Abstract
This review explores the impact of computational modeling on aortic root replacement—a complex and invasive procedure often performed alongside other aortic interventions—by examining two key scenarios. The first scenario involves using computational modeling to predict patient-specific outcomes for additional structural interventions following aortic root replacement, enhancing the likelihood of successful outcomes. The second scenario focuses on simulating and planning aortic root replacements performed in conjunction with aortic valve repair. By simulating various surgical techniques, these models optimize procedure effectiveness, safety, and the repeatability of high-quality clinical outcomes.
This is a visual representation of the abstract.
Keywords
Introduction
The fascination with the aortic valve and its complex biomechanics is not a recent phenomenon. It can be traced back to the Renaissance when Leonardo da Vinci meticulously studied and illustrated the anatomy of the aortic valve. His drawings captured the intricate geometric harmony necessary for proper valve function, highlighting an early appreciation for the valve's sophisticated biomechanics. 1 This historical perspective reminds us that the pursuit of understanding the aortic valve's form and function has been a continuous journey—one that spans centuries of medical and scientific progress. 2
Fast forward to 2024, and our ability to explore the complexities of the aortic valve has evolved significantly. Today, we stand at the intersection of medicine, engineering, and artificial intelligence (AI). The same fascination that drove da Vinci to study the aortic valve now drives us to develop patient-specific computational models using AI, machine learning (ML) algorithms, and computer vision. These models represent a modern evolution of that early curiosity, potentially revolutionizing clinical decision-making for complex aortic and valvular pathologies. Today, AI and ML play a crucial role in enhancing computational models by allowing for the analysis of vast datasets, identifying intricate patterns in patient anatomy, and predicting individual outcomes. For example, ML algorithms have been used to indicate the likelihood of heart failure hospitalizations by analyzing electronic health record data, including patient demographics, comorbidities, laboratory results, and medication history. These models can identify high-risk patients earlier, allowing for timely interventions and personalized treatment plans, ultimately improving patient outcomes and reducing hospital readmissions. 3 Neural networks, such as convolutional neural networks, have been employed to improve image analysis, providing clinicians with greater precision in understanding patient-specific anatomical structures. 4 These applications illustrate the growing use of AI/ML in cardiovascular interventions, where data-driven predictions and simulations now inform and optimize procedural strategies.
The integration of computational modeling outputs into clinical practice is akin to da Vinci's detailed anatomical drawings—both serve as essential tools in advancing our understanding of the aortic valve. However, where da Vinci could only observe and illustrate, today's technologies allow us to simulate, predict, and optimize interventions. AI-enhanced computational models enable simulations that predict how valves will interact with a device or behave after a complex repair specific to an individual patient's anatomy. This would help to reduce the risk of complications and improve patient outcomes in complex procedures.
The development of predictive three-dimensional (3D) models for aortic root and aortic valve interventions promises to significantly impact the future of cardiovascular care, especially given the diverse treatment options available and the variations seen in patient-specific anatomy. These models are poised to enhance the decision-making capabilities of heart teams by supporting shared decision-making and advancing the practice of precision medicine, which can be operationally defined as tailored treatment (based on computer simulations) and prevention strategies based on individual patient characteristics. These individual characteristics may include genetics, lifestyle, and environmental factors beyond anatomical ones. There are various examples of precision medicine utilization in cardiovascular medicine. In patients with familial hypercholesterolemia or individuals who do not respond well to statins, genetic and biomarker testing can be conducted to identify candidates for PCSK9 inhibitors, which can effectively lower LDL cholesterol levels. 5 Additionally, in patients with dilated cardiomyopathy, genome-guided decision-making can be used to guide treatment options. Specific mutations can be identified to help decide between various heart failure medications or even help consider eligibility for clinical trials. 6 Thirdly, in patients suffering from heart failure, biomarkers such as NT-porBNP can be used to help guide treatment paradigms and provide more robust information to decide on medication dosage adjustments. These biomarkers can even help physicians consider advanced therapies like cardiac resynchronization therapy based on patient-specific individual patient profiles.7,8
The integration of predictive 3D models in structural heart-related cardiovascular care aligns with the broader shift towards precision medicine, where treatments are increasingly tailored to individual patient characteristics. This review aims to explore the potential impact of such modeling in aortic root replacement (ARR)—a complex and invasive procedure often performed alongside other aortic interventions—by examining two key scenarios.
The first scenario involves using computational modeling to predict patient-specific outcomes for additional structural interventions following an ARR. Specifically, we will focus on trans-catheter valve interventions post-ARR. This approach enables clinicians to tailor treatment plans more effectively, thereby improving the likelihood of successful outcomes by considering each patient's unique post-root replacement anatomical and physiological characteristics.
In the second scenario, we explore the use of computational modeling to simulate and plan ARRs performed in conjunction with aortic valve repair. By simulating various surgical techniques, these models can potentially help optimize procedure effectiveness and safety, as well as the repeatability of high-quality clinical outcomes. This enables surgeons to better anticipate challenges and select the most appropriate surgical methods based on the patient's specific needs and condition. Both scenarios described above involve comprehensive pre-procedural planning by multidisciplinary heart teams, typically composed of interventional cardiologists, cardiac surgeons, and imaging specialists. The planning phase aims to optimize patient outcomes by carefully assessing anatomical structures, determining valve sizing, and identifying potential procedural risks. This planning is currently conducted using commercially available software, such as 3Mensio (Utrecht, Netherlands) or TeraRecon (Durham, North Carolina), which allow clinicians to visualize and analyze preoperative imaging, including CT scans.
However, while these platforms provide detailed anatomical data and allow for some degree of procedural planning, they are limited to 2-dimensional (2D) analysis. This restricts the ability of heart teams to fully understand the complexities of patient-specific anatomical interactions during valve implantation. For example, these tools typically provide overlays or projections of the valve position in relation to the native aortic annulus or other surrounding structures. Notably, current planning tools do not simulate how the valve interacts with the patient's calcified native or prosthetic valve leaflets, the aortic root, or the coronary ostia. They are also not designed to predict possible complications such as coronary obstruction, paravalvular leak (PVL), or the risk of aortic root injury—issues that can arise from the complex interplay of anatomical forces during valve deployment. As a result, clinicians must often rely on their experience and intuition to anticipate potential challenges. In contrast, emerging technologies that leverage 3D computational modeling and AI/ML have the potential to overcome these limitations. These advanced tools can simulate valve deployment in patient-specific anatomy, offering a more accurate prediction of procedural outcomes and improving patient safety and procedural success by allowing for personalized, data-driven decision-making.
Current State of Computational Modeling
A revolution of pre-planning paradigms is on the horizon now that many groups have been able to harness the power of AI and incorporate it into 3D reconstructions of patient-specific anatomy. Multi-slice CT scans can be used to create a 3D digital reconstruction of patient-specific cardiac anatomy, which improves the appreciation for the complex anatomical features present in the heart.9–12 Various methods have been developed to create 3D images of the heart and its structures, such as manually selecting key points and using interpolation to fill in the gaps. 12 In addition, semiautomatic techniques have been used to reconstruct the patient's anatomy by analyzing standard pre-procedural imaging. These techniques work by using differences in imaging intensity to distinguish the heart valves from the surrounding blood, helping to create accurate models of the anatomy.9,11–13 One study by Yeats et al (2024) focused on patients with bicuspid aortic valves (BAV) and severe aortic stenosis. They created a 3D model of both the aortic valve and the aortic arch, allowing for detailed measurement and analysis of the shape and structure of the valve and arch. 14 Using this data, they developed a new classification system to help better understand and categorize the different forms of BAV and aortic arch configurations in these patients. This new system could help improve how doctors approach treatment planning for patients with these complex conditions.
The future of this work is directed at taking the information generated from these reconstructions and combining them with predictive models that leverage the power of AI to predict stent and tissue interactions, in addition to the development of computationally derived radiologic biomarkers that can provide insight into the anatomical and physiological impacts of various treatment options. These advancements are crucial for improving the safety and efficacy of complex transcatheter heart valve (THV) procedures. Many studies have demonstrated the power computational modeling has to assess the risk of various complications associated with trans-catheter aortic valve replacements (TAVRs) in a patient-specific manner.
Coronary obstruction is one of the complications that can be experienced during a TAVR procedure. Although coronary obstruction during native TAVR has an incidence of 0.6%, it is associated with a 30-day mortality of 40–50%. 15 As such, accurate assessment of the risk for coronary obstruction is of vital importance, and there is evidence to demonstrate that coronary obstruction risk prediction can be enhanced with computational modeling. In a study by Heitkemper and colleagues, 16 FE computational modeling methods and DLC/d were used to assess the coronary obstruction risk for 28 patients out of 600 who were considered high risk based on their coronary ostium height being < 14 mm and/or their sinus of Valsalva diameter < 30 mm. Based on the results of the post-simulation DLC/d measurements, 22 of the 28 high-risk patients (78.5%) successfully underwent TAVR without experiencing a coronary obstruction. Notably, the DLC/d measurements in patients cleared for TAVR via computational modeling were significantly different from those in the six patients who were unable to undergo TAVR due to a high risk of coronary obstruction (P < .00078). In contrast, there was no statistically significant difference in coronary ostium height or sinus of Valsalva diameter between the two groups (P > .32). 16 The authors concluded that computational modeling has the potential to expand the pool of TAVR-eligible patients who were previously considered unsuitable, thereby broadening the scope of TAVR utilization. 16
In a separate study, Capelli et al (2012) used finite-element analysis (FEA) to simulate the deployment of a balloon-expanding THV in patients who had a failed bioprosthetic valve, providing the literature with another example to computationally determine the feasibility of valve-in-valve procedures. The authors determined that using these simulation methods can improve patient selection for more complex, non-native THV procedures, which may enhance their safety and clinical effectiveness. 13 Other technologies based on FEA, such as the FEops HEARTguide product (Ghent, Belgium) have also become commercialized software to simulate various structural heart procedures. In a multicenter retrospective study, Halim and colleagues used this assessment tool during the pre-procedural planning stage of TAVR to evaluate device size selections in 140 patients with a borderline annulus size range. 17 Their work illustrated the ambiguous nature of device size selection and stressed the potential importance of simulations to determine THV size in a patient-specific manner. 17
Hokken and colleagues (2023) demonstrated how computational modeling, through the use of FeOps HEARTguide software, can assist heart teams in planning TAVR procedures, particularly in patients with complex bicuspid anatomy, by providing critical insights into the risks of PVL and permanent pacemaker implantation. 18 Similarly, Rocatello and colleagues (2018) explored another dimension of computational modeling, focusing on the role of contact pressure in relation to conduction abnormalities after TAVR. The researchers employed FEA to simulate TAVR in a cohort of 112 patients who had undergone the procedure with a self-expanding valve. 19 The contact pressure and contact pressure index were measured and compared between two groups: one with new conduction abnormalities and one without. The results showed that in the group with new conduction abnormalities, both contact pressure and the contact pressure index were significantly higher (P = .01). 19 These findings suggest that increased contact pressure may be a contributing factor to post-TAVR conduction disturbances, highlighting the potential for computational modeling to aid in identifying patients at greater risk and guiding device selection or implantation techniques to minimize these complications.
The results of these studies underscore the critical role of computational modeling in understanding and potentially mitigating post-procedural complications, such as conduction disturbances. Similarly, AI and ML have emerged as valuable tools in pre-procedural decision-making, particularly in predicting post-TAVR gradients using routine pre-TAVR imaging. This information can assist clinicians in making informed decisions about managing failed surgical valves. As mentioned above, high transvalvular gradients have been associated with poor outcomes and early structural valve deterioration (SVD), in addition to patient-prosthesis mismatch (PPM). 10 The ability to predict the risk of PPM during the pre-procedural planning phase is vitally important because the only operative intervention to combat PPM occurrence is redo surgery or VIV TAVR procedure. 20
Dasi and colleagues (2023) set out to do just that and conducted a retrospective study on patients who had undergone TAVR, in which they trained, validated, and tested an AI model to predict the post-procedural pressure gradient. A second AI model was similarly developed to predict the post-procedural aortic valve area. 21 The pressure gradient predictions had a mean absolute error of 3.0 mm Hg, while the aortic valve area predictions had a mean absolute error of 0.45 cm². 21 Further optimization, such as incorporating simulated stent geometric characteristics as inputs into the deep learning model, is necessary to reduce the mean absolute error. Additional insights into stent-tissue interactions must also be integrated into the training models to enhance prediction accuracy. Although further refinement is needed, this study underscores the potential of AI-based algorithms to improve pre-procedural planning by offering predictive insights that may eventually support clinical decision-making and procedural outcomes.
Much of the research referenced in this review has utilized FEA as the foundation for computational modeling. FEA has traditionally been a powerful tool for simulating complex interactions between medical devices, like THVs, and human anatomy, providing detailed insights into stress, strain, and deformation under various conditions. However, despite its widespread use in research, FEA has several limitations that may explain its slower integration into routine clinical practice and heart team decision-making processes. One major drawback is that FEA using commercially available software, such as Abaqus (Dassault Systèmes, Vélizy-Villacoublay, France), can be highly computationally intensive. This can result in impractical turnaround times for fast-paced clinical environments, where decisions must be made quickly. Additionally, the material properties of the THVs being simulated are often proprietary information, which poses another significant challenge. Without access to the precise mechanical properties of the THVs, such as the specific behavior of the stent frame or leaflet materials under stress, FEA models may rely on approximations, which can reduce the accuracy and clinical relevance of the results. This lack of transparency can limit the utility of FEA models when heart teams need highly reliable predictions to guide procedural planning.
In response to these challenges, other types of computational modeling, such as physics-driven reduced-order models, have emerged as promising alternatives. These models prioritize computational efficiency, allowing for faster simulation times without sacrificing too much accuracy. Reduced-order models can often provide real-time or near-real-time insights, making them far more compatible with the clinical workflow. They are beneficial when rapid turnaround is essential for decision-making during heart team meetings or for complex procedural planning. Moreover, these models often require less detailed input on material properties, allowing them to be more flexible in the absence of proprietary data while still offering a reasonable degree of accuracy in predicting device performance and patient outcomes.
Recently, the FDA-cleared PrecisionTAVI software as a medical device (DASI Simulations, LLC, Dublin, Ohio) has shown promise in its integration into the heart team decision-making paradigm. The physics-driven, AI-based algorithm was trained, tested, and validated on hundreds of pre-and post-operative TAVR pairings to predict the device and tissue interactions between the patient-specific anatomy and the THV biomechanics. In an additional study utilizing the same software, Holst et al (2024) describe the prospective utilization of computational modeling analysis on a cohort of 116 patients who were considered high risk for coronary obstruction using traditional risk determination measurements (valve to coronary distances [VTC], coronary heights, Sinus of Valsalva diameter) from January 2020 to December 2022. Based on the predicted stent deformations and the computationally derived radiologic biomarker DLC/d (Figure 1), the heart team was able to predict an increased risk of CO in 39 of 116 patients. Twenty-nine of those patients proceeded with receiving a TAVR in conjunction with procedural modifications to mitigate CO such as bioprosthetic or native aortic scallop intentional laceration to prevent iatrogenic coronary artery obstruction (n = 10), coronary chimney stents (n = 8), and coronary access without stents (n = 3). By integrating the computational modeling outputs into their heart team discussion, there were no CO events both in patients who were predicted to be low risk for TAVR or high risk for TAVR with procedural augmentation. 22

A depiction of the heart team paradigm with the integration of physics driven computational modeling, artificial intelligence, and machine learning in conjunction with heart team clinical experience to enhance decision making and improve patient outcomes.
Overall, while FEA remains a valuable tool for in-depth biomechanical analysis, its limitations in terms of computational load and reliance on proprietary data have hindered its widespread adoption in clinical practice. Emerging computational models, such as reduced-order approaches, are gaining traction due to their ability to balance accuracy with efficiency, potentially offering a more practical solution for integrating computational modeling into routine cardiovascular care.
The integration of computational modeling and AI into the field of structural heart care represents a transformative leap forward in patient care and pre-procedural planning paradigms. 3D reconstructions, predictive models, and AI-based algorithms allow for a nuanced understanding of complex cardiac anatomy to help tailor and optimize clinical outcomes. For example, the use of 3D modeling to assess dynamic changes in the aortic root has shown promise in reducing procedural risks and improving patient outcomes. Moreover, the development of computationally derived radiologic biomarkers offers new avenues for better understanding the physiological impacts of various treatment options, paving the way for precision medicine in the cardiovascular specialty.
Scenario #1: Computational Modeling to Predict Patient-Specific Outcomes for Additional Structural Interventions Following an Aortic Root Replacement
For patients with aortic stenosis and a concomitant aortic root aneurysm or aortic dissection, the standard surgical treatment involves a complete replacement of the aortic root and aortic valve.23,24 This procedure, known as a Bentall procedure with a composite valve graft (CVG), was first introduced by Bentall and De Bono in 1968. Since then, it has undergone various modifications and has become the standard treatment for various aortic root pathologies.25–27
In patients undergoing concomitant valve replacement, there are options for mechanical and biological valve conduits. Mechanical valve conduits require lifelong anticoagulation therapy, which was one of the driving factors that led to the development of biological conduits that do not require anticoagulation medication. 28 Several studies have compared mechanical versus biological valve conduits in patients undergoing a modified Bentall procedure. Lechiancole and colleagues (2019) conducted a study that found hospital mortality to be similar between patients receiving mechanical and biological prostheses in those aged 65 years or older. The 4-, 8-, and 12-year Kaplan-Meier survival probabilities for mechanical valves were 83%, 72%, and 58%, respectively, while for biological valves, the survival probabilities were 85%, 69%, and 36%, respectively. 29 There was no statistically significant difference in survival probability between the two valve conduits at any time point.28 Risk factors for late mortality in this cohort included male sex, age, pre-operative serum creatinine, and EuroSCORE II.28–30
In a separate study that compared the outcomes of surgical aortic valve replacement (SAVR) in patients between the ages of 50 and 70 over nearly 20 years, patients who received a mechanical valve demonstrated better survival than those who received a bioprosthetic valve. 30 In the cohort of 1580 patients, there was no significant difference in early mortality between the two groups (0.9% for mechanical and 1.7% for bioprosthetic, P = .177). However, after inverse probability of treatment weighting adjustments were made, there was a significantly higher risk for all-cause mortality in the bioprosthetic group compared to the mechanical group (hazard ratio [HR], 1.39; 95% confidence interval, 1.07–1.80, P = .014). It should be noted that the mechanical valve cohort did experience a higher risk of stroke and anticoagulation bleeding (P < .001 for both complications), while the bioprosthetic group had a higher risk of re-intervention (P < .001). 30
A meta-analysis conducted by Bashir and colleagues (2019) concluded that mechanical conduits offer no superiority over biological conduits, but there is a significant increase in bleeding complications in patients receiving an ARR with a mechanical valve conduit. 31 While mechanical valves necessitate lifelong anticoagulation therapy, biological valves are susceptible to SVD. 31 Nonetheless, biological conduits are increasingly used in older patients to avoid bleeding complications.31,32 The literature indicates that both valves carry risks, and selection remains controversial. However, because there is not a clear age threshold for the use of mechanical valves in European or American guidelines, it is consistently emphasized that valve selection should be patient-specific, considering the patient's age and lifestyle to determine a tailored approach.33–35
One of the primary limitations of bioprosthetic valves is the risk of SVD in the short to medium term, which presents limited options for subsequent interventions and repeat surgical procedures in a frailer patient population. 36 However, it is notable that bovine pericardial valves have shown a 15-year freedom from SVD at 82.3%, and porcine valves have demonstrated a 20-year freedom from SVD at 63.4%.37,38This demonstrates that while bioprosthetic valves have inherent limitations, they still offer significant longevity in certain contexts.
The literature suggests that a biological valve conduit is preferred in patients over the age of 75 because they can avoid complications from anticoagulation therapy. In this cohort, there is a reduced risk of the patient outliving the valve's lifespan and requiring a secondary intervention due to SVD.29,39–41 This preference reflects the balance between valve durability and patient-specific factors, such as age and health status, which are crucial in clinical decision-making.
However, it is clear these treatment decisions are complex and multi-faceted. Additionally, there are cohorts of patients where clear treatment guidelines do not exist (ie, patients under the age of 65 who require both their valve and ascending aorta to be replaced. 29 As a result of potential SVD with a bio-Bentall, patients who outlive their index valve replacement may need a second intervention when they are significantly older and pose a higher surgical risk. 42 Thus, the ViV TAVI procedure is a viable option for these patients.
The international multicenter prospective Global Valve-in-Valve Registry reports that the mean time to re-intervention after initial surgery was about nine years. 43 Additionally, the registry indicates that the mortality rate was 7.6% at 30 days and approximately 16.8% after one year. 43 Eggebrecht and colleagues (2011) found that one-year mortality was associated with high transvalvular gradients following a valve-in-valve procedure. In their study, 44% of the patients had transvalvular gradients ≥ 20 mm Hg, with a reported 17% mortality rate, primarily among elderly high-risk patients with multiple co-morbidities. 44
Due to the challenging nature of ViV TAVR procedures, computational modeling can potentially aid heart teams by predicting stent frame deformation to highlight device and tissue interactions that would not be evident using traditional planning techniques. One such challenge is the significantly increased risk of coronary obstruction during ViV TAVI, which is six times higher than in native TAVI procedures. 45 A less common complication is the potential deformation of the stent frame with self-expanding THVs and valve migration due to the interaction between the TAVI stent frame and the supracoronary graft anastomosis. 46 In a study by Sirset-Becker and colleagues (2024), the FDA-cleared PrecisionTAVI (DASI Simulations, LLC, Dublin, OH, USA) physics-driven AI algorithm was used to analyze the feasibility of ViV TAVR after ARR with CVG. The authors hypothesized that using predictive computational modeling would empower operators with more advanced pre-procedural planning techniques to assess the complex anatomical characteristics of patients receiving these procedures. 47 When the authors compared traditional two-dimensional (2D) planning techniques to the 3D computational modeling outputs, they found that the 2D VTCs manually obtained from pre-operative CT scans failed to capture dynamic changes to the aortic root after THV deployment. 47 Additionally, the 3D modeling demonstrated the inadequate contact of the stent outflow in a self-expanding THV, possibly explaining the potential risk of THV migration or embolization observed as a possible complication in these procedures (Figure 2). 47

A demonstration of how the measurement DLC/d is calculated from the simulated THV deployment for a patients received a VIV TAVR.
While computational modeling has shown significant potential in predicting patient-specific outcomes and guiding the decision-making process for additional structural interventions following an ARR, it is equally valuable in planning aortic valve repair procedures. In both cases, these advanced modeling techniques offer clinicians the ability to anticipate complications and optimize procedural outcomes based on the unique anatomical characteristics of each patient. This is particularly important in cases involving complex ARRs with valve repair, where precise planning can mitigate risks and enhance surgical precision.
Scenario #2: The Use of Computational Modeling for Planning Aortic Root Replacements With Aortic Valve Repair
This progress is not limited to TAVR procedures but also extends into the planning and optimization of ARRs concomitant with aortic valve repair. The ability to simulate these incredibly complex surgeries empowers physicians to have a detailed plan prior to performing a sternotomy and placing a patient on coronary artery bypass.
In patients with an aortic root aneurysm and a non-stenotic aortic valve, a valve-sparing root replacement (VSRR) combined with an aortic valve repair is an option. Sparing the aortic valve has been shown to have improved hemodynamics compared to replacement of the valve with either a biological or mechanical conduit. 32 In a study of 178 patients who underwent a VSRR, 33.1% of patients underwent a concomitant aortic valve repair and had an estimated five-year survival probability of 95.2 ± 1.8%. In the under-65 age group mentioned above, younger patients who require a mechanical prosthesis can avoid lifelong anti-coagulation therapy if they receive a VSRR and aortic valve repair instead of a replacement. 48 However, both VSRR and aortic valve repair are incredibly complex procedures, and it is suggested that they be performed by surgeons at centers of excellence, which may limit the generalizability of high-quality outcomes in patients who do not have access to this level of proficiency in clinical care.49,50
As a result, there has been a concerted effort to understand and model the technical nuances of these operations to establish a more consistent and replicable surgical technique among practitioners, with many highlighting the complementary role 3D modeling and reconstructions can have in patient selection and pre-surgical planning. 49 While the most common method of perioperative assessment for these surgical procedures is echocardiography, it is limited by the adequacy of the acoustic windows and patient size; thus, image quality and resolution are not always ideal. 51 Conversely, computer tomography datasets allow for high-quality and detailed 3D reconstruction of cardiac anatomy via a volume in any plane that is relevant to the heart team, with Professor Robert H. Anderson deeming it the “gold standard” for the analysis of cardiac anatomy in patients. 52
However, visualization of cardiac anatomy is just the beginning of the capabilities computational modeling can offer clinical practice. Taking it one step further to analyze the biomechanics and physiology of the heart can help heart teams predict the potential success of their interventions. Ranga and colleagues (2006) utilized MRI velocity mapping to examine valve dynamics in healthy volunteers. They developed computational models to assess and evaluate the function of a normal valve, a valve that had undergone a remodeling procedure, and one that had undergone a reimplantation. 53 Simulations were conducted under three conditions: commissural displacements, displacements at the tips of the leaflets, and systolic stresses across the leaflet surfaces. 53 The peak flow velocities were measured for each scenario at the valve's center. The researchers found that these simulations enhanced their understanding of how different surgical techniques influence leaflet dynamics, such as the locations of peak stresses, which could have significant long-term clinical implications and affect patient outcomes. 53
Marom et al (2012) employed a numerical model to evaluate the predictive power of the aortic annulus diameter on various metrics for assessing aortic valve performance, including principal stress, strain energy density, coaptation area, and effective height. By constructing 3D models of the aortic valve and root, they were able to calculate coaptation height and area. 54 The results showed that a smaller aortic annulus diameter could lead to increased coaptation height and area, thereby improving the effective height during procedures. Improved effective height has been linked to greater coaptation and better valve performance. 54
In a separate study by Marom et al (2013), the research team used a different numerical model to analyze the influence the aortic annulus (AA) diameter and the ratio of the sinotubular junction (STJ) diameter to the AA diameter had on the hemodynamics of the aortic valve. Their results demonstrated that the optimal combination of factors for AA diameters between 24 and 26 mm was large coaptation, low stress in tissues during the diastolic phase, and low flow shear stress during the systolic phase. Additionally, the researchers were able to suggest that valve-sparing procedures that prevent AA expansion may have increased durability. 55 These findings highlight the role computational modeling can have in identifying a patient-specific optimal configuration to ensure long-term valve repair durability.
Hammer and colleagues (2015) analyzed computer simulations to better understand how a specific surgical repair method would treat congenital aortic regurgitation in children. The researchers developed a fluid-structure interaction model and were able to perform simulations to understand the hemodynamic characteristics in a diseased model, a normal model, and three operative models. 56 The results indicated that reconstructive surgery improves the effective orifice area and flow pattern of the aortic valve and demonstrated that specific surgical manipulations during the aortic valve repair may not be necessary to achieve a favorable clinical outcome. 56 Thus, using computational modeling to plan procedures may be helpful in reducing the time a patient is required to be on bypass by demonstrating that a more streamlined surgical approach is sufficient to achieve a successful procedural outcome.
In a separate study by Gu and colleagues (2016), researchers employed FEA to create five models of neo-aortic valves with different-sized STJs. They compared the maximum stress and cusp contact forces for each of the five models and found that when compared to healthy adult patients, the neo-aortic valve of an infant has lower stress; additionally, altering the size of the STJ can increase or decrease the stress experienced by the neo-aortic valve. 57 This study further demonstrates how computational modeling can provide valuable insights into different anatomical factors considered in the repair of the aortic valve.
Brinkman and colleagues (2022) have demonstrated the proof of concept for a computational modeling framework for aortic valve repair (Figure 3). The proposed model is rooted in a physics-based AI-driven algorithm that can reveal anatomical defects under fully pressurized conditions. 58 This allows surgeons to have an appreciation of commissural heights, aspect ratio, the full extent of the aortopathy, and the relationship to the ascending aorta. It also allows them to view the predicted results of various repair techniques, such as the addition of grafted pericardium.56,59 Thus, the model's predictive capabilities can help suggest potential surgical repair approaches and predict quantitative physiological performance metrics (ie, transvalvular gradient, EOA, coaptation, regurgitant fraction, and future valve remodeling). 58 A preliminary proof of concept validation in one patient demonstrated fairly good qualitative agreement between simulated model outputs and 3D reconstructions from a post-operative CT scan (Figure 4 and 5). Future directions for this work would be to test, train, and validate a model on a more robust dataset.

This figure demonstrates the prediction of contact of the self-expanding valve in patients who had a simulated VIV TAVR procedure after an ARR with CVG. This image illustrates the potential to predict the contact between the stent-outflow in the SE THV, which may be a predictor of THV migration or embolization.
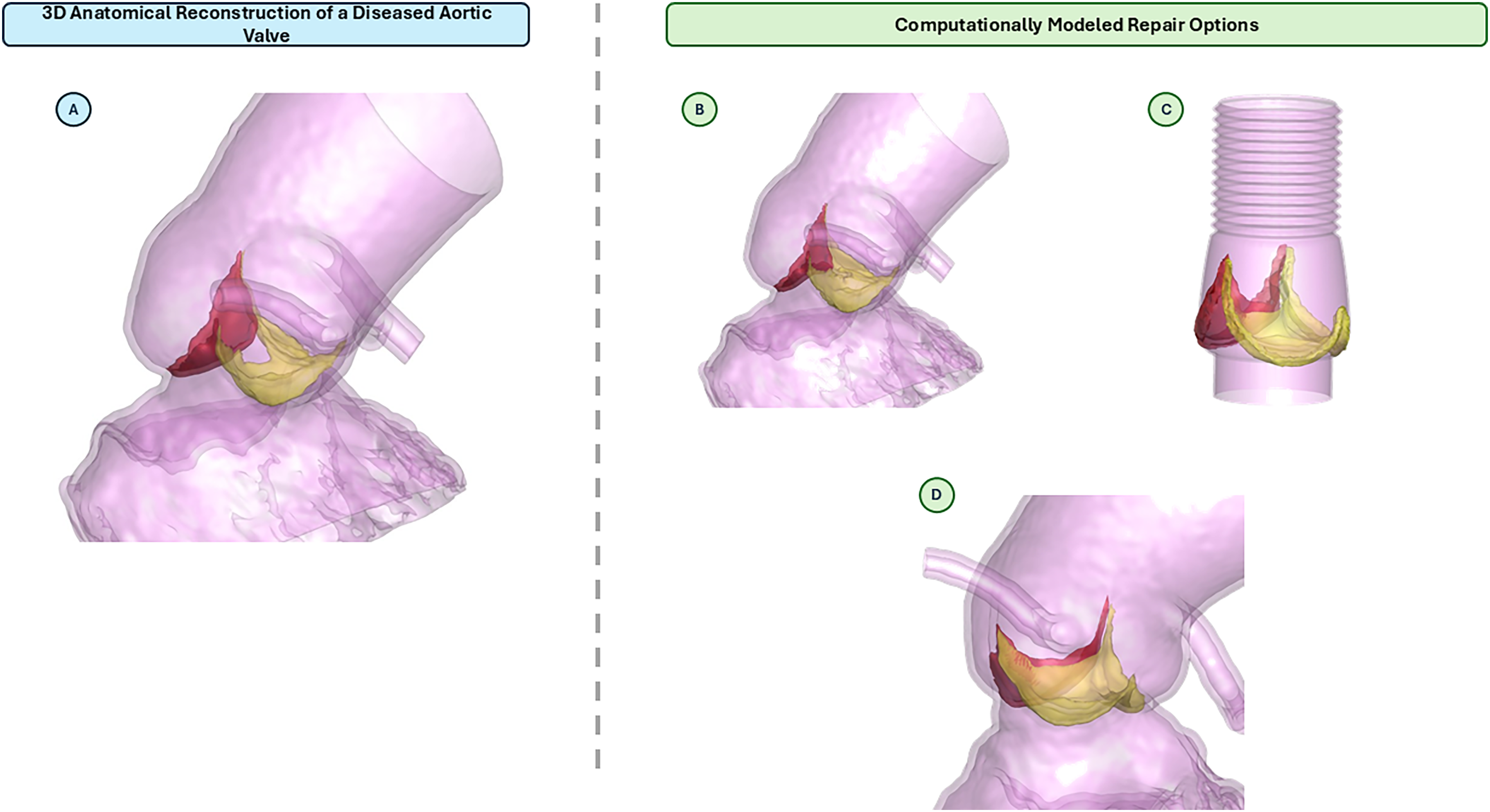
(A) A 3D anatomical reconstruction of patient's diseased aortic root and bicuspid aortic valve. (B) A computational model showing the results of a leaflet plication to repair the aortic insufficiency. (C) A computational model showing the results of root graft and leaflet re-suspension. (D) A computational model showing the results of a leaflet re-suspension to address the aortic insufficiency.
Current Challenges
While computational modeling holds great promise for transforming cardiovascular medicine, several significant limitations and challenges must be addressed before it can be fully integrated into routine clinical practice. One of the primary hurdles is the availability of high-quality data to support and refine these models. For computational modeling to be accurate and predictive, it requires large datasets of patient-specific imaging and procedural outcomes, which are often limited. This scarcity is particularly problematic when attempting to develop biomarkers for predicting rare but life-threatening complications, such as aortic root rupture or coronary obstruction during procedures like TAVR.
A potential solution to this data scarcity would be the establishment of a centralized registry containing CT scans and other relevant imaging from patients who have experienced procedural complications. Such a registry could serve as a valuable resource for researchers and clinicians, providing the necessary data to enhance model accuracy and enable the identification of subtle anatomical features that may predispose patients to complications. This would help to bridge the gap between theory and clinical application, allowing for the development of more robust and reliable models.

(A) A simulation of an aortic root replacement and leaflet re-suspension in a patient with a bicuspid aortic valve. (B) A 3D reconstruction of the same patient from the post-CT scan from their aortic root repair surgery.
Another major challenge in validating these computational models is the lack of post-operative imaging. For models to be useful in predicting outcomes and guiding decision-making, they need to be validated against real-world patient data, including post-TAVR CT scans. However, obtaining these post-operative images can be expensive for hospitals, particularly if there is no clear clinical indication for them, such as hypo-attenuated leaflet thickening. In many cases, post-TAVR imaging is not reimbursed by insurance, which makes it financially burdensome for institutions to obtain them routinely.
Additionally, institutional review boards (IRBs) are often hesitant to approve post-TAVR CT scans for research purposes. Their concerns typically center around the risks associated with the contrast dye used in the scans, which can be harmful to patients with kidney issues, as well as the additional radiation exposure. These safety concerns create a significant barrier to collecting the data needed to validate computational models. Without widespread access to post-TAVR imaging, it becomes challenging to verify whether these models accurately reflect real-world outcomes, limiting their adoption in clinical practice.
Addressing these challenges will require a multi-pronged approach. Initiatives like establishing data registries for procedural complications, developing alternative methods for model validation that don’t rely on post-operative imaging, and exploring ways to minimize risks associated with post-TAVR CTs could help overcome the current barriers. Furthermore, collaboration between researchers, clinicians, and regulatory bodies will be essential in shaping the future of computational modeling in cardiovascular interventions, ensuring that these models are not only innovative but also practical and safe for everyday clinical use.
Broader Implications and Future Directions
The broader implications of computational modeling extend beyond the specific scenarios discussed in this review, with the potential to transform various structural heart interventions. As AI-driven predictive models advance, they promise to revolutionize cardiovascular medicine by offering unparalleled precision in personalized care. These technologies will enable clinicians to perform more comprehensive patient risk assessments and refine pre-procedural planning, tailoring interventions to the unique characteristics of each patient's anatomy, health status, and procedural risks. By utilizing these models, clinicians can anticipate complications, avoid adverse outcomes, and optimize procedural strategies, delivering more data-driven, individualized care.
However, the widespread adoption of computational modeling in clinical practice requires ongoing refinement and validation. These models must undergo rigorous testing to ensure they accurately predict real-world outcomes across diverse patient populations and clinical settings. Collaboration between medical professionals and technology developers will be crucial to addressing limitations and advancing model reliability. As computational power and AI algorithms improve, models will become faster, more accurate, and accessible, facilitating their integration into routine practice. Regulatory frameworks must also evolve to address data usage, validation, and safety, ensuring the safe incorporation of these technologies. Ultimately, computational modeling will play a central role in shaping the future of precision medicine and cardiovascular care.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
Dr Becker reports having patents filed on computational modeling for structural heart procedures. Dr Becker is also a consultant for DASI Simulations, LLC. Dr Dasi reports having patents filed on 3-dimensional (3D) computational modeling for structural heart procedures. Dr Dasi is also the co-founder and CTO of DASI Simulations, LLC. S. Chris Malaisrie reports a relationship with Edwards Lifesciences Corporation that includes consulting or advisory, funding grants, and speaking and lecture fees; with Medtronic that includes: consulting or advisory, funding grants, and speaking and lecture fees; with Artivion that includes: consulting or advisory, funding grants, and speaking and lecture fees; and with Terumo Aortic that includes: consulting or advisory, funding grants, and speaking and lecture fees.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
