Abstract
Objective:
To present the rehabilitation considerations in prehabilitation and the rehabilitation phase post spinal cord stimulator implantation; and to evaluate the efficacy of synergistic combination of epidural spinal cord stimulation (SCS) with motor imagery, trunk rehabilitation, robotic and overground gait training in improving mobility in persons with sensorimotor complete spinal cord injury (SCI).
Design:
A non-controlled before-after clinical trial.
Methods:
Epidural spinal cord stimulators were implanted into 3 participants with complete thoracic SCI (Thoracic 4, Thoracic 2 and Thoracic 10 respectively). They underwent 1 month of prehabilitation, followed by 7 months of daily rehabilitation post-implantation, consisting of SCS, motor imagery, trunk rehabilitation, robotic therapy and overground gait training. Pre-post assessments include the American Spinal Injury Association Impairment Scale (AIS) examination, Spinal Cord Independence Measure version III (Mobility subscale) (SCIM-III), Walking Index for Spinal Cord Injury II (WISCI-II), Trunk Assessment Scale for Spinal Cord Injury (TASS), Five Times Sit-to-Stand (5×STS) and 2-minute Walk Test (2mWT).
Results:
Post rehabilitation, there were improvements in WISCI-II, TASS, 5×STS and 2mWT in all 3 participants; and no change in AIS sensory scores. Only participants 2 and 3 exhibited slight improvement in SCIM-III. The WISCI-II of all participants improved, ranging from 3 levels to 9 levels, which was considered a clinically meaningful change. Furthermore, the change in TASS exceeded the minimal clinically important difference (MCID) of 4 points for two participants. The change in 5×STS exceeded the MCID of 2.27 seconds for all participants. These significant improvements translated into the participants’ ability to perform sit-to-stand independently and ambulate with a rollator frame under supervision of a carer.
Conclusion:
This clinical trial demonstrated proof-of-concept and promising findings that support our rehabilitation protocol in improving functional mobility of people with sensorimotor complete SCI, which may be replicated in future studies.
*The study protocol was registered with Clinical-trials.gov (NCT05644171; Registered 13 November 2022. https://clinicaltrials.gov/ct2/show/NCT05644171)
Keywords
Introduction
Spinal cord injury (SCI) is a profoundly debilitating condition resulting from injury to the spinal cord, leading to motor, sensory and autonomic dysfunctions. 1 In patients with motor complete SCI, regaining volitional control of movements and walking are the major immediate goals. 2 However, only 5% to 8.5% of people with sensorimotor complete SCI lesion (American Spinal Injury Association Impairment Scale [AIS] Grade A) regain some ability to walk. 2 To date, rehabilitation strategies, such as robotic therapy, 3 functional electrical stimulation 4 and motor imagery, 5 have been applied for rehabilitation of gait in SCI patients. Although these rehabilitation techniques have been used independently or in combination; there appears to be a lack of an organised rehabilitation protocol to effectively restore functional neurological function in patients with SCI.
Epidural spinal stimulation (ESS) delivered via a spinal cord stimulator (SCS) placed on the dorsal surface of the spinal cord has emerged as a promising approach for functional recovery in SCI patients.6,7 ESS involves the use of electrical impulses to stimulate the spinal cord and modulate its activity.6,7 Histopathological sections have determined that despite clinical diagnosis of motor complete spinal cord injuries, there may still be spared active neural connections across the site of injury but they are not robust enough to generate clinically observable functions. 8 ESS may thus potentially allow the exploitation of these spared but functionally silent descending pathways in order to produce movements of paralysed limbs 9 and translate task-specific sensory information into activity-dependent plasticity.8,10
Motor imagery is a dynamic cognitive process in which a person imagines that he/she performs a movement without any actual motor output. 11 Wang et al 5 demonstrated that motor imagery, evokes strengthened activation in sensorimotor areas in patients with complete SCI. Motor imagery also activates premotor cortex, supplementary motor area, cingulate and parietal cortical areas, basal ganglia and cerebellum, that are involved in movement preparation and execution. 12 Hence, motor imagery leads to movement-specific central activation patterns 11 ; facilitating the organisation of central motor commands and thereafter, the execution of active volitional movements. 12
Trunk muscles are essential for stability and mobility as they are involved in movements requiring an upright posture, such as sitting, standing or walking. 13 Studies have demonstrated absent or reduced trunk control in approximately 70% to 80% of people with SCI, 14 leading to negative interference with their daily activities, such as bed mobility, transfers, sitting balance and reaching. 15 Hence, rehabilitation techniques to promote activity of the trunk muscles is crucial to postural control and provide necessary movement for functional mobility.
Individualised, high repetitions and high-intensity gait training has the potential to activate the sensory inputs and central neuronal circuits, promote neuroplasticity and facilitate recovery in motor complete SCI.16 -18 However, manual assistance to facilitate every step of the motor complete SCI patient in therapeutic gait training sessions can be extremely labour-intensive, and often requires 2 or more therapists. Growing body of evidence supports the use of robotic technology to improve locomotor ability of people with SCI. 19 Robotic technology can aid patients to practise up to 1000 steps in 30 minutes of therapy compared to conventional physiotherapy in which patients with moderate to severe level of dependence achieve an average 100 steps in 30 minutes of therapy. 20
In our pilot trial (RESTORES trial: RESToration Of Rehabilitative function with Epidural spinal Stimulation), we explored the synergistic combination of multi-modality treatment involving ESS, motor imagery, trunk rehabilitation, robotic exoskeleton and overground gait training to restore functional mobility in 3 participants with chronic sensorimotor complete thoracic level SCI. In this manuscript, we present the rehabilitation considerations in prehabilitation and post ESS implantation. We shall discuss our rehabilitation protocol based upon this paradigm; and the clinical outcome post 7 months of daily intensive rehabilitation.
Methods
Study Design and Participant Selection
Our pilot trial, an uncontrolled before-after clinical trial, was performed in accordance with local Institutional Review Board approval (IRB reference number: CRIB 2022/2496). The 3 participants identified in this study, from a nationwide recruitment drive, had provided informed consent and authorisation to present publicly identifiable information for research purposes. The study protocol was registered with Clinical-trials.gov (NCT05644171; Registered 13 November 2022. https://clinicaltrials.gov/ct2/show/NCT05644171).
The inclusion criteria were: (i) aged 21 years or older; (ii) chronic (greater than 1 year) motor complete (AIS classification A or B) SCI; (iii) spinal injury between the levels of Thoracic 1 (T1) to Lumbar 1 (L1); (iv) segmental reflexes that remain functional below the level of lesion; and (v) able to perform the perioperative rehabilitation program as judged by the research team. Exclusion criteria were (i) significant medical co-morbidities that would significantly increase the risk of the operation; (ii) severe dysautonomia with systolic blood pressure fluctuation less than 50 mmHg or more than 200 mmHg on tilt table testing; (iii) painful musculoskeletal dysfunction, contractures, unhealed fractures, pressure sores, severe spasticity (Modified Ashworth Scale score 3-4), and osteoporosis; (iv) significant psychological issues or ongoing drug abuse; (v) pregnancy and lactating patients; and (vi) progressive spinal cord disease.
Rehabilitation Protocol
The clinical trial consisted of prehabilitation and post-operative rehabilitation phases. The rehabilitation protocol of the trial is detailed in the Appendix Figure 1 to Figure 29.
Prehabilitation is defined as the practice of enhancing a patient’s functional capacity prior to surgery to improve post-operative outcomes. 21 This has been shown to improve patients’ post-operative pain, disability, self-efficacy, psychological behaviours and satisfaction with surgical outcomes. 22 The prehabilitation phase was critical to enable adequate physical and mental conditioning of the participants before the post-operative rehabilitation phase. Our prehabilitation protocol consisted of twice-weekly rehabilitation, for a period of 4 weeks. In each week of the prehabilitation phase, the participants received one conventional physiotherapy session and one robotic exoskeleton therapy session, each lasting one hour.
The conventional physiotherapy focused primarily on core muscle strengthening and trunk control. A standing regime was essential to promote verticalisation and weightbearing tolerance by using the THERA-Trainer Balo (Medica Medizintechnik GmbH, Germany) (Appendix Figure 14). In addition, the THERA-Trainer comprised of virtual reality games, to engage the participants in fun and interactive ways, whilst actively practising weight shifts and trunk control in all directions. The ability to perform dynamic weight shifts is an integral component in overground gait training. The MOTOmed viva2 (RECK-Technik GmbH & Co. KG, Germany) electrical bicycle was used for maintaining the range of motion of all limbs. In addition, the participants need to practise motor imagery of the lower limb movements while cycling.
EksoGT (Ekso Bionics, Richmond, California, USA) is the wearable robotic exoskeleton used in this trial (Appendix Figure 25). The exoskeleton has actuators at the hip and knee joints, with a back support for the trunk. The software of EksoGT enables physiotherapists to customise step length, step height, swing time and percentage of robotic assistance in order to encourage active participation from the user. 16 The participants received weekly overground gait training with the EksoGT and practised dynamic weight shifts whilst familiarising with the robot’s gait pattern. This is key as the patients would need to perform dynamic weight shifting for overground walking in post-operative rehabilitation.
All participants underwent surgical implantation of a 3-column (5-6-5), 16 contact SCS (Specify SureScanTM 5-6-5 Medtronic) paddle leads on the dorsal dural surface of the spinal cord at Thoracic 11(T11) to Thoracic 12 (T12) for Participant 1; and Thoracic 12 (T12) to Lumbar 1 (L1) for Participant 2 and Participant 3 (Figure 1). Intraoperative stimulation (2 Hz, 450 microseconds, 0-10 mA) was performed to confirm the location of the paddle at the midline and to ensure adequate coverage of the intended muscles (rectus femoris, vastus lateralis, medial hamstring, tibialis anterior, medial gastrocnemius, soleus) using surrogate electrophysiological monitoring. The lead wires was connected to the implantable pulse generator (Medtronic Intellis AdaptiveStim).
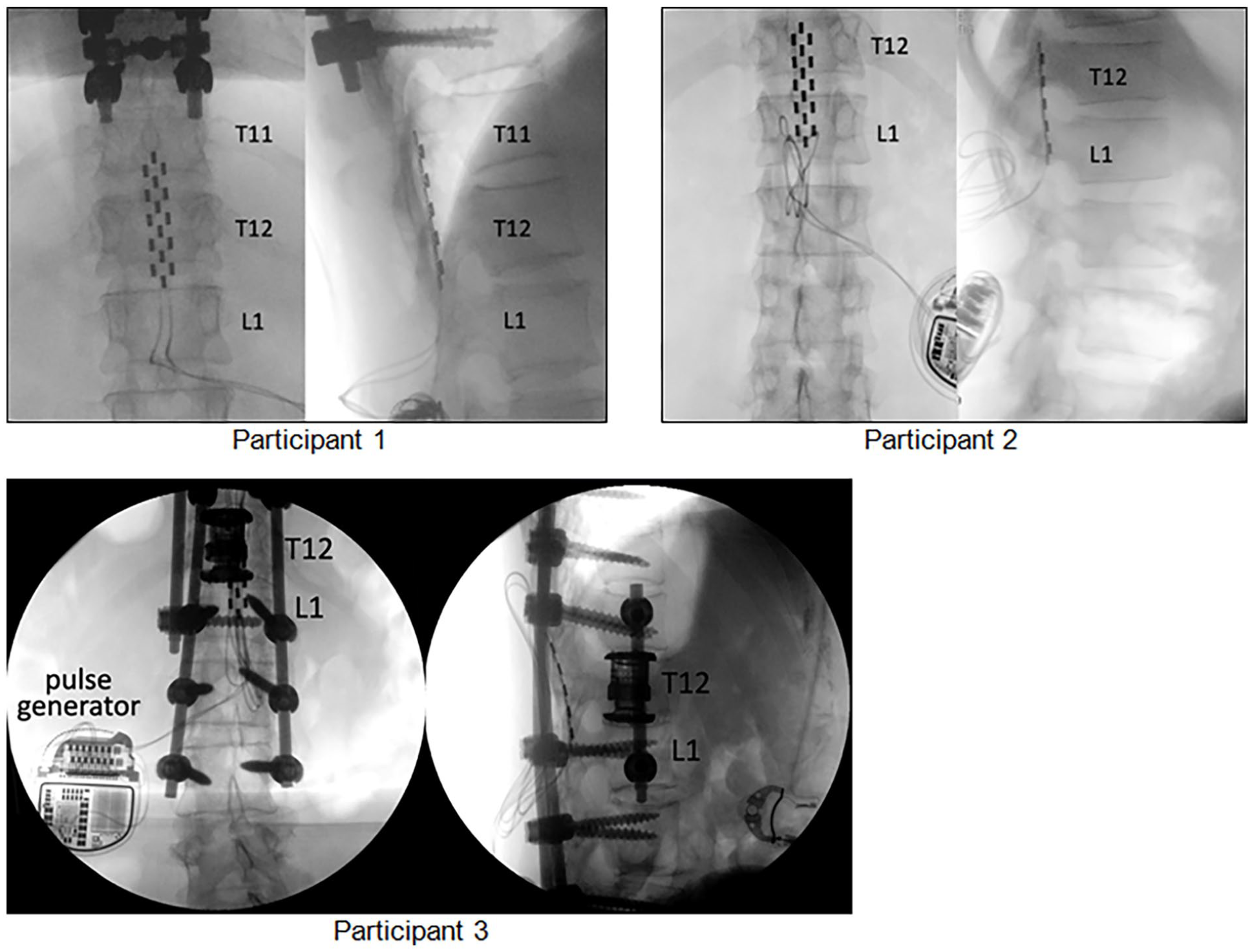
Intraoperative X-ray of the final placement of spinal cord stimulator (SCS) implants in the 3 participants.
Post-operative rehabilitation commenced 1 month post-implantation of the SCS to allow adequate surgical wound healing. The participants attended rehabilitation therapy 5 times per week for a period of 7 months. Such intensive rehabilitation was provided in view that the participants, who had chronic SCI, had not been standing or walking for numerous years post injury. They would require daily therapy to relearn different skill sets, such as performing sit-to-stand, weight shifting in standing, postural control, active attempts at volitional control and coordination of their lower limbs during overground gait training. The task-specific repetitive rehabilitation exercises were aimed at facilitating motor relearning of complex skills; hence, promoting neuroplasticity and motor recovery. Additionally, the daily therapy aimed to condition their bodies for the demands of functional mobility training, given their long-term dependence on a wheelchair, with the goal of gradually improving their cardiovascular fitness.
Each rehabilitation therapy session lasted 1 to 1.5 hours, depending on fatigue level of the participants. The spinal cord stimulator was switched on throughout the rehabilitation sessions. The rehabilitation programme consisted of the following:
The initial primary aim was to achieve targeted SCS programming to train volitional control of key individual muscle groups, such as rectus femoris, vastus medialis, vastus lateralis, hamstrings, tibialis anterior, gastrocnemius and soleus, with the epidural stimulator switched on. These key muscles are essential for sit-to-stand, 23 standing 24 and walking. 25
The spinal cord stimulation parameters, namely, intensity, pulse width and frequency, were customised per participant and during each rehabilitation session. Titration and customised mapping of the stimulation (2-5 Hz, 250-350 microseconds, 0-10 mA) was performed by palpating the targeted muscle groups for contractions and observed for active volitional movements of the intended muscles groups (Appendix Figure 1–3). During the mapping of stimulation, the participants were instructed to visualise the intended movement and practised motor imagery.
In our study, the participants coupled motor imagery with observation of the lower limbs in order to attempt movement practice (Appendix Figure 4–5). As part of the rehabilitation programme, the participants were instructed to practise motor imagery at home, with the spinal cord stimulator switched on, to build upon the movement practice in the daily therapy at the outpatient clinic. Key movements for motor imagery include: hip flexion, hip extension, knee flexion, knee extension, ankle dorsiflexion and ankle plantarflexion. They were also encouraged to imagine stepping movements in preparation for overground gait training.
We focused on trunk rehabilitation as many high thoracic level SCI patients tend to have difficulty maintaining trunk control due to trunk muscle weakness below the level of injury. As trunk control is closely associated with upper and lower limbs, it was essential to work on trunk control in order to improve the participants’ chances of ambulation.
Trunk rehabilitation for the participants included trunk control exercises in lying, sitting, balancing on gym balls, simulated hippotherapy and simulated kayak training (Appendix Figure 6–13). In the simulated hippotherapy session (Appendix Figure 10), the physiotherapist provided random perturbation and oscillation of the therapy thumbler in vertical and horizontal directions while the participant sat straddled across the thumbler. The random perturbation was aimed at challenging trunk control and participant’s alertness to respond to perturbation as part of balance training. Another variation of simulated hippotherapy includes strapping the therapy thumbler on 3 brackets of ceiling hoists so that the participant experienced simulation of sitting on a moving horse (Appendix Figure 11). As the 3 ceiling hoists were pushed to move anteriorly, it provided visual-vestibular stimulation for the participant; hence, simulating a horse moving forward in hippotherapy. Taken together, the simulated hippotherapy could provide fun, interactive and engaging therapy session for the participants to work on trunk control and balance reactions.
Simulated kayak training was provided to further train the trunk control and balance reactions due to the continuous paddling actions and trunk rotation. The participant wore the safety harness secured to the ceiling hoist and sat on the inverted Bosu ball placed on the ground (Appendix Figure 12). The participant held a long pole and simulated paddling strokes similar to kayaking actions. As part of therapy progression, the participant was positioned to sit on the Bosu ball placed on top of the therapy stool (Appendix Figure 13). The physiotherapist would provide random perturbation of the Bosu ball to simulate movements on the kayak on waters; and pushed it anteriorly simulating the modified kayak moving forward, while the participant paddled; hence, providing visual-vestibular stimulation for the participant while paddling.
Sit-to-stand (STS) transition is a precursor to walking, hence critical for performing daily life activities and is a prerequisite for functional independence. 26 In our study, the participants practised STS training with a rollator frame so that they could relearn movement transition of their centre of mass and control of STS (Appendix Figure 17). A mirror provided feedback to promote postural awareness of upright posture. In addition, the participants practised STS using the EksoGT training feature to improve eccentric and concentric muscle contraction of the trunk and lower limbs. Squatting exercises with the EksoGT was practised at 3 angles, namely, 45 degrees, 65 degrees and 90 degrees knee flexion (Appendix Figure 16).
Participants received the EksoGT robotic therapy 2 to 3 times a week on various terrain types, to ensure sufficient repetitions of reciprocal gait stepping pattern were achieved (Appendix Figure 25–26). They practised walking using the ProStep mode in the EksoGT software. In this mode, the participants need to attempt active weight shifting to actuate the contralateral motors to swing the limb forward, hence facilitating motor control and motor learning. 16 Average number of steps practised with the EksoGT ranged from 1000 to 1800 steps in 45 minutes to 1 hour session. The high intensity and step repetitions achieved with robotic exoskeleton have been shown to promote activity-dependent neuroplasticity.27,28 The robotic exoskeleton sessions were interspersed with conventional physiotherapy sessions to minimise fatigue and reduce monotony of sessions.
Overground gait training, with bodyweight support, was conducted using the ceiling hoist (Maxi Sky 2 ceiling hoist, Arjo, Sweden) and rollator frame (Appendix Figure 20–21). The safety harness of the ceiling hoist enabled the participants to practise dynamic weight shifting and volitional control of swinging of lower limbs in reciprocal gait pattern. The spinal cord stimulation parameters (50 Hz, 100-400 microseconds, 5-20 mA) were customised to aid the participants’ ability to swing the lower limbs. Some degree of ankle dorsiflexion was activated with the spinal cord stimulator which aid in the foot clearance during swing phase of gait cycle. Participants were cued to monitor their upright posture, initially with the use of mirror, during overground gait training to improve postural awareness.
Motor imagery technique, by using an eye shade, was also included during overground gait training when the participants were able to demonstrate consistent lower limb movements during the gait cycle (Appendix Figure 22–24). They were instructed to imagine and visualise the dynamic weight shift, weightbearing on the lower limb; followed by flexion of the contralateral hip and swinging leg forward.
Attempts to ambulate without the ceiling hoist were explored based on the participants’ ability. The aim was to wean off the ceiling hoist eventually by the end of the research trial so that the participants can ambulate within home environment in the future. Between 1 and 4 months of rehabilitation, bilateral knee gaiters were applied as assistive devices to support the knees as the quadriceps and hamstrings control were still relatively weak to support the full body weight of the participants during stance phase of gait. The graded approach to weaning off the ceiling hoist included gait training with a lax ceiling hoist, to ensure safety, by lowering the hoist till the safety harness straps were laxed. Once the physiotherapist determined that the participant could maintain an erect posture in standing and walking with the bilateral knee gaiters consistently, without veering, for a period of 1 week, the ceiling hoist support was removed for the subsequent gait training. To ensure safety during gait training without the ceiling hoist, the physiotherapist would hold onto a gait belt and the wheelchair/chair was pulled along behind the participant as he/she walked. Overground gait training without safety harness support occurred both on even ground in therapy gym; and on uneven terrain in the outdoor environment in the final 2 months of rehabilitation (Appendix Figure 27–29).
Outcome Measures
The outcome measures used for evaluation at the end of prehabilitation phase and subsequent monthly assessment (Table 1), with the spinal cord stimulator switched on:
Clinical outcomes of Participant 1 (T4 SCI).

Daily 7 months rehabilitation commenced after resting 1 month post spinal cord stimulator implantation for wound healing.
(i) American Spinal Injury Association (ASIA) Impairment Scale (AIS) motor and sensory examination is a standardised examination of the physical impairment of people with SCI. 29
(ii) Spinal Cord Independence Measure version III (Mobility subscale) assesses the mobility components such as mobility in bed, transfers, mobility indoors and mobility outdoors. 30
(iii) Walking Index for Spinal Cord Injury II (WISCI-II) is a functional capacity scale designed to measure improvements in walking ability in SCI. 31 The score ranged from 0 to 20; with Level 0 as ‘patient is unable to stand and/or participate in walking’ to Level 20 as ‘ambulates with no devices, with brace and no assistance’.
(iv) Trunk Assessment Scale for Spinal Cord Injury (TASS) consists of 9 seated tasks to evaluate trunk function of people with SCI. 32 The maximum score is 44 points; with higher scores indicating better trunk function.
(v) Five Times Sit-to-Stand (with rollator frame) (5×STS) assesses functional lower limb muscle strength and functional change of transitional movements. 33 The average time required from the command ‘go’ to the participant’s back touching the backrest of the chair on the fifth repetition of sit-to-stand over 2 trials was captured.
(vi) 2-minute Walk Test (2mWT) assesses self-paced walking ability and functional capacity. 34 The average distance walked over 2-minute period would be captured.
The pre-post assessments were conducted by the study team doctors and physiotherapists; hence, non-blinded.
Results
Summary of the Participants
Two male and one female participants with chronic thoracic AIS A SCI were recruited into this study. Participant 1 (P1) was a 47-year-old male ex-jockey with Thoracic 4 (T4) AIS A SCI after a horse-riding accident in 2017. He suffered a Thoracic 6 (T6) burst fracture with multiple thoracic vertebral fractures spanning from Thoracic 3 (T3) to Thoracic 7 (T7). He underwent Thoracic 1 to 9 (T1-T9) spine fixation and decompressive laminectomy of Thoracic 3 to 7 (T3-T7).
Participant 2 (P2) was a 26-year-old male with Thoracic 2 (T2) AIS A SCI after a motorcycle accident in 2018. He suffered a complex Thoracic 3-4 (T3-4) fracture subluxation and underwent a Thoracic 3-4 (T3-T4) laminectomy and a Cervical 7 (C7) to Thoracic 2 (T2) spinal instrumentation.
Participant 3 (P3) was a 38-year-old female with Thoracic 10 (T10) AIS A SCI after falling down an escalator shaft at a subway station in 2013. She suffered multiple rib fractures, bilateral pelvic fractures and complex triple column fracture involving the Thoracic 12 (T12) vertebral body with near complete obliteration of the spinal canal and cord compression. She underwent a Thoracic 11-12 (T11-T12) decompressive laminectomy and a Thoracic 9 (T9) to Lumbar 3 (L3) spinal instrumentation.
Functional Clinical Outcomes
The clinical outcomes of the participants are presented in Tables 1 to 3. The granularity of the results revealed the recovery trajectory per individual as they had different levels of complete thoracic SCI, namely T4, T2 and T10.
Clinical outcomes of participant 2 (T2 SCI).

Daily 7 months rehabilitation commenced after resting 1 month post spinal cord stimulator implantation for wound healing.
Clinical outcomes of participant 3 (T10 SCI).

Daily 7 months rehabilitation commenced after resting 1 month post spinal cord stimulator implantation for wound healing.
All 3 participants demonstrated improvements in their WISC-II, TASS, 5×STS and 2mWT; and no change in AIS sensory scores post 7 months rehabilitation. The WISCI-II of the participants improved, ranging from 3 levels to 9 levels. The TASS improved by 11 points for P1; 2 points for P2; and 13 points for P3. Hence, implying improvement in overall trunk control.
The 5×STS and 2mWT results for the 3 participants were remarkable – from inability to perform sit-to-stand and walking at prehabilitation phase to a level whereby they were able to perform sit-to-stand independently; and ambulate on level and unlevel terrains with a rollator frame with supervision of a carer at the end of 7 months of daily rehabilitation.
Functional Motor Recovery
All participants were able to achieve volitional control of bilateral hip flexion, knee flexion-extension and ankle dorsiflexion-plantarflexion, with the spinal stimulator switched on.
P1, P2 and P3 were able to achieve sit-to-stand independently with the rollator frame on the 30th therapy session (week 6), 15th therapy session (week 3) and 13th therapy session (week 3) respectively.
With the support of the ceiling hoist and rollator frame, the participants were able to initiate swing and stance phases of the gait cycle; with P1 achieving this on the 17th therapy session (week 4), P2 achieving this by the 6th therapy session (week 2) and P3 achieving this by the 5th therapy session (week 2).
At week 3 (14th session), P3 was able to stand up independently with knee gaiters by pushing through the rollator frame and assume an erect posture in static standing. Hence, the physiotherapists progressed the gait training without ceiling hoist by end of week 3 for her. She was able to walk an average of 5 metres with the rollator frame without ceiling hoist under supervision. Such level of functional mobility was only achieved at the end of week 18 for P1 and P2.
With regular rest breaks, P1 and P2 was able to ambulate between 30 metres to 38 metres cumulatively by week 28. As for P3, she achieved a significant milestone by walking non-stop for 252 metres on Day 109 (week 22) of the therapy session. In addition to the gait training in the gym, the physiotherapists brought them out of the therapy gym to practise walking in the real world environment, such as on tar road, uneven terrains and gentle slope, from weeks 20 to 28 (Appendix Figure 28–29). This was aimed at transferrable of skills learnt in the clinical therapy gym to functional walking for daily living. At the end of week 28, all the participants achieved ambulation with the rollator frame under supervision from their carers.
Discussion
Results from this clinical trial demonstrated significant functional neurological motor improvements in 3 participants with chronic AIS A SCI after our RESTORES trial protocol. All the participants exhibited improvement in the WISCI II, TASS, 5×STS and the 2mWT. The improvement in WISCI of 3 to 9 levels is considered a clinically meaningful change as Burns et al 31 showed that a change of 1 WISCI level can be considered a real difference in a clinical context. Furthermore, the TASS of P1 and P3 exceeded the minimal clinically important difference (MCID) of 4 points. 32 All participants improved on the 5×STS and exceeded the MCID 2.27 seconds. 33 These improvements translated into the participants’ ability to perform sit-to-stand independently and ambulate with a rollator frame with supervision of a carer post 7 months of rehabilitation. This remarkable outcome was attributed to the synergistic integration of SCS, motor imagery, robotic therapy and customised physiotherapy, which focused on trunk rehabilitation and overground gait training. One of the key factors to succeed in overground gait training was the customised and precise SCS programming to target key muscles essential for locomotion during the swing and stance phases of the gait cycle.
Epidural Spinal Cord Stimulation
In this clinical trial, epidural spinal cord stimulation has demonstrated to be a promising neurotechnology to activate and control lower limb muscles to stand, step and walk through neuromodulation of lumbosacral circuitry.9,35 Post intensive, task-specific rehabilitation, the 3 participants with chronic complete SCI achieved positive functional outcomes of independent sit-to-stand, standing and supervised ambulatory status. They could execute volitional control of the movement of each leg during gait training, despite absent sensation and proprioceptive feedback from their trunk below the level of injury and the lower limbs. Improvements in their volitional lower limb movements were in congruent with evidence that ESS may boost muscle recruitment via the activation of Ia muscle spindle afferents 36 ; strengthen connections between neurons by enhancing the efficacy of existing synapses and descending pathways 37 ; and drive local lumbar circuit plasticity via the afferent inputs. 38 Taken together, the improvements in the participants suggest the huge potential of ESS to facilitate functional recovery in people with SCI.
Motor Imagery
Motor imagery can strengthen motor commands by potentiating recruitment and synchronisation of motoneurons, which leads to improved recovery. 39 Therefore, in our trial, we chose to use active functional movements to perform kinestheic motor imagery. 40 The participants were able to couple motor imagery with observation of lower limbs in order to attempt movement practice in lying, standing and during gait training. They reported that visualising weight shifting, movement of stepping and swinging of lower limb during gait training facilitated the volitional control of each leg in stance and swing phase of gait. This was further enhanced when they were trained to walk blindfolded with eye shade and they could focus on motor imagery without distraction from the busy therapy gym environment. Hence, consistent stepping movements were observed during overground training.
Trunk Rehabilitation
For ambulation to occur in paraplegic patients, they need to rely heavily on upper limb and trunk muscles to assist with propulsion forwards to compensate for the lower limb weakness. 41 Hence, our prehabilitation protocol included trunk control training and upper limb strengthening in preparation for overground gait training. In our study, when the participants pushed down on the rollator frame during dynamic weight shifting, stance and swing phase of gait cycle, their latissimus dorsi, trapezius, pectoralis major and serratus anterior muscles might be activated to stabilise the upper and lower trunk and pelvis. As there were no EMG captured in this clinical trial currently, the hypothesis of probable muscle activation was inferred from other studies.42,43 Results from the EMG studies42,43 indicated that people with high complete thoracic SCI used their latissimus dorsi, trapezius, pectoralis major, serratus anterior muscles, erector spinae, rectus abdominis, external oblique, internal oblique and transversus abdominis, both in voluntary efforts and in reaction to random balance perturbations.
In people with thoracic SCI who are clinically classified as complete injury, there may be residual neural connections that might have been reinforced during trunk rehabilitation. Another possible explanation for the activation of lower trunk muscles is induced indirectly by contractions of upper trunk muscles during perturbation. 43 Such contractions might lead to mechanical stretching and a subsequent reflex muscle activation of the lower trunk muscle groups.42,43 Hence, the EMG results42,43 support the importance of trunk rehabilitation in our trial to improve trunk stability and postural control. Studies on hippotherapy simulator44,45 and kayaking46-48 have demonstrated positive improvements in postural control and balance of neurological patients. Therefore, our team provided these therapies to improve trunk control and balance reactions in the participants. Hippotherapy simulator can lead to various activity changes in the trunk muscles and improve balance ability. 44 Maintaining the sitting posture and balancing the trunk requires continuous compensation for perturbations to the upper body by motion of the simulated kayak (on an unstable Bosu ball), as well as of the dynamic arm movements and paddle in the air. 48
Post trunk rehabilitation, the improvement in trunk control in all the participants aided in their trunk stability and hence, independent sit-to-stand (their 5×STS exceeds the MCID of 2.27 seconds 33 ) and locomotor ability on different terrain. In addition, P1 and P3 reported increased lateral flexion of the trunk which aid in better pressure relief of ischial tuberosity, toileting and perineal hygiene; hence improving their quality of life.
Robotic Exoskeleton Therapy
Robotic exoskeleton aids in minimising users’ abnormal posture and movement; enabling them to experience walking in a normal physiological gait pattern as part of motor relearning. 16 In contrast to tethered treadmill-based robotic gait training systems, robotic exoskeletons enable gait training in non-gym settings such as common spaces and outdoor terrain. Furthermore, visual spatial and vestibular components are entrained as the user navigates on different environmental surfaces. 49
Dynamic weight shifting ability of the participants improved after EksoGT therapy as they experienced over 1000 steps of practice in 45 to 60 minutes of therapy. Repeated activation of sensory feedback circuits during standing and walking promotes activity-dependent reorganisation of neural connections that ameliorates locomotor performance. 50 Despite the sensorimotor complete thoracic SCI, the participants were likely able to utilise residual upper trunk proprioceptors to relearn the degree of dynamic weight shift required during walking with the EksoGT. Dense muscle spindles in paraspinal rotators are crucial for monitoring the range of motion of the trunk. 51 Hence, intensive training on the exoskeleton aided in their overall trunk proprioceptive feedback, motor control and facilitate motor learning.27,28 During overground gait training, the participants reported that they learnt to gauge the boundaries of appropriate weight shift, in part, due to the intensive training with EksoGT; and in part, using their ‘body to sense’, possibly using trunk proprioceptors, visual and vestibular systems, as they could not sense the ground. Additionally, they could gauge how to transit their centre of mass anteriorly from initial contact (heel strike) position at the beginning of stance to terminal stance; thus aiding them to walk forward.
While training with the EksoGT, the participants’ trunk muscles were possibly activated, based on their subjective reports of trunk muscle fatigue post training. This is congruent with the findings by Alamro et al 17 that overground training with EksoGT elicited greater trunk muscle activity compared to Lokomat robotic treadmill training system in people with high-thoracic motor complete SCI. Challenging participants in our study to work out their trunk muscles intensively during EksoGT training had translated to improvement in functional mobility. Intensive training on EksoGT also worked on improving their cardiovascular fitness as studies have shown that patients exercised at a moderate-intensity with exoskeleton gait training.52,53 The participants in our study reported that they did not experience fatigue easily after the initial 4 weeks of EksoGT training. They felt that they could tolerate longer therapy sessions, from the initial 1 hour to 1.5 hours. This was also observed in their ability to progress to walking, without ceiling hoist support, for longer distance from 5 metres to 50 metres before they required a rest break. P3, who was 11 years post SCI, even achieved a significant milestone and personal best performance by walking non-stop for 252 metres without rest breaks, due to improved cardiovascular fitness. Overall, that improved their self-efficacy as they were motivated to participate more in therapy sessions.
Limitations
In our pilot trial, we explored the synergistic combination of multi-modality treatment involving epidural spinal cord stimulation, motor imagery, robotic exoskeleton, trunk rehabilitation and overground gait training to restore functional mobility in 3 participants with chronic sensorimotor complete thoracic level SCI. With such combinatory therapies, it was difficult to evaluate which treatment modality was effective, and which was not, to optimise the functional outcomes in the participants. However, we utilised the principle of applying the best available scientific evidence per treatment modality with good clinical reasoning to develop the rehabilitation protocol for post spinal cord stimulator implantation, in a stepwise staged fashion. The detailed rehabilitation protocol (refer to Appendix) is aimed at providing a reference guide for clinicians who support such intensive rehabilitation post spinal cord stimulator implantation.
As this clinical trial reports the outcomes post 7 months of rehabilitation with SCS, there is a need to follow-up with the participants to evaluate if the gains made will be maintained over the next 1 year and thereafter. The long-term follow-up will provide insights into the real benefits of the SCS with rehabilitation. If the functional mobility gains of standing and walking are well maintained beyond a year, it will facilitate a further push for such innovative combinatory therapies to be offered to people with complete spinal cord injury as a clinical service because of the real-world translation of functional gains from hospital to home. Another vital aspect that should be investigated include interviewing the participants to gain deeper insights into the impact of the improved functional mobility on their quality of life as well as for their carers. Additionally, future studies should consider capturing EMG and kinematic data of the trunk in standing and during walking to shed more insight into trunk control in people with SCI as trunk control affects different facets of a person’s physical functioning in daily life.
Our team recognised the huge amount of resources, in terms of manpower, equipment and therapy costs, required over the 7-month period. It may present a challenge to replicate such clinical rehabilitation programme in some rehabilitation facilities. One possible solution may include refining the dosage of the combined therapies to suit the operational needs of the facility; for example, providing daily rehabilitation therapy daily for the initial 3 months and then reduce the frequency of therapy to 2-3 times per week for the remaining 4 months. The clinical outcomes can then be tracked, analysed and the rehabilitation protocol can be further adjusted to yield optimal patient outcomes. The 3 participants in our study feedbacked that having daily therapy for the initial 3 months was critical for them to relearn complex skills of sit-to-stand, postural control in standing, balancing, weight shifting and walking once again, given their dependence on a wheelchair for the past years. The physiotherapists concurred with their feedback; recognising the need to scaffold the motor learning of the complex skills via part task practice and subsequently, whole task practice. In other words, rehabilitation facilities should not consider trimming the frequency of therapy in the initial 3 months. Our participants added that they had even forgotten how to stand, let alone attempting to swing their legs to walk again. Hence, the motor imagery training within our rehabilitation protocol aided them to relearn movements again. The daily therapy also helped them improve their cardiovascular fitness and rebuild their confidence on getting back onto their feet again, considering that they had no sensory feedback due to their complete SCI.
Conclusion
We have detailed our rehabilitation protocol of RESTORES trial in a stepwise staged fashion. The results from this trial appear to support our rehabilitation protocol for post ESS implantation. The rehabilitation focus on motor imagery, trunk rehabilitation, gait training and neuroplasticity via robotic therapy, coupled with ESS, has helped our participants with AIS A SCI to achieve remarkable improvements in their functional mobility. They had achieved independent sit-to-stand ability and functional walking with rollator frame over a period of 7 months; considering that P1, P2 and P3 were wheelchair dependent for 6 years, 5 years and 11 years respectively. More time is needed to fully evaluate the true extent of restoration of motor power and functional improvements in these participants. The next challenge would be making such intensive rehabilitation therapy with SCS sustainable and affordable to more people with SCI. This trial suggests the potential of synergistic integration of SCS with advanced robotic therapy and customised neurorehabilitation to facilitate functional neurological recovery and enhance the quality of life in people with sensorimotor complete SCI.
Footnotes
Appendix
REHABILITATION POST EPIDURAL SPINAL CORD STIMULATOR IMPLANTATION

| Aims of physiotherapy | |||
|---|---|---|---|
| To conduct detailed mapping and programming of epidural spinal cord stimulator for volitional control of key lower limb muscle groups - stimulator parameters include: (i) Frequency (ii) Intensity (iii) Pulse width |
1) Hip flexors-extensors |
Figure 1: Practising hip flexion-extension. |
Key individual muscle groups that were targeted during programming: i) rectus femoris ii) vastus medialis iii) vastus lateralis iv) hamstrings v) tibialis anterior vi) gastrocnemius vii) soleus These key muscles are essential for sit-to-stand, standing, and walking. |
| 2) Knee flexors-extensors | Figure 2: Practising knee flexion-extension. |
||
| 3) Ankle dorsiflexors-plantarflexors |
Figure 3: Practising ankle dorsiflexion-plantarflexion. |
||
To enhance motor learning via motor imagery |
Visualise the intended movement and rehearsed the movement required: i) Hip flexion-extension ii) Knee flexion-extension iii) Ankle dorsiflexion-plantarflexion |
Figure 4: Motor imagery of hip-knee flexion in preparation of overground locomotor training. Figure 5: Motor imagery of ankle dorsiflexion-plantarflexion. |
After motor imagery exercises, the participant would attempt moving the limbs. These key movements shown in Figure 4 and 5 are essential for overground gait training. |
| To improve trunk control | 1) Trunk control exercises in lying • small ball placed underneath the sacrum • practise lower trunk and pelvic rotation by rolling knee side to side |
Figure 6: Practise lower trunk and pelvic rotation |
Practise slow and fast movements - includes isometric contraction to hold position before relaxing after few seconds. |
| 4) Trunk control |
|||
| Resistive theraband tubing acting as the rein and stirrups. |
|||
| Physiotherapist would provide pushing force to move the therapy thumbler anteriorly (gliding on ceiling hoist) + rolling the thumbler side-to-side: provides visual-vestibular stimulation for the participant; hence, simulating a horse moving forward in modified hippotherapy | |||
| Physiotherapist would provide random perturbation of the Bosu ball to simulate movements and pushing it anteriorly while the participant was kayaking: provides visual-vestibular stimulation for the participant; hence, simulating the modified kayak moving forward. | |||
| Aid in weightbearing on lower limbs as well. |
|||
| Learning how to utilise the trunk proprioceptors above the level of injury to gauge the amount of weight shift in standing |
|||
| To improve eccentric and concentric muscle contraction of lower limb; and improve sit-to-stand ability using the Ekso training feature | Figure 16: Practise sit-to-stand with the EksoGT | ||
| Figure 17: Practise sit-to-stand with the rollator frame | |||
| Practise hip-knee flexion to place foot on wooden block in preparation to step up |
Figure 18: Practise single leg stepping up-down wooden block. |
||
| Practise swing-to and swing-through gait pattern. |
|||
| Nordic walking with support of ceiling hoist | Figure 21: Nordic walking | ||
| Observe that the lower limb movements are similar with and without eye shade. Hence, supporting the hypothesis that motor imagery prior to volitional attempt of swing through of lower limb will aid in locomotor training |
|||
| Intensive overground gait training with Ekso robotic exoskeleton |
Figure 25: Practise walking with Ekso robotic exoskeleton with mirror feedback. | Mirror feedback was gradually weaned off once the participant exhibited consistent steps. | |
| Figure 27: Practise walking with bilateral knee gaiters and rollator frame on even terrain in the therapy gym. | The aim of this approach was to wean off the ceiling hoist eventually by the end of the research trial so that the participants can ambulate within home environment with supervision by a carer in the future. | ||
| To train ambulation in real world environment with rough and uneven terrain |
Figure 28: Practise walking with bilateral knee gaiters and rollator frame on rough and uneven terrain. |
Practise walking on |
|
Acknowledgements
The authors would like to thank our participants for their valuable contribution to this study. We would also like to gratefully acknowledge Medtronic for their generous support in this trial.
Ethical Considerations
This research has been approved ethics approval from the SingHealth centralised institutional review board (CIRB 2022/2496)
Consent to Participate
Informed consent obtained.
Consent for Publication
The participants are agreeable for their photos to be used for publication.
Author Contributions
KRW provided all oversight for this study. KRW, NWH, JKRX, SKW, NZYV, WLL, MWP and FM designed the study. KRW, NWH, JKRX performed the critical surgical procedures of the study. SKW, MWP, KRW, NZYV and WLL provided prehabilitation and post-operative rehabilitation. All authors provided critical feedback and approved the final manuscript.
SKW: Writing – original draft. KRW, NZYV, WLL, MWP, NWH, JKRX, FM: Writing – review & editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by the National Medical Research Council of Singapore (NIG grant MOH-001083-00) and the Ng Teng Fong Healthcare Innovation Programme (NTF-HIP grant NTF_FY2022_2_C2_CR_05). The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The authors had full access to all the data in the study and had overall responsibility for the decision to submit for publication. No funding was received from the drug manufacturer or distributor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
