Abstract
Background
Calcific aortic stenosis (AS) and peripheral artery disease (PAD) share common pathophysiological mechanisms and risk factors related to atherosclerosis. However, the prevalence of PAD and its association with mortality in patients with AS remains heterogeneous and require further investigations.
Objectives
The aims of this study were (a) to determine the prevalence of PAD in patients with severe AS and (b) to assess the impact of PAD on mortality in these patients.
Methods
From June 2000 to December 2019, patients who underwent surgical aortic valve replacement (SAVR) at the Limoges University Hospital were enrolled. Over a median observational period of 7.6 years [4.9–11.4], 1462 patients were studied. PAD was defined by mention of clinical diagnosis, significant stenosis on ultrasound, or history of lower-limb revascularization.
Results
Patients with PAD accounted for 8.9% (
Conclusions
Almost one out of 10 patients with severe AS presents PAD. This comorbidity was independently associated with traditional risk factors and markedly reduced survival, increased mortality. Similarly to patients with CAD, patients with AS and concomitant PAD should be considered at high risk of poor outcome.
Keywords
Key Point box
Peripheral artery disease (PAD) and calcific AS share cardiovascular risk factors and have common pathways. In patients with AS, PAD is under-screened. The association of both PAD and AS seems be associated with worse outcome but is still understudied. Around 9% of patients undergoing surgical aortic valve replacement (SAVR) had PAD. Male gender, dyslipidemia, type-2 diabetes mellitus (DM), chronic kidney disease (CKD) at stage ≥ III and obstructive sleep apnea syndrome (OSAS) were associated with the presence of PAD. Patients with PAD had a subsequent 1.6-fold increase in risk of long-term overall mortality than patients without PAD. This over mortality was even more increased among patients with both CAD and PAD.
Introduction
In high-income countries, the prevalence of aortic stenosis (AS) is growing and represents a major public health burden with a significant impact on morbidity and mortality. 1 Calcific AS is the most common valvular disease in Europe and North America and the most common indication for valve replacement, 2 affecting 10% of the population aged 70 years and older. Furthermore, AS is expected to rise with the demographic aging and could double by 2040 and triple by 2050 to 2060.3–5 The leading pathophysiologic mechanisms involved include inflammation, calcification, fibrosis, and angiogenesis. Many of these mechanisms, as well as their common risk factors, are shared with other atherosclerotic diseases such as peripheral artery disease (PAD).6–8 Indeed, similarly to AS, PAD is considered as one of the most prevalent cardiovascular diseases in the elderly and is associated with a specific profile of risk factors such as hypertension and metabolic disorders.9,10 Consequently, PAD reflects severe damage to the vascular beds of the lower limbs and is associated with an increased risk of overall and cardiovascular deaths.9,11 PAD is under-detected in the general population and in patients with calcific AS. Therefore, its prevalence and its association with mortality remain poorly reported and heterogeneous.12,13 This study aimed to (a) determine the prevalence of PAD and other atherosclerotic lesions in severe AS, (b) identify factors associated with PAD, and (c) assess the impact of PAD on long-term mortality.
Methods
Study Cohort
From June 2000 to December 2019, 1592 patients with severe AS underwent surgical aortic valve replacement (SAVR) at the Limoges University Hospital (Figure 1). In order to focus on patients with severe calcific AS and no prior valvular conditions, we excluded patients who met at least one of the following criteria (
Data Collection
The database was built using a pre-specified methodology for data extraction from the Limoges University Hospital’s database. This database was developed in the fields of medicine, surgery, and obstetrics, based on the International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM), and the International Classification of Health Interventions (ICHI) codes. Additionally, the entire medical record of the cohort up to the date of surgery was reviewed to address any missing data, correct coding errors in the hospital information system, or include variables that were not captured in the ICD-10-CM.
Definitions and Outcomes
Patients with PAD were identified if they met at least one of the following criteria prior to SAVR: a clinical diagnosis documented in medical records, an ankle-brachial index ≤0.90, significant stenosis on ultrasound (>50%), a history of thromboendarterectomy or angioplasty of the lower limbs (excluding acute limb ischemia), lower-limb revascularization through endovascular techniques or bypass surgery (except for acute limb ischemia), or amputation due to PAD (excluding sepsis). Cardiovascular comorbidities such as coronary artery disease (CAD) and cerebrovascular disease (CVD) were also collected. Briefly, CAD was defined as the presence or history of myocardial infarction, acute coronary syndrome, coronary revascularization (previous angioplasty, coronary artery bypass graft), or presence of atheromatous stenosis >50% of coronary artery. Similarly, CVD was defined as the presence of at least one of the following criteria: prior ischemic stroke or transient ischemic attack, previous carotid thromboendarterectomy or carotid artery stenting, and carotid artery stenosis (>50%) detected with Doppler. Other comorbidities or risk factors (eg hypertension, smoking status, dyslipidemia, type-2 diabetes mellitus (DM), chronic kidney disease (CKD)) were collected using a combination of ICD-10-CM codes or direct capture in the medical records. Smoking status includes active and former smokers. CKD was defined by an estimated glomerular filtration rate ≤ 59 mL/min/1.73 m2 (stage ≥ III). Overall mortality data was collected from hospital medical records or through official registrations from the French National Institute of Statistics and Economic Studies, available under an open license.
Statistical Analysis
Data were reported as frequencies and percentages for categorical variables. To test significance between groups, variables were compared using independent Pearson

Flowchart. *Patients could have more than one reason for non-inclusion. SAVR: surgical aortic-valve replacement; TAVR: transcatheter aortic valve replacement.
Results
The overall included population was 1462 patients (median age: 75.0, IQR: 68.0-79.0, male gender:

Venn diagram representing the prevalence of PAD and its distribution with other vascular bed involvements. Data are in frequencies and percentages. PAD: peripheral artery disease; CAD: coronary artery disease; CVD: cerebrovascular disease.
Baseline Characteristics of the Study Cohort According to PAD Status.

PAD: peripheral artery disease; BMI: body mass index; CKD: chronic kidney disease; COPD: chronic obstructive pulmonary disease; OSAS: obstructive sleep apnea syndrome; NYHA: New York Heart Association; LV: left ventricular.
Population Characteristics and Factors Associated With PAD
Table 1 summarizes the demographic and clinical characteristics of the study population according to the presence of PAD. Patients with PAD were more frequently male and had a higher proportion of hypertension than those without PAD, 76.9% versus 59.1%,
The independent risk factors associated with PAD are shown in Table 2. The fully adjusted multivariate regression model showed that patients with PAD were at greater odds of being male (ORa = 2.15, 95% CI: 1.40-3.31,
Univariate and Multivariate Analysis Assessing Factors Associated with PAD.

OR: odds ratio; ORa: adjusted odds ratio; surgical aortic-valve replacement; PAD: peripheral artery disease; BMI: body mass index; CKD: chronic kidney disease; COPD: chronic obstructive pulmonary disease; OSAS: obstructive sleep apnea syndrome.
A similar model that included the variables CAD and CVD separately showed that CAD, CVD, CKD stage ≥ III, DM, dyslipidemia, and male gender were independently associated determinants of PAD (Appendix 1).
PAD and Long-Term Mortality
Over a median follow-up period of 7.6 years (IQR: 4.9-11.4), 710 patients (48.6%) died. This resulted in 5-year survival of 80.8% (95% CI: 78.7-82.8) and 10-year survival of 51.4% (95% CI: 48.6-54.4). Survival according to the PAD status is reported in Figure 3. Patients with PAD had significantly reduced survival compared to those without PAD. Respectively, the 5-year and 10-year survival were 69.1% (95% CI: 60.4-76.3) and 36.0% (95% CI: 27.1-45.0) for patients with PAD versus 81.9% (95% CI: 79.7-83.9) and 53.0% (95% CI: 50.0-56.0) for patients without PAD; log-rank

Kaplan–Meier analysis of long-term survival according to the presence of PAD. Log-rank test compare differences between the resulting curves. PAD: peripheral artery disease; HR: hazard ratio; CI: confidence interval.
Cox Proportional Hazards Model Assessing the Relationship Between PAD and Long-Term Overall Mortality.

HR: hazard ratio; HRa: adjusted hazard ratio; CI: confidence interval; PAD: peripheral artery disease; CAD: coronary artery disease; CVD: cerebrovascular disease; BMI: body mass index; CKD: chronic kidney disease; COPD: chronic obstructive pulmonary disease; OSAS: obstructive sleep apnea syndrome.
Survival according to the presence of PAD and CAD is reported in Figure 4. Patients without CAD but with PAD have significantly lower survival than those with neither CAD nor PAD. Respectively, the 5-year and 10-year survival were 72.0% (95% CI: 58.7-88.4) and 41.9% (95% CI: 27.1-64.9) for patients without CAD but with PAD versus 83.9% (95% CI: 81.3-86.6) and 57.7% (95% CI: 53.8-61.8) for patients with neither CAD nor PAD; log-rank
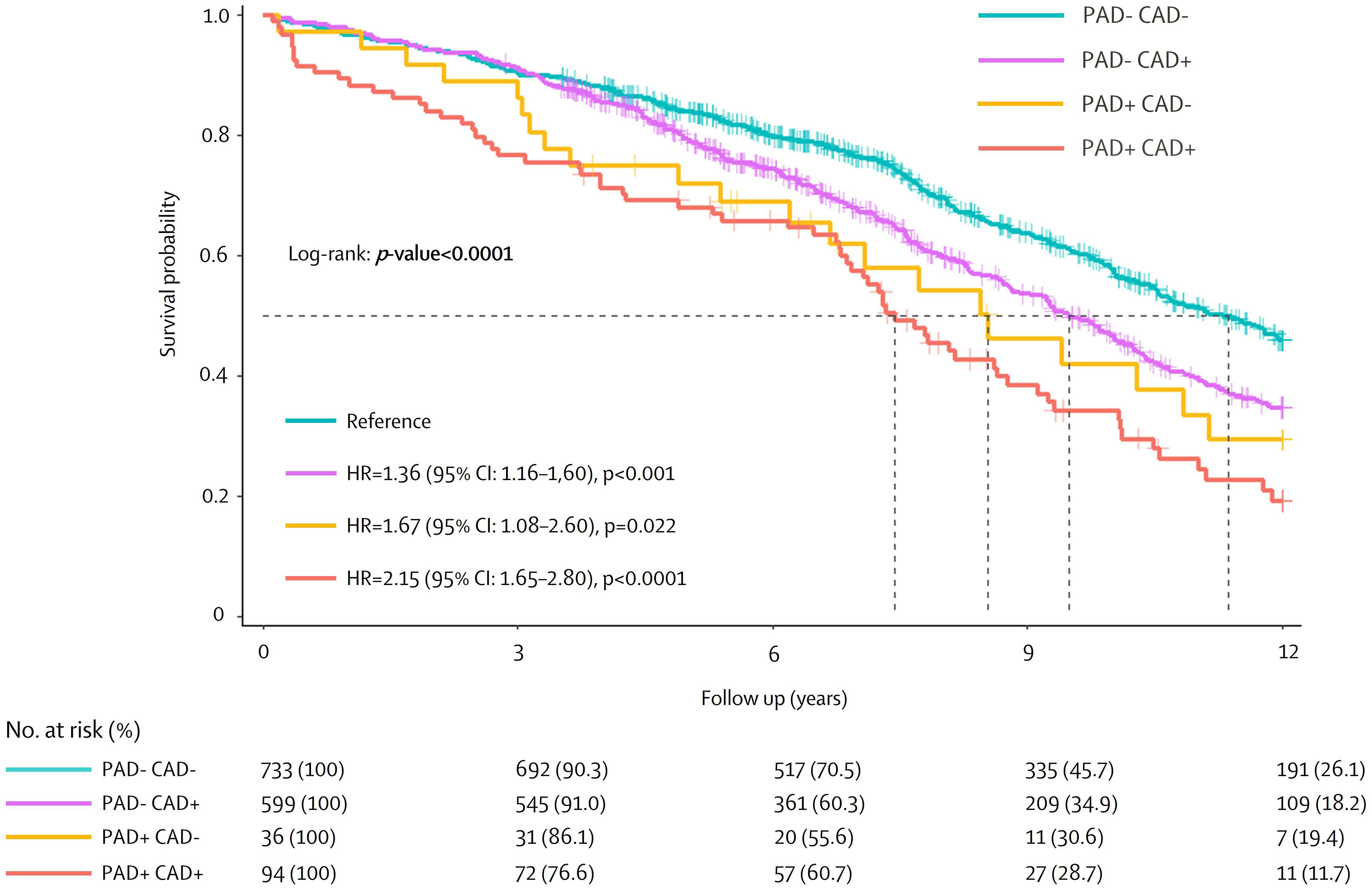
Kaplan–Meier analysis of long-term survival according to PAD and CAD status. Log-rank test compare differences between the resulting curves. PAD: peripheral artery disease; CAD: coronary artery disease; HR: hazard ratio; CI: confidence interval.
Cox Proportional Hazards Model Assessing the Relationship Between PAD/CAD Status and Long-term Overall Mortality.
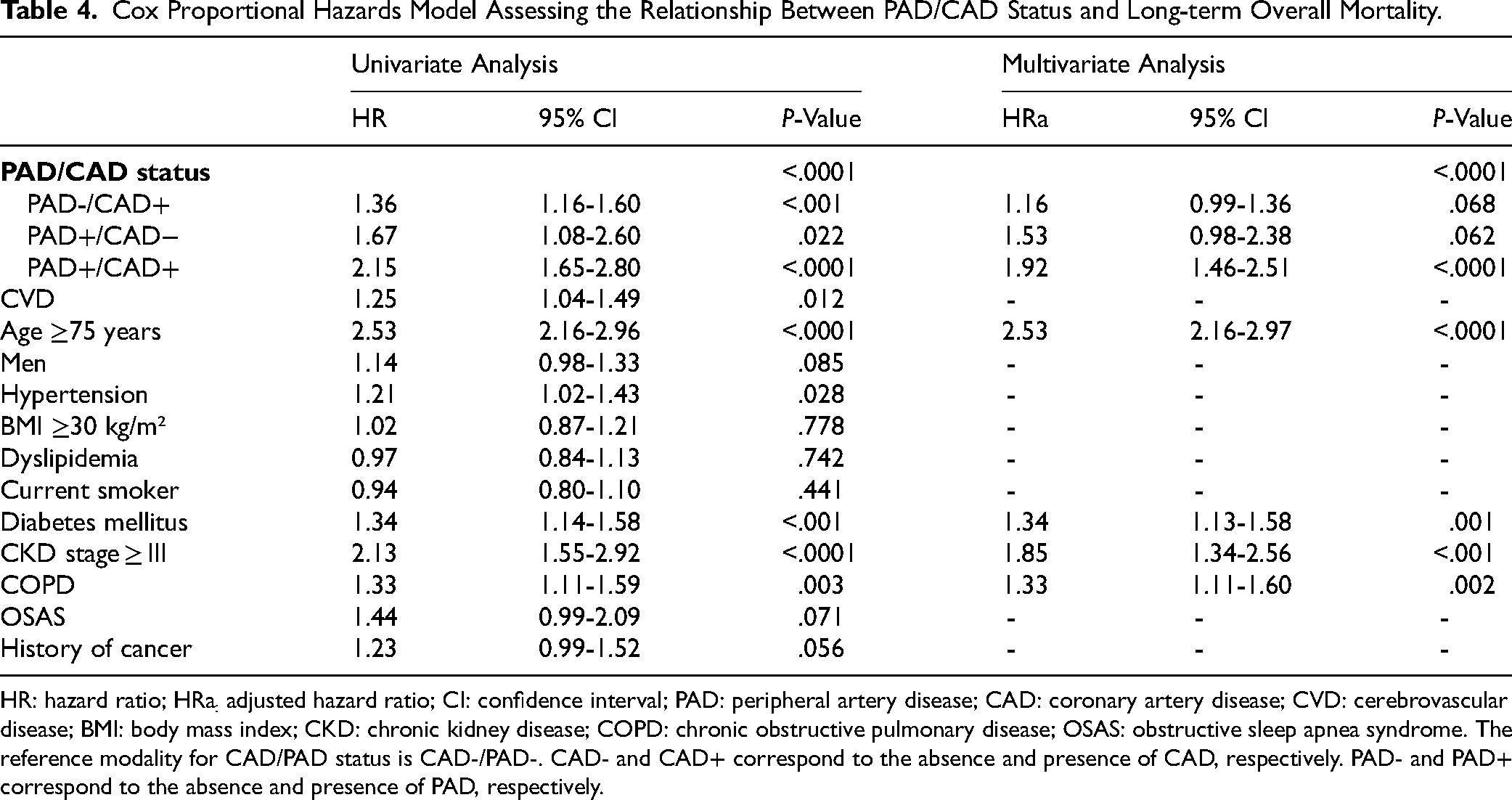
HR: hazard ratio; HRa: adjusted hazard ratio; CI: confidence interval; PAD: peripheral artery disease; CAD: coronary artery disease; CVD: cerebrovascular disease; BMI: body mass index; CKD: chronic kidney disease; COPD: chronic obstructive pulmonary disease; OSAS: obstructive sleep apnea syndrome. The reference modality for CAD/PAD status is CAD-/PAD-. CAD- and CAD+ correspond to the absence and presence of CAD, respectively. PAD- and PAD+ correspond to the absence and presence of PAD, respectively.
Patients who underwent concomitant CABG had lower long-term survival than those who did not undergo concomitant CABG (log-rank
The proportion of PAD was 5.2% in women and 11.3% in men. Men with PAD had significantly lower long-term survival than those without PAD (log-rank
Discussion
The main findings of this study involving patients with severe calcific AS were that (a) the prevalence of PAD was 8.9%, (b) the associated factors for PAD were male gender, dyslipidemia, type-2 DM, CKD at stage ≥ III and OSAS, and (c) patients with PAD had an independent 1.6-fold increase in risk of long-term overall mortality compared to patients without PAD. In addition, the risk of mortality was even more higher among patients with type-2 DM, regardless of CAD status.
Prevalence of PAD in AS
In this large-scale retrospective cohort, the prevalence of PAD in severe AS was lowest than previously reported by other studies.14,15 However, the vast majority of studies are retrospective or post-hoc analysis, with relatively small sample size and not specifically designed to investigate the prevalence of PAD in patients with AS. The lack of data from prospective studies with systematic screening for PAD limits its prevalence estimation. Furthermore, PAD is often investigated in selected patients receiving TAVR, ie, older patients with generally more comorbidities, in whom coexisting PAD may be more frequent. Altogether, these factors limit the accurate estimation of PAD prevalence’ from literature. However, in our study, the period of inclusion started in 2000. At that time, awareness about PAD screening was weak which may have lead to marked underestimation of PAD prevalence. Of note, PAD often coexists with other vascular conditions, such as CAD and CVD. 16 Among patients with PAD, 33.1% presented a triple bed affect, and, only 19% were free from CAD or CVD. It is important to emphasize that PAD may be seen as a marker of advanced and severe atherosclerotic disease. Hence, the presence of PAD in patients with AS should raise concerns about more harmful atherosclerotic disease with consequential outcomes. 17
PAD Associated Risk Factors in Severe AS
In this study, male gender, dyslipidemia and type-2 DM were associated with PAD among patients with severe AS. These factors are well-established risk factors and comorbidities associated with PAD among patients with AS.2,8 Indeed, type-2 DM is recognized to be more prevalent in patients with AS compared to the general population. 18 In addition, Arita et al. showed that PAD was independently associated with AS in subjects with CKD. 19 These associations may reflect a cardiovascular-kidney-metabolic syndrome (CKM) involving complex interrelations, especially in calcific AS. 20 Patients with severe AS include individuals with cardiovascular diseases and clinical expression such as CAD and PAD, ie, stage 4 CKM. However, hypertension, smoking status and obesity were not associated with PAD among patients with severe AS in our study. This may suggest that their potential associations with PAD in the context of AS have not been identified due to their integration within the CKM spectrum. To note, some studies show no association between obesity and PAD, or even an inverse relationship.7,8
Long-Term Mortality of Patients with PAD
In our study, patients with PAD had significantly lower overall survival with a subsequent 1.6-fold increased risk of mortality than those without PAD. These results are similar to two meta-analyses which showed that PAD is apparently an independent risk factor of mid-term mortality in severe AS.14,21 This over mortality among patients with PAD remained significant after adjustment for a range of traditional cardiovascular risk factors. Despite the considerable overlap of cardiovascular risk factors between PAD and other atherosclerotic diseases, PAD may present a greater susceptibility to the development of severe vascular occlusion. In addition, patients with PAD have a poorer prognosis with increasing severity of lower extremity vascular bed involvement. 22 The impact of PAD on mortality may reflect profound metabolic impairment and a greater burden on the cardiovascular system, supported by an advanced CKM. In our subgroup analysis, patients with PAD and simultaneous CAD had a significantly 2.2-fold increased risk of mortality. Importantly, patients with PAD but without CAD had also poorer long-term survival than patients with neither CAD nor PAD with a 1.7-fold increased risk of mortality (Figure 4). Indeed, it is reported that mortality is proportional to the severity of PAD, and above all, this is not modified by the presence of CAD.23,24 The American Heart Association also reported that patients with PAD but without CAD had a higher risk of mortality than patients with CAD but without PAD. 25 In our study, there was no significant difference in mortality between patients with PAD but without CAD and patients with CAD but without PAD. Furthermore, patients with PAD and concomitant CAD had the highest risk of mortality, confirming the results of the FOURIER trial. 26 It is worth noting that, in the overall population and subgroups, we also show an interaction exposing patients with PAD and concomitant type-2 DM to a subsequent 3-fold increased risk of long-term overall mortality. Indeed, PAD is closely related to type-2 DM and is an independent prognostic marker of mortality among patients with severe calcific AS.27,28
Finally, it remains of importance to note that patients with PAD but no CAD seem to have similar survival than those with CAD but no PAD. In patients with AS, CAD is a well-established risk factors for mortality and these patients are considered at high risk with subsequent more aggressive therapeutic strategies and investigations. In patients with PAD, there was no survival difference between patients who underwent SAVR with concomitant CABG and those who had isolated AVR. This result is consistent with findings from patients with PAD according to CAD status, and suggests that the impact of PAD on mortality is independent of coronary lesion. Our results emphasize that patients with PAD should also be considered at high risk and may benefit from screening and early management strategies similar to those for patients with CAD. Indeed, it may be beneficial to screen for PAD in patients with early stages of AS (ie, mild to moderate AS) to prevent adverse outcomes related to established risk factors and to enhance medical therapy.
Study Limitations
The main limitations are those inherent to a retrospective observational study, such as selection bias, unmeasured confounding variables and classification bias. The prevalence of PAD in this study is most likely underestimated compared to the actual prevalence of PAD in severe AS. Despite using the Limoges University Hospital database for screening patients undergoing SAVR and affecting by PAD using a large ICD-10-CM and ICHI codes, medical record rarely included a history of a lower extremity artery disease, an intermittent claudication or measurements of the ankle–brachial index. In addition, similar to previously published series, serial systematic screening was not performed in this cohort, limiting our estimation of the prevalence of PAD. The fact that this study focused solely on the SAVR constitutes a limit because of the expansion of TAVR. Since 2010, the role of TAVR in the treatment of severe AS has sharply risen. Besides, Limoges University Hospital was granted the right to perform TAVR in 2013 only. Thus, until 2013, which represents two-thirds of the period studied, all patients underwent SAVR for severe AS at our institution. Furthermore, the trigger for intervention (ie, indication for CABG, SAVR or both) is unknown in our database.
Additionally, patients who underwent SAVR may not fully represent the broader population of those diagnosed with severe AS. There are no specific ICD-10-CM diagnostic codes to identify patients with AS at severe stage. The only reliable method to identify patients with severe AS is to use medical classification codes for clinical procedures. Indeed, patients are referred by external cardiologists specifically for this procedure, with follow-up care at Limoges University Hospital initiated through pre-operative assessments. Importantly, to our knowledge, there are few studies investigating the impact of PAD on long-term mortality in severe calcific AS over an observation period of more than 10 years.
Conclusion
The findings of this study suggest that patients with PAD who are operated on for AS have increased long-term overall mortality compared to patients without PAD. This risk is even higher among patients with concomitant CAD and type-2 DM. These results suggest that patients with PAD should be considered at higher risk, and may benefit from specific management. Screening for PAD would be helpful in the early stages of AS, as the prevention of modifiable risk factors may be optimized and strengthened. The prognosis for patients with concomitant PAD and AS may improve only when these conditions are identified. Nevertheless, further studies are needed to investigate the efficacy of such a strategy.
Supplemental Material
sj-tif-1-hvs-10.1177_30494826241296674 - Supplemental material for Association and Prognostic Implications of Peripheral Arterial Disease and Calcific Aortic Stenosis
Supplemental material, sj-tif-1-hvs-10.1177_30494826241296674 for Association and Prognostic Implications of Peripheral Arterial Disease and Calcific Aortic Stenosis by M. Faure, J. Jouan, P. Lacroix, J.D. Blossier, P.M. Preux, L. Chastaingt, M. Boukhris, M. CanCané, G. Lux, V. Aboyans and J. Magne in Journal of the Heart Valve Society
Supplemental Material
sj-tif-2-hvs-10.1177_30494826241296674 - Supplemental material for Association and Prognostic Implications of Peripheral Arterial Disease and Calcific Aortic Stenosis
Supplemental material, sj-tif-2-hvs-10.1177_30494826241296674 for Association and Prognostic Implications of Peripheral Arterial Disease and Calcific Aortic Stenosis by M. Faure, J. Jouan, P. Lacroix, J.D. Blossier, P.M. Preux, L. Chastaingt, M. Boukhris, M. CanCané, G. Lux, V. Aboyans and J. Magne in Journal of the Heart Valve Society
Supplemental Material
sj-tif-3-hvs-10.1177_30494826241296674 - Supplemental material for Association and Prognostic Implications of Peripheral Arterial Disease and Calcific Aortic Stenosis
Supplemental material, sj-tif-3-hvs-10.1177_30494826241296674 for Association and Prognostic Implications of Peripheral Arterial Disease and Calcific Aortic Stenosis by M. Faure, J. Jouan, P. Lacroix, J.D. Blossier, P.M. Preux, L. Chastaingt, M. Boukhris, M. CanCané, G. Lux, V. Aboyans and J. Magne in Journal of the Heart Valve Society
Footnotes
Abbreviations
Acknowledgments
The authors wish to express their gratitude to G. Gschwind, M. Eymeri, and A. Rejasse at the Department of Medical Information, Limoges University Hospital (Limoges, France), for their assistance with access to the Hospital Database developed in the fields of medicine, surgery, and obstetrics, as well as the data extraction methodology. They also thank S. Juge at the Department of Research and Innovation, Limoges University Hospital (Limoges, France) for regulatory procedures.
Authors’ Contributions
MF, JM, and VA designed the study. MF drafted the first version of the manuscript. MF, GL, and MC collected and computerized all the data. MF and JM analyzed and interpreted the data. JJ, JDB, VA, PL, LC, and MB participated in the drafting of the final version of the manuscript. All authors read and approved the final manuscript.
Ethics Approval
The Limoges University Hospital's ethics committee approved the study protocol under contract number: 34-2023-05.
The database has been registered with the institution's research department under contract number: MR004/23-04-1.
This study is registered in the Limoges University Hospital's public registry for studies not involving human subjects, and an information poster regard the right to object to the reuse of health data has been created.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data that support the findings of this study are available on reasonable request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
