phase of the disease process.
In patients diagnosed with autoimmune disorders, concerns about autograft durability have arisen, particularly due to reports of recurrent autograft inflammation and associated mortality. 126 To date, no cohort studies have evaluated outcomes after the Ross procedure in these patients. This issue is particularly pertinent for young women, who arguably stand to benefit most from a Ross procedure compared to other valve replacement options. Therefore, the Ross procedure may be considered for this younger demographic. However, surgery should be postponed during active inflammatory phases, and it is crucial to maintain inflammatory quiescence.
Several connective tissue disorders (CTD) are associated with a weakened vascular wall due to defects in the structure of collagen, elastin, or other essential vessel wall components. These systemic diseases will also affect the pulmonary artery wall, altering the decision-making algorithm for their valvular disease. A Ross procedure should thus be cautiously offered to patients with diagnosed CTD and familial aortopathy.
15
Nevertheless, for these younger patients with CTD and unrepairable AI, modifications that exclude the native autograft sinuses or their ability to dilate by either reimplanting or external wrapping have been proposed.
127
The decision to offer these patients a Ross procedure should be individualized and prosthetic AVR may be a valid option considering their CTD.
Age limit?
It is generally acknowledged that the Ross procedure should be offered to all eligible patients <50 years in comprehensive valve centers. Two recent studies set out to examine the outcomes of the Ross procedure in patients ≥50 years.75,128 Both studies demonstrated that the Ross procedure was associated with excellent survival and freedom from reintervention in this age category. This suggests that selected older individuals do benefit from a Ross procedure. This was confirmed in a cohort of patients who underwent the Ross procedure using a root inclusion technique.
129
Therefore, in patients 50–60 years, the Ross procedure should also be considered. In a randomized trial, 12% of Ross recipients were 50–60 years and nearly 10% >60 years –with excellent long-term survival and freedom from reintervention outcomes.
12
The survival benefit of the Ross over prosthetic AVR is more significant in those with a longer life expectancy. Therefore, instead of focusing on the chronological age of the patient, it is more appropriate to assess their anticipated life expectancy (>15-20 years), fitness for surgery, overall activity levels and aortic root anatomy (annulus size). In patients aged 60–70 years, the decision must therefore be individualized, considering these factors. From a technical standpoint, one could even argue that older patients are ideal Ross candidates for they commonly present with pure AS and smaller annuli than young patients with for example bicuspid AI.
Future outlook
The true challenges ahead are to better define the reference centers performing the Ross operation, provide longer-term comparisons between substitutes, explore viable alternatives to pulmonary homografts, and disseminate high-quality evidence to the community and patients. The question then remains how the mounting evidence will translate to guideline recommendations in line with the observations and outcomes from these high-volume, experienced centers.
Despite the ever-growing body of evidence favoring the Ross procedure, there exists serious misalignment with current practice recommendations. Current ACC/AHA guidelines state a Class IIb for the Ross procedure in younger patients in whom anticoagulation should be avoided, mentioning that the operation is complex and ≥50% of pulmonic homografts will require reintervention between 10–20 years 5 (Table 2). In 2024, the operation is standardized and can be broken down in several teachable and reproducible steps. Perioperative risk of the Ross procedure is not different from conventional AVR in reference centers experienced in aortic root surgery.27,94,104 Moreover, contemporary pulmonary homografts have a better expected durability, with <5% reintervention at 10 years –in large part done percutaneously. 89 The durability of the pulmonary autograft was acknowledged in the 2021 ESC/EACTS guidelines, which noted its survival and valve-related reoperation outcomes comparable to mechanical aortic valves 6 (Table 2). However, several studies have now consistently demonstrated superior survival following a Ross procedure compared to mechanical AVR.4,9,11,99 Notably, no comparative studies were cited and no recommendations for the Ross procedure listed in this guideline (Table 2). The wealth of evidence and profound understanding of the factors contributing to the operation's success, published since the latest guidelines, should encourage a reconsideration of the Ross procedure as a viable option for adults.
Current American (2021) and European Guideline Recommendations (2022) Concerning the Ross pcrocedure.

Abbreviations: AHA, American Heart Association; ACC, American College of Cardiology; ESC, European Society of Cardiology; EACTS, European Association for Cardiothoracic Surgery; COR, class of recommendation; LOE, level of evidence; NR, non-randomized.
Above all, these recommendations, the indications, and reference centers should be well-defined to avoid past errors. This approach is crucial for ensuring that the Ross procedure can meet its promising expectations.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
