Abstract
Background
We aimed to systematically assess the effectiveness and tolerability of erenumab in a clinical setting, specifically a tertiary headache center.
Methods
This was a retrospective cohort study at the John Graham Headache Center of Brigham and Women’s Hospital. All patients who received erenumab from a headache specialist at the Center from 17 May 2018 to 31 January 2019 were included. Patients were contacted and underwent a structured clinical interview including information about erenumab use, perceived benefit, adverse events (AEs), and a global assessment of benefit versus drawbacks. Chart review was performed for patients who could not be contacted.
Results
Four hundred and forty-four patients were initially identified and 418 were eligible. Two hundred and ninety-five participants completed the structured clinical interview portion of the study (response rate 70.6%). Seventy-four patient charts were additionally reviewed. Two hundred and forty-one participants had used erenumab. One hundred and sixty nine (70%) of participants experienced at least one adverse event, with constipation (43%), injection site reaction (24%), fatigue (15%), worsening headache (12%), and dizziness (11%) the five most commonly reported AEs. One hundred and sixty-eight participants (69.7%) felt that the benefits of erenumab outweighed any drawbacks. One hundred and fifty-one participants (62.7%) planned to continue using erenumab. Cost of treatment was cited by 12% of participants as a reason for either not starting or for stopping erenumab. Of patients who had an increase in dose due to lack of or partial efficacy, 46.5% felt that the dose increase was helpful. We identified one case of unintended pregnancy in our study population.
Conclusions
This large “real-world” study validates the findings of benefit of erenumab observed in clinical trials. Although adverse events were far more common in this population than in clinical trials, the planned continuation rate was relatively high. The substantial discrepancy between adverse events seen in clinical practice compared with clinical trials suggests systematic differences between clinical trial participants and patients who receive the treatment in clinical practice, or may indicate suboptimal ascertainment of adverse events in the trials. Clinicians should continue to be vigilant for adverse events in clinical practice.
Introduction
Erenumab is a monoclonal antibody that antagonizes the calcitonin gene-related peptide (CGRP) receptor (1). It was approved by the United States Food and Drug Administration in May 2018 for the preventive treatment of both episodic and chronic migraine (2). Migraine is a common, chronic, and highly disabling condition (3). The safety and effectiveness of erenumab were assessed in three pivotal studies in the prevention of episodic and chronic migraine (4–6).
Clinical experience of the effectiveness and adverse events of migraine preventive medications may differ from results reported in clinical trials (7). This difference can be due to many factors that apply to clinical trials but not to day-to-day practice, including the vigilant tracking of headache days in clinical trials, small sample size, study design and outcomes reported, short duration of treatment, and the strict patient selection criteria, which may lead to exclusion of non-adherent patients, those with certain comorbid conditions, or those using certain medications (8).
We therefore aimed to systematically assess the effectiveness and tolerability of erenumab in the real-world setting of a tertiary headache center. We specifically aimed to assess patient perceptions of effectiveness, prevalence and nature of adverse events, barriers to use, and patient global impression of erenumab.
Methods
We conducted a retrospective cohort study at the John Graham Headache Center at the Brigham and Women’s Faulkner Hospital, a tertiary headache center with over 10,000 patient visits per year. This study was approved by the Partners Institutional Review Board (IRB).
Using our electronic medical record, we obtained a list of patients who had been prescribed erenumab by a physician or physician’s assistant on staff at the Graham Headache Center between 17 May 2018 and 31 January 2019. We used the reports feature in EPIC to construct a report, specifying each of the eligible providers by name as well, and searching for erenumab 70 mg and 140 mg. Since erenumab was not added as a coded entry in our EMR until a month after product availability, we also conducted a separate report of patients who received a free-text order for a medication by the same providers in the same period. We conducted a brief chart review of patients who received a free-text prescription and identified those for whom the free-text order was a prescription for erenumab.
Subjects were deemed eligible if they received a prescription for erenumab for the treatment of migraine from one of the headache specialists at the John Graham Headache Center. Subjects were excluded if they did not receive the medication prior to the structured clinical interview. They were also excluded if they lacked the means of communication due to traveling abroad, language barrier, or death. Where uncertain, study staff determined eligibility based on consensus.
Recruitment letters and study information sheets were mailed to identified patients. Patients could contact study staff to decline participation. After 2 weeks, subjects who did not decline participation were considered enrolled in the study. Enrolled patients who participate in Epic’s MyChart features (an electronic communication modality) were sent a message to encourage them to call to participate or decline participation. Between March 2019 and September 2019, enrolled patients were called by either SK or GH to confirm eligibility, obtain informed consent, and administer the structured clinical interview using our study questionnaire. We took care to ensure that participants who were patients of an investigator were directly contacted by a different member of the study staff. Patients were offered an opportunity to be entered into a draw for a gift card at the end of the study.
The structured clinical interview followed a questionnaire developed for this study. The structured clinical interview script was reviewed by five headache specialists and piloted by five patients. The interview consisted of questions about patient characteristics (self-identified gender, date of birth, age, weight, height, body mass index, and self-identified race), erenumab use (whether erenumab was obtained and used, intent to continue taking erenumab or not), erenumab adverse events (open-ended question followed by reviewing a checklist of possible adverse events), and effectiveness (perceived benefits, whether benefits outweighed any problems, total headache days in past month, any dose change, reason for dose change, and whether dose change was helpful). We did not assess data on headache days prior to starting erenumab due to concerns about accuracy of patient recall beyond 30 days. Free-text notes were also taken during the interview. Answers to the structured clinical interview questions were entered by the interviewer into Research Electronic Data Capture (REDCap), hosted by Partners Healthcare.
The structured list of possible adverse events was developed from clinical trial results, theoretical concerns raised in the basic science literature (including vascular complications), and clinical observation of erenumab adverse events in our clinic (9). Adverse events were categorized according to the Medical Dictionary for Regulatory Activities (MedDRA) (10). Two independent reviewers (RB and SK) independently used our institutional subscription to the proprietary MedDRA database to code and enter free-text descriptions of adverse events. Disagreements were resolved by consensus. Following administration of the formal interview questions, participants were asked whether they had any additional comments. We reviewed these comments, identified major themes, and selected quotations that illustrated these themes.
Finally, for enrolled subjects who were unreachable by phone and who did not respond to the electronic message to participate or withdraw, a chart review was conducted to gather available information about erenumab tolerability and effectiveness. The data gathered via chart review was limited to available information documented in the medical records. Chart review endpoints were whether they took the medication, reason(s) for not taking it, plan to continue, reason for discontinuation, adverse events, whether erenumab was helpful in treating headaches, dose change, reason for dose change, and whether the dose change was beneficial to the patient.
We did not systematically investigate serious adverse events. However, any hospitalizations or deaths discovered by or reported to the study staff were evaluated through chart review by two headache specialists (SK and RB) to identify the event and its possible relevance to erenumab use.
Statistical analysis
We present descriptive statistics in this paper. We did not pre-specify any statistical analyses and the sample size was determined by the available clinical population.
Results
Eligibility and enrolment
We identified 444 patients who had been prescribed erenumab by one of the 11 headache specialists at the John Graham Headache Center during the study period. Eligibility and enrolment are depicted in Figure 1. Twenty-six subjects (5.9%) were determined to be ineligible and excluded from the study. Ineligibility was attributed to the subject verbally denying receipt of a prescription for erenumab (n = 7), having received the prescription from outside our headache center (n = 7), having a diagnosis other than migraine (n = 4), having a language barrier (n = 2), not having obtained the medication from the pharmacy yet (n = 2), physician having rescinded the prescription immediately due to the patient reporting a latex allergy (n = 1), being deceased (n = 1), or lacking the means of communication due to living abroad (n = 1). By consensus, we agreed to exclude one additional participant who had received the first dose shortly prior to being contacted for the interview and was unable to provide meaningful information about their experience.

Flow of patients through study.
We therefore included 418 eligible subjects for our study. Forty-nine subjects (11.7%) declined participation and the remaining 369 (88.3%) were considered enrolled for the final analysis. The majority did not provide a reason for declining (41/49), with the remainder reporting lack of effectiveness (2), time constraints (2), prescribing physician’s departure from the practice (2), subject’s relocation (1), and adverse events (1) as reasons to decline. Of the enrolled subjects, 295/418 (70.6%) completed the telephone structured clinical interview whereas data for the remaining 74/418 subjects (17.7%) were obtained from chart review.
Demographics
Table 1 displays the demographics of the 418 eligible patients. Since the sample was based on the available data, statistical analysis was not performed to compare demographic characteristics among the different groups of eligible patients (participants, declined to participate, and chart review). However, the groups were generally similar in their demographics and baseline characteristics with most patients being white females with a mean age of 41.7–48.6 years of age across the three groups and a mean BMI range of 26.3–27 kg/m2 across the groups.
Demographic characteristics of study population (n = 418).

Telephone structured clinical interview
Usage patterns and outcomes
Table 2 summarizes responses from the 295 participants in the telephone structured clinical interview. Of the participants who completed the telephone structured clinical interview, 241/295 (81.7%) had used erenumab. Among those who had used the treatment, the mean number of headache days in the 30 days prior to the telephone structured clinical interview was 15.3 (range 0–30). In determining the monthly headache days, 108/241 (44.8%) referred to a diary, whereas the remaining 133 (55.2%) relied on recall.
Results of the telephone survey (n = 295).
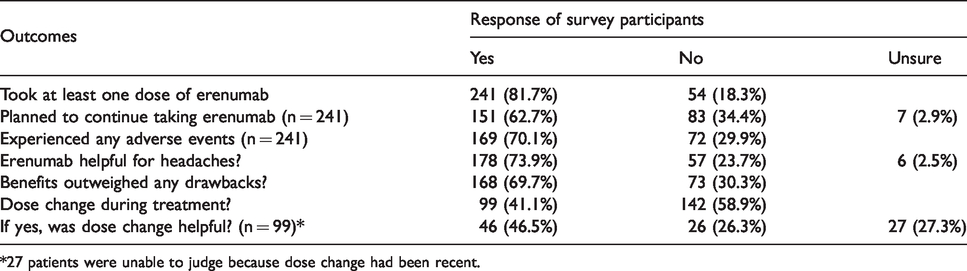
*27 patients were unable to judge because dose change had been recent.
Seventy percent of participants who used erenumab (169/241) reported experiencing at least one side effect, and 12% of the participants discontinued the treatment for that reason (Table 3). Participants who discontinued erenumab reported taking a mean of four doses (range 1–12). One hundred and sixty-eight participants (69.7%) reported that they had more benefits from taking erenumab than problems. One hundred and fifty-one participants (62.7%) planned to continue to take the medication. Some participants who reported more benefits than problems did not plan to continue the medication, due either to cost or to partial effectiveness (leading to the decision to try a different CGRP monoclonal antibody).
Factors affecting use of erenumab (n = 241 survey respondents).
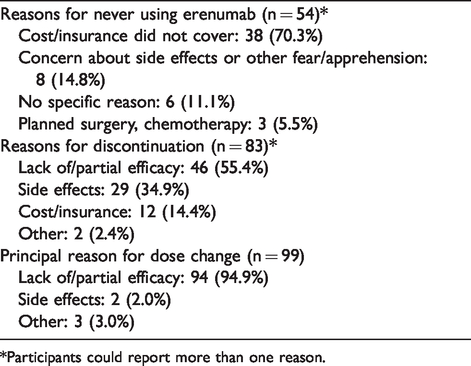
*Participants could report more than one reason.
Dose change included increases or decreases from the starting dose during treatment. Most participants who had a change in dosage reported lack of effect or partial effect as the reason for the dose change (94/99, 94.9%). The remainder were either due to adverse events (2, 2%) or for an unknown reason (3, 3%). Forty-six participants (46.5%) found the dose change to be helpful.
Factors affecting use of erenumab
Participants who were prescribed erenumab but never used it reported cost or inadequate insurance coverage as the main reason (38/54, 70.3%) while those who received the medication but discontinued it reported lack of or partial effectiveness as the main reason for discontinuation. Table 3 details factors affecting treatment with erenumab. Including participants who did not use the medication due to cost as well as those who discontinued the medication for this reason, cost was a prohibitive factor for 50/295(16.9%) of all participants who received a prescription for erenumab.
Adverse events
Adverse events reported by telephone structured clinical interview participants are listed in Table 4, organized according to MedDRA category. The 169 participants who reported adverse events described 384 individual instances. Constipation was the most commonly reported specific adverse event (104/241, 43.2%), followed by injection site reaction (n = 58, 24.0%).
Adverse events categorized by MedDRA (Medical Dictionary for Regulatory Activities) groups.

One participant reported a serious adverse event of hospitalization for small bowel obstruction, which we felt was related to erenumab use. The participant was hospitalized after starting erenumab, and the timing was therefore felt to be supportive of a causal relationship. None of our structured clinical interview participants had experienced a vascular event (myocardial infarction, stroke, or transient ischemic attack) by the time of the structured clinical interview.
Chart review
Seventy-four patients could not be reached by phone and were assigned to the chart review group. Results from the chart review population are summarized in Table 5. Fifteen patients had no available data in the medical record and we could not determine whether they took the medication or not. Fifty-one patients took the medication, of which 36 (70.6%) were planning to continue taking it at the time of the chart review, while the remainder had discontinued it. Reasons for discontinuation were lack of benefit (7/15, 46.7%), adverse events (4, 26.7%), cost (1, 6.7%), unexpected pregnancy (1, 6.7%), and unknown (2, 13.3%).
Erenumab outcomes in chart review population (n = 74).

Adverse events were noted in the charts of 13 patients (25.5%), who reported 20 individual adverse events. There was clear documentation that there were no adverse events for 12 patients (23.5%) and no documentation of the presence or absence of adverse events in 26 patients. We chose to code records that did not mention side effects as missing data, rather than evidence of lack of side effects. As in the structured clinical interview participant population, the most commonly reported adverse events were constipation in five (9.8%) patients and injection site reaction in three (5.9%) patients. Erenumab was recorded as helpful in 38/51 (74.5%) of the patients. Details about dose changes in this population are presented in Table 5.
We identified one death in the group of patients using erenumab. Although this patient was excluded from the outcomes study population, we performed a chart review to gather details about the cause of death. It was not possible to determine whether this death was related to erenumab use as no details were available in the chart. We did not identify any vascular events during chart review.
Spontaneous comments
One hundred and fifty-five people offered spontaneous comments about erenumab. Table 6 lists several themes that emerged, along with selected quotations that illustrate common or noteworthy experiences.
Themes emerging from spontaneous patient comments.

*Patient comments are summarized and paraphrased; verbatim remarks are indicated with quotation marks.
Discussion
Roughly two-thirds of patients prescribed erenumab thought it was helpful in treating their headaches and that the benefits outweighed any problems. Among patients who reported a dose change, the majority found the increased dose more effective. This suggests that although there was no statistically significant difference between the 70 mg and 140 mg doses in the clinical trials, individual patients in clinical practice may benefit from the higher dose (4). At the time of the structured clinical interview, most patients were planning to continue the treatment. Our findings regarding effectiveness are consistent with efficacy data from the phase 3 clinical trial (4).
An important finding was that the cost of the medication or inadequate insurance coverage influenced initiation or continuation of erenumab in about 1 in 7 eligible patients. This is an early and important signal of the role of cost as a barrier to treatment. This bears out early concerns and projections that not all patients who need it will have access to new treatments (11,12).
Notably, our study found a high incidence of adverse events, with 70% of participants reporting at least one side effect. Our study suggests that the burden of adverse events seen in clinical practice may be much higher than seen in the phase 2 and 3 clinical trials (4–6). Table 7 shows a composite of all adverse events reported across all the phase 2 and 3 clinical trials for comparison. Consistent with results from erenumab clinical trials, gastrointestinal disorders were the most commonly reported adverse events, followed by the major grouping that includes injection site reactions. Interestingly, participants in our structured clinical interview frequently reported symptoms categorized as nervous system disorders, a pattern that differs from clinical trial results. This group of symptoms includes worsening headache, reported by 10% of participants. Worsened headache is implied by the average headache day versus responder results in the erenumab clinical trial results. In the clinical trials, 15–20% of the patients on the 70 mg dose, 12–21% on the 140 mg dose, and 28–30% of the placebo group had “no change or more migraine days per month” (17). Dizziness was also commonly reported by participants in our study, and participants also reported sleep disturbances. These findings are particularly interesting because the CGRP mAbs are not thought to cross the blood brain barrier (13). Other symptoms that may warrant further monitoring and investigation include musculoskeletal complaints, particularly muscle spasm and joint aching. Reassuringly, we did not identify any signal of increased risk of vascular events.
Adverse events reported in erenumab phase II/III clinical trials.

On 8 October 2019, the product label for erenumab was updated to include a statement of safety concern for “constipation with serious complications” (14). Our findings support that this is a frequent and potentially serious complication. The August 2019 Quarterwatch report, a publication of the non-profit Institute of Safe Medication Practices, summarized available MedWatch data regarding CGRP monoclonal antibodies (15). According to the report, erenumab ranked first in the number of adverse event reports among 33 new drugs approved in 2018. The report attributes this finding partly to the high number of patients using this medication, which was estimated at 93,000 patients monthly. A total of 10,508 adverse events were reported, of which 1458 were with a serious outcome. Unsurprisingly, constipation was the most commonly reported side effect, but a clear signal for alopecia, which was not reported in clinical trials, was identified with 376 reports. The Quarterwatch analysis raised concerns that musculoskeletal adverse events could have been underestimated in clinical trials given the frequent MedWatch reports of muscle spasms, arthralgia, and myalgia (16). Potentially life-threatening anaphylactic shock was also reported in 35 cases, and a risk of hypersensitivity reaction was added to the erenumab label in March 2019. No cases of life-threatening anaphylaxis were identified in our study, but several allergic reactions were identified.
The difference in adverse events between our study and phase 2 and 3 clinical trials of erenumab could be attributed to many factors, including patient characteristics, the methodology for detecting and reporting adverse events, the sample size and duration of treatment, and patient expectations. Our patients were, on average, older than those in the clinical trials and may have had a higher burden of comorbid medical conditions and concomitant medications. The specific methodology for eliciting adverse events in the phase 2 and 3 clinical trials of erenumab is not clear and could not be determined from the primary publications. A review of FDA documents also notes that this could not be determined (17). It is possible that adverse event ascertainment in erenumab clinical trials involved asking open-ended questions only, which is known to be suboptimal and in particular may lead to underreporting of psychiatric, sexual, or other adverse events (18). Clinical trials are typically designed to focus on efficacy endpoints rather than adverse event endpoints. Therefore, these studies may be underpowered or have follow-up periods of insufficient duration to detect such adverse events compared to the long duration of follow-up in clinical practice (19). Lastly, it is possible that clinicians’ discussion of common adverse events with patients leads to more vigilant tracking of specific adverse events or even produced a “nocebo” effect (20). All providers in our headache center discussed potential risks of erenumab and limitations in available data with patients prior to starting erenumab.
One unintended pregnancy occurred among the 369 patients in the study, which corresponds to an incidence of 0.3% (95% confidence interval 0–1.5%). This is of concern, since product labeling specifically warns against pregnancy in those receiving treatment based on the unknown effects of the antibody on pregnancy outcomes (21). Our study suggests that unintended pregnancy is likely to occur in patients using erenumab, and the effect of CGRP blockade on pregnancy outcomes should be a research priority. We also identified a patient death in the group of patients who had received a prescription for erenumab. Although we are unable to link the death to erenumab use, we are also unable to rule out a relationship.
We are aware of two other fully published studies of erenumab use in a real-world setting (22,23). In the first, the investigators reported outcome in 220 patients with chronic migraine who had received at least one dose of erenumab (22). In this retrospective study, 58% of patients reported at least 30% improvement in headache frequency. As in our study, the prevalence of adverse events was much higher compared to clinical trials (constipation 20%, nausea 7%, increased headache 5%, fatigue 5%, depression 3%, joint pain 3%, injection site reaction 2%, diarrhea 2%, and anxiety 2%). Three severe adverse events were identified: A migraine-related stroke, severe neurologic symptoms, and a case of severe fatigue and joint pains. The second paper reports on 78 patients with migraine (13 episodic migraine (EM), 65 chronic migraine (CM)) treated with at least one dose of erenumab in an Italian clinic (23). In this population, 100% of those with EM and 87.5% of those with CM reported benefit. Only one adverse event, injection site erythema, was reported. The early experience of two Australian headache centers was presented in poster format at a recent conference (24). Among 65 patients who had used erenumab for at least three months, 19 (29%) reported a 50% or greater decrease in overall headache days and 27 (46%) reported a 50% or greater reduction in migraine days. Twenty-seven (42%) and 14 (27%) reported no improvement in headache or migraine days, respectively. The investigators reported “there were few reported side effects” but did not provide information about how that was determined. It seems likely that the reported effectiveness of the treatment was high and the prevalence of side effects was low because the study was limited to those who had continued the medication for 3 months.
A major strength of our study is the high response rate to our structured clinical interview. The systematic, individual questioning of adverse events combining open ended and checklist methods may be more sensitive than other methods of ascertainment. The high response rate is reassuring that these results are likely to be representative of our tertiary headache population. Our assessment of global perception of benefits compared with drawbacks provides information that clinicians and patients often seek when considering a new treatment option. We believe that the information about dose change and the outcome of that change can also provide guidance to clinicians in their day-to-day practice.
Our study has some limitations. It was carried out in a single site and consisted of a population that was 85% white, which may affect generalizability of the results. The retrospective study design limited our ability to objectively track treatment response, and we focused on patient perception of benefit. Additionally, some patients discontinued treatment due to partial effectiveness and in some cases their physician decided to switch them to a different medication (mostly another CGRP monoclonal antibody) in hopes that they would get more benefit. This may lead to the underestimation of benefit from erenumab in our study. Hospitalizations were only captured if they were reported by participants and we did not formally query them about this in the structured clinical interview. Based on discussions with colleagues in our headache center, we believe that not including this question on the structured clinical interview resulted in underestimation of such serious adverse events. Finally, the duration of erenumab use varied among study participants, since information was not collected at a standard time after the medication had been used. Thus, our findings constitute a snapshot of early patient experience with the treatment, but do not provide information about longer-term use.
Conclusions
Erenumab is an effective treatment option for patients with episodic and chronic migraine. Most patients in our study were planning to continue to take the medication, as benefits were judged to outweigh the drawbacks. However, post-marketing studies, including ours, identified new adverse events and a much higher rate of known adverse events than identified in the erenumab phase 2 and 3 clinical trials. Clinicians should be cautious about underestimating the rate of adverse events or dismissing adverse events reported by patients as irrelevant to erenumab, and should instead report, or encourage their patients to report these to the manufacturer and the FDA (25). Our study also provides early quantification of the frequency of unintended pregnancy in real-world use.
Key findings
In a real-world clinical setting, erenumab was effective in the majority of patients. The majority of patients felt that benefits outweighed adverse events. Adverse events were much more common in the clinic-based study population compared to clinical trial results. Cost was a barrier to treatment for 12% of participants.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SK and GH have nothing to disclose. EL receives salary support from the BMJ for her services as an editor. RB is an Associate Editor for the journal Headache. RB’s spouse is an employee of CarePort Health, a subsidiary of AllScripts.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a generous donation from the Bill and Joan Alfond Foundation to the John Graham Headache Center.
