Abstract
Introduction:
Obesity is a growing global challenge with rising prevalence and incidence rates associated with economic burdens. Despite the presence of a variety of approaches to managing obesity, care remains fragmented, and the transparency of care methodologies used by obesity providers remains limited. Hybrid models of care, which blend both digital tools and in-clinic visits, offer a promising and patient-centered approach. However, the lack of outcome-reporting standards remains a limitation. This study evaluates the effectiveness of Semaglutide and Tirzepatide in managing obesity using the newly developed International Consortium for Health Outcomes Measurement (ICHOM) Obesity standard set.
Methods:
This study employed a retrospective study design including 528 adults enrolled in a 6-month hybrid care model (meta[bolic], Zone.Health), to manage obesity. Participants were prescribed either a glucagon-like peptide (GLP)-1 receptor agonist (Semaglutide) or dual GLP-1/GIP receptor agonist (Tirzepatide) as part of the program alongside behavioral monitoring and virtual engagement. Outcomes were collected based on the ICHOM Obesity standard set from medical records at baseline, 3 months, and 6 months, and were analyzed using mixed for repeated measures. Subgroup analyses based on dosage bands were done using the Kruskal-Wallis test.
Results:
Both groups demonstrated significant improvements in weight, body mass index, waist circumference, fat mass, and glycemic control. However, within 6 months, on average, the Tirzepatide group had a greater reduction in weight (13.15 kg), waist circumference (11.34 cm), and HbA1c (0.33%) when compared with the Semaglutide group, with average reductions as 9.54 kg, 7.24 cm, and 0.23%, respectively. In the context of Tirzepatide dosage, all three groups (2.5–5 mg, 5–7.5 mg, and 7.5–15 mg) experienced significant weight loss.
Conclusion:
This study is among the first to apply the ICHOM Obesity Standard Set to evaluate Semaglutide and Tirzepatide in managing obesity within a hybrid care model. Tirzepatide was associated with superior clinical outcomes when compared with Semaglutide. The integration of standardized outcome reporting enhances transparency and comparability of hybrid interventions. Future studies should explore cost-effectiveness and long-term outcomes to support broader adoption.
Keywords
Background
Obesity and overweight are considered one of the most demanding public health challenges, with their prevalence rates rising at an unprecedented pace. While overweight is merely defined as an excess of fat deposits, obesity has a more complicated measure. According to the World Health Organization, obesity is defined as a chronic condition where excessive fat accumulation can impair the overall health of individuals and put them at risk of many more comorbidities, with body mass index (BMI) cut-off points set at 25 kg/m2 for overweight and 30 kg/m2 for obesity. 1 As of 2022, one in eight people are living with obesity worldwide. 1 To further elaborate on this, 2.5 billion adults were classified as overweight, and of these, 890 million fit the obesity criteria. 1
Specifically in the United Arab Emirates, according to a study published in 2023, 39.8% of residents of Dubai were overweight, and 17.8% of all residents were obese. 2 The shift in socioeconomic status and rapid urbanization in many developing countries, including the UAE, has been correlated with the rising prevalence of obesity. 3 This is mainly due to the increased adoption of sedentary lifestyles and unhealthy eating behaviors. 4
Obesity is one of the main risk factors for a range of noncommunicable diseases, including cardiovascular diseases, type 2 diabetes, hypertension, and in severe cases, cancer.1,4 The increase of these diseases not only burdens individual health but also exerts an enormous strain on health care systems, quality of life, as well as workforce productivity across populations.5,6 According to the World Obesity Federation, the economic burden of obesity and overweight is estimated to reach 4.32 trillion dollars annually by 2035, with 51% of the population being overweight or obese. 7
Despite the increasing recognition of the obesity epidemic, clinical care remains inconsistent with treatment approaches. Treatment options in all aspects, ranging from lifestyle modifications, pharmacological interventions, behavioral tactics, and even surgeries, remain varied. Conventional methods, such as BMI measurements and weight loss strategies, although helpful, do not adequately reflect the complex nature of obesity and its effects on individuals’ lives. 8 These measures, including mental health, treatment satisfaction, and long-term sustainability, are often neglected, although they are crucial aspects of care.
The rapid expansion of digital health technologies has initiated the emergence of hybrid care that integrate in-clinic visits with remote digital monitoring. 9 Several studies have evaluated the use of these models and reported an improvement in clinical outcomes, including glycemic control, quality of life, as well as cardiovascular risk.10–14 Specific to the obese population, a study explored the role of hybrid care model combined with pharmacotherapy and lifestyle interventions and reported significant decreases in waist circumference, triglycerides, and blood pressure. 15 However, despite the promise of digital health, not all implementations have yielded favorable outcomes. The Peterson Health Technology Institute’s 2023 report critically evaluated digital diabetes management solutions and found limited evidence supporting their cost-effectiveness and long-term clinical impact. 16 Many digital platforms failed to produce sustained improvements in glycemic control when deployed as standalone interventions, raising concerns about their real-world utility. Hybrid care models have the potential to overcome these shortcomings by integrating the accountability, clinical oversight, and continuity of traditional care with the scalability and personalization of digital tools. Unlike purely digital solutions, hybrid care models enable in-clinic interactions, dynamic monitoring, timely clinical intervention, and multidisciplinary support, factors that are especially critical in managing complex conditions like obesity. However, the success of these models relies on the ability to consistently measure and compare outcomes across various settings.
To ensure the consistent evaluation of such outcomes across diverse clinical settings, the implementation of standardized outcome frameworks is essential. In 2024, the International Consortium for Health Outcomes Measurement (ICHOM) released standardized, meaningful, and inclusive outcome measures specific to obesity. 17 The importance of adopting such standards is further underscored by the rapid rise in the use and cost of antiobesity medications in recent years. Despite their demonstrated clinical benefits, these treatments remain largely excluded from insurance coverage in many regions, and their long-term return on investment is often questioned due to the high likelihood of weight regain following treatment discontinuation.18,19 This rebound effect, combined with the financial burden of pharmacotherapy, highlights the need for transparent and standardized outcome reporting to justify treatment strategies and inform policy decisions.
Transparent, outcomes-based evidence is essential to support payer engagement, advocate for reimbursement inclusion, and ensure accountability in clinical practice. Standardized sets such as ICHOM’s provide a critical pathway for consistent, credible, and patient-relevant reporting across care models. This study is among the first to report obesity-related outcomes using the ICHOM Obesity Standard Set to evaluate the effectiveness of a hybrid care model. By aligning with this global framework, the study contributes to a more advanced and evidence-driven approach to obesity management. This study aims to report obesity outcomes using the ICHOM obesity standard and assess the difference in effectiveness between dual and single glucagon-like peptide (GLP)-1 receptor agonists in a hybrid care model for managing obesity to contribute to a more advanced approach to obesity care.
Methodology
The meta[bolic] Program (branded as Zone.Health)
The meta[bolic] weight loss program (Zone.Health) is a 6-month, technology-enabled, hybrid model of obesity care launched in 2023 (www.metabolic.health). This structured program combines traditional in-clinic care with continuous digital support to deliver hyper-personalized, multidisciplinary obesity management. Each participant is engaged through a hybrid care model framework that integrates remote monitoring, real-time data sharing, and bi-weekly virtual coaching with a team of physicians, dietitians, sports scientists, and health coaches.
Individualized care plans are developed based on patients’ clinical profiles, metabolic status, and behavioral needs, with dynamic adjustments made throughout the program. The hybrid structure facilitates high-touch, continuous engagement, ensuring timely medication titration, lifestyle reinforcement, and psychosocial support. As patients progress, touchpoints are tapered to monthly follow-ups, maintaining accountability while supporting long-term sustainability.
Participants are provided with access to a mobile application for food logging, behavioral tracking, and integration of biometric data from continuous glucose monitors, activity trackers, and sleep monitoring devices. The data from these wearables were limited to patient support and not reported in the analysis. This real-time feedback loop allows the care team, including physicians, clinical dietitians, lifestyle coaches, sports scientists, and cardiology specialists, to proactively intervene, personalize education, and optimize outcomes. Each team member monitors specific parameters: glucose levels, dosing, body composition, sleep quality, stress, activity, and cardiovascular markers, as part of a hybrid care model previously described in New England Journal of Medicine Catalyst. 13 The hybrid care model thus provides a robust infrastructure for delivering pharmacotherapy within a behaviorally supportive and outcomes-driven ecosystem.
Study design
A retrospective study looked at 528 individuals who were on the program for 6 months with complete datasets and were prescribed Semaglutide or Tirzepatide as part of their treatment. Before starting the medication, participants underwent a thorough evaluation and dietary assessment. Medications were chosen based on medical appropriateness and included options like Semaglutide (Ozempic® or Wegovy®), Tirzepatide (Mounjaro®). Although allocation was not randomized, baseline demographic and metabolic characteristics were broadly comparable. No significant age or HbA1c differences were observed. Only participants with complete 6-month data were included in the analysis. Those without full follow-up records were excluded prior to analysis. All participants signed up for a value-based contract, committing to regular engagement and data sharing. Compliance was high, with everyone meeting the program’s criteria for interaction and data submission.
Titration approach using monthly in-clinic body composition analysis
GLP-1 titration in this program was guided by monthly in-clinic body composition analysis rather than weight alone. These assessments allowed for regular evaluation of changes in fat mass, skeletal muscle mass, and overall body composition. Medication dosing decisions, including whether to escalate, maintain, or adjust, were based on these objective metrics in combination with patient-reported tolerability. For Semaglutide, doses were typically initiated at 0.25 mg and titrated up to a maximum of 2.4 mg once weekly (Ozempic® up to 2 mg or Wegovy® up to 2.4 mg), depending on the specific indication and patient response. For Tirzepatide, dosing was initiated at 2.5 mg and could be escalated in 2.5 mg increments every 4 weeks up to a maximum of 15 mg once weekly (Mounjaro®). The dosage bands used in this study (Semaglutide: 0.25–0.5 mg, 0.5–1 mg, and 1–2.4 mg; Tirzepatide: 2.5–5 mg, 5–7.5 mg, and 7.5–15 mg) reflect these real-world titration protocols and were used to evaluate dose-dependent treatment effects. This approach aimed to support appropriate weight loss trajectories while monitoring for unintended reductions in lean mass, thereby facilitating individualized and data-informed treatment adjustments throughout the program. However, the dropout rate and the side effects associated with the dosage were not collected.
Gathering data and participants
Data were extracted from patients’ records both at baseline and of the 6 months, utilizing the electronic medical record system. Information pertaining to patients’ gender, age, medications, weight, and BMI was recorded. Laboratory variables including lipid profile (total cholesterol, low-density lipoprotein [LDL], high-density lipoprotein [HDL], and triglycerides), and HbA1c were collected. Assessment of fat mass and skeletal muscle mass was conducted utilizing a body composition analyzer. A substantial portion of the ICHOM-defined metrics, such as anthropometric, metabolic, and liver-related measures, were already being captured prior to implementation. Additional data elements, including patient-reported outcomes and other parameters specified by the ICHOM framework, are currently being integrated into the clinic’s ongoing data collection process to ensure full alignment with the standard set moving forward.
Ethical approval
Ethical approval was obtained from the Dubai Health Authority (DHA) (Ethical approval number DSREC-02/2025_09), and patient data were anonymized to maintain confidentiality. Written informed consent was obtained from all patients during their initial visit.
Statistical analysis
Baseline demographic and clinical characteristics of participants in the Semaglutide and Tirzepatide groups were summarized. Categorical variables were presented as frequencies and percentages, while continuous variables were reported as means with standard deviations (SD) for normally distributed data or as medians with interquartile ranges (IQR) for non-normally distributed data.
Changes from baseline in anthropometric, body composition, glycemic, and lipid parameters at 3 and 6 months in both groups were analyzed using a mixed model for repeated measures. Results were presented as estimated marginal means with corresponding 95% confidence intervals (CIs) for continuous outcomes. The absolute and relative changes in body weight and waist circumference from baseline to 6 months were evaluated among overweight and obese patients across the different medication groups. The 95% CIs are not adjusted for multiple comparisons. Participants were also categorized based on average dosage bands, and the absolute changes in the anthropometric and body composition parameters were assessed using the Kruskal–Wallis test, and a two-sided P value of <0.05 was considered to indicate statistical significance. All statistical analyses were done using R version 4.4.0.
Results
Basic demographics and baseline characteristics
At baseline, the demographic and clinical characteristics of participants were generally comparable between the (n = 120) and Tirzepatide (n = 408) groups (Table 1). The mean age was similar across both groups (41.07 ± 9.11 years vs. 41.13 ± 9.51 years). A higher proportion of females was observed in both groups. Participants in the Tirzepatide group exhibited higher mean body weight, BMI, waist circumference, and fat mass. HbA1c levels were similar in both groups (5.37 ± 0.38 vs. 5.37 ± 0.41), suggesting comparable glycemic status at baseline. Lipid profiles also showed minimal differences, with slightly lower mean LDL and total cholesterol levels in the Tirzepatide group. Median triglyceride levels were higher in the Tirzepatide group (118.05 mg/dL) than in the Semaglutide group (105.2 mg/dL).
Baseline Demographic and Clinical Characteristics of Participants in the Semaglutide and Tirzepatide Groups

Anthropometric and body composition outcomes
At 3 months, the Tirzepatide group achieved a mean weight reduction of 8.28 kg (95% CI: 7.73, 8.83), compared with 6.62 kg (95% CI: 5.61, 7.63) in the Semaglutide group. By 6 months, this difference widened further, with a reduction of 13.12 kg (95% CI: 12.19, 14.05) in the Tirzepatide group versus 9.54 kg (95% CI: 7.84, 11.24) in the Semaglutide group. BMI decreased by 4.59 units (95% CI: 4.27, 4.91) in the Tirzepatide group and 3.40 units (95% CI: 2.81, 4.00) in the Semaglutide group at 6 months. Waist circumference reduction was more pronounced in the Tirzepatide group (11.34 cm; 95% CI: 8.95, 13.73) than in the Semaglutide group (7.24 cm; 95% CI: 2.96, 11.52). Fat mass reduction at 6 months was 9.67 kg (95% CI: 8.83, 10.52) for the Tirzepatide group compared with 7.54 kg (95% CI: 6.00, 9.08) for the Semaglutide group (Table 2).
Changes from Baseline in Anthropometric and Body Composition Parameters at 3 and 6 Months in the Semaglutide and Tirzepatide Groups (Mean Change with 95% CI)

Patients receiving Tirzepatide exhibited consistently greater reductions in both absolute (kg) and relative (%) weight compared with those on Semaglutide treatment. This difference was more pronounced at the 6-month mark, particularly among obese individuals. At 6 months, overweight individuals receiving Tirzepatide therapy had greater weight loss (−11.1 kg, −13.7%) and waist circumference reduction (−7.74 cm, −8.37%) compared with those on Semaglutide (−9.25 kg, −11.8%; −8.06 cm, −8.4%). In obese patients, Tirzepatide also led to greater weight loss (−13.8 kg, −13.2%) and waist circumference reduction (−10.2 cm, −9.29%) than GLP-1 alone (−11.3 kg, −11.8%; −9.46 cm, −8.79%) (Fig. 1).

The mean absolute (left panel) and mean relative (right panel) changes in body weight and waist circumference over time (baseline, 3 months, and 6 months) among overweight and obese patients, stratified by treatment groups: Semaglutide treatment (black) and Tirzepatide (blue).
Gender-based subgroup analysis
To evaluate whether gender differences influenced treatment response, a subgroup analysis stratified by gender was performed. Supplementary Tables S1 and S2 present changes in anthropometric and body composition outcomes for male and female participants, respectively. Across both sexes, participants receiving Tirzepatide showed greater improvements in weight, BMI, waist circumference, and fat mass compared with those receiving Semaglutide. Detailed gender-specific results are provided in Supplementary Tables S1 and S2.
Comparative glycemic and lipid responses between treatment groups
Both Semaglutide and Tirzepatide groups exhibited favorable changes in glycemic and lipid profiles over the 6-month period, with the Tirzepatide group generally showing greater improvements (Table 3). Glycemic control, as measured by HbA1c reduction, was more pronounced in the Tirzepatide group, with a decrease of 0.28% at 3 months and 0.33% at 6 months, compared with 0.19% and 0.23%, respectively, in the Semaglutide group.
Changes from Baseline in Glycemic and Lipid Parameters at 3 and 6 Months in the Semaglutide and Tirzepatide Groups

Improvements in lipid parameters were also noted. LDL cholesterol levels decreased in both groups, with slightly higher changes seen in the Tirzepatide group at 3 months (17.89 mg/dL; 95% CI: 13.73, 22.10) and similar changes at 6 months. Total cholesterol also decreased in both groups, with the Tirzepatide group showing a 22.23 mg/dL decrease at 3 months and a 20.25 mg/dL at 6 months. Triglyceride levels depicts more decrease in the Tirzepatide group at both timepoints, with a 6-month decrease of 35.55 mg/dL (95% CI: 19.38, 51.70) compared with 30.19 mg/dL (95% CI: 0.44, 59.90) in the Semaglutide group.
Dose–response analysis: Changes in anthropometric and body composition parameters by dosage
Participants were stratified into dosage groups, and outcomes such as weight, BMI, waist circumference, fat mass, fat percentage, and skeletal muscle mass were compared (Table 4).
Change in the Anthropometric and Body Composition Parameters with Respect to the Average Dosage Bands from Baseline to 3-Month Among the Semaglutide and Tirzepatide Group
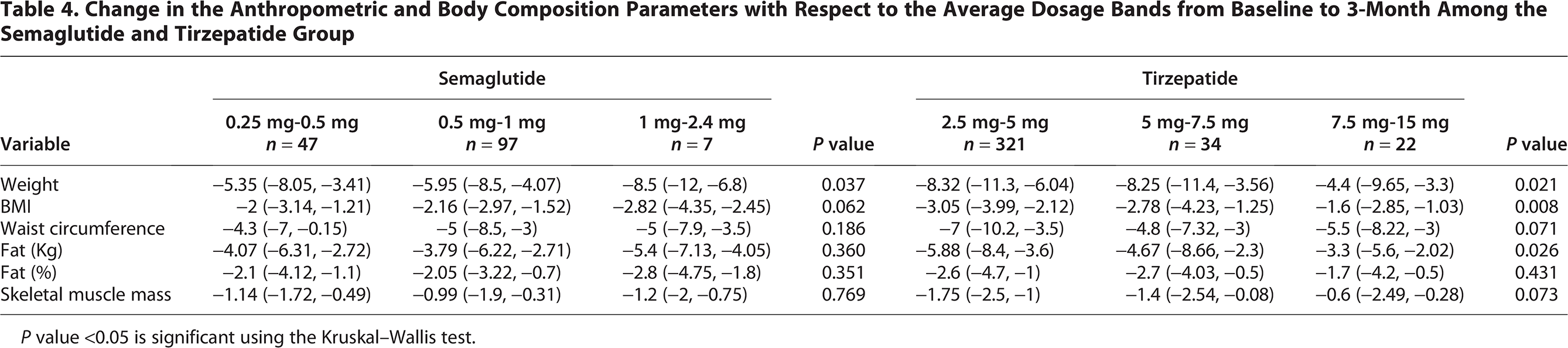
P value <0.05 is significant using the Kruskal–Wallis test.
In the Semaglutide group demonstrated a statistically significant reduction in weight was demonstrated across dosage bands (P = 0.037), with median weight change ranging from −5.35 kg (IQR: −8.05 to −3.41) in the 0.25–0.5 mg group to −8.5 kg (IQR: −12 to −6.8) in the 1–2.4 mg group. Pairwise comparisons confirmed that the reduction was significantly greater between these two dosage bands (P = 0.035), indicating a dose-dependent trend.
Similarly, the Tirzepatide group showed significant differences in weight loss across dosage levels (P = 0.021), with the greatest reduction observed in the 2.5–5 mg group (−8.32 kg, IQR: −11.3 to −6.04), followed by −8.25 kg (IQR: −11.4 to −3.56) in the 5–7.5 mg group. Interestingly, the highest dose group (7.5–15 mg) exhibited a smaller median weight reduction (−4.4 kg, IQR: −9.65 to −3.3), suggesting a plateau or potential diminishing returns at higher doses. It is important to note that the apparent plateau at higher Tirzepatide doses likely results from small subgroup size rather than a pharmacodynamic ceiling.
Discussion
This study demonstrates the effectiveness of Tirzepatide compared with Semaglutide receptor agonists (Semaglutide) when administered within a structured, digitally enabled hybrid care model for obesity management over both 3- and 6-month periods. Most obesity programs do not measure extensive metabolic parameters and simply focus on BMI, and hence the adoption of the ICHOM Obesity Standard Set as a structured framework is used to enhance outcome reporting and promote benchmarking across care models. 17 The findings indicate that Tirzepatide results in significantly greater improvements across multiple outcomes, especially in weight loss, body composition, and glycemic control. Baseline differences in adiposity may explain part of the larger absolute reductions; however, relative percentage changes confirm consistent superiority of Tirzepatide.
These outcomes are interpreted within the context of the program’s comprehensive hybrid framework. The Zone.Health model was specifically designed to enhance engagement and continuity of care by combining digital tools with personalized clinical input. This infrastructure likely contributed to the favorable results observed in both groups, particularly in supporting adherence, timely treatment adjustments, and the preservation of skeletal muscle mass during weight loss. The integration of pharmacotherapy within a digitally supported care model offers a practical and scalable approach to optimizing treatment outcomes. In this context, the observed differences between Tirzepatide and Semaglutide may reflect not only the underlying pharmacologic mechanisms but also the synergistic benefits of consistent clinical support, behavioral reinforcement, and remote monitoring. This study highlights the value of combining advanced therapeutics with a structured care delivery model to achieve meaningful and sustainable obesity outcomes.
Participants utilizing Tirzepatide achieved consistently greater weight reduction than those who received Semaglutide therapy, where the mean difference at 3 months for the former was 8.28 kg, while the latter was 6.62 kg. By 6 months, the difference increased to 13.12 kg and 9.54 kg, respectively. These findings are not only statistically significant but also clinically relevant, given that a weight loss of 5%–10% is linked to improved metabolic outcomes and lower cardiovascular risk. 20 Recognizing that body composition and treatment response may vary by gender, we performed a gender-based subgroup analysis. While baseline characteristics differed between males and females, the overall trends observed in the main analysis were preserved. Both male and female participants receiving Tirzepatide demonstrated greater reductions in weight, BMI, and fat mass compared with those on Semaglutide.
BMI and waist circumference followed a similar trend, further supporting the effectiveness of Tirzepatide. At 6 months, the BMI in the Tirzepatide group decreased by 4.59 kg/m2 compared with 3.40 kg/m2 in the Semaglutide group. Waist reductions were at 11.34 cm and 7.24 cm, respectively. A similar finding was reported in another study, where the waist circumference reduction was −18.08 cm compared with −13.04 cm for Tirzepatide and Semaglutide, respectively. 15 These results indicate a reduction in central fat distribution, which is an important factor in cardiovascular and metabolic risk.21,22
Compared with published studies in which GLP-1-based therapies such as Semaglutide have been associated with lean mass reductions of up to 6.9 kg, representing approximately 13.2% of total weight loss over 6 to 12 months in individuals with a baseline BMI of 37.8 ± 6.7, 23 and where Tirzepatide-induced weight loss has consistently resulted in a lean mass loss of approximately 25% relative to total weight loss in cohorts with a baseline BMI of 33.7 ± 4.8, 24 our findings at 6 months showing skeletal muscle mass reductions of only −1.10 kg in the Semaglutide group and −1.96 kg in the Tirzepatide group suggesting favorable lean mass preservation. These results are consistent with our prior 3-month analysis from the hybrid care model, which demonstrated a mean skeletal muscle loss of −1.63 ± 5.86 kg alongside preferential fat mass reduction. 25 The maintenance of skeletal muscle mass is essential for obesity management. 26 The observed decline in muscle mass necessitates further research to address the muscle loss associated with pharmacological weight reduction methods.
With regards to glycemic control, both groups experienced a reduction in 6 months (0.33% in the Tirzepatide group vs. 0.23% in the Semaglutide group). Although both groups had normal HbA1c levels at baseline, the Tirzepatide group demonstrated a greater improvement, indicating its effectiveness in enhancing insulin sensitivity, even among non-diabetic individuals. This finding is consistent with existing literature on the dual GLP-1/glucose-dependent insulinotropic polypeptide (GIP) (Tirzepatide) mechanism, which may provide greater glycemic benefits compared with GLP-1 agonists alone (Semaglutide). 15
The changes in lipid profile were mixed. Although HDL cholesterol levels showed a slight improvement in the Tirzepatide group at 3 months, this improvement was not maintained at 6 months. An increase was observed with LDL and total cholesterol levels, particularly at 3 months. While this increase may seem concerning, it could indicate changes in lipoprotein metabolism resulting from weight loss or alterations in dietary intake. 27 In contrast, triglyceride reductions were consistently higher in the Tirzepatide group, underscoring its potential to improve metabolic syndrome markers.
This study also analyzed the dose-response effects of both treatments. In the Semaglutide group, greater weight loss was associated with higher doses, indicating a dose-dependent effect. Participants who received doses ranging from 1 to 2.4 mg experienced a significant reduction in weight and BMI compared with those on lower doses.
In the Tirzepatide group, all three dosage bands (2.5–5 mg, 5–7.5 mg, and 7.5–15 mg) experienced weight loss. However, the highest dose group (7.5–15 mg) exhibited less weight loss compared with the moderate dose groups (2.5–5 mg and 5–7.5 mg). This finding suggests a plateau or diminishing returns at higher doses. This trend was also observed in measurements of BMI, fat mass, and skeletal muscle mass. An explanation for this might relate to side effects or decreased adherence at higher doses, although this data was not captured in the analysis.
The decline in effect at high doses could also be related to the sample size. The 2.5–5 mg group has the largest sample (n = 321), whereas the 5–7.5 and 7.5–15 mg groups have a smaller one. The greater variability in all variables among the larger group likely influenced the higher observed median, which may not reflect a true dose-response plateau or reversal. This observation should be acknowledged as a limitation when interpreting the dose–response relationship in the Tirzepatide group. Optimum improvement was noticed in the 5–7.5 mg group in the context of weight, BMI, and fat mass. This result suggests that moderate dosing may be the best approach; however, without data on adherence and side effects, a definitive conclusion cannot be drawn. The effectiveness of Tirzepatide over Semaglutide across multiple domains of clinical outcomes underscores the value it has as a therapeutic option in managing obesity, as supported by this study and by prior clinical trials.28,29 Nonetheless, the apparent muscle loss observed in both therapies, especially Tirzepatide, requires future trials to focus on strategies to optimize muscle mass preservation and assess long-term sustainability.
Clinical Implications and Policy Relevance
The findings from this real-world hybrid care program suggest that combining GLP-1/GIP receptor agonist therapy with a digitally supported model can optimize clinical outcomes while supporting muscle preservation. Standardized reporting through the ICHOM Obesity Standard Set enhances transparency, facilitates benchmarking across clinics, and enables outcome-based evaluation of obesity interventions from a health system perspective, integrating such hybrid models may improve long-term cost-effectiveness by reducing downstream complications of obesity and minimizing medication waste through continuous engagement and dose monitoring. These standardized outcome metrics can also support value-based reimbursement frameworks and strengthen the case for insurance coverage of antiobesity pharmacotherapy as part of structured, outcome-monitored care pathways. Future studies incorporating cost–utility and payer analyses will be essential to guide broader policy adoption.
Limitations
Several limitations should be acknowledged in this study. First, the study design was observational, and treatment allocation was not randomized, contributing to a potential selection bias. Secondly, the baseline body composition in both groups might have influenced the magnitude of change observed. Third, data on dietary intake, medication adherence, and physical activity were unavailable, which limited the ability to contextualize the outcomes; integration into future analyses is planned. Differences in baseline weight and BMI between the treatment groups may have contributed to the observed outcome magnitudes. As no multivariable adjustments or propensity matching were performed, this introduces potential confounding that should be considered when interpreting the results. The larger Tirzepatide cohort may have influenced outcome magnitudes; this imbalance was addressed by presenting both absolute and relative changes. Lastly, the groups in the study were unbalanced, indicating that larger randomized studies need to be conducted to clarify the plateau observed at high doses. Future studies should focus on collecting behavioral data, ensuring group balance, and incorporating randomization of treatment.
Conclusions
In conclusion, this study demonstrates that Tirzepatide therapy delivered within a structured hybrid care model yields significantly greater improvements in weight loss, body composition, and glycemic control compared with Semaglutide therapy, while maintaining relatively preserved skeletal muscle mass. The hybrid model integrating pharmacotherapy with continuous digital monitoring and multidisciplinary support likely contributed to these favorable outcomes. By aligning with the ICHOM Obesity Standard Set, this study enhances transparency and comparability in obesity care reporting. Future research should focus on validating these findings through randomized trials, exploring long-term weight maintenance, optimizing dosing strategies, and incorporating behavioral data and patient-reported outcomes. Additionally, cost-effectiveness analyses and multi-center benchmarking studies are needed to guide broader implementation and policy adoption of hybrid care models to manage obesity.
Ethics Approval and Consent to Participate
This study was reviewed and approved by the Dubai Scientific Research Ethics Committee under the Dubai Health Authority (DHA) (Approval number: DSREC-02/2025_09). Written informed consent was obtained from all participants at the start of the program.
Authors’ Contributions
H.Z. conceptualized the study, methodological design, coordinated data analysis, and led article writing. K.C.T., J.A., and K.C. contributed to data collection and clinical oversight. G.P. was responsible for statistical analysis and figure/table generation. A.A. and A.H. contributed to article revisions. I.A. supervised the project and provided critical review of the final draft. All authors read and approved the final article.
Footnotes
Author Disclosure Statement
H.Z., G.P., K.C.T., J.A., K.C., A.A., A.H., and I.A. are all affiliated with GluCare.Health or Zone.Health, which deliver hybrid care models to manage obesity. I.H. and A.H. are co-founders of GluCare.Health. The authors declare no other competing interests.
Funding Information
No external funding was received for this study.
Availability of Data and Materials
The datasets generated and/or analyzed during the current study are not publicly available due to institutional policies protecting patient confidentiality. However, they are available from the corresponding author (H.Z.) on reasonable request.
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
