Abstract
Aortic valve repair has now been shown by long-term evidence to reduce valve-related mortality while restoring quality and life expectancy similar to the general population. However the rate of repair worldwide remains low (around 15%-20%), while 80% of regurgitant valves can be repaired. The commonest cause of aortic insufficiency (AI) in the western world is dystrophic AI with thin and pliable tricuspid, bicuspid, unicuspid, or quadricuspid valves. Three phenotypes of the aorta can be distinguished: dilated aortic root; dilated ascending aorta; and isolated AI. Characteristic lesions leading to dystrophic AI include: a dilated annulus > 25 mm, dilation of the sinotubular junction (STJ) and cusp prolapse. Sub- and supravalvular aortic annuloplasty (reduction of the annulus), restoring the ratio between annulus and STJ, as well as cusp effective height resuspension are all critical components of aortic valve repair and valve-sparing root surgery, helping to increase the surface of cusp coaptation and protecting the repair for long-term durability.
This is a visual representation of the abstract.
Keywords
Key points
Aortic valve repair reduces valve-related mortality compared to prosthetic valve replacement, as well as restoring life expectancy to that of the general population. AI can be associated with 1 of 3 aorta phenotypes: (1) dilated aortic root; (2) dilated ascending aorta; and (3) isolated AI with nondilated aorta. The majority of cases of AI involve a dilated annulus > 25 mm. Calibrated annuloplasty should be performed at sub- and supravalvular levels in order to restore the annulus:STJ ratio and should be adapted according to the phenotype of the root and ascending aorta. Uniform clinical reporting of all available AV repair and replacement techniques, such as in the HVS Aorta Registry, will be key to improving long-term patient outcomes.
Introduction
Many different schools of aortic valve and root repair have achieved long-term or very long-term outcomes, up to 40 years, with excellent valve stability and repair durability after valve-sparing root replacement (VSRR) operations.1-12 Further studies comparing repair of the aortic valve versus composite valve and graft operations show better event free survival in the repair group.13,14 As a result, VSRR is the procedure of choice for patients with aortic root dilatation and pliable tricuspid or bicuspid aortic valves irrespective of the degree of aortic insufficiency (AI), as recommended by international guidelines class I level B evidence. 15 Moreover, standardization of surgical techniques using circumferential sub- and supravalvular annuloplasty as well as systematic assessment of cusp effective height have further improved VSSR as well as isolated AI repair, which are now a IIb recommendation in the 2021 European guidelines.9,15,16
However, despite the breadth of evidence and recommendations in international guidelines, rates of VSRR remains low and flat over time and less that 20% of indications (STS data base, Euro heart survey) are repaired as well as isolated AI repair which remains anecdotal while 80% of the valves are repairable.9,17-19
Aortic insufficiency: A diameter disease combined with cusp lesions
According to Euro Heart Survey for valvular heart disease, two-third of cases of AI (50% degenerative, 15% congenital) involve a pliable tricuspid or bicuspid dystrophic valve with or without aneurysm. 17 These patients represent good candidates for aortic valve repair, while those with restrictive AI caused by cusp retraction or destruction are relative contraindications for repair.
In order to help plan any repair procedure, multimodality imaging including echo and synchronized computed tomography (CT)/magnetic resonance imaging (MRI) should aim to identify 3 parameters: the aorta phenoptype; the mechanism of AI; the valve type.
Aorta phenotype
Baseline analysis of the aorta should include diameter of the annulus, sinus of Valsalva, sinotubular junction and ascending aorta. Depending on whether the sinuses of Valsalva and/or the tubular ascending aorta are dilated, 3 distinct phenotypes of AI can be individualized. According to the phenotype, different aortic repair strategies and operative risk can be applied: the first phenotype is normal root and ascending aorta (<40-45 mm) which will require isolated AV repair without aorta replacement; the second is dilatation of the aortic root (sinus of Valsalva ≥ 45 mm) which will require valve-sparing root replacement with coronary reimplantation as well as aortic valve repair in most cases; the third is dilatation of the ascending aorta (≥45 mm) which will require tubular aorta replacement without coronary reimplantation, along with aortic valve repair. Dilatation of the annulus +/− STJ are almost always associated with all 3 aorta phenotypes (even when the aorta is not significantly dilated, as in the case of isolated AI) as a combined mechanism of AI (Figure 1).

Phenotypes of the proximal aorta and mechanisms of aortic insufficiency. Drawing by Pavel Zacek (used with kind permission).
Mechanism of AI
Three types of lesions can induce AI (Figure 1). Type 1 is caused by dilatation of the aorta 20 and produces a central jet. The aortic cusps are stretched within the enlarged aorta (root or tubular or both) and therefore can present some degree of restricted motion (cusp tethering). Type 2 AI corresponds to aortic cusp prolapse with an eccentric jet. Prolapse can be due to an abnormal elongation of the free margin of the cusp or rupture of a fenestration. Finally, type 3 AI is characterized by cusp retraction or destruction due to fibrosis, calcification or infective processes leading to a large central or eccentric regurgitation. AI due to acute infective endocarditis or restrictive AI without annular dilation (< 25 mm) are contraindications for AV repair.
Valve type and cusp parameters
Valve type is defined by the number of cusps, which is based on the number of functional commissures (ordered by incidence) (Figure 2): (1) A tricuspid (tri commissural) valve has 3 fully developed commissures; (2) a bicuspid (bi commissural) aortic valve (BAV) has 2 fully developed commissures and 0 or 1 raphe on the fused cusp. 21 A fused cusp is named according to the type of fusion, for example, R-L (right-left fusion) represents the most common type of BAV. (3) A unicuspid (unicommissural) aortic valve (UAV) has 1 fully developed commissure and 2 raphes. (4) A quadricuspid valve has 4 commissures, one of which is often underdeveloped corresponding to a raphe.
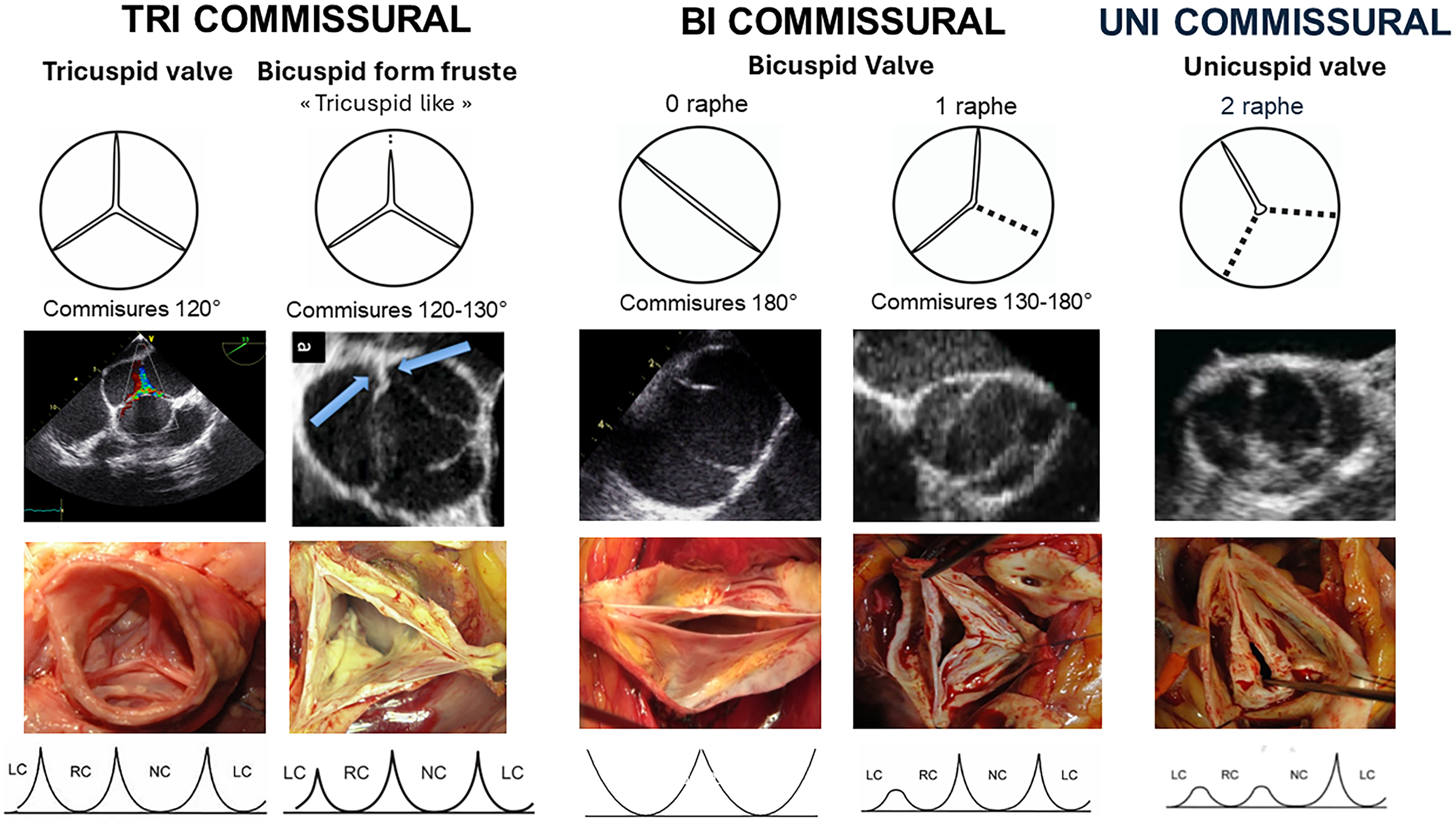
Valve type including commissural orientation and variation of aortic annulus morphology in relation to the leaflet insertion line.
A raphe of the fused or conjoined cusp, also called a nonfunctional commissure (underdeveloped commissure with a hypoplastic interleaflet triangle), is the area of fusion between 2 abnormally developed aortic cusps. The raphe is characterized by a certain length of fusion between the 2 cusps and a certain height of insertion on the aortic root wall. The presence of 1 or 2 raphes, even if incomplete, makes the valve anatomically bicuspid or unicuspid. Absence of a visible raphe on echo does not mean that there is no raphe but just perfect fusion without “the keloïd scar” of a visible raphe. A small raphe (1-2 mm) with 3 functional commissures is characterized as the “tricuspid like” form frustre of BAV.
Commissure orientation is defined as the angle formed by the lines joining the commissures to the central axis of the valve. The angle measured is the one on the nonfused cusp side. It varies between 120° (tricuspid valve or form fruste “tricuspid like” BAV configuration) and 180° (symmetrical bicuspid configuration) (Figure 2).
The cusp free margin is the free border or edge of the aortic cusp, running from one commissure to another. It participates along its entire length in the coaptation of the valve (Figure 3).

Diagram of aortic valve and root illustrating valve parameters. Abbreviations: cH, coaptation height; Com angle, commissure angle; Com Height, commissure height; eH, effective height; FML, free margin length; gH, geometric height; STJ, sinotubular junction diameter.
The geometric height (gH), also called the cusp height, is defined as the distance between the cusp nadir and the middle of the free margin. Intraoperatively, the gH is measured with a straight ruler along the aortic side of the cusp by applying gentle traction on the free margin to straighten the cusp tissue along the ruler. In adults, the cusp is considered retracted when the gH is 16 mm or less in tricuspid aortic valves and 19 mm or less in the bicuspid nonfused aortic cusp 22 (Figure 3).
The coaptation height (cH), also called the coaptation length (cL), is defined as the distance of cusp apposition in diastole. It can be measured echocardiographically on the long axis view of the aortic valve. The normal range is 4 to 5 mm (Figure 3).
The effective height (eH) is the orthogonal distance from the annulus to the middle of the free margin of the cusp. The eH can be measured by echocardiography and intraoperatively with a dedicated caliper. The normal eH in the adult population is close to 9 mm (8–10). 23 Standard 3D TOE assessment in aortic valve repairability should include eH of all 3 cusps for tricuspid valve and eH of the nonfused and fused cusp for bicuspid valve 24 (Figure 3). Cusp prolapse is defined by an effective height lower that the respective other cusps leading to an eccentric jet of AI. The most common type of cusp prolapse is an incomplete prolapse, where effective height of the cusp is lower than the other cusps, but still remains above the plane of the annulus (ie, the eH still remains a positive number in millimeters). Less commonly, there may be complete prolapse, or a flail cusp, where the free margin of the cusp prolapses below the plane of the annulus (in which case the eH will be a negative number in millimeters). Cusp prolapse can be related to an elongated free margin, a rupture of fenestration, commissural disruption or induce by surgical distortion of the commissures.
Principles of aortic valve repair: Annuloplasty and cusp effective height resuspension
From valve dynamics to anatomical landmarks of aortic annuloplasty
The systolic expansibility of the aortic root starts during the pre-ejection phase and occurs through the interleaflet triangles (Figure 4). This allows a dynamic clover shaped opening of the valve which confers less stress on the leaflets. Closure of the aortic valve starts during ejection as a recoil process combined with retrograde vortex backflows within the sinuses of Valvalsa. 25 These delicate physiological dynamics explain the excellent durability of the valve throughout a lifespan. Expansibility of the root is documented to be around 6.2% at the annulus and 5.7% at the STJ in adults. 26

Aortic valve and root dynamics on an ovine model. (a) Systolic expansibility of the root at each level (one cardiac cycle). (b) Cross-sectional area diagram of the aortic root at maximum expansion during ejection showing the clover-shaped orifice of the aortic valve. (c) Tilt angle between the annulus and commissural plane at end diastole. (d) MRI aortic blood flow showing vortex within the sinus of Valsalva. Abbreviations: AA, ascending aorta; Ao, aortic pressure; B, annulus; C, commissures; L, cusp; LV, left ventricular pressure; SoV, sinus of Valsalva; STJ, sinotubular junction.
The aortic annulus has been described in different ways, with terms such as virtual ring, basal ring, or ventriculoaortic junction.27-32 The term “annulus” is synonymous with the inflow of the aortic root. This is the plane passing through the nadir of the aortic cusps. Measurement of the diameter of the annulus can be made either on echo long axis view or by direct intubation intraoperatively. Using the term “annulus” also avoids confusion with “ring” repair prostheses. Large pooled echocardiographic studies have shown that the mean STJ [27.2 mm (range, 24.7-29.5 mm)] is larger than the aortic annulus [22.3 mm (range, 20.5-24.5 mm)] diameter with an STJ/aortic annular base ratio of 1.2.23,33 Therefore, an aortic annulus diameter larger than 25 mm and an STJ diameter larger than 30 mm are deemed as functionally dilated.
The importance of root geometry on valve competence has been demonstrated by a number of finite element studies. Marom et al 34 showed that reduction of STJ induces a symmetrical prolapse by lowering the effective height (eH) of all cusps while dilation of aortic annulus diameter reduces mostly the coaptation height (cH). As a result, an annuloplasty will essentially increase the cH with almost no effect on eH. External dissection down to the subvalvular plane is very important in external ring annuloplasty or reimplantation root procedures. On tricuspid AVs with normal sized aortic roots, external dissection of the annulus may be achieved down to the subvalvular level below the nadir of the left and the noncoronary cusps (basal ring) and in 80% of cases below or within 3 mm of the nadir of the right cusp27,35-39 (Figure 5). Fully reaching down to the subvalvular plane in the region below the right-noncommissure is difficult. Here, the membranous septum limits the dissection plane. The base of the right-noninterleaflet triangle corresponds externally to the insertion of the membranous septum, right atrium wall, infundibulum, and septal leaflet of the tricuspid valve. 36 The annulus is not of equal thickness around its circumference. Its thickest portion (with a mean thickness of 2.5 mm) is the muscular part. Therefore, an external annuloplasty would produce a reduction in the annulus of at least 5 mm.28,40

Internal view of external dissection limits of the subvalvuar plane. The aortic root has been opened with the cusps removed. The blue line indicates the sinotubular junction. The green line indicates the aortic annulus. The dotted line shows the subvalvular dissection plane. The schematic 3-dimensional aortic annular views at the bottom show the cusp insertion points (red line), plane of dissection (thick black line), and the aortic annulus (dotted line) viewed from each sinus.
Valve-sparing root replacement: Reimplantation or remodeling with aortic annuloplasty ?
In 1983, Magdi Yacoub was the first to describe VSRR using the remodeling technique whereby a tube graft is tailored to create 3 neosinuses or scallops.1,41 Ten years later, Tyrone David described VSRR using the reimplantation technique where the valve is contained within the tube graft. 42 There has been much debate as to the pros and cons of each technique over the years. The remodeling technique has the benefit of offering preserved aortic root dynamics and physiological cusp motion by allowing the root to expand through the interleaflet triangles. But several reports have shown that a dilated annulus (>25–28 mm) was a risk factor for recurrent AI due to annular dilatation.12,43-45 On the other hand, the reimplantation technique addresses the annulus and supports it through the proximal suture line. But valve dynamics may involve rapid cusp closure and impaction on the tube graft. 46 The consequences of these different findings were that both techniques were adapted taking into account their respective weaknesses. Neosinuses were created using different techniques or Valsalva type graft in the reimplantation procedure to improve blood flow and cusp motion, and a circumferential external annuloplasty was added to the remodeling technique10-12,47 (Figure 6). The effect of this annuloplasty was to restore the normal annulus diameter, which is almost always enlarged in patients with dystrophic root aortopathy (formerly called annuloaortic ectasia) as well as isolated AI while maintaining systolic expansibility of the annulus in order to preserve valve dynamics. 48 Although annuloplasty is provided by both the reimplantation technique and the remodeling + ring technique, in cases of repair failure where further attempts at repair are futile, the external ring can be cut and removed, thereby leaving a large annulus once again to insert a large prosthetic valve into.

Algorithm of management of the aorta in aortic valve repair for aortic insufficiency. Drawing by Pavel Zacek (used with kind permission).
The technical modifications of the VSRR procedures were not the only reason they gained in reproducibility and durability. The introduction by Schafers et al of the systematic intraoperative measure of eH permitted one to assess objectively the cusp geometry and to diagnose pre-existing or induced cusp prolapse during VSRR. In the case of cusp prolapse, the central cusp plication technique, by reducing the length of the free margin, allows the eH to be increased to approximately its normal value of 9 mm. Good alignment of all cusp free margins with the intraoperative eH measure has been shown to significantly improve the durability of the VSRR. 12
Long-term data accumulated on VSRR techniques have shown that, in experienced centers, freedom from reoperation can be excellent after long-term follow up. The Tyrone David historical cohort achieved a 96% freedom from reoperation after almost 20 years. 10 The remodeling technique associated with the external calibrated expansible extra-aortic aortic ring annuloplasty showed a survival equal to the age and sex-matched general population with a 98% freedom from valve-related reoperation at 10 years.7,11 Moreover, patients who have VSRR report an excellent quality of life and an extremely low rate of valve-related events compared to mechanical valve or biological valve replacement.11,13 In accordance with these excellent long-term results, since 2014, the international guidelines recommend that one consider “aortic valve repair, using the reimplantation or remodeling with aortic annuloplasty technique, in young patients with aortic root dilation and tricuspid aortic valves” (Class Ic indication). 21 In the 2021 ESC Guidelines, a Class Ib recommendation for “aortic root or tubular ascending aortic aneurysm (irrespective of the severity of aortic regurgitation),” to perform valve-sparing root replacement. 15 Further recommendation is given to consider aortic valve repair for isolated aortic regurgitation (Class IIBc). 15
Despite the fact that more than 80% of aortic root aneurysm operations are undertaken for dystrophic AI with a tricuspid or bicuspid valve, a recent analysis of the Society of Thoracic Surgeons database as well as Euro heart survey II revealed that less than 20% of these patients had a VSRR procedure while 80% of these valve were repairable.7,9,17,18 In most centers, VSRR surgery remains highly selective for young patients with TAV and with no or only limited central AI. As would be expected, those cases of root aneurysm not requiring cusp repair are the simplest to manage and have the best outcomes. However, it has been shown extensively that remodeling with annuloplasty or reimplantation and systematic intraoperative cusp assessment also yield excellent outcomes and repair durability in patients with severe, eccentric AI and also in patients with BAV.10-12,47,49
Isolated aortic valve repair: A matter of diameter
AI associated with sinuses of Valsalva and ascending aorta both ≤40 to 45 mm is termed isolated AI. Even though the aorta may not be significantly enlarged, isolated dystrophic AI is almost always associated with an enlarged annulus and/or STJ (annulus ≥25 mm, STJ ≥30 mm). Managing the aortic annulus as part of treating AI is not new. Taylor and colleagues first performed an aortic annuloplasty in 1958, named “aortic circumclusion,” by placing silk sutures as a circumferential annuloplasty running underneath the coronary arteries on a beating heart (Figure 7). 51 The first aortic valve replacement was performed only 2 years later, largely leading to the abandonment of this technique. Over the years different techniques of annuloplasty have been utilized, each with different results. Cabrol described the first internal annuloplasty technique in 1966 using sub- and supracommissural plication sutures. 52 This posed the potential benefit of addressing the annulus and the STJ. However despite the initial good outcomes reported by the Duran and Cosgrove groups,53,54 mid- and long-term outcomes revealed high rates of recurrent AI when the annulus was enlarged.20,47 The Homburg group have shown that the Cabrol subcommissural annuloplasty has been shown to be a predictor of reoperation when combined with remodeling root repair in BAVs. 55 The Brussels group also showed similar results in a bicuspid cohort, with 77% freedom from AI ≥3 at 4 years for subcommissural annuloplasty, compared to 100% for the reimplantation technique. 47 Further studies have shown the subcommissural annuloplasty to also be a predictor of reoperation and redilatation of the aortic annulus in cases of tricuspid aortic valve also. 56 An alternative annuloplasty in the form of a continuous U-shaped internal suture along the cusp insertion line, tied using a Hegar dilator inside the annulus has been used by Carpentier, 57 Haydar et al, 58 and Scholhorn et al 59 (Figure 7). Haydar et al also reinforced the suture line with glutaraldehyde-soaked pericardial strips. 58 In 2005, Lansac et al described an external ring annuloplasty as a critical component of aortic valve repair and valve-sparing procedures.44,45 In the context of isolated aortic valve repair, they proposed that double sub- and supravalvular annuloplasty techniques using 2 external rings placed at the annulus and STJ levels protect the repair and increase the surface of coaptation (Figure 6). This is logical when compared to valve-sparing root procedures, since the root procedure the graft automatically provides an annuloplasty at the STJ level by bringing the commissures to the diameter of the tube graft. Therefore it makes sense to also address the STJ with an annuloplasty when performing isolated AV repair. Indeed absence of STJ stabilization has been identified as an independent risk factor of reoperation in isolated AI as well is BAV repair.9,16 As a result, in order to achieve a good cH and long-term competency of the valve, the physiological ratio of the annulus:STJ diameters (ie, 1:1.2) must be re-established as part of the repair process. Thus, a separate expansible annuloplasty at the supravalvular STJ level in addition to a subvalvular annuloplasty at the annular level using a standardized sizing system would provide both a reduction in respective diameters as well as maintaining the geometric ratio of annulus/STJSTJ and systolic expansibility.

Different annuloplasty techniques by Taylor 1958 (A), Cabrol 1966 (B), Carpentier 1983 (C), Duran 1993 (D), Haydar 1997 (E), Lansac 2003 (Coroneo, Inc. Extra-Aortic Ring) (F), Schafers 2009 (Suture Annuloplasty) (G), Fattouch 2011 (H), Rankin 2011 (HAART Ring) (I). Reprinted by permission from Springer Nature, Gen Thorac Cardiovasc Surg. Kunihara et al. 60
In cases of a dilated ascending aorta with normal root size, the subvalvular annuloplasty at STJ level will be performed by the supracoronary tube (Figure 6). In cases of borderline root diameters (close to 45 mm), the decision to perform a valve-sparing root replacement would be indicated by the position of the coronary ostia being higher than the STJ (hence the STJ ring would be contra-indicated as it would cause coronary ischemia).
A similar idea of a home-made double annuloplasty was also described by Fattouch et al in 2011. They used both an internal and external annuloplasty combined with a crown shaped ring for STJ annuloplasty. 61 When compared to subcommissural annuloplasty (the Cabrol technique), this showed midterm improvement in freedom from AI grade ≥2 62 (Figure 7).
After having identified a dilated annulus over 27 mm as a risk factor of reoperation, Schäfers et al described circumferential suture annuloplasty using Ethibond and subsequently polytetrafluoroethylene Gore-Tex 0 suture, 43 with results initially showing improved outcomes compared to no annuloplasty, particularly for bicuspid isolated valve repair 63 (Figure 7). However long-term results have shown no difference to no annuloplasty for both bicuspid and tricuspid valve sparing remodeling root replacement.5,64
Schomburg and Rankin described in 2011 a rigid internal ring HAART (Hemispherical Aortic Annuloplasty Ring Technology),65,66 although the internal ring was first used in 1993 by Carlos Duran in a small number of patients (Figure 7). Midterm results however have shown higher rates of recurrent AI at only 4 years, particularly in tricuspid aortic valves (21% ≥ moderate). 67 Potential issues that have been highlighted have been interaction of the valve with an internal rigid ring of predetermined geometry, potential left ventricular obstruction of the subvalvular ring, incomplete STJ stabilization and ring dehiscence. 67 Long-term results are needed to assess outcomes.
Cusp prolapse is defined as a free margin level that is lower than its adjacent cusps, or an eH that is below the normal value of 9–10 mm in an adult. Prolapse repair includes a central plication suture to shorten the free margin and to increase its effective height. Extension of the plication from the free margin toward the cusp belly for a few millimeters prevents the cusp from billowing. Free margin resuspension with a running suture of Gore-Tex 7/0 is an alternative technique to treat cusp prolapse; it can also be used to reinforce the free margin or to close a small fenestration. This technique is used less frequently because it may be associated with overcorrection or late stenosis in BAV.
Cusp fenestrations, also called commissural fenestrations, are a congenital variance comprising a gap with no cusp tissues in a small area of the cusp near the commissure and just below the free margin. Most fenestrations within the coaptation area do not induce AI; therefore, their presence does not contraindicate valve repair and does not necessitate any specific management during AV repair with not impact on long-term results. 19 However, in certain cases, fenestration can be involved in the cusp prolapse mechanism (ie, by elongation or rupture of the free margin in relation to the fenestration). In those cases, the area of the fenestration should be closed and reinforced using a small pericardial patch, or if very small, with a direct running suture (taking care not to shorten the free margin length).
In addition to a ruptured fenestration, several other cusp lesions may need patch repair. In infective healed endocarditis, for example, when cusp destruction is limited, the valve can be repaired using a pericardial patch to close a perforation or to reconstruct part of one cusp. A unicuspid valve can be transformed into a symmetrical BAV (bicuspidization) using a patch to reconstruct a functional commissure. Finally, a rheumatic valve can be repaired if the valve cusps are not to retracted and if the annulus is dilated over 25 mm. Patch cusp extension within the belly of the cusp after disinsertion of cusp on its insertion line combined with sub- and supravalvular annuloplasty can be perform in selected case of isolated AI. In growing patients, AV repair, even with a patch, seems to be a good option because it allows the annulus to grow normally and it also allows the eventual Ross operation to be delayed to an advanced age where the pulmonary homograft is better tolerated. 68 Although treated autologous and xenopericardium have shown similar rates of degeneration in AV repair, the focus is currently on biological matrix scaffolds (ie, decellularized xenopericardium) that are under clinical investigation for AV repair.
To summarize, isolated AI repair is best stabilized with circumferential sub- and supra (STJ) valvular annuloplasty or ascending aorta replacement to restore both sub- and supravalvular diameters (Figure 6).
Bicuspid aortic valve repair
BAV disease affects 1% to 2% of the population. 69 Although BAV can occasionally be associated with normal lifelong valve function, its presence is associated with a risk of early valve degeneration and aortopathy leading either to aortic insufficiency or stenosis and aortic aneurysm formation during adulthood. Young adults, who generally present with predominant AI, tend to have a higher rate of aortic root dilatation. In contrast, older patients, who typically present with predominant aortic stenosis, tend to have a higher rate of ascending aorta dilatation. Aortic valve repair is an attractive alternative to replacement in young adults with regurgitant BAV. During the last 2 decades, increased knowledge about BAV disease and refinement of surgical techniques have led to improved standardization and reproducibility of BAV repair.63,70
BAV phenotypes fall along a spectrum. Over the years, many different classification systems have been proposed for BAV morphology. To aid in the dissemination of surgical techniques and uniform research reporting, an international consensus statement on nomenclature and classification of BAV is now available. 71 The “symmetrical” BAV with a commissural orientation of 180° is at one extremity and the “form fruste” of BAV with a commissural orientation close to 120° “tricuspid like” at the other extremity. If the valve is bicommissural with a rudimentary commissure (symmetric or asymmetric BAV) the valve will be repaired as bicuspid valve in a symmetrical design. The tricommissural forme fruste of BAV will be repaired as a tricuspid valve. In addition to having cusp lesions such as prolapse, regurgitant BAVs also present with a systematically large annulus (mean diameter: 30-32 mm in regurgitant BAV vs 27-28 mm in regurgitant TAV vs 23-24 mm in normal TAV) and eventually dilatation of the aortic root or the ascending aorta.
Even if BAV repair techniques still vary among different centers of excellence with large case numbers,47,63,70 over the years the approaches have progressively reached similar goals comprising (Figure 8) the following: (1) restoring cusp configuration with central cusp plication, raphe closure, and intraoperative measurement of the eH; (2) reduction and stabilization of the aortic annulus with circumferential annuloplasty; (3) valve-sparing root replacement using reimplantation or remodeling plus the annuloplasty technique when the root diameter is >45 mm. Both VSRR techniques (remodeling + annuloplasty or reimplantation) allow reinsertion of the commissures at 180°, which ensures the most stable repair with the lowest gradient.9,72-73 When the root size is normal (as in the case of isolated AI, or AI with enlarged ascending aorta), a sinus plication stitch on the side of the fused cusp, as described by Schäfers et al, combined with a 180° placement of the commissures on an STJ ring, can increase the orientation of the commissure toward a more favorable symmetrical valve geometry. As long as the ratio between the annulus and STJ is restored in BAV repair, excellent long-term durability has been shown irrespective of the aorta phenotype (95% freedom from reoperation at 9 years).12,47,63,70

Bicuspid aortic valve repair according to aorta phenotype: (a) alignment of the free edge of the fused cusp along the length of the nonfused cusp; (b) placement of the 2 commissures at 180° for management for different BAV phenotypes: reimplantation valve-sparing root replacement or remodeling valve-sparing root replacement with subvalvular annuloplasty for root aneurysm; hemiremodeling root replacement by replacing the noncoronary sinus (in R-L fusion BAV) with subvalvular annuloplasty and sinus plication; isolated BAV repair with double annuloplasty (STJ and subvalvular) along with sinus plication. Drawing by Pavel Zacek (used with kind permission). Abbreviations: BAV, bicuspid aortic valve; STJ, sinotubular junction.
Peroperative echocardiographic evaluation
In order to ensure good long-term durability, it is important to adhere to strict intraoperative criteria as a determinant of repair success or failure. Based on 3D-TEE performed in the OR, the result of AV repair is satisfying in cases of: ϖ No residual AR or minimal central AR jet (trace); ϖ Individual cusps eH ≥9 mm; ϖ intercusp coaptation length ≥4 mm; mean transaortic pressure gradient < 10 mm Hg. 24
A repair is only satisfactory and able to give good long-term durability if one has achieved a postrepair result of AI ≤ grade I central (<3 mm at the origin of the jet). If there is any residual eccentric jet of AI, even if mild, this indicates residual cusp prolapse and can lead to recurrent AI in the future.
Conclusions
Current medical evidence shows that AV repair is safe, reduces valve-related mortality compared to prosthetic valve replacement, produces better quality of life and provides a similar life expectancy to that of the general population. As was the case for mitral valve repair, dissemination of AV repair techniques will improve with standardization, thereby increasing the rate of AV sparing/repair for both tricuspid and bicuspid valves, even in patients with severe AI. Calibrated annuloplasty should be performed at sub- and supravalvular levels in order to restore the STJ/annulus ratio dynamics and should be adapted according to the phenotype of the root and ascending aorta while respecting valve dynamics. Uniform clinical reporting of all available AV repair and replacement techniques, such as in the HVS Aorta Registry, will be key to improving long-term patient outcomes.
Footnotes
Acknowledgment
Thank you to Dr Pavel Zacek for their artistic contributions and editorial assistance.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Emmanuel Lansac has consultant agreements with the company CORONEO, Inc. (www.coroneo.com), in connection with the development of an aortic ring bearing the trade name “Extra Aortic.”
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
