Abstract
Background:
Secukinumab, a fully human monoclonal antibody, was approved in Korea for the treatment of moderate to severe psoriasis in September 2015.
Objectives:
To assess the safety and effectiveness of secukinumab in patients with moderate to severe psoriasis in Korea.
Design:
Multicenter, real-world, noninterventional study conducted over 6 years.
Methods:
Adults with moderate to severe psoriasis were enrolled. Safety was assessed by evaluating adverse events (AEs), treatment-related AEs, and serious AEs (SAEs). Effectiveness was assessed using the change in absolute Psoriasis Area and Severity Index (PASI) score, percentage of patients achieving PASI 75/90/100 and PASI ⩽2; at weeks 12 and 24.
Results:
Overall, 829 and 542 patients were included in the safety and effectiveness sets, respectively. AEs, treatment-related AEs, and SAEs occurred in 29.0%, 9.5%, and 4.1% of patients, with incidence rates of 39.43, 12.98, and 5.59 per 100 patient years, respectively. The absolute PASI score decreased from 16.1 ± 7.1 (baseline) to 1.6 ± 2.4 (week 24), with a similar reduction in biologic-naïve (16.4 ± 7.3 to 1.5 ± 2.2) and biologic-experienced (14.8 ± 5.9 to 2.4 ± 3.2) groups. At week 24, PASI 75/90/100 was achieved by 95.1%, 62.4%, and 24.9% of patients. At week 24, PASI 75/90 were higher in biologic-naïve (96.6%/65.8%) than biologic-experienced patients (88.3%/48.6%), whereas PASI 100 was similar in both cohorts (24.1% and 28.6%). A similar trend in PASI ⩽ 2 was observed in both cohorts.
Conclusion:
Secukinumab showed sustained effectiveness and favorable safety profile in adult patients with moderate to severe psoriasis in Korea.
Introduction
Psoriasis is an immune-mediated chronic inflammatory disease characterized by relapsing scaly plaques, with a substantial impact on patients’ quality of life. 1 The increased feed-forward activation of the adaptive immune system is a pathophysiological feature of psoriasis.2,3 The activated dendritic cells result in the release of interleukin-12 (IL-12) and IL-23, which facilitates the differentiation of T helper1 (Th1), Th17, and Th22 cells. The Th1, Th17, and Th22 cells release interferon (IFN)-γ, tumor necrosis factor-α (TNF-α), IL-17, and IL-22, which cause keratinocytes to undergo intracellular signal transduction, resulting in an inflammatory loop leading to psoriatic disease.2,3
As per the World Psoriasis Day consortium, psoriasis has a worldwide prevalence of 2–3%. 4 In the USA and Europe, 2–4% of the population is affected by psoriasis, 5 but the prevalence is low in East Asian countries (0.14%), including Korea (0.44–0.45%).6,7 Plaque psoriasis is the most common manifestation of psoriasis, accounting for 83.8% of cases in Korea. 8 The evolution of psoriasis treatment has mostly revolved around treating plaque psoriasis; with the biologics entering the market, the therapeutic outcome has significantly improved owing to their effectiveness and favorable safety profile.
Secukinumab is a fully human monoclonal antibody that selectively neutralizes IL-17A and has proven to be effective in the treatment of multiple manifestations of psoriatic disease, including psoriasis localized to the nails, scalp, palms, soles, and joints.9,10 Besides psoriatic disease, secukinumab has recently been approved by the European Commission and the U.S. Food and Drug Administration (USFDA) for the treatment of moderate to severe hidradenitis suppurativa in adults.11,12 Secukinumab has shown sustained long-term efficacy and a favorable safety profile in adult patients with moderate to severe plaque psoriasis in four pivotal randomized controlled trials (RCTs).13–15 Other biologics for the treatment of moderate to severe psoriasis include IL-12/23 inhibitors (ustekinumab), IL-17 inhibitors (ixekizumab, bimekizumab, and brodalumab), IL-23 inhibitors (ustekinumab, guselkumab, risankizumab, and tildrakizumab), and TNF-α inhibitors (etanercept, infliximab, and adalimumab).16–18
Since patients’ clinical characteristics may differ from those enrolled in RCTs, the results from these RCTs cannot be generalized and may not match the real-world patient population. 19 For instance, older populations or patients with comorbidities may not be included in the clinical trial setting. Real-world studies provide valuable insights for various patient groups to further support the outcomes of the clinical trials.20–22 Therefore, it is essential to investigate the clinical responsiveness of secukinumab in a real-world setting. Many investigators have studied the effectiveness and safety of secukinumab in real-world practice.23–25 However, routine clinical practice differs from country to country, and limited data are available for the safety and effectiveness of secukinumab in a Korean clinical setting.19,26 Secukinumab (300 mg) was approved in Korea by the Ministry of Food and Drug Safety on 24 September 2015 for the treatment of moderate to severe plaque psoriasis in adults who require phototherapy or systemic therapy. 27 The granted re-examination period was 6 years from the date of approval up to 23 September 2021; this postmarketing surveillance (PMS) would, therefore, evaluate the safety and effectiveness of secukinumab for its approved indication in the real-world setting of Korea.
Methods
Study design and patients
This was a multicenter, noninterventional study conducted for up to 24 weeks from 24 September 2015 to 23 September 2021 at 70 participating institutions in Korea. Adult patients aged ⩾18 years with plaque psoriasis who have been prescribed and continuing secukinumab treatment before the site initiation or who were first prescribed after the site initiation were enrolled into this surveillance after signing a consent form. Patients with contraindications to secukinumab described in the approved package insert (e.g. hypersensitivity to secukinumab or severe hypersensitivity to its ingredients and/or clinically significant active infection) and/or patients who were participating in another clinical trial were excluded from this study.
Secukinumab (300 mg) was administered in accordance with the drug label: subcutaneous injections with initial dosing at weeks 0, 1, 2, 3, and 4, followed by monthly maintenance dosing (every 4 weeks). 27 The data of enrolled patients were collected by the investigators through electronic case report forms from the start of the PMS period until the target number of cases were achieved. This PMS was designed in accordance with the International Society of Pharmacoepidemiology Guidelines for Good Pharmacoepidemiology Practices (GPP), Pharmaceutical Affairs Act, Enforcement Regulation on the Safety of Pharmaceuticals, etc., Regulation on Control of Safety Information of Pharmaceuticals, etc., Standard for Re-examination of New Drugs, etc., and with the ethical principles laid down in the Declaration of Helsinki.
The written IRB/EC (Institutional Review Board/Ethics Committee)-approved informed consent was obtained from all eligible patients before collecting any data.
Data collection and outcome measures
Data were collected at visit 1 (baseline), visit 2 (week 12 ± 4 weeks), and visit 3 (week 24 ± 4 weeks). At baseline, data on demographic factors (age, sex, weight, and smoking history) and disease-related factors (disease severity, disease duration, family history of disease, current medical history), comorbidities, baseline Psoriatic Area and Severity Index (PASI), and history of previous biologic treatment were collected.
The safety of secukinumab was assessed by evaluating the adverse events (AEs), treatment-related AEs, and serious AEs (SAEs). AEs were coded using Medical Dictionary for Regulatory Activities terminology. Incidence rates were reported as the number of events per 100 patient-years (PYs) with 95% confidence intervals (CIs). In case of discontinuation of treatment in <12 weeks during the PMS period, the safety information was collected for 4 weeks after the discontinuation.
Effectiveness was assessed by evaluating the following: (a) change in absolute PASI score over a period of 24 weeks; (b) percentage of patients achieving at least 75%, 90%, and 100% improvement in PASI score from baseline (PASI 75, PASI 90, and PASI 100, respectively) at week 12 and week 24; (c) percentage of patients who had PASI ⩽2 at week 12 and week 24 in biologic-naïve and biologic-experienced patients. Only patients who adhered to criteria per local label and reimbursement guidelines (i.e. patients who had a PASI score of ⩾10 at baseline) were included in the assessment of effectiveness.
Sample size calculation and statistical analysis
Since the Pharmacovigilance Risk Assessment Committee has requested update to the European label with the term inflammatory bowel disease (IBD), it was identified as an important AE to be observed. In the recently published pooled safety analysis of 21 clinical trials, the incidence of new-onset IBD was 0.4%. 28 Poisson probability distribution formula was applied to calculate the sample size required to show statistically significant difference for an AE with an incidence rate of 0.4%. As a result, at least 749 patients were required to find one or more AE of interest (IBD) with an expression rate of 0.4% with a 95% probability of success. In this study, the dropout rate was considered 10% and proceeded to 821 cases.
Statistical analyses were performed using the SAS (version 9.4; SAS Institute, Cary, NC, USA).
The missing values were handled using observed case analysis method. The descriptive statistics for continuous variables were presented as mean, standard deviation, first quartile, median, third quartile, minimum, and maximum, and the categorical data were presented using frequency and percentage. The amount of change in the measured values at each time point and at the last visit was displayed graphically. Univariate analysis using categorical data analysis method (Chi-square test or Fisher’s exact test) was performed to identify the impact of previous biologic exposure status, sex, and weight (kg) on effectiveness and safety of secukinumab.
Results
Baseline characteristics of patients
Overall, 829 patients with plaque psoriasis were included. Of these, 66 (8%) discontinued the treatment. The major reason for discontinuation was onset of AEs [n = 41, 62.12%; which includes discontinuation due to lack of efficacy (n = 19/41)], followed by patients not visiting the hospital (n = 16, 24.24%), and others (n = 9, 13.64%).
The baseline demographics and disease characteristics of the patients are summarized in Table 1. Patients were predominantly men (67.7%); the mean ± standard deviation (SD) age of patients was 46.1 ± 13.3 years, and the mean duration of disease was 7.85 ± 9.6 years. The mean ± SD weight (kg) of the patients was 72.4 ± 14.6, with 68.9 ± 10.8 being in the <90-kg group and 101.0 ± 9.6 in the >90-kg group. At baseline, a majority of the patients (71.9%) exhibited moderate to severe psoriasis, whereas the remaining (28.1%) exhibited mild psoriasis with a mean ± SD PASI score of 12.0 ± 8.7 and a median PASI score (min–max) of 12.0 (0–54.6). The distribution of the density of PASI scores for all patients with psoriasis at baseline, week 12, and week 24 has been indicated in Figure 1. The most frequent comorbidity at baseline was hypertension (12.7%), followed by diabetes mellitus (7.6%). Overall, 19.4% of patients had prior exposure to biologic treatment, whereas 80.6% of patients were biologic-naïve.
Baseline characteristics of patients.
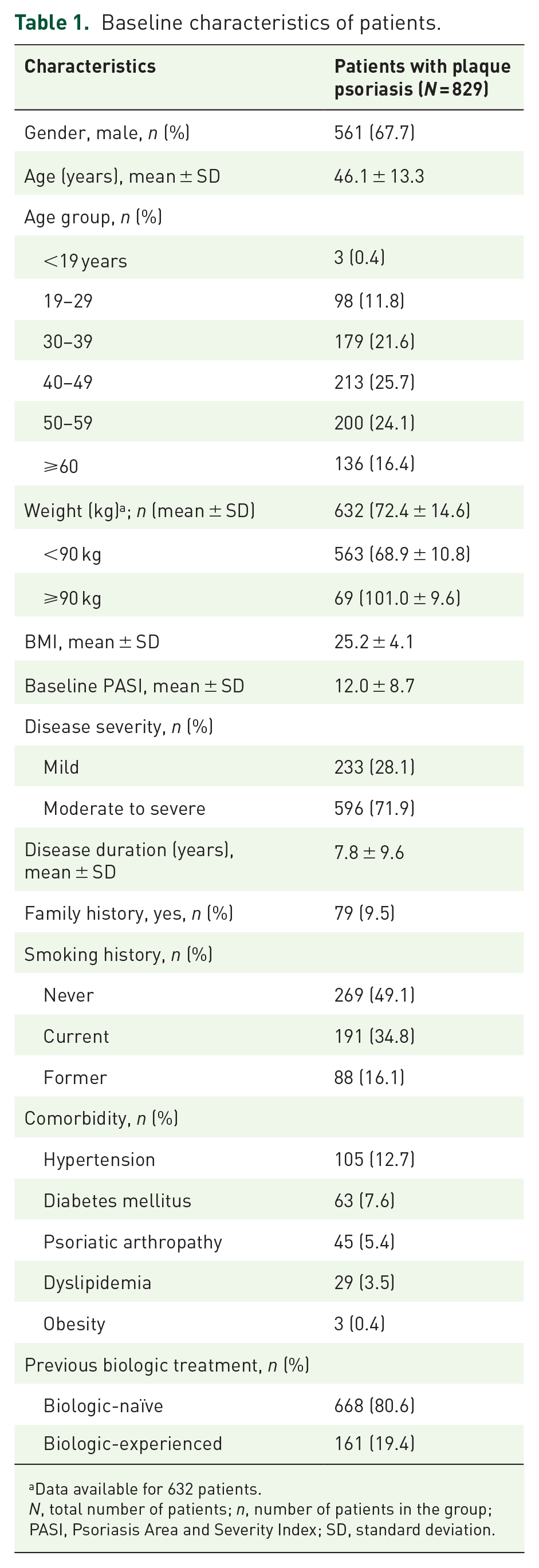
Data available for 632 patients.
N, total number of patients; n, number of patients in the group; PASI, Psoriasis Area and Severity Index; SD, standard deviation.

Distribution of the density of overall PASI scores at baseline, week 12, and week 24 in total patients (N = 829).
Safety
Incidence rate of AEs per 100 PYs
The total secukinumab exposure was 608.7 PYs, and the mean secukinumab treatment duration was 268.2 days (Table 2). Overall, AEs occurred in 240 patients [29.0%; exposure-adjusted incidence rate (EAIR)/100 PYs: 39.43]. The most commonly reported AE was pruritus, which occurred in 18 patients (2.2%; EAIR: 2.96/100 PYs). Treatment-related AEs were reported in 79 patients (9.5%; EAIR: 12.98/100 PYs), whereas SAEs were reported in 34 patients (4.1%; EAIR: 5.59/100 PYs).
Summary of the safety profile.

ADR, adverse drug reaction; AE, adverse event; CI, confidence interval; EAIR, exposure-adjusted incidence rate; IBD, inflammatory bowel disease; MACE, major adverse cardiovascular events; N, total number of patients; n, number of patients; PYs, patient-years; SD, standard deviation; SOC, system organ class.
The AEs of special interest included two patients with IBD without previous history (one case of Crohn’s disease and one case of ulcerative colitis; EAIR: 0.33/100 PYs). The incidence of ulcerative colitis was attributed to secukinumab administration and led to permanent discontinuation of the secukinumab treatment. There was one case of vulvovaginal Candida infections and one case of serious infections reported, with EAIRs being the same for these patients (EAIR: 0.16/100 PYs). There were no reports of major adverse cardiovascular events and malignancy. There were no deaths during the period of this surveillance. Weight, biologics experience, and gender didn’t show a significant influence on safety (Supplemental Table S1).
Effectiveness
In total, 542 patients (who had baseline PASI ⩾10) were included in the effectiveness assessment, of which 83.0% (n = 450) of patients were biologic-naïve and 17.0% (n = 92) were biologic-experienced. Overall, the absolute PASI (mean ± SD) score decreased from 16.1 ± 7.1 at baseline to 2.2 ± 2.7 at week 12 and 1.6 ± 2.4 at week 24. Similar improvements in absolute PASI score were reflected in both biologic-naïve and biologic-experienced cohorts [Figure 2(a)]. The percentage reduction in PASI score in the overall cohort was 89.4%, with a similar reduction observed in biologic-naïve and biologic-experienced cohorts [Figure 2(b)]. Overall, a high proportion of patients achieved PASI 75 at week 12, which was maintained until week 24 (95.1%).

Change in PASI over time. (a) Absolute PASI score. (b) Percentage change from baseline in PASI score.
A similar trend was observed in both the biologic-naïve and biologic-experienced cohorts (Figure 3). Likewise, PASI 90 and PASI 100 were achieved by 62.4% and 24.9% of patients overall, respectively, at week 24. A greater proportion of patients in the biologic-naïve cohort achieved PASI 90 at week 24 than biologic-experienced cohort, whereas the proportion of patients achieving PASI 100 at week 24 were somewhat similar in both biologic-naïve and biologic-experienced cohorts (Figure 3). Overall, the proportion of patients achieving PASI ⩽2 was 74.8% at week 24 with a greater proportion of patients in the biologic-naïve cohort achieving PASI ⩽2 than biologic-experienced cohorts (Figure 3). Weight, biologics experience, and gender didn’t show a significant influence on the efficacy (Supplemental Table S2).

Percentage of patients achieving PASI 75, PASI 90, PASI 100, and percentage of patients who had PASI ⩽2 over time.
Discussion
Despite reports of several real-world studies on secukinumab in psoriasis, there is a paucity of evidence pertaining to studies in Korea. This PMS evaluated the safety and effectiveness of secukinumab in patients with psoriasis in a real-world setting in Korea. No new safety concerns were identified during this PMS. Moreover, clinical benefits of secukinumab were demonstrated in all effectiveness assessments. It is noteworthy that majority of the patients remained under the secukinumab treatment during this study. Patients from over 70 institutions in Korea with moderate to severe psoriasis were included in the present study, indicating that these centers cater to patients with more severe forms of psoriasis in a routine clinical setting.
A majority of the population consisted of men, with a mean age of approximately 46 years, which is similar to that in a previous Korean study with ustekinumab, 29 and patients had a shorter mean disease duration than an epidemiologic study conducted in Korea where patients recruited at baseline exhibited 9 years of disease duration. 30 In line with another study, which evaluated the safety and effectiveness of ustekinumab for plaque psoriasis in Korea (biologic-naïve patients: 88.5%), 29 the majority of the population was biologic-naïve (80.6%) in the current study. This indicates that the use of biologics is limited or not widely accessible to a greater population in Korea. According to reports from 2017, only 5.7% of the patients with psoriasis were receiving biologics, 30 indicating that the use of biologics is limited to or not widely accessible to a greater population in Korea. Although the usage appears to be higher in the current study, the gap is evident with respect to another Asian study, where more than 50% of the enrolled patients were reportedly using biologics at baseline. 31 The percentages of biologic-experienced patients enrolled were, however, much higher in European studies assessing effectiveness, safety, and drug survival of secukinumab.32,33 Other studies assessing the effectiveness of secukinumab on patients with psoriasis also had higher percentage of patients exposed to biologics at baseline.9,34,35 These studies indicated much higher accessibility of biologics by the Western population compared to the Korean population.
Overall, the safety profile of secukinumab was consistent to that in previously reported real-world studies, and no new safety concerns were identified.19,24,33,36,37 The most common AE was pruritus, which is similar to the earlier reports in Korea. 29 Occurrence of treatment-related AEs was found to be much less than in a real-world study conducted in Japan (9.5% versus 24.2%). 31 In patients with psoriasis treated with secukinumab, EAIR per 100 PYs of approximately 0.3 was previously reported for ulcerative colitis and Crohn’s disease, 25 which is similar to the current study. A previous study reported that the use of biologics increases the risk of infections 38 and that secukinumab is associated with a slightly increased risk of infections compared to the other biologics. 24 However, in the current study, infections were reported in only two patients.
In the current study, there was an improvement in all effectiveness parameters analyzed in the overall patient population. The mean PASI at baseline in our study (12.0) was lower than that in the RCTs [CLEAR (21.7), ERASURE (22.5), and FIXTURE (23.9) studies].12,39 This may be due to the inclusion criteria of these studies, where patients with PASI scores⩾12 were recruited. The mean PASI was also different in patients who received secukinumab in HOPE (Australia), SUSTAIN (Australia), and DERMBIO (Denmark) studies (21.8, 13.0, and 7.3, respectively).9,24,36 This difference might reflect differences in treatment guidelines among countries and use of biologic treatment in real-world settings. 29 In the current study, the absolute PASI (mean ± SD) score decreased from 16.1 ± 7.1 at baseline to 1.6 ± 2.4 at week 24. Chiricozzi et al. 40 observed similar outcomes where the absolute mean PASI score decreased from 16.65 at baseline to 1.96 at week 24. A real-world study conducted in Italy showed a PASI score improvement over a 240-week period of secukinumab treatment, with 99.6% of patient’s achieving PASI <10 at week 24, which showed no patients discontinued the treatment over the long follow-up time in line with other real-world study enrolled across Europe.37,41 In this study, more biologic-naïve patients were included (80.6%) and showed numerically higher reduction in absolute PASI scores from baseline up to week 24 than the biologic-experienced patients. These results are in line with the therapeutic outcomes (PASI, body surface area, Investigator’s Global Assessment, Dermatology Life Quality Index) of patients with psoriasis of other real-world studies conducted in the USA, Italy, and Australia.9,33,42
The current study also reported that 74.8% of patients achieved PASI ⩽2 at week 24. This value is slightly higher than that reported in another European real-world study, 40 where 60.6% of patients achieved PASI ⩽2 at week 24 of secukinumab treatment. This difference could be because the current study has majority of biologic-naïve patients (80.6%) compared to the European study (47.6%). The proportion of patients achieving PASI 75/90/100 is consistent with those reported in the other real-world studies, albeit the duration of studies reported was diverse, ranging from 12 to 78 weeks,5,12,31,43 including the real-world data from a recent study conducted in Korea. 19 The PASI 75 response rates in the current study were comparable to those reported in ERASURE and FIXTURE, whereas PASI 90/100 response rates were lower in the current study. 12 A study by Galluzzo et al. 44 showed that PASI 75/90/100 was achieved more frequently by biologic-naïve patients at weeks 12 and 24. The same is corroborated by other real-world studies, wherein higher proportion of biologic-naïve patients achieved PASI 75/90/100 responses than biologic-experienced patients.9,31 The current study also demonstrated the same trend for PASI 75 and 90; however, the higher proportion of biologic-experienced patients achieved PASI 100 than biologic-naïve patients. Interestingly, a stark difference in the proportion of patients achieving PASI 100 in the biologic-naïve cohort versus biologic-experienced cohort was observed in another study from Japan. 31
We acknowledge the few limitations of the study. The study was subject to limitations inherent to observational real-world studies where no experimental conditions can be applied, and factors that might affect the safety and effectiveness of a drug are difficult to control, unlike in regulated clinical trials. Further, as the underlying conditions in patients with past medical histories and concomitant medications may make them vulnerable, the difference in the incidences of AEs is predictable. However, in an uncontrolled routine setting, the clinical significance of underlying conditions is difficult to determine with the limited data. Moreover, since there is evidence regarding the association of IL-17 inhibitors and IBD,45,46 investigators may have excluded the patients who are prone to develop IBD; therefore, the patient population may be biased.
Conclusion
In this large-scale PMS, secukinumab showed rapid effectiveness and favorable safety profile in adult patients with moderate to severe psoriasis, consistent with the previously conducted real-world studies across the globe. Moreover, this is one of the few real-world studies reporting the assessment of safety and effectiveness of secukinumab in patients with psoriasis in Korea.
Supplemental Material
sj-docx-1-taj-10.1177_20406223241230180 – Supplemental material for Real-world safety and effectiveness of secukinumab in adult patients with moderate to severe plaque psoriasis: results from postmarketing surveillance in Korea
Supplemental material, sj-docx-1-taj-10.1177_20406223241230180 for Real-world safety and effectiveness of secukinumab in adult patients with moderate to severe plaque psoriasis: results from postmarketing surveillance in Korea by Byung Soo Kim, Dong Hyun Kim, Bong Seok Shin, Eun-So Lee, Seong Jin Jo, Chul Hwan Bang, Yeojun Yun and Yong Beom Choe in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors thank the patients who participated in this study and the study investigators. The authors thank Debashree De, PhD, Rajib Kishore Hazam, PhD, and Nivedita Jangale, PhD (Novartis Healthcare Pvt Ltd, Hyderabad) for editorial and medical writing support, which was funded by Novartis Korea Ltd in accordance with the Good Publication Practice (GPP 2022) guidelines (![]() ).
).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
