Abstract
Background
A subset of individuals with episodic migraine (EM) and chronic migraine (CM) does not respond to anti-calcitonin gene-related peptide (CGRP) monoclonal antibodies (mAbs), even after switching agents. Although atogepant is effective in EM and CM, its role in individuals unresponsive to anti-CGRP mAbs remains to be elucidated. We hypothesize that atogepant may be a viable preventive option in these cases.
Methods
We conducted a retrospective cohort study in EM and CM individuals who discontinued anti-CGRP mAbs due to lack or loss of efficacy and were subsequently treated with atogepant. The primary endpoint was the ≥50% response rate in monthly headache days (MHDs). Secondary outcomes included ≥30% and ≥75% response rates in MHDs, response rates in monthly moderate-to-severe headache days (MSHDs), and changes in MHDs, MSHDs, acute medication days (AMDs) and Headache Impact Test (HIT-6). Patient Global Impression (PGI) scores and adverse events (AEs) were recorded.
Results
Of 213 screened records, 44 participants met inclusion criteria; 39 (88.6%) were female and 39 (88.6%) had CM. Prior use of anti-CGRP mAbs included erenumab in 11.4% of participants, galcanezumab in 52.3% and fremanezumab in 86.3%. After three months, 18.2% achieved a ≥50% reduction in MHDs; ≥30% and ≥75% response rates were 25.0% and 6.8% respectively. For MSHDs, ≥30%, ≥50% and ≥75% response rates were 47.6%, 33.3% and 19.0%, respectively. Median MHDs decreased from 24.5 (interquartile range (IQR) = 16.0–30.0; range 9.0–31.0) to 21.5 (IQR = 10.0–30.0; range 3.0–31.0; p = 0.011), and median MSHDs from 15.0 (IQR = 10.0–25.5; range 5.0–30.0) to 12.0 (IQR = 5.9–19.0; range 0.0–30.0; p = 0.001). AMDs and HIT-6 scores also showed significant reductions. According to the PGI scale, 59.1% of individuals reported some degree of improvement. AEs occurred in 50.0% of participants, most commonly constipation (31.8%). Five (11.4%) participants discontinued treatment due to side effects.
Conclusions
After three months of treatment, atogepant led to a clinically meaningful improvement in a subset of participants. It may be a valuable preventive option for individuals unresponsive to anti-CGRP mAbs and warrants further investigation in prospective studies.
This is a visual representation of the abstract.
Introduction
Although anti-calcitonin gene-related peptide (CGRP) monoclonal antibodies (mAbs) have revolutionized the preventive treatment of migraine, real-world evidence indicates that a substantial proportion of individuals do not achieve meaningful clinical benefit. According to an observational Italian study, 28.8% of individuals discontinue anti-CGRP mAb treatment, with lack of efficacy accounting for 70.6% of these discontinuations (1). Furthermore, one study reported that 13.1% of individuals exhibit no response even after six months of therapy (2). In clinical practice, treatment failure rates with these therapies have been estimated to reach as high as 30–40% (3), highlighting the need for alternative strategies in this subgroup of migraine individuals.
Switching between mAbs may be an effective strategy for individuals who do not respond to initial anti-CGRP therapy. A systematic review of 19 studies found that transitioning between different anti-CGRP mAbs can be beneficial for non-responders, reporting a median reduction of 12.8 monthly migraine days (MMDs) after six months of follow-up, and up to 48% of participants with episodic migraine (EM) and 36% of those with chronic migraine (CM) achieved a ≥50% response rate after switching (4). Interestingly, one retrospective study suggested that treatment outcomes may be independent of whether the switch involved a different mechanism of action, the number of doses received of the initial mAb or the inter-treatment interval (5).
However, the studies included in the systematic review were heterogeneous, encompassing switches due to inefficacy, individuals’ preference or tolerability issues. In studies where switching was prompted solely by lack of efficacy, the proportion of participants achieving a ≥50% reduction in monthly headache days (MHDs) ranged from 5% to 35% (6–11), highlighting that a considerable number of individuals remain without effective preventive options.
Recent studies have demonstrated the efficacy and safety of atogepant for the preventive treatment of EM and CM (12–16). Although there is currently no direct evidence supporting the use of gepants in migraine individuals who have failed anti-CGRP mAbs, this therapeutic approach has been proposed as a reasonable and potentially beneficial alternative (17).
The aim of this retrospective study was to evaluate the effectiveness of atogepant in individuals with EM or CM who had previously discontinued anti-CGRP mAbs due to lack or loss of effectiveness. From a broader real-world cohort of 213 individuals treated with atogepant, we analyzed a subgroup of 44 who met these criteria. We hypothesize that a proportion of these individuals may achieve a ≥50% reduction in MHDs.
Methods
This is a monocentric retrospective cohort study conducted at a tertiary headache center, utilizing data from medical records and paper headache diaries of migraine individuals treated with atogepant from June 2024 to December 2024.
Participants
Migraine individuals were selected through manual review of medical records. Inclusion criteria were a diagnosis of migraine according to the International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria (18); prior treatment with anti-CGRP mAbs; and initiation of atogepant at any point after mAb discontinuation. Participants were included if they had discontinued mAb therapy due either to ineffectiveness or to loss of effectiveness. Ineffectiveness was defined as a reduction of ≤30% in MHDs after receiving at least six doses. Loss of effectiveness was defined as a drop in treatment response to ≤30% improvement in MHDs from baseline, sustained for over three months, following a prior ≥30% response maintained for at least three months.
Participants were excluded if baseline data for the primary outcome were missing or if atogepant was initiated due to adverse events related to mAb treatment. Because selection of participants was conducted manually, no validation of automated codes or algorithms were required. Given the retrospective nature of the study, the decision to initiate atogepant, including timing and indication, was made at the discretion of the treating physician.
Variables
This study did not include data linkage across multiple databases. All data were obtained from a single source, comprising the hospital electronic health record system, through a manual review of the medical records. Demographic and clinical variables were collected.
Headache diaries included data on headache intensity (on a 1–3 scale: 1 = mild pain, 2 = moderate pain and 3 = severe pain), duration (in hours) and use of acute medication. However, information on accompanying symptoms characteristic of migraine was not recorded, preventing a reliable distinction between MHDs and MMDs according to ICHD-3 criteria. Therefore, MHDs defined as any day on which a participant recorded any type of headache in the paper diary, were used to evaluate the primary outcome.
Additionally, data were collected on monthly moderate-to-severe headache days (MSHDs), the number of days the participants used acute medication (AMDs) and the type of treatment used. Medication overuse headache (MOH) was assessed according to the ICHD-3 criteria (18). The following variables were also recorded: migraine type, presence of aura, age at migraine onset, number of previous and concomitant preventive treatments (documented by compound and classified according to the evidence-based guidelines developed by the Italian Society for the Study of Headache and the International Headache Society), (19) the duration of concomitant preventive treatments prior to the initiation of atogepant, the type and number of anti-CGRP mAbs used, total number of doses received, the reasons for discontinuing biological therapy, and the Headache Impact Test (HIT)-6 scores (20). Participants-reported outcomes were assessed using the Patient Global Impression (PGI) scale (21): 1 = very much improved, 2 = much improved, 3 = minimally improved, 4 = no change, 5 = minimally worse, 6 = much worse and 7 = very much worse. The occurrence of adverse events (AEs) with atogepant and/or prior anti-CGRP mAbs were also documented based on the information available in each medical record.
The investigators had full access to the medical records database used to create the study population. Access was granted under the institution’s data use agreement, ensuring compliance with privacy regulations. All participants data were deidentified to maintain confidentiality. Data cleaning was performed by cross-referencing with original medical records. All steps were thoroughly documented to ensure reproducibility.
Endpoint
The baseline period was defined as the month preceding the initiation of atogepant. The primary endpoint was the proportion of participants achieving a ≥50% reduction in MHDs from baseline to month 3.
Secondary endpoints included the proportion of participants achieving a ≥50% reduction in MSHDs, response rates of ≥30% and ≥75% reduction in MHDs and MSHDs at month 3, changes in MHDs, MSHDs, AMDs and HIT-6 scores, and patient-reported global improvement assessed using the PGI scale. In addition, the rate of AEs was evaluated.
Statistical analysis
A priori statistical power calculation was not conducted due to the sample size being determined by available data. An exploratory analysis was performed to delineate demographic and baseline characteristics. Categorical variables were summarized using frequencies and percentages, while continuous variables were presented as the mean and standard deviation (SD) or median, interquartile range (IQR) and range, depending upon their distribution as determined by the Kolmogorov–Smirnov test. Changes in MHDs, MSHDs and AMDs from baseline to month 3 were analyzed using the Wilcoxon signed-rank test, while changes in HIT-6 scores were assessed using the paired t-test. Frequencies and percentages were calculated for each PGI category to describe participants perceived global change. Statistical significance was determined by two-tailed tests with p < 0.05 considered statistically significant. This is the primary analysis of the collected data and it was preplanned. No previous reports or similar analyses of the data used in the present study have been published. Analyses of the primary outcome were conducted following the intention-to-treat principle, allowing the inclusion of all participants who initiated treatment, regardless of adherence or early discontinuation. For missing follow-up data related to treatment, we used the last observation carried forward method because it effectively handles intermittent missing entries in longitudinal headache diaries, provides a conservative estimate of treatment effectiveness and reflects a pragmatic approach to estimating treatment outcomes in the presence of fluctuating symptom patterns. This approach allowed us to handle missing data effectively and maintain the integrity of our statistical analyses. Data analysis was performed using SPSS, version 25 (IBM Corp., Armonk, NY, USA).
Ethical approval
The research protocol for this study was approved by the institutional research ethics committee of the Hospital Fundación Jiménez Díaz (PA_EO066-25_FJD) and the study was conducted in adherence to the ethical principles outlined in the Declaration of Helsinki. Due to the retrospective nature of the study, the requirement for written informed consent was waived. Furthermore, our report adheres to the guidelines set forth by the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement.
Results
Cohort characteristics
In total, 213 medical records of migraine individuals receiving treatment with atogepant were identified. Of these, 46 had previously been treated with anti-CGRP mAbs. Two participants were excluded because the reason for mAb discontinuation was adverse events. Therefore, the final cohort for analysis comprised 44 participants.
The mean (SD) age of the cohort was 49.1 (12.9) years, with a predominance of female participants (39 out of 44; 88.6%). CM was observed in 39 out of 44 (88.6%) participants (Table 1). Migraine aura was reported by 20 out of 42 (45.5%) participants, with data missing for two individuals (4.5%). Unilateral pain was reported by 30 out of 43 participants (68.2%), with one case of missing data (2.3%). The median age at migraine onset was 15 years (IQR = 12.0–19.5; range = 5.0–55.0). The median number of prior preventive drugs used was 6.0 (IQR = 4.3–7.0; range = 3.0–11.0). MOH was present in 26 out of 40 (65.0%) participants, with data missing for four individuals (15.4%). The overused medication was non-steroidal anti-inflammatory drugs in 18 out of 26 participants (69.2%), triptans in two out of 26 (7.7%) and a combination of both drug classes in six out of 26 (23.1%). In total, 12 out of 44 (27.3%) participants were concurrently receiving an oral medication with a preventive effect on migraine, with a mean (SD) duration of prior use of 43.8 (20.0) months (range 12–76 months) and no changes in dosage were observed during the study period (Table 2).
Cohort characteristics.

Abbreviations: CGRP = calcitonin gene-related peptide; IQR = interquartile range; MOH = medication overuse headache; mAb = monoclonal antibody; N = number of participants with available data for each variable; NSAID = non-steroidal anti-inflammatory drug; SD = standard deviation.
Percentage of patients with a concurrent preventive medication.
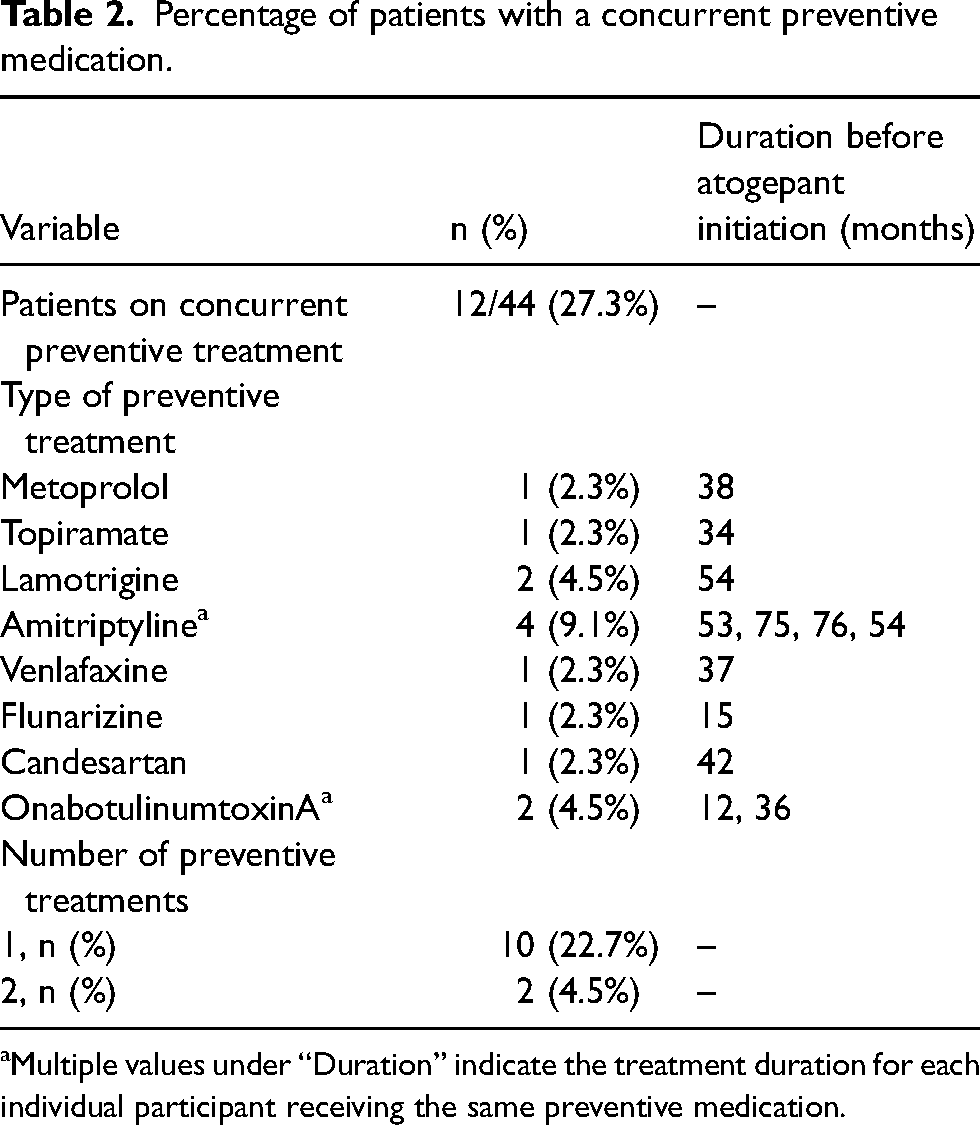
Multiple values under “Duration” indicate the treatment duration for each individual participant receiving the same preventive medication.
Prior anti-CGRP mAb treatment
Among the 44 participants, five (11.4%) had previously received erenumab, with a median of 12 doses administered (IQR = 8.0–13.0; range = 6.0–14.0), 23 (52.3%) received galcanezumab, with a median of 15 doses (IQR = 6.0–18.3; range = 6.0–25.0) and 38 (86.3%) received fremanezumab, with a median of eight doses (IQR = 6.0–12.3; range = 6.0–36.0). Regarding the number of anti-CGRP mAbds previously tried, 23 participants (52.3%) had received one, 20 (45.5%) received two and one (2.3%) received all three. Reasons for discontinuing the last anti-CGRP mAb included ineffectiveness in 27 cases (61.4%) and loss of effectiveness in 18 cases (40.9%).
Effectiveness
The median number of MHDs at baseline was 24.5 (IQR = 16.0–30.0; range = 9.0–31.0), which decreased to 21.5 at month 3 (IQR = 10.0–30.0; range = 3.0–31.0; p = 0.011) (Figure 1). Data at month 3 were missing for five participants (11.4%) who discontinued treatment during the first month due to AEs. A ≥50% response rate was achieved by eight out of 44 participants (18.2%) (Figure 2). Response rates of ≥30% and ≥75% were observed in 11 (25.0%) and three (6.8%) participants, respectively (Table 3).

Box plot illustrating the monthly headache days, monthly moderate-to-severe headache days and acute medication days at baseline (MHDs-B, MSHDs-B and AMDs-B, respectively) and at month 3 (MHDs-3, MSHDs-3 and AMDs-3, respectively).

Bar chart illustrating the cumulative response rates at month 3. MHDs = monthly headache days; MSHDs = monthly moderate-to-severe headache days.
Changes in migraine-related variables from baseline to month 3.

Note: p-values were obtained using the Wilcoxon signed-rank test (for non-normally distributed variables) or paired t-test (for normally distributed variables).
Abbreviations: AMDs = acute medication days; IQR = interquartile range; HIT = Headache Impact Test; MHDs = monthly headache days; MSHDs = monthly moderate-to-severe headache days; SD = standard deviation.
The median number of MSHDs at baseline was 15.0 (IQR = 10.0–25.5; range = 5.0–30.0), based on data from 42 out of 44 participants. This decreased to 12.0 (IQR = 5.9–19.0; range = 0.0–30.0) at month 3 (p = 0.001). Data at month 3 were missing for four participants (9.5%) due to treatment discontinuation related to AEs. A ≥50% reduction in MSHDs was achieved by 14 of 44 participants (33.3%), with ≥30% and ≥75% responses in 20 (47.6%) and eight (19.0%) participants, respectively.
The median number of AMDs at baseline was 17.5 (IQR = 12.3–29.8; range = 0.0–31.0) based on data from 40 participants, and decreased to 12.0 (IQR = 4.3–26.8; range = 0.0–31.0) at month 3 (p < 0.001). Follow-up data were missing for three participants (7.5%).
Finally, the mean (SD) HIT-6 score at baseline was 67.9 (6.5) based on 39 participants, and decreased to 64.9 (9.1) at month 3 (p = 0.011). Follow-up data were missing for three participants (7.7%).
Participant's reported outcome
At follow-up, 26 out of 44 participants (59.1%) reported some degree of improvement according to the PGI scale: three (6.8%) were very much improved, eight (18.2%) were much improved and 15 (34.1%) were minimally improved. Thirteen participants (29.5%) reported no change, while five (11.3%) experienced some degree of worsening (Figure 3).

Patient Global Impression (PGI) scores at month 3 (n = 44). PGI scale values: 1 = very much improved; 2 = much improved; 3 = minimally improved; 4 = no change; 5 = minimally worse; 6 = much worse; 7 = very much worse.
Safety and tolerability
AEs were reported in 22 out of 44 participants (50.0%) during atogepant treatment (Table 4). The most frequently observed events were constipation (14 participants, 31.8%), nausea (8 participants, 18.2%), fatigue (5 participants, 11.4%), sedation (5 participants, 11.4%) and cognitive difficulties characterized by difficulty concentrating, bradypsychia and memory lapses (2 participants, 4.5%). In total, five participants (11.4%) discontinued treatment due to AEs. Specifically, two participants suspended treatment due to constipation, one due to nausea, one due to cognitive difficulties and one due to a combination of nausea, constipation and cognitive difficulties.
Adverse events reported during treatment with anti-CGRP mAbs, atogepant, or both.

Abbreviations: CGRP = calcitonin gene-related peptide; mAb = monoclonal antibody; NA = not applicable.
During previous anti-CGRP mAb treatment, AEs were reported in 11 participants (25.0%), with injection-site reactions being the most common (6 participants, 13.6%). Notably, participants who experienced non-injection site-related adverse events during any of the anti-CGRP mAb treatments also reported similar events with atogepant: constipation in four cases (9.0%) and nausea in one case (2.3%).
Discussion
Our study provides one of the first evidence supporting atogepant as an effective alternative for migraine individuals who have not responded or have experienced a loss of efficacy with anti-CGRP mAbs. After three months of treatment, 18.2% of participants achieved a ≥50% reduction in MHDs.
Our findings align with those of the recent STAR study, (22) a real-world, prospective, multicenter Italian cohort that evaluated the effectiveness of atogepant over 12 weeks in individuals with EM and CM. In that study, 56 participants had previously received anti-CGRP mAbs, and 46.4% of them achieved a ≥50% reduction in MMDs. However, no data were reported on MHDs, nor was it specified whether any participants had discontinued anti-CGRP mAbs due to AEs. Differences in population characteristics between the STAR study and our cohort, such as the proportion of CM (69.8% vs. 88.6%, respectively) and outcome measures (MMDs vs. MHDs, respectively), may partly explain the variations in response rates observed between studies. Interestingly, in the STAR study, prior failure to anti-CGRP mAbs was not significantly associated with atogepant response in multivariable analysis, which is consistent with our findings and supports the notion that atogepant may be effective even in migraine individuals with limited benefit from previous CGRP-mAbs therapies.
A noteworthy aspect of our cohort is the relatively high prevalence of migraine with aura (45.5%), which deserves further consideration. Although a population-based study reported that individuals with migraine with aura tend to have a higher number of MHDs (23), which could partly account for this finding, previous data from our center showed a lower prevalence of aura (27.1%) in a similar population (5). In addition, there is no current evidence suggesting that the presence of aura negatively influences the response to anti-CGRP therapies (24). Taken together, these findings suggest that the difference observed in the present study is likely due to selection bias or random variability rather than a clinically meaningful distinction.
The differential efficacy of atogepant in migraine individuals unresponsive to anti-CGRP mAbs may be explained, at least in part, by distinct mechanisms of action at the trigeminovascular level. Experimental data suggest that mAbs such as fremanezumab preferentially inhibit Aδ fiber activation, but not C fibers (25), thereby suppressing the activation of high-threshold dura-sensitive trigeminovascular neurons in the trigeminocervical complex, while failing to inhibit wide dynamic range neurons (26). By contrast, atogepant has demonstrated the ability to inhibit both Aδ fibers and, to a lesser extent, C fibers (27), suppressing both activation and sensitization of high-threshold neurons (28). Although it does not directly inhibit wide dynamic range neuron activation, it has been shown to prevent their central sensitization, a key mechanism in migraine chronification (28). These differences may contribute to atogepant's broader efficacy across distinct nociceptive pathways, potentially explaining its therapeutic benefit in migraine individuals who previously failed mAbs therapies.
The safety profile of gepants is similar to that of mAbs and is associated with few adverse effects (29). According to a post hoc analysis of pooled data from four clinical trials, 56.3–64.5% of atogepant participants experienced one or more treatment AEs, with this the most commonly reported being upper respiratory tract infection (5.3–7.7%), constipation (5.0–6.0%), nausea (4.6–6.6%) and urinary tract infection (3.4–5.2%) (30). In the STAR study, the overall AEs rate was 44.3%, with constipation (25.5%), decreased appetite (16.0%) and nausea (15.0%) reported most frequently (22). In our cohort, 50.0% of participants reported AEs, with constipation (31.8%) and nausea (18.2%) being the most frequent, both occurring at higher rates than those previously reported in clinical trials, but consistent with real-world evidence. Notably, no cases of upper respiratory tract infection were documented, likely due to under-reporting in routine clinical practice. Importantly, five participants (11.4%) discontinued treatment due to AEs, comprising a proportion that is not negligible and warrants consideration in clinical decision-making. Interestingly, most participants who experienced non-injection- site-related AEs during previous mAbs treatment also reported similar AEs during atogepant treatment. These findings may indicate a potential individual susceptibility to gastrointestinal AEs in migraine individuals exposed to different CGRP-targeted therapies. Therefore, when such AEs occur with anti-CGRP mAbs, switching to atogepant may not be a suitable option. However, given the limited sample size, definitive conclusions cannot be drawn.
In interpreting our findings, it is important to consider the national and institutional context in which treatment decisions are made. In Spain, anti-CGRP treatments are publicly funded for the preventive treatment of individuals with high-frequency EM (8–14 migraine days per month) and CM who have failed at least three preventive treatments, including onabotulinumtoxinA in the case of CM, administered at appropriate doses and for a minimum duration of three months. However, the decision regarding which preventive treatment to initiate first, and whether or when to switch therapies, is ultimately made by each hospital's pharmacy and therapeutics committee.
Our study has several limitations that should be acknowledged. First, although the phase III ADVANCE trial (31). showed that atogepant demonstrated effectiveness as early as the first day after treatment initiation and maintained a sustained reduction in MMDs over three months, the follow-up period in our study was limited to three months. Therefore, it is possible that some participants might have responded beyond this time window. Second, the retrospective nature of the study and reliance on medical records, as originally created for clinical rather than research purposes, introduce potential information bias and incomplete data. Furthermore, due to the limited detail in these records, it was not possible to reliably differentiate between headache days and migraine days according to ICHD-3 criteria. Although headache frequency was assessed using paper diaries, not all participants completed them consistently. For missing outcome data, we applied the last observation carried forward method. While this conservative approach is useful in managing intermittently missing values in fluctuating conditions, it assumes stability that may not reflect actual individuals experience and can lead to either underestimation or overestimation of the treatment effect. Moreover, the lack of a control group may have introduced bias due to placebo effects and natural variability in migraine patterns. Although 27.3% of participants were receiving a concomitant preventive treatment, it is unlikely that the observed response was primarily attributable to these agents because such treatments had been initiated prior to the study and their dosages remained stable throughout the follow-up period. Lastly, we did not stratify participants according to the presence of MOH, which may have influenced treatment response.
Despite these limitations, our study has several strengths. To our knowledge, it is the first real-world study specifically evaluating the effectiveness of atogepant in migraine participants unresponsive to anti-CGRP mAbs, which is a population often excluded from clinical trials and with limited treatment alternatives. The use of clearly defined inclusion criteria and standardized headache diaries strengthens the reliability of outcome measures. Additionally, the inclusion of both EM and CM individuals, along with detailed clinical variables such as HIT-6 scores, prior preventive treatments and acute medication use, provides a comprehensive overview of treatment response in routine clinical practice.
Our sample was predominantly composed of individuals with CM, which likely reflects the clinical profile of individuals referred to tertiary headache units, where more complex and treatment-resistant cases are typically managed. The limited number of participants with EM restricts the generalizability of our findings to this subgroup. As a result, our conclusions may be more applicable to individuals with a higher migraine burden and a lower likelihood of response to previous preventive therapies. Although the single-center, retrospective design may limit external validity, the inclusion of a real-world, difficult-to-treat population offers a more accurate representation of routine clinical practice than that provided by controlled trial settings.
Conclusions
This study provides one of the first real-world evidence supporting the use of atogepant in migraine individuals who have failed to respond or have lost response to anti-CGRP mAbs. A meaningful proportion of participants achieved clinically significant reductions in MHDs after three months of treatment. These findings suggest that atogepant may represent a valuable therapeutic option for this difficult-to-treat population, warranting further investigation in prospective and controlled studies.
Clinical implications
Non-response to anti-CGRP mAbs is common, even after switching.
After three months, atogepant achieved a ≥50% reduction in monthly headache days in 18.2% of participants.
Improvement on the PGI scale was reported by 59.1% of participants.
Adverse events were reported in 50.0%, mainly constipation (31.8%); 11.4% discontinued treatment.
Footnotes
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alex Jaimes has received honoraria from Lilly, TEVA, Organon, Allergan-Abbvie and Lundbeck. Jaime Rodríguez-Vico has received honoraria from Lilly, TEVA, Novartis, Allergan-Abbvie and Exeltis, as well as research support from Allergan-Abbvie. Andrea Gómez has received honoraria from Organon. Olga Pajares, Ignacio Eguilior, Lena Nystrom and Jesús Porta-Etessam declare no conflicts of interest.
Ethical statement
The study was conducted in adherence to the ethical principles outlined in the Declaration of Helsinki. Due to the retrospective nature of the study, the requirement for written informed consent was waived.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
