Abstract
Objective
This study aims to assess the incidence of prosthesis–patient mismatch (PPM), the hemodynamic performance, as well as the clinical outcomes of the Carpentier-Edwards Magna Ease (CEME) bioprosthesis in the aortic position while addressing sex-based disparities and variations in valve size.
Methods
Between 2012 and 2022, 1062 patients underwent an AVR with a CEME bioprosthesis in two high-volume cardiac surgery centers. Predicted PPM was assessed using manufacturer and Lancellotti et al. chart values, alongside measured values. Echocardiographic measurements were evaluated over a 5-year period.
Results
Predicted PPM based on the manufacturer chart was noted in 2.1% of women and 0.28% of men (P < .001). When employing definitions by Lancellotti et al., PPM was detected in 45% of females and 18% of males (P < .001). Notable differences were observed in terms of severe measured PPM in favor of male patients (F: 30%, M: 17%, P < .01). Regarding prosthesis sizes, the comparison of PPM from the manufacturer's chart with Lancellotti et al. values revealed significant disparities: 2% versus 58% in the 19 to 21 mm group, 0.27% versus 13% in the 23 mm group, and 0% versus 1.1% in the ≥25 mm valve sizes (P < .001). Measured severe PPM was encountered in 30% of patients with 19 to 21 mm prosthesis, 20% in the 23 mm group and 12% in the >25 mm prosthesis. Despite these differences, mortality rates remained comparable across sexes and valve sizes.
Conclusion
Female patients and those with 19 to 21 mm prostheses experience more severe PPM compared to men or individuals with larger valves. In this contemporary cohort, PPM does not impact short-term mortality.
This is a visual representation of the abstract.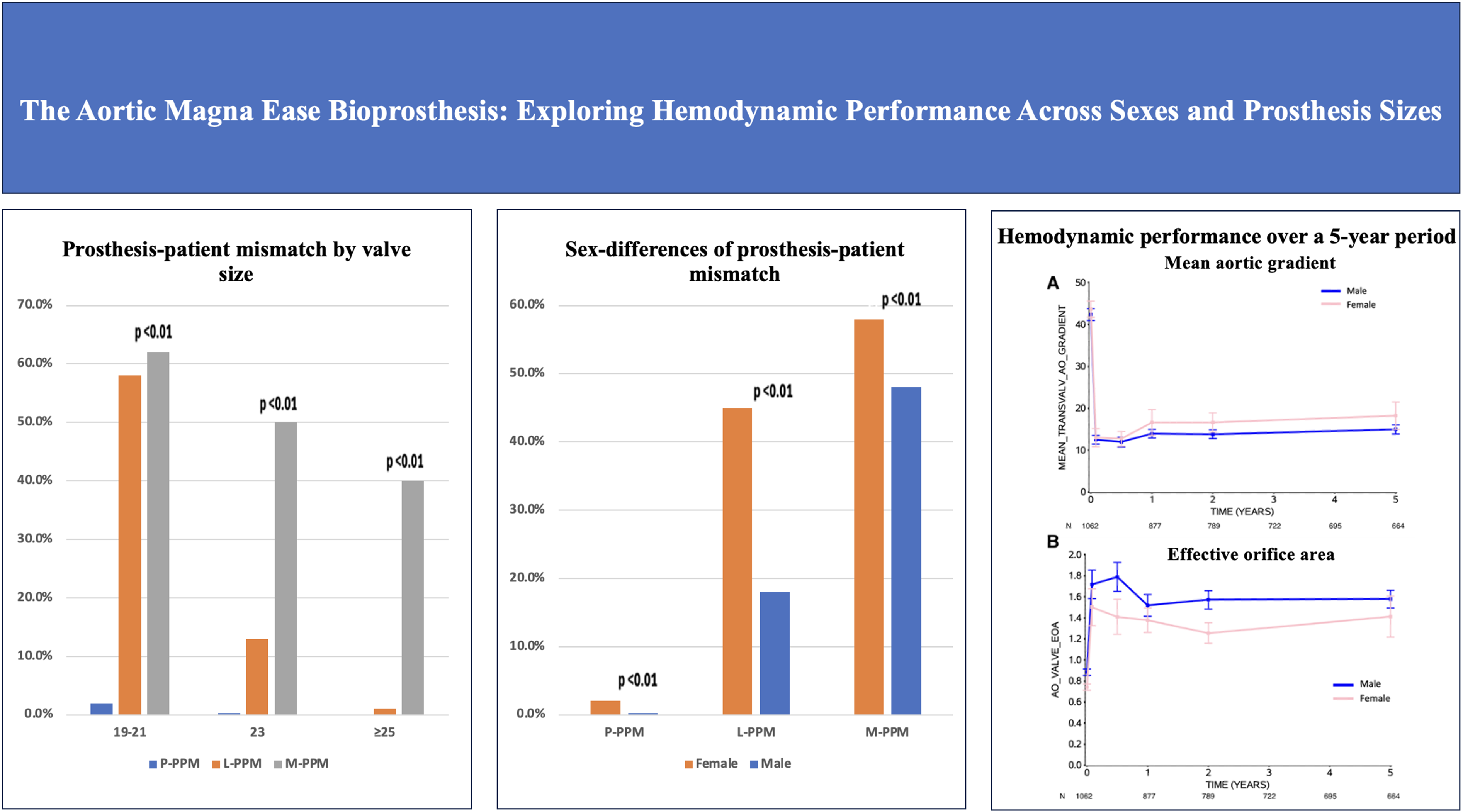
Keywords
Key Points
Female patients and those with small prostheses exhibit elevated aortic gradients, reduced effective orifice areas, and higher rates of prosthesis-patient mismatch. This mismatch is more frequent in women and in patients with smaller valves, compared to men or individuals with larger prostheses. A more proactive approach to annular enlargements in female patients and individuals with small annuli to achieve equivalent hemodynamics to male patients and larger prosthesis is needed.
Background
Aortic stenosis (AS) is one of the most prevalent form of valvular heart disease in the developed world with a prevalence of 0.2% among quinquagenarians, but increasing to 9.8% among octogenarians. 1 Aortic valve replacement (AVR) remains the gold standard treatment for severe AS to improve survival, quality of life and left ventricular function. There has been a notable rise in the utilization of bioprosthetic valves in recent years primarily due to their advantageous hematologic profile, increasing durability and the compelling potential for upcoming transcatheter valve-in-valve replacement. 2 Among the multiple prosthetic options available, the Carpentier-Edwards Magna Ease (CEME) bioprosthesis is one of the most frequently utilized heart valves.
While advancements in prosthetic valve technologies have significantly improved surgical outcomes, the phenomenon of prosthesis–patient mismatch (PPM) remains an important consideration, prompting a deeper exploration into its implications for the aortic CEME bioprosthesis. PPM, a phenomenon where the effective orifice area (EOA) of the prosthetic valve is smaller than the patient's physiological needs, has been recognized as a factor influencing postoperative outcomes.3–5 Earlier research has indicated that PPM can be anticipated during the operation with the projected indexed EOA (IEOA).6–8 Therefore, when comparing bioprostheses with similar durability, the preference should be for the model that provides the largest EOA relative to the patient's annulus size to prevent PPM and thereby minimize postoperative gradients.9,10
While the impact of PPM on postoperative outcomes is often documented, the influence of sex on surgical outcomes, particularly in the context of SAVR, remains a subject of debate and warrants further investigation. The majority of SAVR studies focused on short-term results leaving the impact of sex on other long-term outcomes unexplored. Some studies suggest that women may experience worse outcomes than men, while others find no difference between the sexes.11–13
Therefore, the primary objective of this study is to evaluate the incidence of PPM in AVR using the CEME bioprosthesis and its implications on clinical outcomes. The study also aims to examine potential variations in PPM incidence based on patient sex and valve size. Additionally, a comparison between predicted and measured PPM values will be conducted.
Methods
Study Population
This study included adult patients (≥18 years) who underwent AVR with a CEME bioprosthesis, including Bentall procedures, between 2012 and 2022. Additional inclusion criteria consisted of having echocardiography images at baseline, within 30 days post-surgery, and at least one longer-term follow-up echocardiography. Patients with pure aortic regurgitation (AR) were excluded. These criteria were fulfilled by 1062 patients. Retrospective data collection was conducted using harmonized databases from two high-volume cardiac surgery centers in the province of Quebec. These databases centrally collect all preoperative, perioperative, postoperative, and echocardiographic data in a prospective fashion. Approval for the study was obtained from the local ethics committee on November 23rd 2020 (IRB 20.261).
Study Endpoints
The primary endpoint is the incidence of moderate or severe PPM and was assessed using three methods: measured PPM (M-PPM) and predicted PPM based on the manufacturer's chart (P-PPM) and on a recently published chart by Lancellotti et al. (L-PPM). 5
Secondary endpoints included mortality, structural valve deterioration (SVD), valve thrombosis, permanent pacemaker implantation, endocarditis, stroke, acute renal failure, readmission, aortic valve reintervention, as well as mid-term hemodynamic data such as EOA, IEOA, mean aortic gradient (MAG), paravalvular leaks (PVL), and left ventricular ejection fraction (LVEF).
Measurement of PPM involved ultrasound assessment within the initial 30 days post-surgery, while echocardiographic measurements were monitored for up to 5 years postoperatively. Echocardiograms were conducted as routine follow-up or when a change in the patient's clinical status mandated it.
Definitions
Predicted PPM was determined based on EOA derived from both the manufacturer's chart provided and a chart published by Lancellotti et al. 5 The third method (M-PPM) involved measuring the EOA using the continuity equation: EOA = CSALVOT × (LVOTVTI ÷ AoVTI), where EOA is in cm2, CSA is the cross-sectional area of the left ventricular outflow tract (LVOT), LVOTVTI is the subaortic velocity-time integral and AoVTI is the aortic velocity-time integral, both in cm. Measured post-operative EOA values were available in 72% of echocardiography reports.
Moderate PPM is defined by an IEOA of 0.66 to 0.85 cm2/m2 and severe PPM by an IEOA ≤0.65 cm2/m2. Different thresholds were applied when the body mass index (BMI) exceeded 30, as per Lancellotti et al.'s publication, with IEOA values between 0.56 and 0.70 cm2/m2 classified as moderate and those ≤0.55 cm2/m2 as severe. 5
The Kidney Disease: Improving Global Outcomes definition was used to define acute renal failure which is characterized by an absolute increase in the serum creatinine level of 26.4 μmol/L or more or an increase in the serum creatinine level of more than 150% to 200%. The definitions of hemodynamically significant SVD, valve thrombosis and endocarditis were based on the VARC-3 criteria. 14
Statistical Analysis
Comparisons based on sex and valve size were conducted, with valve sizes categorized into three groups: small (19 and 21 mm), intermediate (23 mm), and large (≥25 mm). Continuous variables were expressed as mean ± standard deviation or median [interquartile range] and were compared using ANOVA or Kruskal–Wallis tests, as appropriate. Categorical variables were expressed as frequencies (%) and were compared using Pearson's χ2 test or Fisher's exact test, as appropriate. Mixed effects models were used to assess changes in echocardiographic measures over time and account for the correlation between repeated follow-up measurements (the MIXED procedure in SAS software, version 9.4; SAS Institute, Cary, NC). Piecewise linear random effect models using time as a continuous measure were constructed with time knots at baseline(preoperatively), 1 month, 6 months, 1 year, and 2 years. Estimates were obtained from a piecewise linear random effect model with a repeated measures statement with compound symmetry covariance. No random slopes were introduced in this model. Univariable analyses were conducted to determine whether PPM served as a predictor of mortality using a Cox proportional hazard model. A log-rank test was used to determine the freedom from SVD. Statistical significance was considered at P-values < .05.
Results
Among the 1062 patients included, 32.0% were female, and the average age of the cohort was 72 ± 9 years. Female patients were slightly older than men (F: 74 ± 8 years, M: 71 ± 9 years, P < .01). Males exhibited a larger body surface area (BSA) compared to women (M: 1.9 ± 0.2, F: 1.7 ± 0.2, P < .01). No significant differences were found in terms of BMI and the prevalence of bicuspid aortic valves between the two sexes. Eighty-one percent of women received 19 mm and 21 mm prostheses, whereas only 16.3% of men received the same sizes. Predicted operative mortality by the STS score was higher in the female group 2.96 ± 2.73% in comparison to 2.44 ± 2.99% in the male group (P < .01). The STS predicted mortality for the overall cohort was estimated at 2.61 ± 2.92%. Additional baseline characteristics are detailed in Table 1. The median echocardiographic follow-up was estimated at 2.4 years [0.0, 5.94].
Baseline Characteristics.

Values are expressed as n (%), mean ± SD and median [IQR]. Chronic renal failure as defined by estimated glomerular filtration rate (eGFR) less than 60 ml/min/1.73 m2, CABG: coronary artery bypass graft, CPB: cardiopulmonary bypass, EOA: effective orifice area, LVEF: left ventricular ejection fraction, NYHA: New York Heart Association.
Sex-Based Comparison
Hemodynamics and Prosthesis–Patient Mismatch
Analysis of hemodynamic parameters and PPM revealed notable sex-based differences and are shown in Table 2. According to the manufacturer chart, the P-PPM incidence was 2.1% among women and 0.28% among men. Utilizing Lancellotti chart definitions, L-PPM was identified in 45% of women and 18% of men. After adjusting for a BMI > 30, these values were halved in both groups. All patients presented with moderate PPM, with none demonstrating severe PPM. Female patients exhibited more severe M-PPM (30%) compared to males (17%), with a minimal difference noted in moderate M-PPM.
Short-Term Hemodynamics Data—Overall Cohort and by Sex.

Values are expressed as n (%) and mean ± SD.
BMI: body mass index; EOA: effective orifice area; PPM: prosthesis–patient mismatch.
Overall, women demonstrated higher MAG and smaller IEOA. Mild PVL were comparable between groups, while the LVEF was higher in females, albeit not clinically significant as both groups had a LVEF within the normal range (F: 59 ± 8, M: 54 ± 11, P < .01) (Table 2).
Clinical Outcomes
Clinical outcomes are showcased in Table 3. Procedural, 30-day, and 1-year mortality rates were similar between female and male patients. Males had a higher incidence of acute renal failure (M: 21% vs F: 14%, P < .01) and endocarditis (M: 6.7% vs F: 1.6%, P < .01). No significant differences were observed in stroke, valvular thrombosis, and permanent pacemaker implantation between the two sexes. Table 3 indicates that overall male patients experienced slightly more reinterventions compared to female patients (M: 5.7%, F 5.0%, P = .03). Female patients underwent more valve in valve procedures, while male patients had more surgical reinterventions, the reason being endocarditis.
Short-Term Clinical Outcomes Overall Cohort and by Sex.

Values are expressed as n (%).
SVD: structural valve deterioration; Viv: valve-in-valve; TAVR: transcathether aortic valve replacement.
Valve Size Comparison
Hemodynamic Characteristics and Prosthesis–Patient Mismatch
Table 4 presents the hemodynamic characteristics for each prosthesis size-groups. According to the manufacturer's chart, the incidence of P-PPM was 2% in the 19 to 21 mm valves, 0.27% in the 23 mm group, and 0% in the ≥25 mm group. Lancellotti chart values showed L-PPM in 58% of 19 to 21 mm prostheses, 13% of 23 mm, and 1.1% of ≥25 mm valves. Adjusting for BMI reduced mismatch but maintained the same trend across groups. Specifically, 33% of patients in the 19 to 21 mm group experienced mismatch, 1.6% in the 23 mm group, and none in the ≥25 mm group. All patients exhibited moderate PPM, with none demonstrating severe PPM. Moderate M-PPM was slightly higher in smaller prostheses compared to larger valves. However, a significant disparity was observed among the three groups in cases of severe M-PPM. Severe M-PPM occurred in 30% of cases in the small group, 20% in the intermediate group, and 12% in the large group.
Short-Term Hemodynamics Data—by Prosthesis Sizes.

Values are expressed as n (%) and mean ± SD.
BMI: body mass index; EOA: effective orifice area; PPM: prosthesis–patient mismatch.
The post-operative MAG was lower in larger valve sizes. PVL did not show a significant difference among the three groups. The LVEF was slightly higher in smaller prostheses but remained within the normal range across the three groups (Table 4).
Clinical Outcomes
Table 5 demonstrates that there was no significant difference in procedural and 1-year mortality among the three valve size groups. However, mortality at 30 days was higher in the smallest and largest valves compared to the 23 mm valves. The three groups were comparable in terms of other perioperative clinical outcomes. Readmissions for cardiovascular causes and reintervention rates were similar between groups (Table 5). Freedom from SVD is comparable between sexes across different valve size groups, as illustrated in Figure 1. At 5 years, female patients experienced freedom from SVD in 86.7%±3.3% of cases compared to 100%±0% for their male counterparts (P = .07) in the 19 to 21 mm group. In the 23 mm group, 100%±0% of females and 96.3 ± 1.5% of males (P = .18) were free from SVD. Lastly, in the large prosthesis group (≥25 mm), 100%±0% of females and 96.3 ± 1.9% of males (P = .74) had no SVD. The results for the 23 mm and ≥25 mm groups must be interpreted with caution due to the small number of female patients in those groups.

Freedom from SVD according to valve size group.
Short-Term Clinical Outcomes by Prosthesis Sizes.

Values are expressed as n (%).
SVD: structural valve deterioration; Viv: valve-in-valve; TAVR: transcathether aortic valve replacement.
Overall Hemodynamic Performance
Panel A in Figure 2 illustrates a marginal increase in the MAG during the first year, followed by a stabilization thereafter with higher MAG in females. In contrast, panel B depicts an inverse pattern for the EOA, with a slight decrease at 1 year and then maintaining stability up to 5 years with lower EOA in females. The LVEF remains consistently stable within the normal range throughout the 5-year period, as demonstrated in panel C.

Hemodynamic performance over a 5-year period.
In the univariable analysis, PPM was not identified as a predictor of mortality, whether assessed through the IEOA (HR 0.27 [0.06-1.32]) or the binary presence of PPM (HR 1.19 [0.56, 2.53]). Age was not shown to be a risk factor for mortality in this cohort (HR 1.02 [0.99-1.05]; P = .181). When comparing patients with and without PPM, those with PPM were more prone to have a reintervention, either surgical or a valve-in-valve procedure, as opposed to those without PPM (PPM: 7.3%, no PPM 2.8%, P < .01).
Discussion
To our knowledge, this is the first study on the CEME prosthesis reporting on sex-based and size-based differences in hemodynamic performance and clinical outcomes. Few valve-related complications were encountered, which is consistent to the numbers previously published in CEME series, ascertaining the safety of this prosthesis.15–17 The same applies to MAG and EOA which remained stable over a 5-year period.15,16 The procedural contemporary and 30-day mortality rates were comparable to those published in a series of similar size to the present one. 15 The operative mortality was well under the STS-predicted mortality (Tables 1, 3, and 5).
Sex-Based Comparison
Female patients receiving a CEME prosthesis presented a higher incidence of severe PPM, consistent with findings by Astudillo et al., where 82% of women had severe mismatch but lower rates of moderate PPM compared to men. 18 Despite the increased mismatch in female patients, mortality was unaffected, though SVD had a tendency of being higher in female patients with small prosthesis without attaining statistical significance. The impact of PPM on mortality in SAVR remains controversial. Multiple studies reported that the presence of PPM does not influence mortality.19–21 Das De et al. have not found a significant difference in patient survival when patients with moderate PPM, severe PPM and no PPM were compared, while Moon and colleagues came to the same conclusion only in patients >70 years of age.19,21 However, according to a Swedish group, severe PPM is associated with an increase in long-term mortality and an increase in heart failure hospitalizations. 22 The increased residual gradient that can be observed in patients with PPM can lead to less LV mass regression and quicker prosthesis degeneration, explaining the worse outcomes observed in patients with PPM. 23 A meta-analysis of 34 observational studies supports those findings with an increase of all-cause and cardiac-related mortality. 24 In contrast to this recently published study by Dismorr et al., which found no difference in in aortic valve reintervention between patients with and without PPM, our study found a higher proportion of reinterventions in those with PPM. 22 This aligns with the findings of a systematic review and meta-analysis of over 60 studies. 25 Our study noted a higher prevalence of coronary artery disease and concomitant coronary artery bypass graft (CABG) surgery in men, potentially contributing to equivalent mortality rates between sexes despite the higher incidence of severe PPM in women. The trend of higher BMI in women, along with a greater proportion receiving 19- to 21-mm prostheses, contributed to increased PPM. Higher BMI has been linked to increased PPM in other studies26,27 and these findings suggest potential over-indexation of EOA in obese patients, leading to an overestimation of PPM severity in this population. Consequently, BMI-adjustment is suggested.
Valve Size Comparison
The incidence of severe PPM was significantly higher in the 19 to 21 mm prostheses, consistent with findings by other authors.17,28 Despite these findings, aortic valve reintervention rates were similar between the 3 groups. Luthra and colleagues demonstrated poorer survival in patients with smaller valves and with moderate or severe PPM. 28 The authors also determined that valves ≤ 21 mm were predictors of moderate or severe PPM. 28 Interestingly, Yen et al. did not find a difference in PPM, but observed higher MAG in small prostheses with no impact on mortality across different valve sizes, aligning with our results. 29 A sub-analysis of the SURTAVI trial was performed comparing self-expandable TAVR to SAVR to ascertain if differences in hemodynamic performance and clinical outcomes are present when comparing individuals divided into 3 groups: small, medium and large annuli. They concluded that annulus size is a predictor of PPM and that TAVR is associated with lower rates of PPM across all 3 groups. 30
Since TAVR is associated with a lower prevalence of severe PPM compared to SAVR 30 patients with a small annulus may be better served by TAVR if the surgeon is not prepared to do an aortic annulus enlargement, especially in women, in whom a disproportionate benefit with TAVR has been noted. 31 Unfortunately, data on annular enlargements was not available in our database, but we estimated that at that time it would represent approximately 5% of the entire cohort.
Measured Versus Predicted PPM
Our study highlights that measured EOA values tend to be smaller than those from the commercial chart, leading to an overestimation of PPM. Factors such as low flow, the pressure recovery phenomenon, and inter/intra-observer variability contribute to this discrepancy. The presence of low flow is common both before and after AVR, preventing the valve leaflets from fully opening. This can result in a small measured EOA, potentially leading to the incorrect conclusion that severe PPM is present. The pressure recovery phenomenon might also be responsible for the overestimation of the mismatch. Continuous-wave Doppler measures maximum velocity anywhere along the axis of interrogation and cannot take into account pressure recovery. The precision of the echocardiographic measurement of the LVOT can be affected when a prosthetic valve is present due to the shadowing caused by the prosthesis. As a result, the measured values likely overestimate the presence of PPM, while the manufacturer chart tends to underestimate it. The accurate assessment is likely somewhere in between, with the Lancellotti chart and BMI adjustment appearing to be closer to real values. Indeed, predicted EOAi has been shown to be superior to predict hemodynamic and clinical outcomes following AVR. 32
Limitations
This is a retrospective observational study from two cardiac surgery centers. Potential inter-observer variability in measured echocardiographic values is possible, and different intervals for follow-up echocardiography may affect analyses. Additionally, data on annular enlargements were unavailable, and a portion of patients had follow-up conducted at other institutions, unless patients necessitated the expertise of a tertiary-care center, limiting longer-term echocardiographic assessment. Finally, the results presented are at relatively short-term. Event rates or durability may vary at a longer follow-up. This is first and foremost an echocardiographic monitoring database and the clinical data does not seem reliable after 1 year due to patients being followed by cardiologists in other hospital centers, so only 1-year clinical data was included.
Conclusions
The CEME valve offers stable hemodynamics up to 5 years. Female patients and those with 19 to 21 mm valve prostheses experience more severe PPM. Female patients with 19 to 21 mm prosthesis showed a trend towards increased SVD without attaining statistical significance. Measured echocardiographic data tends to overestimate PPM severity compared to predicted values. Adjusting PPM thresholds is recommended for patients with a BMI >30. Finally, PPM in our cohort does not appear to impact short-term mortality. Age was not deemed a risk factor for mortality. Extended follow-up would provide valuable insights into the durability and hemodynamic performance, particularly among female patients and in cohorts with smaller valve prostheses.
Footnotes
Abbreviations
Ethics Committee Approval
IRB 20.261, approved on November 23rd 2020.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Edwards Lifesciences.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Kalavrouziotis is a proctor for Edwards Lifesciences. Dr. Forcillo is a proctor, consultant, speaker and part of the advisory board for Edwards Lifesciences, Medtronic and Terumo. All the remaining authors do not have any disclosures.
Correction (July 2025):
The article has been updated to remove the “Central Message” and “Perspective Statement” sections. Their content has been consolidated under the “Key Points” section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
