Abstract
Mechanobiology investigates mechanotransduction—the conversion of mechanical forces into biochemical signals essential for development, homeostasis, and pathophysiology. While contact-based techniques provided foundational insights, they are limited by surface-centered analysis, substrate artifacts, and insufficient 3D spatiotemporal resolution. This review systematically explores non-invasive optical technologies designed to overcome these constraints. We first overview advancements in high-resolution 3D imaging and high-throughput platforms, followed by a discussion on label-free viscoelastic mapping via Brillouin microscopy and depth-resolved quantification via optical coherence elastography (OCE). We also address molecular-level readouts via fluorescence resonance energy transfer (FRET)-based tension sensors and pN-scale manipulation using optical tweezers, extending the research paradigm to active spatiotemporal control via optogenetics. Finally, we discuss the clinical potential of these tools, emphasizing their role in establishing quantitative biomarkers and mechanistic insights for diverse physiological and pathological conditions.
Keywords
Introduction
Mechanobiology is an interdisciplinary field integrating biology, physics, and engineering to study how cells and tissues perceive physical forces and transduce them into biochemical signals. 1 This process, known as mechanotransduction, is fundamental to regulating cellular functions and tissue behavior, and has emerged as a pivotal research area in life sciences and biomedical engineering.2–4 Cells respond to diverse mechanical stimuli—such as shear stress, tension, compression, and substrate stiffness—which not only induce structural adaptations but also directly modulate gene expression, protein activity, and cell fate determination.5,6 Consequently, mechanical signals are now recognized as key regulators of both the structural and functional/molecular properties of biological systems.3,7
Recent research has transitioned from observing macroscopic structural changes to elucidating specific molecular signaling pathways. This shift has established direct links between force transmission, cytoskeletal dynamics, nuclear remodeling, and genomic regulation, leading to the emergence of “mechano-medicine.” 4 In this framework, mechanical forces are utilized as both diagnostic biomarkers and therapeutic targets. While cellular mechanics research over the past four decades primarily focused on the cross-sectional cellular level, recent breakthroughs in high-resolution imaging and micromechanical analysis now allow for precise tracking of force transmission down to the organelle level, providing a critical turning point in understanding the spatiotemporal dynamics of intracellular mechanical signaling.8,9
Despite these conceptual leaps, the biological significance of mechanical stimuli can only be fully understood through a technological foundation for accurate, quantitative measurement. Early mechanobiology relied on macroscopic, contact-based methods such as atomic force microscopy (AFM) and micropipette aspiration to measure deformation or resistance to infer mechanical properties such as Young’s modulus. 10 While intuitive, these techniques often suffered from limited spatial resolution and lack of direct quantification of intrinsic mechanical parameters (e.g., Young’s modulus, shear modulus) and were heavily influenced by the rigidity of the measurement substrates, which often fail to mimic the complex physiological environment. Furthermore, traditional approaches like traction force microscopy (TFM) often provide qualitative observations rather than the high-resolution quantitative data required at the subcellular level. 10
Comparative analysis of conventional and optical technologies in mechanobiology research.

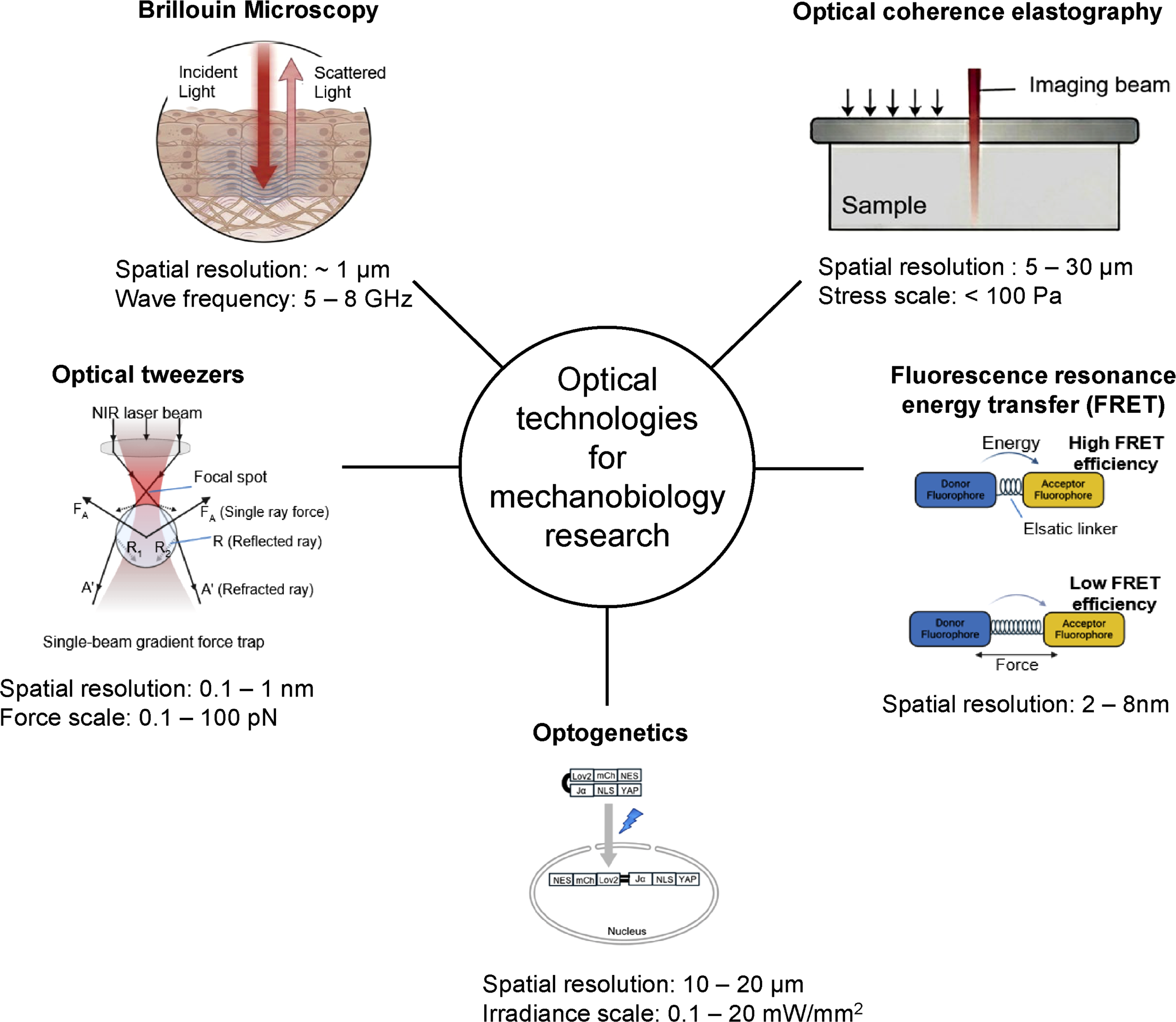
This review introduces the principles and applications of various emerging optical research techniques for mechanobiology and analyzes their contributions to elucidating the complexities of cellular dynamics. Furthermore, it presents the academic and clinical implications of these technologies across diverse fields, including oncology, dermatology, dentistry, and ophthalmology, highlighting their potential for future diagnostic and therapeutic strategies.
High-resolution optical imaging for mechanobiology
The mechanobiological mechanisms by which cells detect and respond to physical stimuli play a fundamental role in regulating cellular processes and are closely associated with diseases such as cancer metastasis and fibrosis. 17 In early studies, cellular mechanics were quantified by directly pressing or pulling cells using physical contact-based approaches. With the advent of non-invasive light-based probes, optical methods now offer the unique capability to measure physical properties deep inside biological samples without direct mechanical contact, minimizing damage to the specimen while enabling fast and large-scale data collection.
The core principle of advanced optical mechanobiology lies in quantifying the stress-strain relationship within living systems. 17 This process follows a three-stage framework: (1) perturbation, where controlled mechanical deformation is induced; (2) detection, where high-resolution optical modalities capture minute structural or refractive index shifts; and (3) reconstruction, where data is computationally processed into mechanical phenotypes like the elastogram.
This integrated approach transforms optical tools into multimodal platforms capable of simultaneously quantifying stiffness and molecular-level changes. 17 To enhance scalability, recent trends focus on user-friendly architectures and artificial intelligence (AI) integration, enabling automated analysis of complex mechanobiological datasets and accelerating their clinical translation.
Isotropic super-resolution via oblique plane microscopy (OPM)
High-resolution imaging of internal cell structures is essential for mechanobiology. Unlike conventional 3D imaging, which often shows blurry or distorted views from certain angles, oblique plane microscopy (OPM) achieves clear and uniform resolution in all directions using a specialized single-lens setup. 18 Without the need to physically move or shake the sample, OPM enables ultra-fast 3D recording. Its minimal light-induced damage and superior resolution have been demonstrated in the long-term 3D observation of the force-generating protein myosin IIA and internal organelle movements, providing critical insights into how mechanical forces travel inside the cell.
Real-time dynamics and multi-angle observation
Despite advancements in resolution, most 3D imaging takes too long to capture each layer (stack), making it hard to see very fast biological movements. To overcome this, projection imaging has been developed, enabling instant 3D observation from multiple angles at once. 19 This strategy allows for the continuous tracking of rapid shape-shifting, such as cancer cell surface bubbling (blebbing), and the direct monitoring of organ-level mechanical movements, including the rapid beating of a live zebrafish heart.
Large-scale and high-speed screening (DaXi & open-top)
Most high-resolution microscopes can only see a very small area at a time. To address this, DaXi technology expands the viewing area significantly without the need to stitch together multiple small images, which often creates artifacts. 20 This allows for the rapid, continuous observation of entire developing organisms, such as Zebrafish and Drosophila embryos. Furthermore, to observe many large samples (like organoids) simultaneously at single-cell detail, an “open-top” dual-view system has been developed. 21 While conventional systems are too slow for large-scale screening, this new platform allows easy access to multiple samples while maintaining high resolution even deep inside thick tissues. By capturing images from two sides and merging them, the system prevents the blurry “shadowing” effect seen in deep layers, enabling long-term 3D tracking of how cells differentiate in intestinal organoids.
Centimeter-scale analysis of entire organs
While small tissue analysis has advanced, capturing how an entire organ interacts across different scales remains a challenge. To overcome this size-versus-detail limit, new imaging techniques using specialized mirror-based optics have been introduced. 22 When combined with chemical treatments that make tissues transparent (optical clearing), these techniques enable nanometer-detail imaging of centimeter-scale samples, such as an entire intact brain, without the need to cut it into thin slices. This approach has facilitated the acquisition of massive 3D maps previously inaccessible, providing a foundation for understanding the physical structure of entire biological systems.
Measuring mechanical stiffness via optical coherence phase microscopy (OCPM)
Images alone cannot tell us how stiff or soft a cell is. To address this, optical coherence phase microscopy (OCPM) has been proposed to “feel” and monitor cell mechanical behavior in real-time without harming them. 23 OCPM can detect microscopic vibrations and shifts at the nanometer scale inside cells caused by slight pressure changes, allowing researchers to map out the relative elastic modulus distribution of cells in 3D. This technique has successfully distinguished cells with lower elastic modulus (MCF-7) from stiffer normal cells (3T3) and demonstrated high sensitivity to stiffness changes caused by drugs. This offers a powerful tool for testing how new drugs affect cell mechanics and studying cancer progression.
Super-resolution fluorescence microscopy and light-sheet microscopy
While OCPM quantifies mechanical properties, super-resolution fluorescence microscopy provides the necessary structural context by revealing how these mechanical signals are converted into biological responses. By enabling the analysis of physical contact points between nanostructures and cells, this technique elucidates the precise mechanisms of molecular deformation under force.24–26 Complementing this, light-sheet microscopy has established itself as an essential tool for long-term, non-invasive observation of living samples. It facilitates the quantitative sensing of physical parameters such as tension while maintaining nanoscale precision within complex 3D environments.27,28 Together, these high-resolution imaging modalities bridge the gap between mechanical force sensing and the visualization of downstream molecular remodeling.
Brillouin microscopy
While traditional imaging techniques excel at monitoring cellular morphology and dynamic movements, they often fall short of directly quantifying the inherent mechanical properties of biological materials. However, these mechanical properties—such as longitudinal modulus and viscoelastic properties —are far more than just a physical backdrop; they are key instructional signals that govern essential biological functions. For instance, mesenchymal stem cells (MSCs) decide their differentiation lineage based on compliance of their extracellular environment. 29 During Xenopus embryonic development, neural crest cells sense a change in the stiffness of nearby tissues to start their migration, a process known as the epithelial-to-mesenchymal transition (EMT). 30 Similarly, tumor cells respond to environments with higher matrix stiffness (i.e., increased effective elastic modulus) as a “green light” to grow faster, invade other tissues, and resist drugs. Furthermore, in cardiovascular health, the specific pattern of stiffness within arterial plaques is a critical indicator of whether they are likely to rupture and cause a stroke or heart attack.31,32 To address the need for measuring these mechanical forces without physically poking or damaging the sample, optical techniques have emerged—with Brillouin microscopy being the most prominent example.
Principles and technical improvements
Brillouin microscopy is a non-destructive and non-contact imaging tool that uses light scattering to probe living cells, overcoming the physical constraints of conventional probe-based measurements such as AFM.12,33 (Figure 1) This technology quantifies viscoelastic properties by detecting interactions with acoustic phonons, which are microscopic natural vibrations occurring within all materials. By analyzing the frequency shifts in light scattered by these internal thermal vibrations, the mechanical state of tissues and cells can be evaluated without any physical intervention. This approach reveals mechanical features that are invisible to standard optical imaging, such as distinguishing the liquid-to-solid phase transitions of intracellular components or mapping the localized mechanical weakening of tissues in corneal diseases. Recently, advanced scattering techniques have been introduced to resolve earlier limitations regarding signal intensity and acquisition speed, and machine learning is now actively utilized to enhance the accuracy of complex mechanical data interpretation. (a) Schematic illustration of Brillouin light scattering within biological tissue. (b) Representative Brillouin spectrum showing Rayleigh and ±νB components. (c) Frequency-dependent mechanical modulus spanning AFM and Brillouin regimes. The figure was created using BioRender.com.
High-resolution 3D mechanical mapping
To overcome the speed limitations of point-by-point sampling, confocal Brillouin microscopy using a virtually imaged phased array (VIPA) was developed. 34 This technology enables high-resolution 3D mapping of stiffness and facilitates real-time monitoring of rapid mechanical changes, such as polymer crosslinking, at 1-s intervals. This high-speed capability allows for tracking dynamic mechanical shifts in cell scaffolds and biological tissues. By measuring rat lenses in their natural state, researchers confirmed a mechanical gradient with increasing longitudinal modulus toward the center. This demonstrates the potential for evaluating biological tissues at the in vivo level without invasive extraction.
Intracellular modulus and mechanical homeostasis
Brillouin microscopy has extended tissue-level observations to the subcellular scale, enabling label-free 3D mapping of the intracellular longitudinal modulus. 35 This technology visualized mechanical signal generation by monitoring shifts in the intracellular solid-liquid volume fraction. A significant correlation between the high-frequency longitudinal modulus and AFM-derived Young’s modulus establishes the Brillouin signal as a quantitative indicator of cellular biomechanical states. Furthermore, research in diverse microenvironments reveals that cell mechanical modulus (e.g., longitudinal modulus) correlates strongly with projected cell area. This suggests that morphological adaptation is a primary mechanism regulating mechanical homeostasis across varying physical contexts.
Mechanical anisotropy in complex tissues
Brillouin microscopy now enables precise analysis of mechanical anisotropy in structurally complex tissues. 36 Using angle-resolved Brillouin microscopy, researchers quantified anisotropic properties driven by collagen fiber alignment. Verification via TEM confirmed that Brillouin signal patterns in corneal lamellae correspond to actual fiber arrangements, proving that extracellular matrix architecture is a primary determinant of tissue biomechanics. Furthermore, noninvasive mapping of anisotropy in living human corneas suggests significant clinical potential. This method allows for the early diagnosis of diseases like keratoconus, where pathological collagen remodeling leads to critical mechanical shifts.
Advanced Brillouin modalities for spatiotemporal and viscoelastic precision
To overcome the speed and phototoxicity constraints of point-scan methods, high-resolution line-scan Brillouin microscopy was developed for precise 4D mapping. 37 By acquiring hundreds of spectra simultaneously via a linear laser profile, this approach significantly enhances imaging speed while minimizing sample damage. This technique successfully captured transient stiffness shifts during morphogenesis in Drosophila and mouse embryos, establishing a direct link between mechanical dynamics and developmental biology. To resolve the dependence of the Brillouin frequency shift on refractive index and density, the Brillouin loss tangent (BLT) index was introduced as a refined viscoelastic indicator. 38 This parameter enables the observation of intrinsic mechanical properties by partially offsetting environmental variables. Using BLT, researchers identified a highly viscous mechanical shell surrounding maturing follicles, which serves as a compartmentalizing structural element. These microscopic viscoelastic changes were found to be directly mediated by ECM remodeling and cytoskeletal reorganization, specifically collagen and actin dynamics.
Optical coherence elastography (OCE)
Mechanical factors are critical in cellular function and disease. While AFM and TFM provide foundational insights, they are primarily limited to surface-level measurements, creating a technological gap in characterizing volumetric 3D biological environments.13,39 Optical coherence elastography (OCE) has emerged as a solution for noninvasive, quantitative 3D mapping of quantitative mechanical parameters (Young’s modulus, shear modulus, viscoelastic parameters) across multiple scales. 13
Building on Schmitt’s early research, OCE enables the quantification of complex parameters—including elasticity, viscoelasticity, anisotropy, and nonlinearity—by detecting nanometer-scale displacements.40,41 These capabilities establish OCE as a vital tool for assessing the mechanical behavior of spheroids, biomaterials, and engineered tissues in their native-like volumetric states.13,41
Principles of OCE
OCE involves three core steps: mechanical loading, OCT-based deformation mapping, and elasticity calculation via mechanical models. 42 Phase stability is maintained through Fourier-domain OCT (FD-OCT), while phase-sensitive detection enables nanometer-scale displacement measurements for quantitative analysis.
OCE is primarily classified by its loading mechanisms and corresponding continuum mechanics models (Figure 2): • Compression OCE: Measures axial strain along the optical axis to estimate Young’s modulus via Hooke’s Law. It is ideal for local stiffness mapping of microstructures due to its high sensitivity.13,42 • Transverse Wave OCE: Estimates shear modulus or Young’s modulus by measuring the velocity of waves induced by air pulses or ultrasonic pressure. Analyzing group or phase velocity allows for a detailed evaluation of viscoelasticity and dispersion properties.
13
• Axial OCE: Categorized into transient, harmonic, and vibrational OCE based on time-dependent load responses. Transient OCE can be used to analyze not only the elastic side of a segment but also the viscous direction, focusing on the degree of damping of the filament, making it useful for understanding time-dependent mechanical behavior. Harmonic OCE utilizes vibration amplitudes or resonant frequencies, while vibrational OCE simplifies the equations of motion to an elastic model when loading frequencies are below the sample’s natural frequency.
13
Principles of optical coherence elastography (OCE). (a) Compression OCE. (b) Transverse wave OCE. (c) Axial wave OCE. The figure was created using BioRender.com.

In conclusion, OCE bridges the resolution gap between micro- and millimeter scales, providing 3D, depth-resolved, and noninvasive quantitative measurements. By converting displacement data into intrinsic properties like Young’s modulus, OCE enables the interpretation of ECM mechanical signals and serves as a foundational technology for quantitative mechanobiology analysis.13,43
OCE measurements in cellular elasticity in 3D
Recent advancements in OCE have overcome the spatial and structural limits of conventional mechano-microscopy. To enhance 3D reconstruction, conditional generative adversarial networks (cGANs) are now used to minimize errors in elastic modulus distribution mapping. 44 Innovative modalities like light-sheet photonic force OCE (LS-pfOCE) enable high-speed in vivo 3D quantification by utilizing optical force for microscopic stimulation. 45 Simultaneously, quantitative micro-elastography (QME) has been applied to characterize cell-ECM interactions within 3D biomaterials, providing insights into mechanical signaling beyond the single-cell level. 46 Furthermore, subcellular mechano-microscopy based on optical coherence microscopy (OCM) allows for high-resolution 3D analysis of individual organelle elasticity, surpassing whole-cell resolution. 47 Integrating these techniques with GelAGE hydrogels has also demonstrated how spheroid stiffness adapts to the surrounding matrix, highlighting the impact of the 3D environment on cell aggregates. 48 These AI-driven and biomaterial-integrated platforms establish OCE as a vital tool for comprehensive 3D mechanobiology research.
Applications of OCE in cancer mechanobiology
OCE provides a high-resolution platform for characterizing mechanical interactions between cancer spheroids and the ECM. By utilizing cGANs for enhanced elasticity estimation, researchers have achieved precise visualization of micromechanical structures and tumor margins. 49 Comparative studies indicate that compression OCE offers superior specificity and accuracy for intraoperative breast tumor evaluation compared to conventional ultrasound elastography. 50 Innovative modalities like magnetomotive OCE monitor melanoma stiffness during hyperthermia therapy, establishing elasticity as a physiological indicator for treatment dosing. 51 In neuro-oncology, GPU-accelerated algorithms enable real-time 3D mechanical mapping, while miniature intraoral OCE systems identify cancerous tongue tissue by its significantly higher measured elastic modulus.52,53 Clinical trials with portable quantitative elastography probes have demonstrated up to 100% sensitivity in detecting residual tumors during surgery, transforming OCE into a robust intraoperative diagnostic platform.54,55
Applications of OCE in tissue-level mechanobiology for clinical diagnosis
OCE serves as a high-resolution tool for establishing quantitative mechanical parameters (Young’s modulus, shear modulus, viscoelastic parameters) as critical clinical biomarkers. 56 In ophthalmology, 3D quantitative mapping of ocular tissue elastic modulus provides insights into ECM reorganization during disease progression, utilizing broadband analysis to decouple geometric effects from intrinsic physical properties.57,58 In dentistry, high-frequency OCE enables non-invasive quantification of gingival biomechanics, revealing that elastic modulus varies by anatomical site and physiological state. The ability to analyze viscoelastic properties in the kHz band offers significant diagnostic potential for periodontal disease and oral tissue engineering. 59 Similarly, wave-based OCE has quantified cerebrovascular mechanics, confirming that arterial effective elastic modulus increases linearly with both pressure and aging. 60 Further, broadband guided-wave OCE has elucidated the nonlinear, anisotropic, and layer-by-layer properties of arterial walls, monitoring viscosity changes related to collagen dynamics. 61 In dermatology, broadband Rayleigh-wave OCE achieved the first depth-resolved quantification of epidermal stiffness through high-frequency dispersion analysis. 62 Collectively, these multi-scale data provide robust clinical indicators across diverse pathologies.
Recent technical advancements in OCE
Ultra-wideband OCE expands the analytical range from soft hydrogels to hard tissues by measuring elastic wave dispersion across extended frequencies. 63 This multiscale approach provides depth-dependent viscoelasticity data, bridging material, tissue, and organ levels. Reverberant 3D-OCE achieves high axial resolution (∼55 μm) by utilizing extended shear wavelengths for stereoscopic elasticity mapping, enabling precise differentiation of corneal layer deformations. 64 Additionally, common-path swept-source OCE enhances phase stability 40-fold by minimizing optical path fluctuations. Achieving ultra-sensitive displacement detection at 300 pm, this system enables precise mechanical measurements under minimal stimulus conditions. 65
Optical tweezers
Mechanical forces generated by cells are essential for cell adhesion, biochemical signaling, and differentiation, ultimately influencing cellular function. Thus, precisely measuring and manipulating them is essential. Optical tweezer (OT), proposed by Ashkin in the 1980s, was a powerful tool for measuring and controlling forces in the pN scale using a highly focused laser beam. 66
Principles of optical tweezers
Optical tweezers (OTs) are a technology that uses a laser beam to capture or move particles. The main forces are the scattering force that pushes particles in the direction of light propagation and the gradient force that pulls particles toward the direction of stronger intensity. The conventional optical levitation trap, introduced in 1971, had limitations due to its stability being affected by the balance between scattering and gravity.
67
In 1983, Ashkin proposed a single-beam gradient force trap, which utilizes gradient force to reliably trap particles. This trap is based on the principle that an axial intensity gradient generates a backward gradient force, pulling particles toward the focus of the light, stably trapping them. This technology enables the trapping of a wide range of particles from particles in the Mie size regime (∼10 µm diameter) to particles in the Rayleigh size regime (∼20 nm). The particle size was measured by comparing the scattered light from a 0.109 µm standard latex particle using the property that Rayleigh scattering is proportional to Ι ∝ (a) Principle of single-beam optical trapping that stabilizes a particle at the focal point. (b) Design of a FRET-based tension sensor module (TSMod) consisting of donor and acceptor fluorophores connected by an elastic linker. (c) Inverse correlation between mechanical force and FRET efficiency, enabling quantitative measurement of molecular tension in living cells. The figure was created using BioRender.com.
Initial trends in optical tweezer technology
Since its inception, OTs have been pivotal in manipulating biological entities without optical damage. Early studies successfully trapped Tobacco Mosaic Virus (TMV) and bacteria, providing size and volume estimates consistent with established structural data. 68 OT’s utility expanded to elucidating the dynamics of kinesin, a molecular motor. Initial research using OT to trap kinesin-coated beads supported the stroke-release model, though the system’s reliance on fixed positions limited its ability to capture transient detachment phases. 69 In 1993, the development of a single-laser optical trapping interferometer enabled nanometer-scale precision, facilitating the discovery of the hand-over-hand model. 70 This high-resolution approach directly observed kinesin moving in discrete 8 nm steps, corresponding to the spacing of α-β tubulin dimers.
Biological applications of optical tweezers
To overcome the limits of single-point trapping for complex morphologies, object-adapted OT was developed. By scanning a laser focus to match an object’s shape, researchers achieved high-precision tracking of helical bacteria (Spiroplasma) with 5–10 nm resolution. 71 This system enables real-time, stain-free 3D super-resolution imaging and quantifies mechanical energy transfer during motility by distinguishing bacterial energy states. OT applications have further extended in vivo, enabling the manipulation of red blood cells (RBCs) within living mouse capillaries. 72 By estimating trapping force constant (trap stiffness, k) at 10 pN/µm, researchers performed non-contact micro-operations to clear clotted capillaries. To maintain efficiency in biological environments, spherical aberration caused by refractive index mismatches was corrected using oil-immersion objectives, establishing OT as a robust tool for precise in vivo micro-manipulation.
Recent advances in measurement technologies of optical tweezers
Ballistic optical tweezers achieve 20 µs temporal resolution for intracellular viscosity measurement by utilizing velocity-based relaxation and structured-light detection. 73 This enables tracking of protein motor movements and cytoskeletal reorganization with femtometer-scale sensitivity. Hybrid systems integrating OT, magnetic tweezers, and fluorescence microscopy now simultaneously measure force and torque in DNA, elucidating the rapid fluctuation of plectoneme structures. 74 Earlier implementations relied on two laser beams—a drive laser and a probe laser—to drive and detect force. 75 In contrast, time-shared OT employs a single reciprocating laser to ensure trap consistency for active microrheology, quantifying liquid-to-solid transitions in biomolecular condensates and age-dependent cytoplasmic fluidization. 76 Furthermore, all-protein-based OT utilizing the Cohesin-Dockerin pair provides a handle-free method for protein folding dynamics. With over 600 pN mechanical stiffness, this system offers a significantly higher single-molecule pick-up frequency than conventional DNA-handle methods. 77
On-chip optical tweezers
On-chip OT has advanced through miniaturization and integration on single substrates. By implementing optical phased array technology, researchers extended the working distance to 5 mm, ensuring compatibility with standard cell culture dishes and coverslips. 78 Beyond simple trapping, automated OT systems now facilitate organelle-level manipulation, such as the noninvasive transplantation of healthy mitochondria into aged stem cells to improve metabolic efficiency and reduce age-related gene expression. 79 Furthermore, flexible and stretchable on-chip OT utilizing TiO2 microlenses on PDMS substrates enables high-throughput trapping of up to 1000 bioparticles. This system’s ability to adhere to curved biological surfaces and control cell-to-pathogen distances provides a versatile platform for studying cell-matrix interactions and immune responses in native-like environments. 80
Fluorescence resonance energy transfer (FRET)
The fluorescence resonance energy transfer (FRET) mechanism, proposed by Förster in 1948, describes the transfer of electronic excitation energy through dipole-dipole resonance interactions between energy donor and acceptor chromophores. 81 The FRET efficiency decreases as the distance between the two fluorophores increases, so this phenomenon can be exploited as a sensor (Figure 3(b) and (c)).
Principles of FRET
FRET sensors work based on the interaction of cholesterol sphingomyelin, two lipid components that play a critical role in the rigidity of cell membranes. A highly rigid membrane has cholesterol and sphingomyelin densely packed together, which shortens the distance between the two fluorophores, resulting in a high FRET signal. On the other hand, in membranes with low rigidity, when the cholesterol concentration decreases, the PFO-D4 domain separates from the membrane or becomes distant, resulting in a decrease or disappearance of the FRET signal. 82
Initial research of FRET for tension sensors
Grashoff et al. developed the Vinculin tension sensor (VinTS) to quantify pN-scale forces within living cells. This sensor utilizes FRET efficiency shifts; increased tension extends the peptide linker between donor and acceptor fluorophores, decreasing FRET efficiency. 81 Studies using VinTS revealed that vinculin undergoes high mechanical tension during focal adhesion (FA) assembly and at cellular protrusions. In contrast, tension decreases during cell contraction and FA disassembly, resulting in recovered FRET efficiency. These findings establish FRET as a vital tool for the real-time, quantitative analysis of intracellular mechanotransduction.
Evolution and recent trends of FRET tension sensors
FRET-based technologies have evolved to offer single-molecule precision and broad in vivo applicability. Molecular tension sensors (MTS) utilizing photostable organic fluorophores can now quantify forces in the 1-5 pN range on single integrins, revealing distinct tension distributions between subcellular and extracellular environments. 83 Internal cellular mechanics have been further elucidated through sensors embedded in the F-actin cytoskeleton. These tools identified mechanical anisotropy, where internal tension distribution varies with the direction of external deformation—a process actively regulated by myosin II. 84 Beyond the cellular scale, the integration of carbohydrate-binding modules with FRET sensors allows for real-time stress mapping in polysaccharide-based biomaterials and living tissues, such as tracking jumping mechanics in locusts. 85 Additionally, genetically encoded phosphatase and tensin homolog (PTEN) biosensors combined with two-photon fluorescence lifetime imaging (2pFLIM) enable the quantification of protein conformational changes in complex environments like the mouse brain. 86 These advancements establish FRET as a robust platform for diagnosing disease mechanisms through the lens of protein dynamics and tissue-level stress.
Optical manipulation in mechanobiology
In mechanobiology, high-resolution sensing is now complemented by the parallel development of active manipulation technologies. Optogenetics, utilizing photoreceptor proteins, enables the precise induction of mechanical signals with millisecond temporal and micrometer spatial resolution. By modulating light parameters, researchers can actively drive changes in the cytoskeleton and adhesion signals, expanding the field toward innovative outcomes in tissue engineering and regenerative medicine.
Molecular-scale control via optogenetics
Optogenetics is a technology that precisely controls cellular activity with light by utilizing ‘opsins’ (light-sensitive proteins). When these opsin genes are expressed in specific cells or tissues using viral vectors, the target cells become responsive to specific wavelengths of light. This allows researchers to activate or inhibit biochemical and mechanical signaling pathways with high spatiotemporal precision, enabling real-time regulation of complex cellular processes.
Initially, research primarily focused on photogated ion channels to modulate neuronal activity. However, modern optogenetics has greatly expanded its scope to encompass diverse genetically encoded systems that utilize light to induce protein-protein interactions or trigger conformational changes. Consequently, this technology has established itself as an innovative tool for the acute manipulation and analysis of dynamic protein functions within living cells.87,88
The constant reconstitution of the actin network and microtubules is central to intracellular force generation. Recent optogenetic studies have moved beyond simple activation to the precise tuning of these elements’ density and position. For instance, the OptoVCA system utilizes an iLID-SspB module to recruit the VCA domain of WAVE1 to the plasma membrane, inducing Arp2/3-mediated actin polymerization.
89
(Figure 4(a)) This system has demonstrated that the physical density of the actin network acts as an independent barrier, controlling the accessibility of protein complexes such as myosin. Similarly, the OptoTrap system overcomes the low spatiotemporal resolution of traditional genetic approaches by using light-induced clustering to temporarily halt the function of endogenous proteins, specifically dissecting the roles of Kinesin-1 and microtubules in neuronal development.
90
Furthermore, the RhoA optogenetics system serves as a molecular switch, coupling endogenous signaling pathways with exogenous modules to reversibly control RhoA—the master regulator of cell contractility—and redistribute traction forces through periodic light stimulation.
91
(a) OptoVCA system for blue-light-induced reversible actin polymerization via iLID–SspB interaction. (b) OptoYAP platform for light-dependent nuclear translocation via LOV2-based NLS exposure. (c) Wireless optogenetic trigger for mechanical-input-independent CGRP release. (d) MscL–cpGFP sensor for monitoring membrane tension via fluorescence quenching. (e) Azobenzene-based system for light-modulated substrate mechanics under 491 nm illumination. (f) Infrared-induced thermoviscous flow (1455 nm) for non-contact manipulation of suspended particles.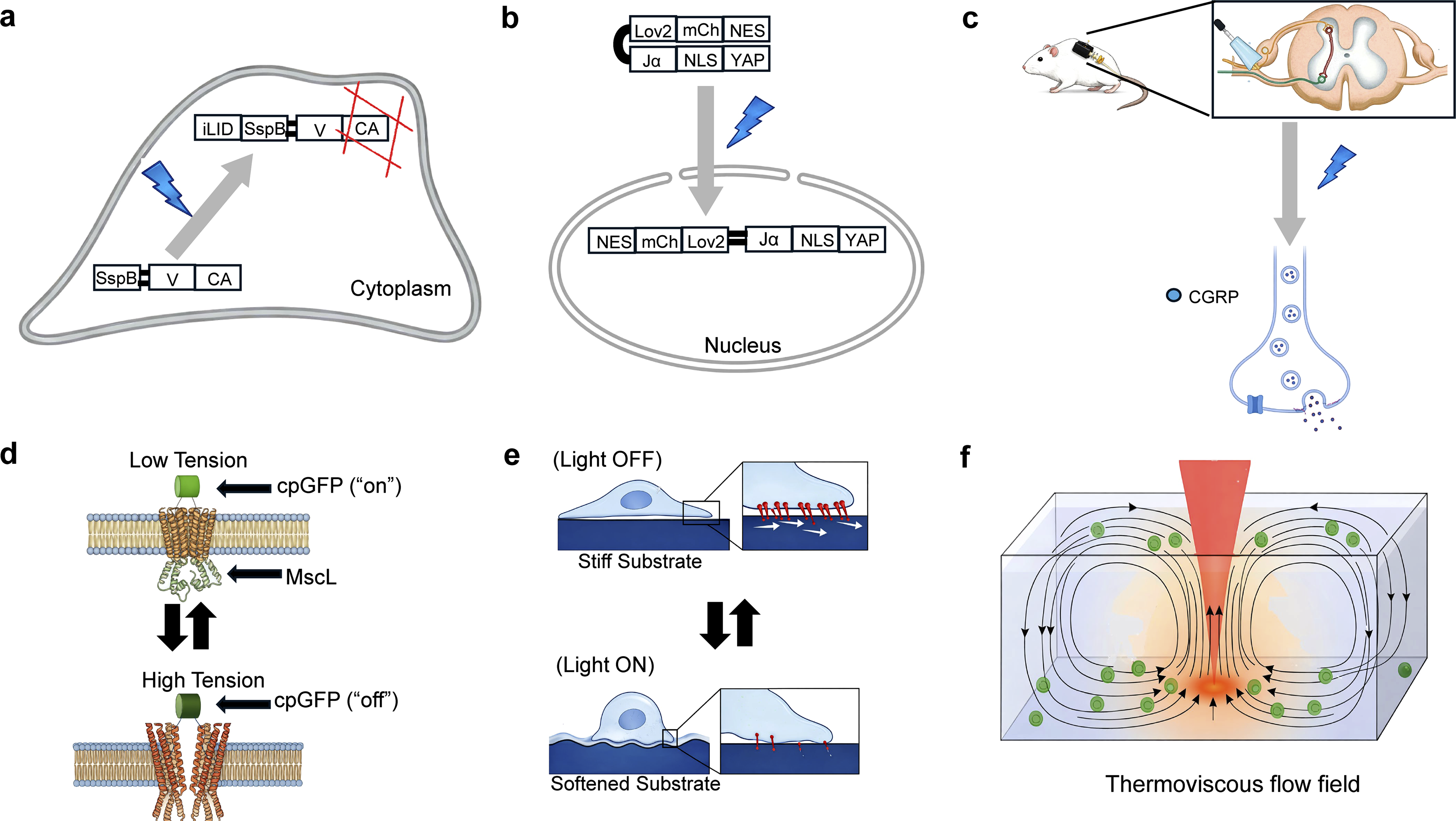
Light-mediated control also extends to focal adhesions (FA), where mechanical signals are converted into biochemical signals and transmitted to the nucleus via transcription factors like YAP/TAZ. The Opto-PXN system utilizes Cry2-mediated oligomerization to induce the phase separation of paxillin, proving that physical condensation alone is sufficient to trigger FA assembly and integrin activation. 92 To investigate downstream effects, OptoYAP technology was developed to directly control the nuclear translocation of YAP by using a light-responsive LOV2 domain that sterically conceals or exposes a nuclear localization signal. 93 (Figure 4(b)) This allows for the regulation of cell fate regardless of the external mechanical environment. Moreover, the integration of genome engineering with digital micromirror devices enables the projection of high-resolution light patterns to regulate cell responses and morphogenesis within complex 3D tissues, overcoming the instability and non-uniformity of transient transfection methods. 94
Physical actuation and environmental modulation
Beyond basic research, optogenetic mechanobiology is expanding into regenerative medicine and implantology. In musculoskeletal research, expressing Channelrhodopsin-2 (ChR2) in skeletal muscles allows for precise, non-invasive muscle loading. By applying blue light (455 nm) to induce isometric contraction, researchers can mimic physiological conditions without the systemic stress and variability of traditional exercise models. 95 This technology has been instrumental in analyzing the stage-specific mechanical adaptation of the Achilles tendon enthesis during development. Furthermore, to address the ‘stress shielding’ phenomenon, researchers developed a wireless optogenetic device to stimulate Piezo2-positive sensory neurons in the dorsal root ganglia. 96 This stimulation promotes the release of CGRP, enhancing bone formation and significantly improving the osseointegration of titanium implants even in the absence of physical loading (Figure 4(c)).
A diverse array of emerging technologies provides innovative solutions for sensing and applying force through light. Quantum-enhanced diamond molecular tension microscopy utilizes the spin characteristics of nitrogen-vacancy centers to monitor integrin traction forces without the photobleaching issues of conventional fluorophores. 97 Complementing this, the MscL(Mechanosensitive channel of large conductance)-based optical membrane tension reporter functions as an “OFF-sensor,” where the opening of mechanosensitive channels under tension causes fluorescence quenching of an inserted cpGFP module. 98 (Figure 4(d)) For tissue-level analysis, laser speckle micro-rheology provides a non-contact, passive method to map viscoelasticity by analyzing laser speckle fluctuations caused by natural thermal movements. 99
In terms of actuation, the nanoscale optomechanical actuator combines gold nanorods and thermoresponsive polymers to convert near-infrared light into pN-scale tensile forces on specific cell receptors. 100 Similarly, azobenzene-based thin films utilize photoisomerization to tune substrate stiffness and shape in real-time, effectively acting as molecular motors that modulate cell spreading and contraction. 101 (Figure 4(e)) Notably, light-driven artificial cell systems have been developed to convert external optical stimuli into autonomous mechanical work, such as the controlled beating of flagella, by using light as a direct energy source. 102 At the system level, optofluidic multiplexed assembly creates thermoviscous flow fields to manipulate particles and assemble microrobots with high degrees of freedom while minimizing cell damage. 103 (Figure 4(f)) Finally, the titanium micro-dish platform integrates high-throughput patterning with pulsed laser-induced nanobubbles to achieve highly efficient biomolecule delivery into thousands of cells. 104
Summary
Advanced optical technologies have introduced a transformative paradigm in mechanobiology, enabling high-precision 3D quantitative mechanical parameter mapping with unprecedented spatiotemporal resolution. Modalities such as OPM, Brillouin microscopy, and OCE have effectively overcome previous bottlenecks in acquisition speed and depth-dependent distortion, particularly through innovations like line-scan imaging and AI-driven signal reconstruction. Furthermore, the synergy between FRET-based molecular sensors and optogenetic actuation, and the high-resolution manipulation capabilities of optical tweezers has expanded the research scope from passive observation to active, dynamic control of mechanotransduction. These technological leaps provide a robust framework for an integrated, multiscale interpretation of biomechanics from the cellular level to entire organ systems. Ultimately, these advancements establish mechanical signals as essential quantitative biomarkers, opening new diagnostic and therapeutic possibilities across diverse physiological and pathological contexts.
Outlook
Compared to traditional measurement methods such as AFM, optical technologies have transformed mechanobiology by offering minimally invasive, multi-scale observation and manipulation. Unlike physical probes, light enables measurements without direct mechanical contact, reaching from molecular-level changes to dynamic whole-cell behaviors. 105
However, the characterization of optical methods as “non-invasive” requires careful qualification, as focused laser systems—such as optical tweezers and Brillouin microscopy—can introduce significant optical artifacts. Localized photothermal effects, radiation pressure, and light–matter interactions may inadvertently perturb cellular structures or alter mechanical properties. To mitigate these photophysical limitations, recent strategies have focused on employing pulsed illumination schemes (pulsed stimulated Brillouin scattering (SBS)), integrating adaptive optical control, and utilizing nonlinear techniques like two-photon microscopy to minimize photodamage while preserving measurement accuracy.106,107
Beyond these technical hurdles, the field is addressing the inherent scope of single-modality measurements through multimodal integration. Combining AFM with fluorescence microscopy or optical tweezers with FRET sensors allows researchers to correlate mechanical stimuli with real-time biochemical signaling, overcoming the trade-offs between resolution and physiological relevance. 105
Looking ahead, mechanobiology is evolving toward a predictive and clinically translatable science. This transition depends on three key trends: (1) the standardization of quantitative mechanical phenotypes for computational modeling; (2) the validation of these phenotypes as actionable biomarkers for diseases like cancer; and (3) the real-time, in vivo integration of sensors within organ-on-a-chip platforms. Ultimately, resolving these spatiotemporal complexities will transform optical tools into indispensable assets for next-generation precision medicine.106,107
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the New Faculty Startup Fund from Seoul National University (860-20250054) and the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (RS-2025-24913047).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
