Abstract
Study Design
Prospective pre–post interventional study.
Objectives
To evaluate the effects of epidural spinal cord stimulation (ESCS) combined with task-specific rehabilitation on mobility, sitting balance, bladder control, lower limb strength, and postural stability in individuals with spinal cord injury.
Methods
Fifteen participants with chronic thoracic SCI received daily ESCS sessions combined with physiotherapy and Tyromotion-based robotic balance training for 8 weeks. Outcome measures included: Timed Up and Go (TUG) test for mobility, Walking Index for Spinal Cord Injury II for ambulatory function, Neurogenic Bladder Symptom Score for bladder control, Manual Muscle Testing for knee extension strength, and anterior–posterior displacement for postural stability and Modified Ashworth Scale (MAS) for spasticity from
Results
All participants successfully completed the study. The Timed Up and Go (TUG) test time significantly improved from 430.25 ± 265.2 seconds to 141.9 ± 92.6 seconds (P < 0.001). The Walking Index for Spinal Cord Injury II (WISCI II) score increased markedly from 0.07 ± 0.26 to 6.07 ± 1.03 (P < 0.001). Antero-posterior (AP) displacement during standing balance decreased from 18.2 ± 3.5 mm to 11.4 ± 2.9 mm (P < 0.001), indicating enhanced static postural control. The Neurogenic Bladder Symptom Score (NBSS) improved significantly, reducing from 24.05 ± 6.91 to 7.4 ± 5.08 (P < 0.001). Manual Muscle Testing (MMT) of knee extension increased from 0.25 ± 0.44 to 3.85 ± 0.37 (P < 0.001). Finally, the Modified Ashworth Scale (MAS) scores were significantly reduced from
Conclusions
ESCS with Tyromotion significantly improves motor strength, trunk balance, gait function, and bladder control in thoracic SCI. These findings support task-specific therapies in SCI recovery programs.
Keywords
Epidural spinal cord stimulation (SCS) has been shown to enhance motor function in spinal cord injury (SCI) patients, with some studies reporting improvements in standing, stepping, and autonomic functions such as bladder control. Epidural spinal cord stimulation (ESCS) has been shown to facilitate voluntary movement, standing, and some autonomic improvements in patients with spinal cord injury (SCI), particularly in those with incomplete injuries. This study Introduces a structured post-stimulation training model that could inform future rehabilitation protocols targeting neuroplasticity across systems in SCI. This is among the first clinical trials to explore multidomain neuroplastic changes (motor, autonomic, and postural) in SCI patients undergoing Tyromotion-based rehabilitation following epidural stimulation.What’s Known
What’s New
Introduction
Spinal cord injury (SCI) is a devastating neurological condition that leads to profound impairments in sensory, motor, and autonomic functions below the level of injury, often resulting in reduced independence and diminished quality of life. Mobility limitations restrict daily activities, while bladder dysfunction and spasticity negatively affect personal hygiene, social participation, and psychological well-being.1,3 Globally, more than 294,000 individuals are living with SCI, with India reporting particularly high rates due to road traffic accidents—estimated at nearly 55 accidents and 17 deaths every hour. 2
Conventional rehabilitation strategies primarily focus on optimizing residual function and minimizing secondary complications. However, in individuals with complete or severe incomplete injuries, these approaches often achieve only modest recovery. Advances in neuromodulation, particularly epidural spinal cord stimulation (ESCS), have demonstrated potential in enhancing motor control, postural balance, and autonomic functions, including bladder regulation. This study investigates the effectiveness of ESCS in improving mobility, postural stability, and bladder function among individuals with SCI.SCI has not only paralysis as its consequences, but also a variety of complications including bladder dysfunction, spasticity, pain and decreased quality of life. Electric spinal cord stimulation (ESCS) refers to the delivery of electrical currents to targeted segments of the spinal cord through implanted electrode leads placed in the epidural space. Targeting such neuronal networks may be used as a route to stimulate spinal neural networks to induce specific cationic flux through membrane, leading to spinal circuitry excitability below the level of injury. Previous studies demonstrate that ESCS can induce voluntary movement in individuals with chronic motor complete SCI.3,4
Epidural spinal cord stimulation (ESCS) has emerged as a promising intervention for restoring motor function after SCI. Previous studies have demonstrated that ESCS can activate central pattern generators (CPGs), facilitate residual supraspinal pathways, and enable recovery of stepping and voluntary movement in individuals with incomplete and even clinically complete injuries (Harkema et al, 2011; Angeli et al, 2014; Gill et al, 2018). Beyond locomotion, ESCS has also shown potential in modulating autonomic and reflex pathways, with reported benefits in bladder control and spasticity management (Herrity et al, 2021; Hofstoetter et al, 2015).
In parallel, task-specific and technology-assisted rehabilitation can provide repetitive, feedback-driven training that enhances neuroplasticity. Virtual reality–based systems such as Tyromotion offer multisensory feedback, motor learning opportunities, and task-specific practice, which may augment the functional gains induced by neuromodulation 3
Despite these advances, there remains limited evidence regarding the combined effect of ESCS and Tyromotion-assisted rehabilitation on functional outcomes in SCI. Understanding how this integrated approach influences motor function, balance, autonomic control, and spasticity may provide important insights for optimizing rehabilitation strategies. 4
Although the effects of ESCS on lower limb motor function have been described in several studies, fewer studies have evaluated the functional mobility, sitting balance, and autonomic functions (e.g., bladder control) achieved with ESCS. In terms of the most relevant SCI complications, bladder dysfunction is one of the most consistently identified as a significant factor affecting quality of life. 5
Rationale
Central Pattern Generators (CPGs) are neural networks in the spinal cord capable of independently producing rhythmic motor activity without sensory input. In SCI patients, CPGs typically remain intact but suffer from insufficient excitatory stimulation. Epidural spinal cord stimulation (ESCS) can activate these supraspinal pathways, potentially improving locomotor function and movement capability.6,12 Bladder function control depends on reflex pathways within the spinal cord. Epidural spinal cord stimulation operates on a distinct mechanism by modulating the spinal circuits to restore voluntary control over the movement and the function of the bladder.7,11 Numerous recent studies have revealed that patients undergoing ESC show a greater ability to start voluntary movements and an improvement in the emptying of the bladder, with marked improvements noted in the mobility assessments.8,9 The research has constantly shown that ESC can lead to observable improvements in the international standards for the neurological classification of the spinal cord lesion (ISNCSCI), highlighting its potential as a transformative intervention in rehabilitation contexts.10,13
Effect of the 8-week ESCS intervention on walking ability, sitting balance, and bladder function among individuals living with thoracic SCI. We expected that ESCS would yield meaningful improvements on these functional domains, as measured by the Timed Up and Go (TUG) test, Walking Index for Spinal Cord Injury II (WISCII), and Neurogenic Bladder Symptom Score (NBSS). Therefore, the present study aimed to evaluate the effects of ESCS combined with Tyromotion-based rehabilitation on walking ability, balance, bladder function, and spasticity in individuals with incomplete SCI.
Objective of the Study
To evaluate the effectiveness of an 8-week intervention combining epidural spinal cord stimulation (ESCS) and Tyromotion-assisted neurorehabilitation in individuals with chronic spinal cord injury (SCI), by comparing pre- and post-intervention changes in. • Functional mobility (Timed Up and Go Test - TUG) • Walking Index for Spinal Cord Injury II - WISCII) • Autonomic function (Neurogenic Bladder Symptom Score - NBSS) • Muscle strength (Manual Muscle Testing - MMT) • Spasticity (Modified Ashworth Scale - MAS) • Postural balance (Anteroposterior and Mediolateral Displacement Measures)
Methods
This was a single-group, pre–post intervention study conducted to assess the effects of an 8-week epidural spinal cord stimulation (ESCS) protocol in individuals with thoracic spinal cord injury (SCI). Baseline assessments were performed prior to the initiation of ESCS, and follow-up measurements were collected immediately after the completion of the intervention period. No control group was included, and each participant served as their own control. The study focused on evaluating within-subject changes in functional mobility, sitting balance, bladder control, and lower limb strength.
S tudy Registration
This study was registered in the ClinicalTrials.gov database.
Participants: Fifteen adult chronic SCI (demographic information not disclosed from original data) participated in this study.
Inclusion Criteria
1. Traumatic SCI with lesions between T6 and T10* 2. Chronic injury (>1 year post-injury) 3. Neurologically stable 4. No contraindications for ESCS
Exclusion Criteria
1. History of seizures 2. Severe osteoporosis 3. Implanted electronic devices 4. Active pressure sores 5. History of uncontrolled infections or cardiovascular instability 6. Severe musculoskeletal deformities affecting locomotion 7. Cognitive impairments that limit adherence to the study protocol
Randomization & Blinding: This clinical study followed a prospective pre-post interventional design to evaluate the effectiveness of Epidural Spinal Cord Stimulation (ESCS) in combination with Tyromotion-based virtual rehabilitation on motor, autonomic, and postural functions in individuals with chronic spinal cord injury (SCI). The study was conducted over a period of 8 weeks and included 15 participants diagnosed with motor incomplete SCI (ASIA C or D), who met the inclusion criteria and provided written informed consent. Given the nature of the intervention, blinding of participants and therapists was not feasible, as both the surgical implantation of the stimulation device and the active engagement with the Tyromotion system required conscious participation and feedback. However, outcome assessors were blinded to the pre- or post-intervention status of the data during analysis to reduce potential bias in evaluation.
Intervention Protocol
Device: Boston Scientific CoverEdge™ X 32-electrode epidural lead.
Stimulation Parameters: - Frequency: 50 Hz - Pulse Width: 300 µs - Amplitude: Tailored to patient tolerance - Duration: 8 weeks, 5 sessions/week.
Co-Intervention: All participants received standardized physiotherapy targeting balance, standing, and stepping strategies.
Mapping in 32-Contact Boston Scientific Epidural Stimulation for Standing in SCI Patients.
Mapping refers to the process of identifying the optimal electrode configurations and stimulation parameters that produce functional motor responses, such as standing, in spinal cord injury (SCI) patients. When using a 32-contact electrode array, like the Boston Scientific CoverEdge™ X or Penta™ 32 surgical lead, the process is systematic and tailored to each individual.
*We limited our inclusion to individuals with spinal cord injury between T1 and T10. Patients with upper cervical lesions were excluded because significant upper limb weakness in this group compromises trunk stability, which was essential given that our primary objective was to establish a walking pattern. Likewise, injuries below T10 were not considered, as these often involve the conus medullaris or cauda equina, where stimulation does not consistently generate effective motor responses due to predominant peripheral nerve involvement. In contrast, injuries between T6 and T10 offered the most appropriate balance—these patients retained adequate trunk stability, produced reliable stimulation responses, and thus were best suited for gait training and evaluation of locomotor recovery.
Intervention Protocol
Each participant underwent surgical implantation of an epidural stimulator (Boston Scientific CoverEdge X 32 Surgical Lead Kit with 32-electrode array, USA). The CoverEdge X Surgical Lead Kit includes one surgical lead and six suture sleeves (two 1 cm, two 2.3 cm tapered, and two 4 cm). The electrode array was positioned over the thoracolumbar spine (T10–L1), which contains neural circuits involved in lower limb motor function and autonomic bladder regulation.
Figure 1. Boston Scientific Epidural Spinal Cord Stimulation User Interface - Left Side Program configuration. Screenshot of the Boston Scientific Stimulation System User Interface Displaying the Left-Side Electrode Array Programming Parameters. The Interface Shows Stimulation Settings including Amplitude, Pulse Width, Frequency, and Electrode configuration Used During the Intervention Protocol Boston Scientific Epidural Spinal Cord Stimulation User Interface - Left Side Program Configuration. Screenshot of the Boston Scientific Stimulation System User Interface Displaying the Left-Side Electrode Array Programming Parameters. The Interface Shows Stimulation Settings Including Amplitude, Pulse Width, Frequency, and Electrode Configuration Used During the Intervention Protocol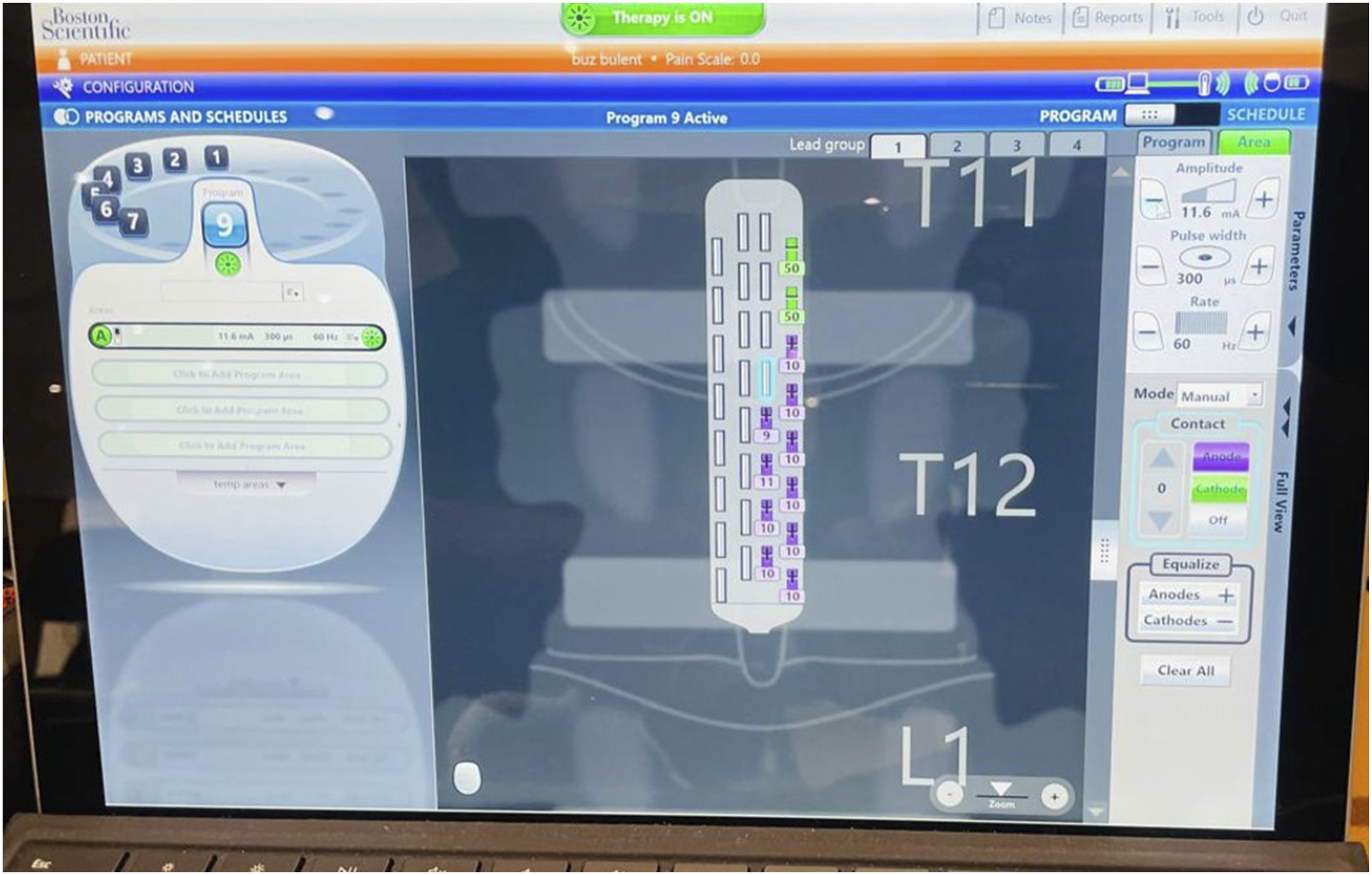
Figure 2. Boston Scientific Epidural Spinal Cord Stimulation User Interface - Right Side Program configuration. Screenshot of the Boston Scientific Stimulation System User Interface Displaying the Right-Side Electrode Array Programming Parameters. The Interface Shows Stimulation Settings including Amplitude, Pulse Width, Frequency, and Electrode configuration Used During the Intervention Protocol Boston Scientific Epidural Spinal Cord Stimulation User Interface - Right Side Program configuration. Screenshot of the Boston Scientific Stimulation System User Interface Displaying the Right-Side Electrode Array Programming Parameters. The Interface Shows Stimulation Settings Including Amplitude, Pulse Width, Frequency, and Electrode Configuration Used During the Intervention Protocol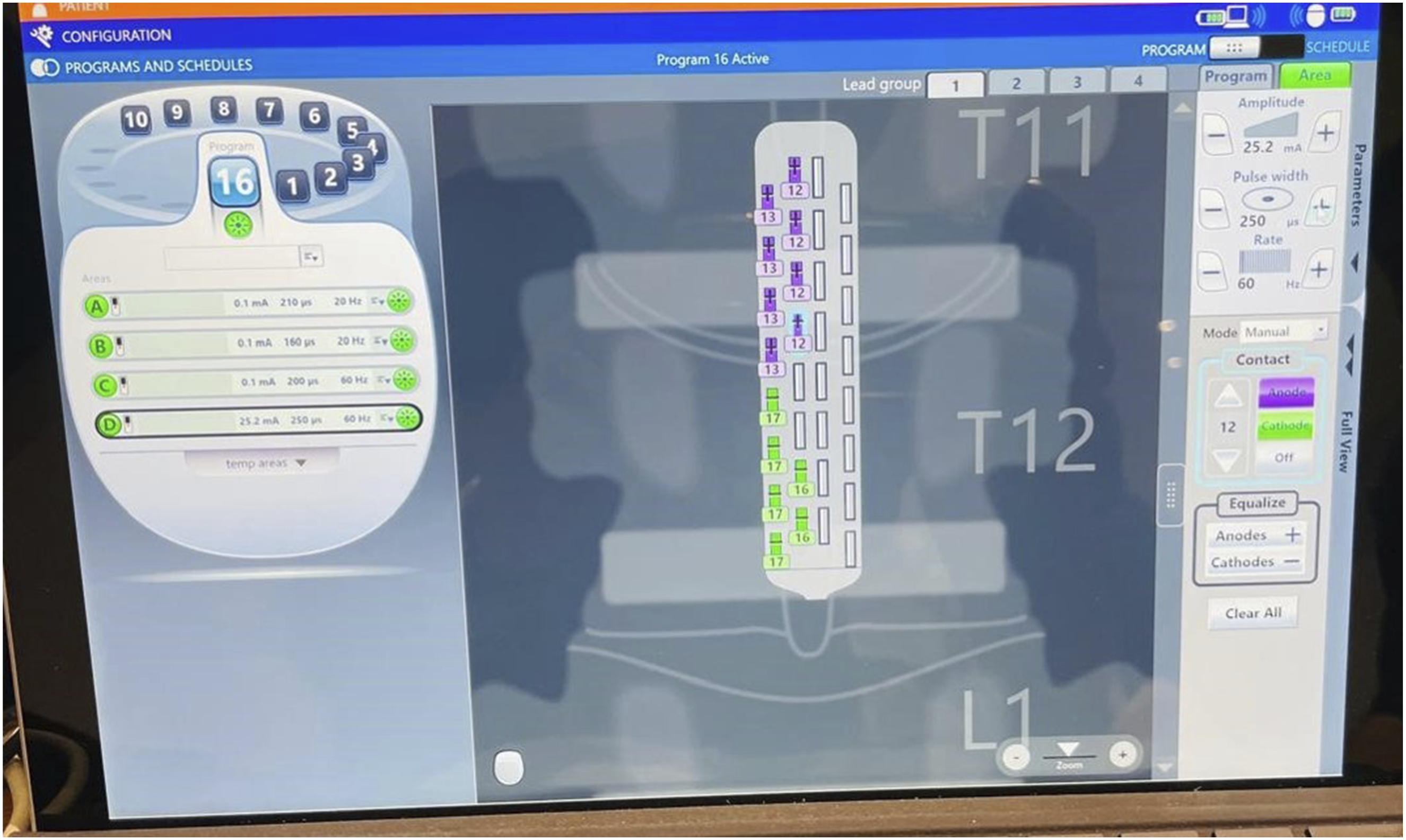
Procedure
Mapping for Standing
Electrode Array Placement
• The 32-contact electrode is surgically implanted over the dorsal aspect of the spinal cord, typically between T10–L1, targeting lumbosacral motor networks that control lower limb muscles. • The rostrocaudal (top-bottom) distribution of contacts allows access to multiple spinal segments responsible for hip, knee, and ankle motor control.
Stimulation Mapping Process
• Mapping begins after surgical recovery (∼1 week post-implantation). • Each contact (or group of contacts) is activated individually or in patterns to identify: o Motor thresholds (minimum amplitude to elicit muscle response) o Muscle activation patterns (e.g., quadriceps, hamstrings, gastrocnemius) o Postural control effects (co-contraction stability)
Following a 2-week recovery period, participants began an 8-week ESCS protocol. Stimulation parameters were individually adjusted based on real-time physiological responses. The following ranges were typically used:
Frequency: 20-60 Hz.
Pulse width: 250-450 μs.
Amplitude: Sub-threshold (0.5-4.0 V).
Frequency & Amplitude:
Frequencies span from 10 Hz to 1 kHz, with sub-perception typically using low-frequency (10-100 Hz).
Functional Outcomes Were Assessed Pre- and Post-intervention Using
Timed Up and Go (TUG) test.
Walking Index for Spinal Cord Injury II (WISCII).
Neurogenic Bladder Symptom Score (NBSS).
Manual Muscle Testing (MMT) for knee extension.
Antero-Posterior (AP displacement) on TYMO for balance standing.
Modified Ashworth Scale (MAS).
Statistical Analysis and Data Analysis.
Walking Ability: Paired t-test for Timed Up and Go test scores.
AP displacement testing: Paired t test for Antero posterior displacement for standing balance.
Bladder Function: Paired t-test for Neurogenic Bladder Symptom Score (NBSS).
MMT manual muscle testing: Paired t test for MMT of knee extension.
Statistical analyses were conducted using IBM SPSS Statistics Version 27.0 (IBM Corp., Armonk, NY, USA). Results were considered statistically significant when P-values were less than 0.05.
Paired sample t-tests were conducted to compare pre- and post-intervention scores using IBM SPSS Statistics V27.0.
Ethical approval and consent to participate The study was conducted according to the guidelines of the Declaration of Helsinki and written informed consent was obtained from each subject, and the center research protocol for this study ethical approval for this study was obtained from the Institutional Ethics Committee of School Ethical committee, approval number SEC/PT/04/24.
Disclosure Statement
This study utilized the Boston Scientific 32-contact epidural stimulation system for mapping and neuromodulation procedures. The device was used solely for clinical and research purposes in accordance with institutional ethical guidelines. Boston Scientific was not involved in the study design, data collection, analysis, or interpretation of results ethical approval for this study was obtained from the Institutional Ethics Committee of School Ethical committee, approval number SEC/PT/04/24.
*Each participant received a surgically implanted epidural stimulator using the Boston Scientific CoverEdge™ X 32-electrode Surgical Lead Kit, positioned over the T10–L1 spinal segments to target lower limb motor and bladder control circuits. Epidural spinal cord stimulation (ESCS) was delivered via the WaveWriter Alpha™ SCS System (Boston Scientific, USA). Devices were obtained through institutional procurement or academic collaboration. Boston Scientific had no role in the study design, data collection, or analysis; all protocols were independently developed and implemented by the research team.
Change in Timed up and Go (TUG) Test Performance
Mean TUG time significantly decreased from 265.25 ± 165.2 seconds at baseline to 82.9 ± 112.6 seconds post-intervention (t = 6.54, P = 2.91 × 10-6), indicating a significant improvement in functional mobility following 8 weeks of epidural spinal cord stimulation combined with neurorehabilitation. Figure 3. Timed up and Go (TUG) Test Performance Scores. Chart Displaying Functional Mobility Assessment Results Using the Timed up and Go Test (Measured in Seconds) for Participants With Chronic Thoracic Spinal Cord Injury. Data Shows Performance Times Before and After Eight Weeks of Epidural Spinal Cord Stimulation Combined With Physiotherapy and Robotic Balance Training. Lower Completion Times Indicate Improved Functional Mobility. Data Presented as Individual Participant Values With Group Mean ± Standard Deviation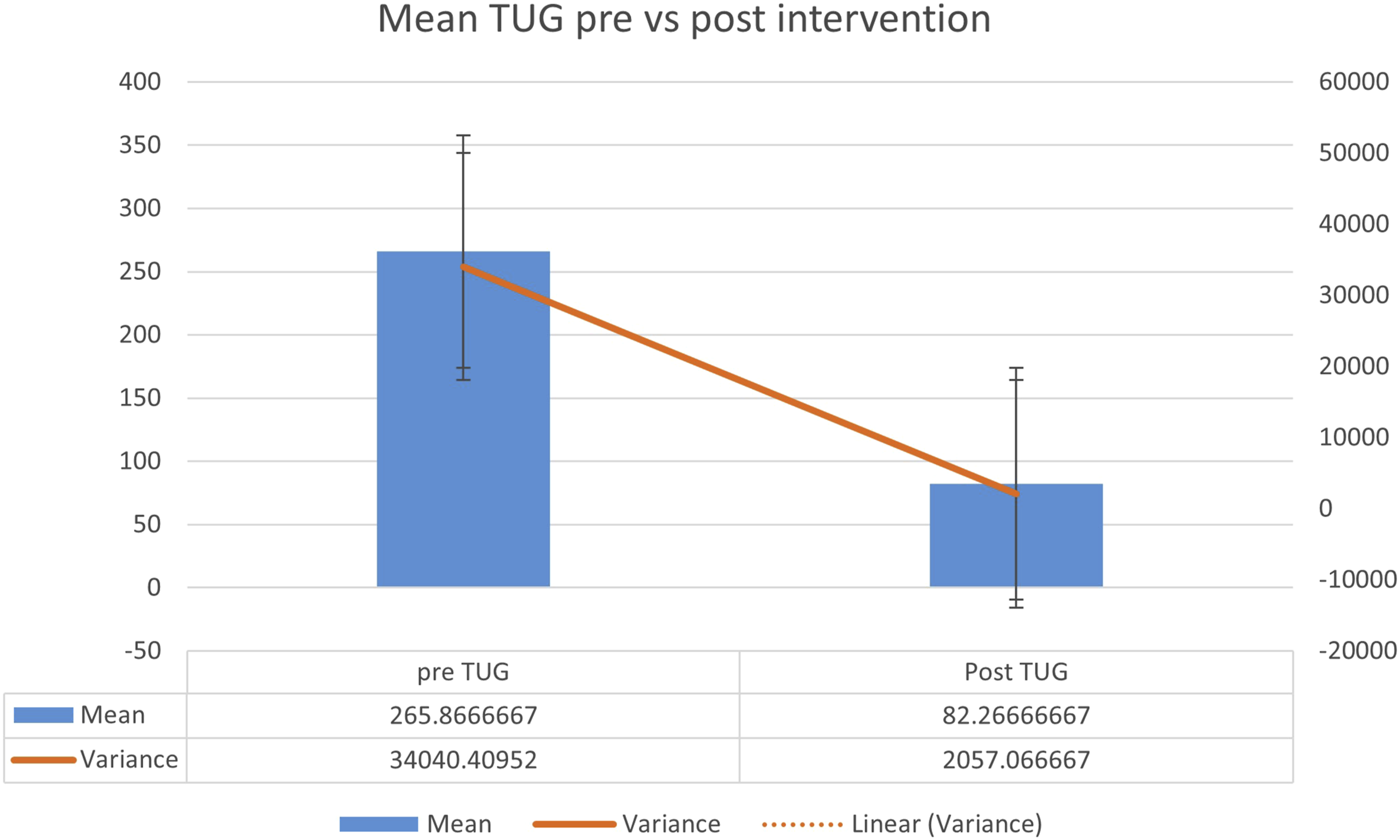
Patient Performing TUG Test During Functional Mobility Assessment.
Photograph demonstrates evaluation of dynamic balance and walking ability during the Timed Up and Go test. Participant Performing the Timed up and Go (TUG) Test. Photograph Documenting a Study Participant With Chronic Thoracic Spinal Cord Injury Performing the Standardized Timed up and Go Test, which Measures the Time Required to Rise From a Chair, Walk 3 Meters, Turn Around, Walk Back, and Sit Down. This Functional Mobility Assessment was Conducted before and after the Eight-Week Intervention Protocol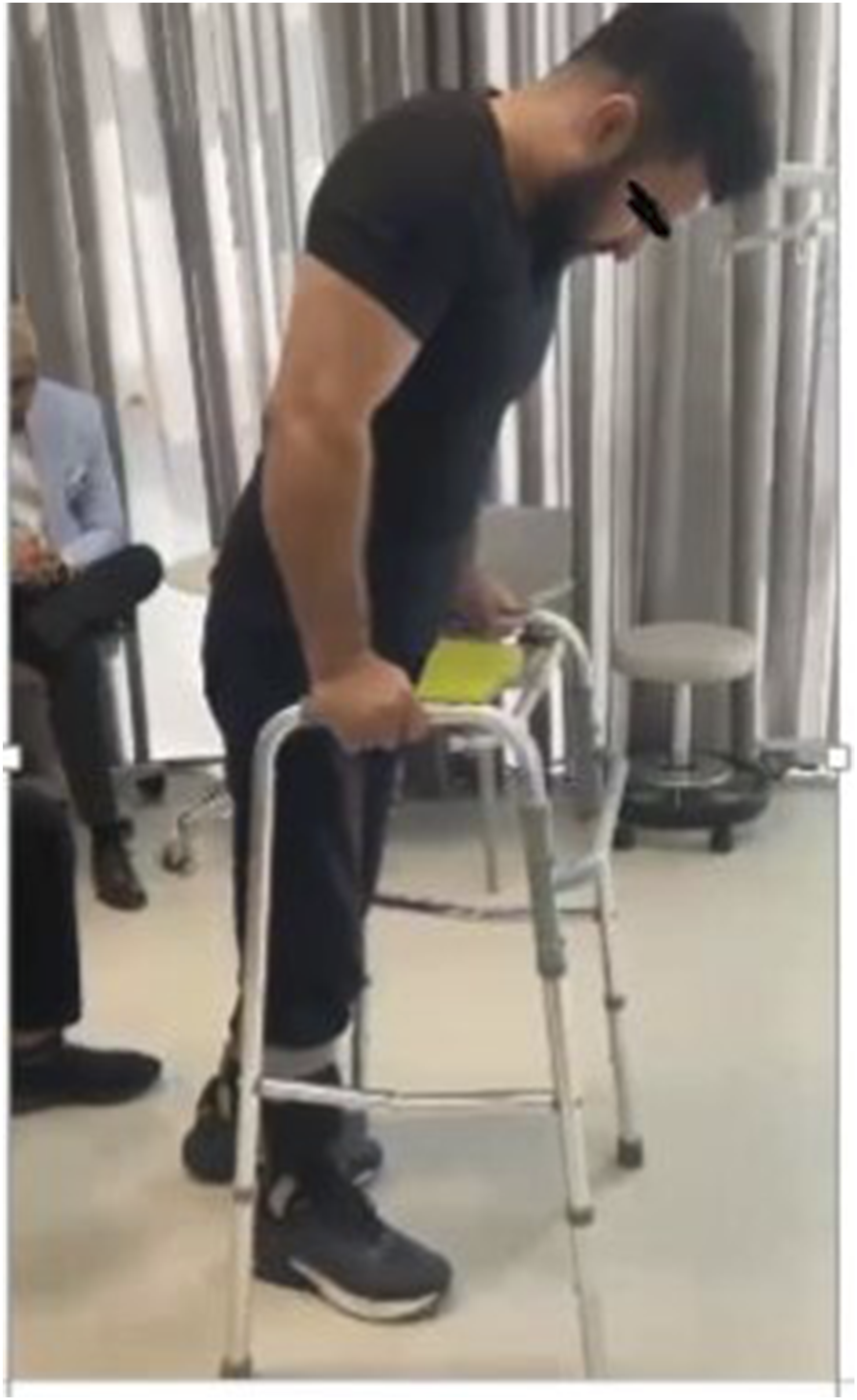
Change in Walking Index for Spinal Cord Injury II (WISCI II) score.
WISCII scores increased markedly from 0.075 ± 5.12 pre-intervention to 6.08 ± 2.69 post-intervention (t = −29.88, P = 1.09 × 10-18), reflecting a significant improvement in walking index reflecting on enhanced ambulatory function among participant. Figure 5. Walking Index for Spinal Cord Injury II (WISCI-II) Ambulatory Function Scores. Comparison of Walking Independence Levels Measured Using the Walking Index for Spinal Cord Injury II (0-20 Scale, where 0 = Unable to Stand and/or Participate in Assisted Walking and 20 = Ambulates With no Devices, no Braces, and no Physical Assistance for 10 meters) in Participants With Chronic Thoracic Spinal Cord Injury. Data Presented Before and after Eight Weeks of Epidural Spinal Cord Stimulation Combined With Physiotherapy and Robotic Balance Training. Higher Scores Indicate Greater Walking Independence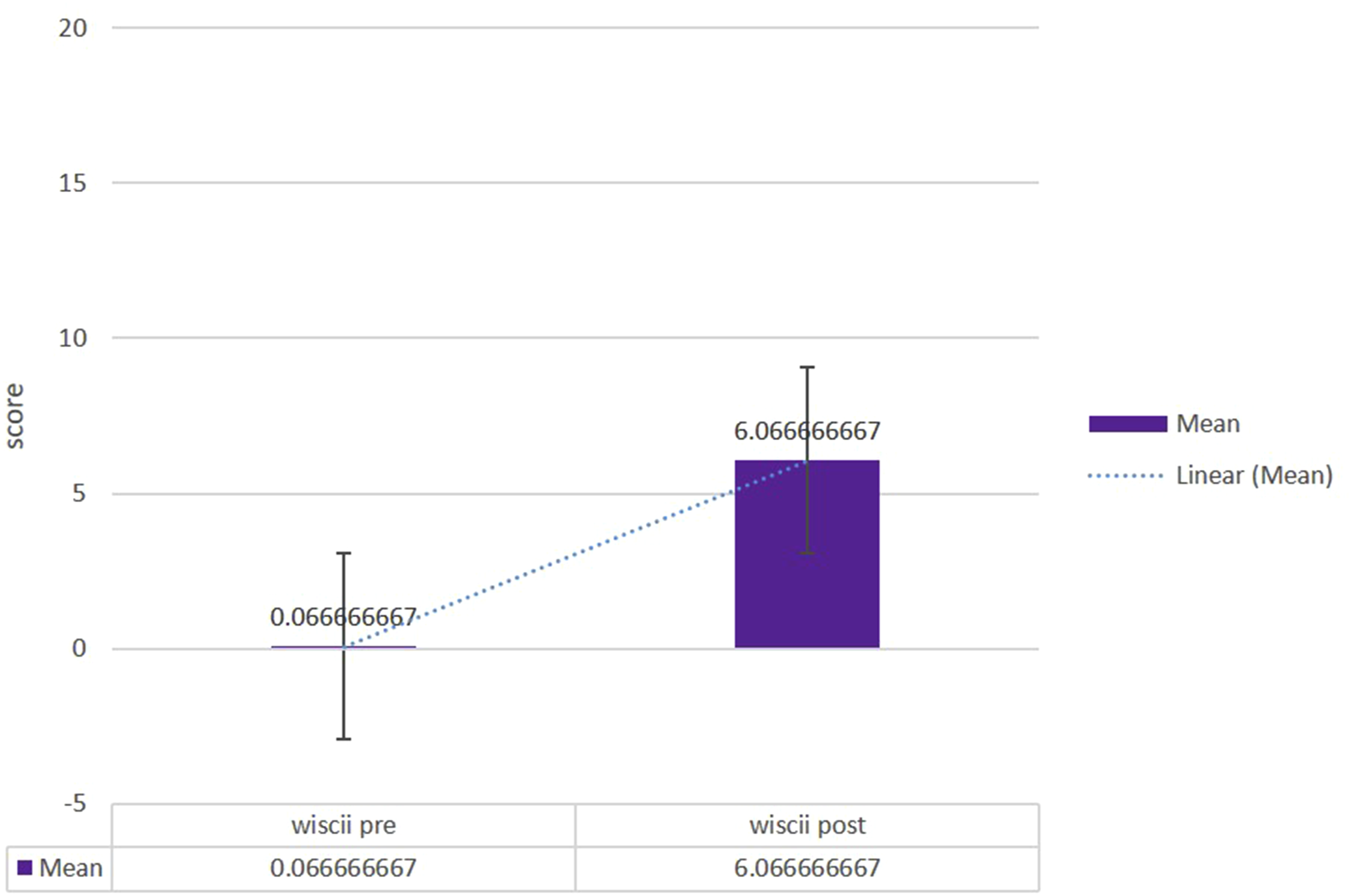
Change in Neurogenic Bladder Symptom Score (NBSS)
Participants showed a significant reduction in NBSS scores from 21.65 ± 6.91 to 8.4 ± 5.08 (t = 12.50, P = 1.30 × 10-10), suggesting notable improvement in autonomic bladder function post-intervention. Figure 6. Neurogenic Bladder Symptom Score (NBSS) before and After Intervention. Assessment of Bladder Control Symptoms Using the Neurogenic Bladder Symptom Score (0-74 Scale, where Higher Scores Indicate More Severe Bladder Dysfunction Symptoms) in Fifteen Participants With Chronic Thoracic Spinal Cord Injury. Data Shows Symptom Severity at Baseline and Following Eight Weeks of Epidural Spinal Cord Stimulation Combined With Physiotherapy and Robotic Balance Training. Lower Scores Indicate Fewer Neurogenic Bladder Symptoms and Improved Bladder Function. Data Presented as Individual Participant Scores With Statistical Analysis Using Paired t-Test (*P < 0.05)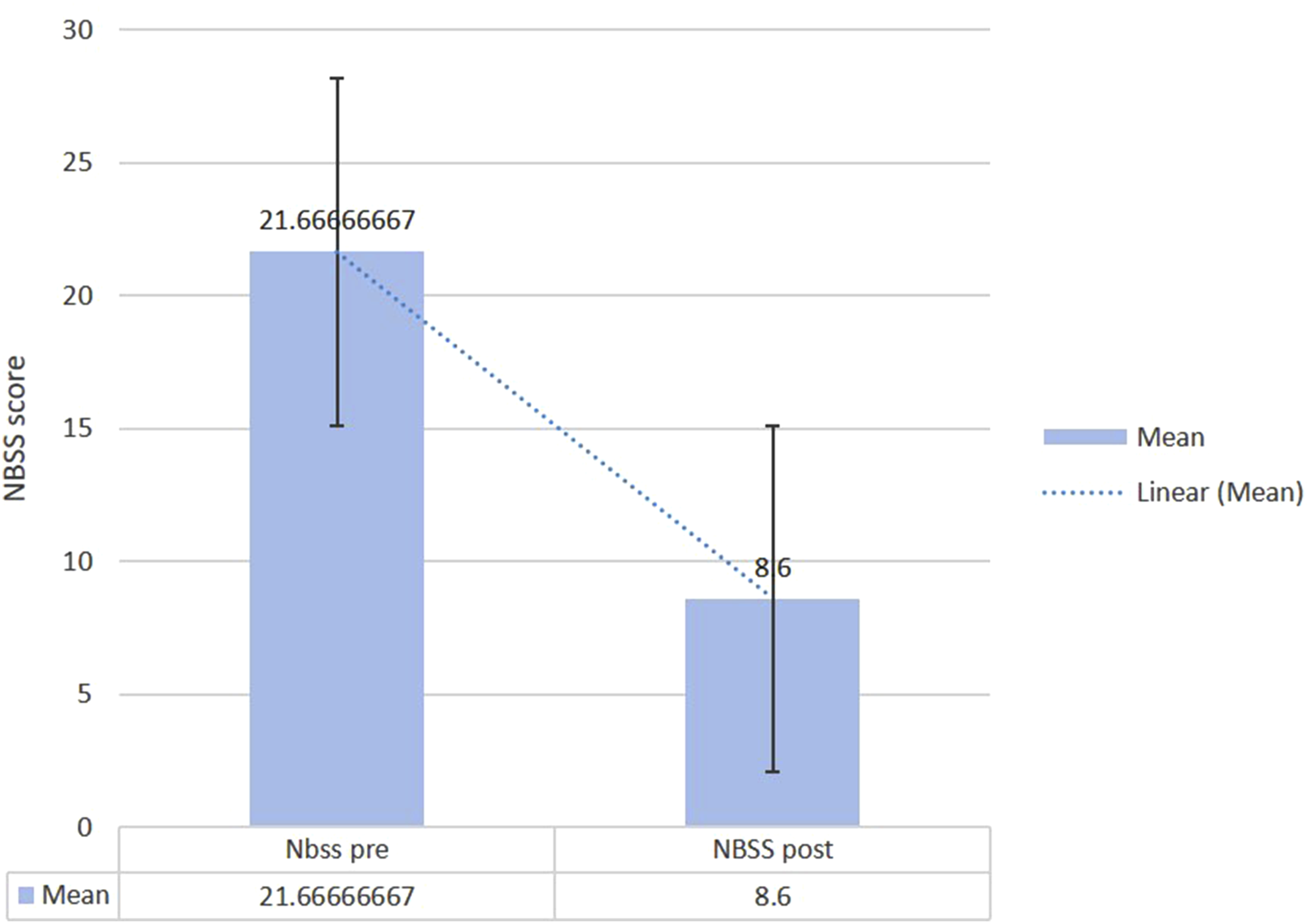
Change in Manual Muscle Testing (MMT) Scores.
MMT scores improved significantly from a baseline mean of 0.25 ± 0.44 to 3.85 ± 0.37 post-intervention (t = −18.25, P = 1.16 × 10-16), indicating a substantial increase in lower limb voluntary muscle strength. Figure 7. Manual Muscle Testing (MMT) Scores for Knee Extension Strength Before and after Intervention. Comparison of Bilateral Knee Extension Strength Measured Using Manual Muscle Testing (0-5 Scale, where 0 = no Contraction and 5 = Normal Strength Against Full Resistance) in Fifteen Participants With Chronic Thoracic Spinal Cord Injury. Data Presented as Individual Participant Scores at Baseline and Following Eight Weeks of Epidural Spinal Cord Stimulation Combined With Physiotherapy and Robotic Balance Training. Higher Scores Indicate Greater Muscle Strength. Statistical Significance Determined Using Paired t-Test (*P < 0.05)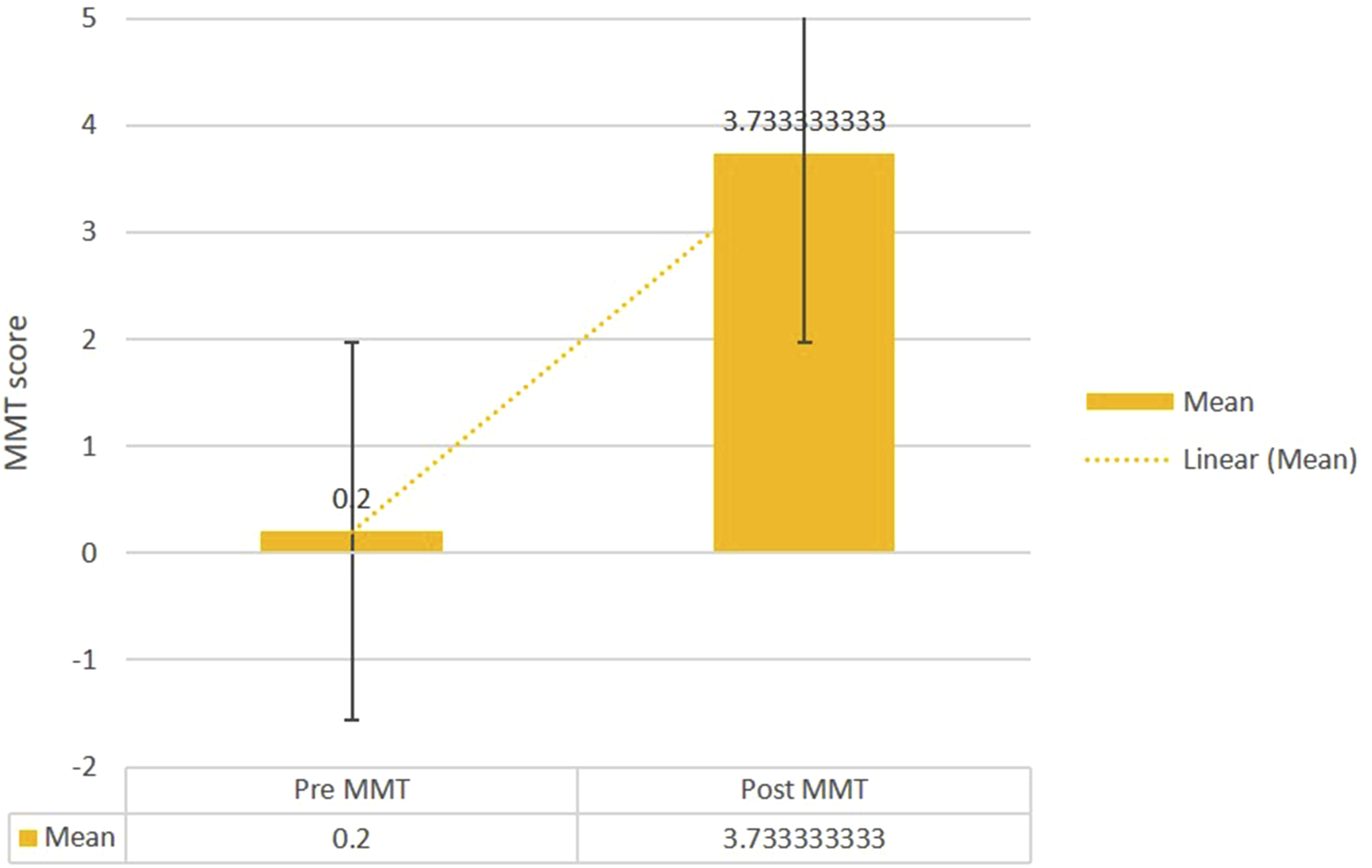
Change in Antero-Posterior Displacement
Antero-posterior displacement reduced from 6.6 ± 0.87 cm pre-intervention to 3.26 ± 1.35 cm post-intervention (t = 7.19, P = 1.73 × 10-10), indicating significant improvement in postural stability and balance control. Figure 8. Anterior–Posterior Postural Sway During Balance Assessment. Graphical Representation of Anterior–Posterior Displacement Measurements (cm) During Postural Stability Testing in Participants With Chronic Thoracic Spinal Cord Injury. Data Shows Balance Performance before and after Eight Weeks of Epidural Spinal Cord Stimulation Combined With Physiotherapy and Tyromotion-Based Robotic Balance Training. Reduced Displacement Values Indicate Improved Postural Stability. Data Presented as Mean ± Standard Deviation With Individual Participant Values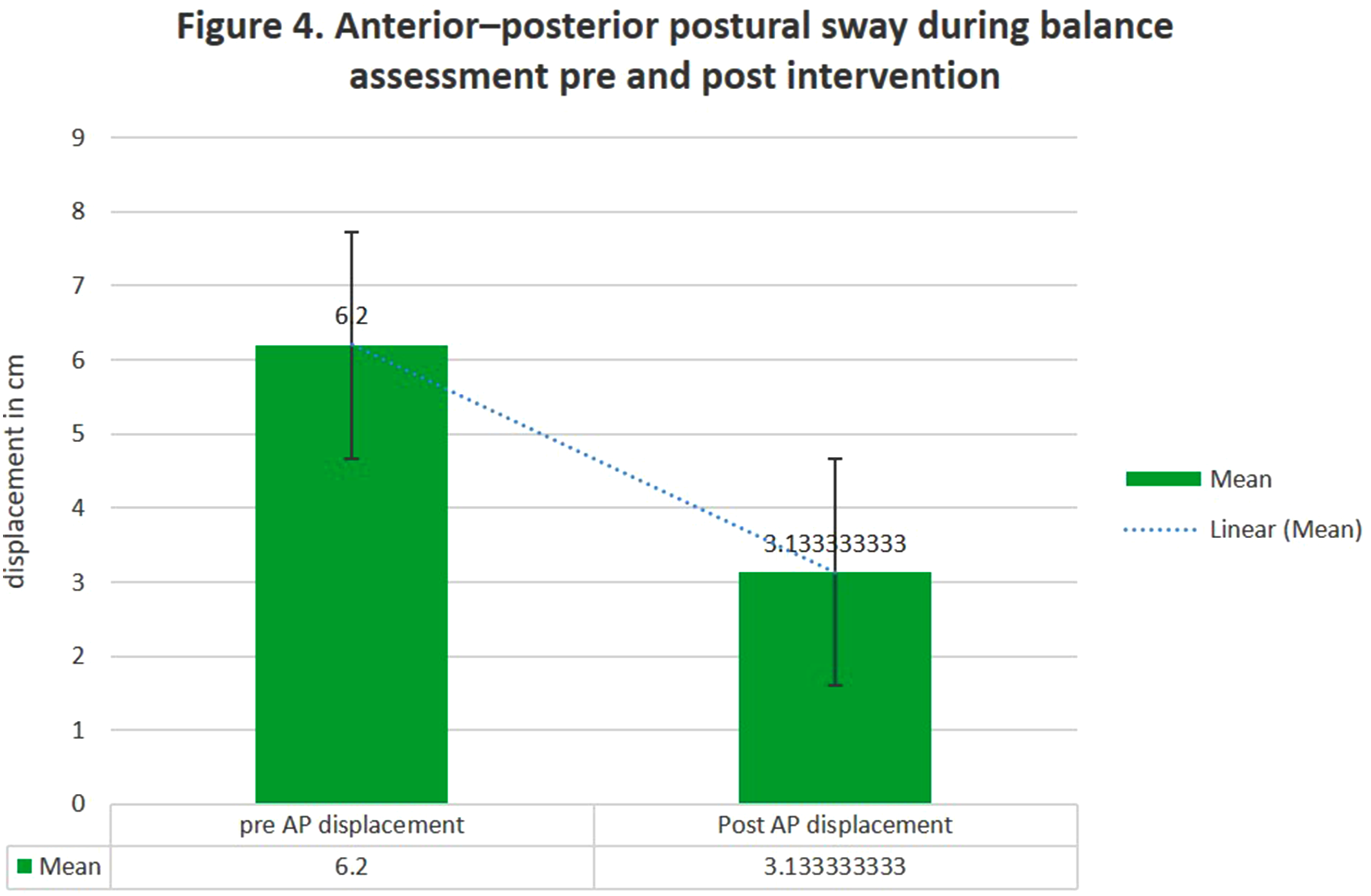
Participant performing balance assessment on Tyromotion balance board Figure 5.
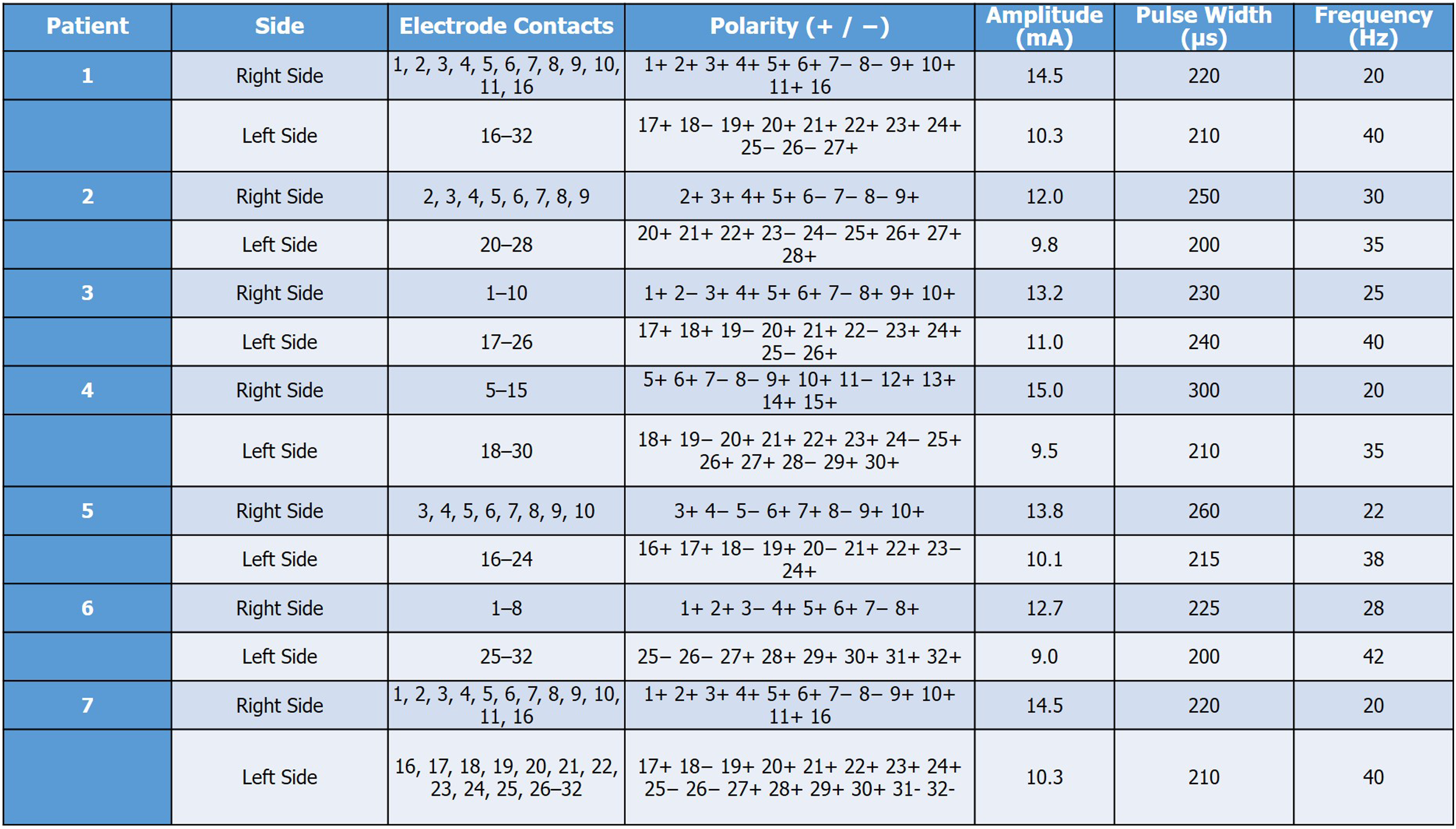
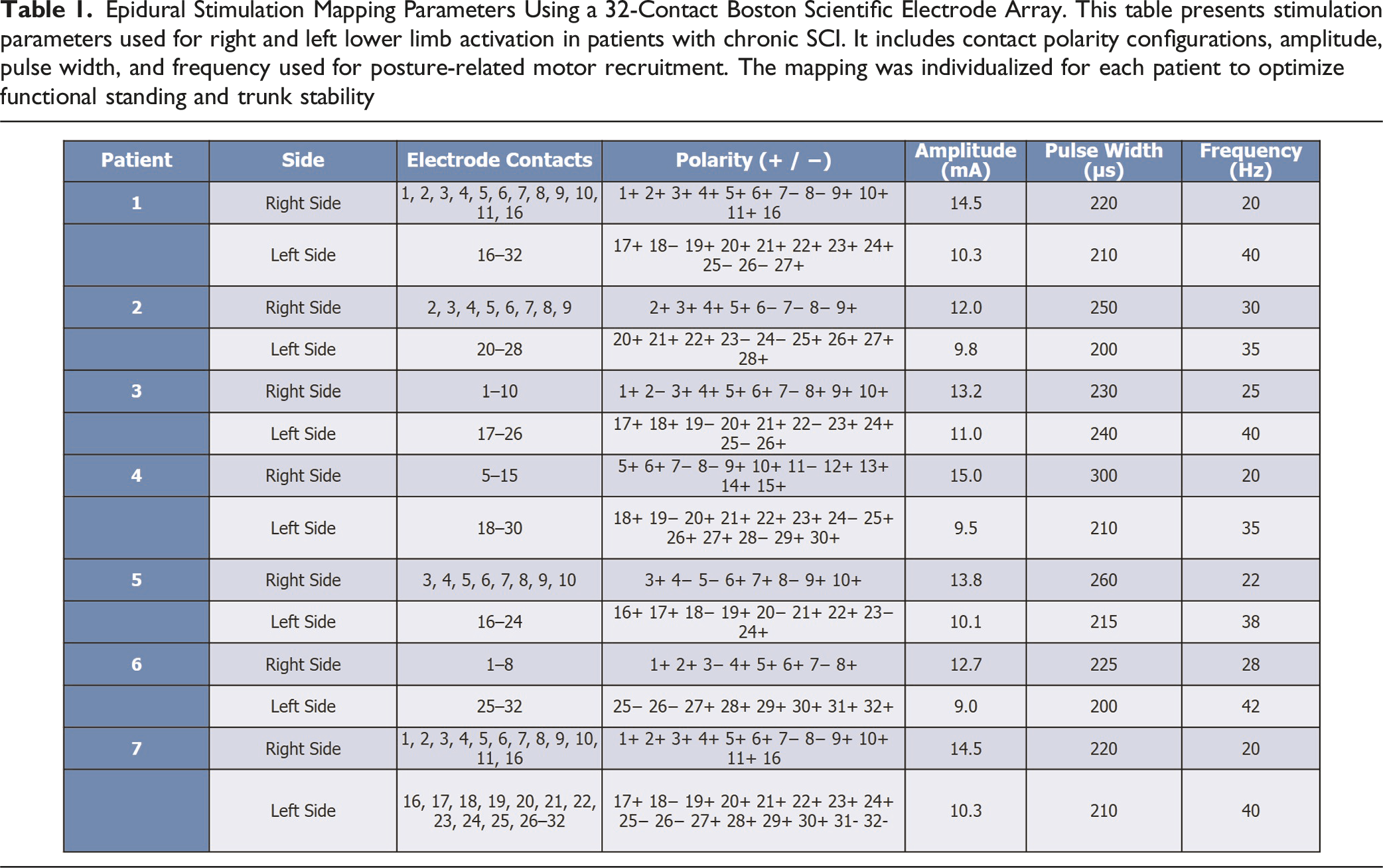
Epidural Stimulation Mapping Parameters Using a 32-Contact Boston Scientific Electrode Array. This table presents stimulation parameters used for right and left lower limb activation in patients with chronic SCI. It includes contact polarity configurations, amplitude, pulse width, and frequency used for posture-related motor recruitment. The mapping was individualized for each patient to optimize functional standing and trunk stability
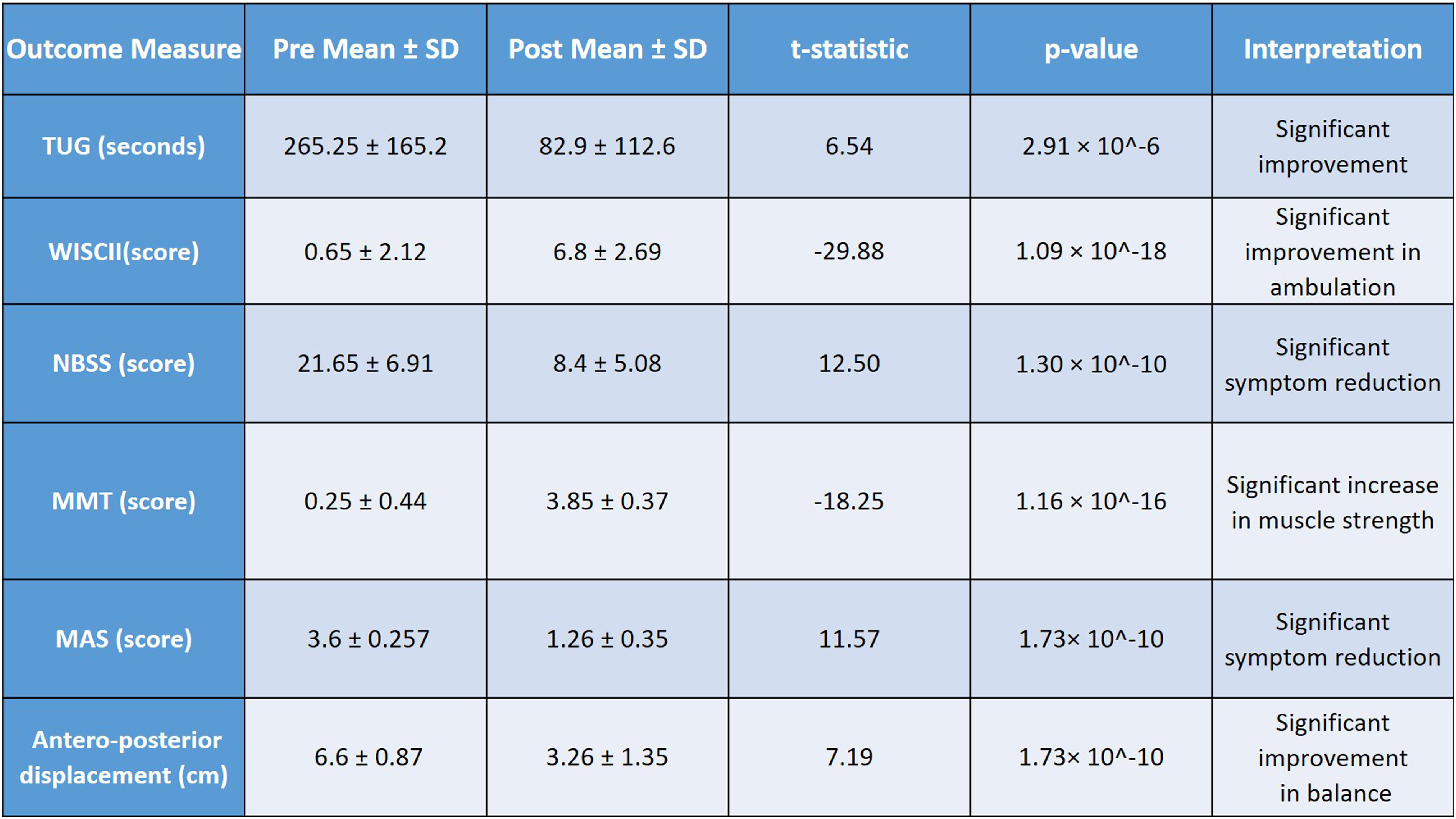
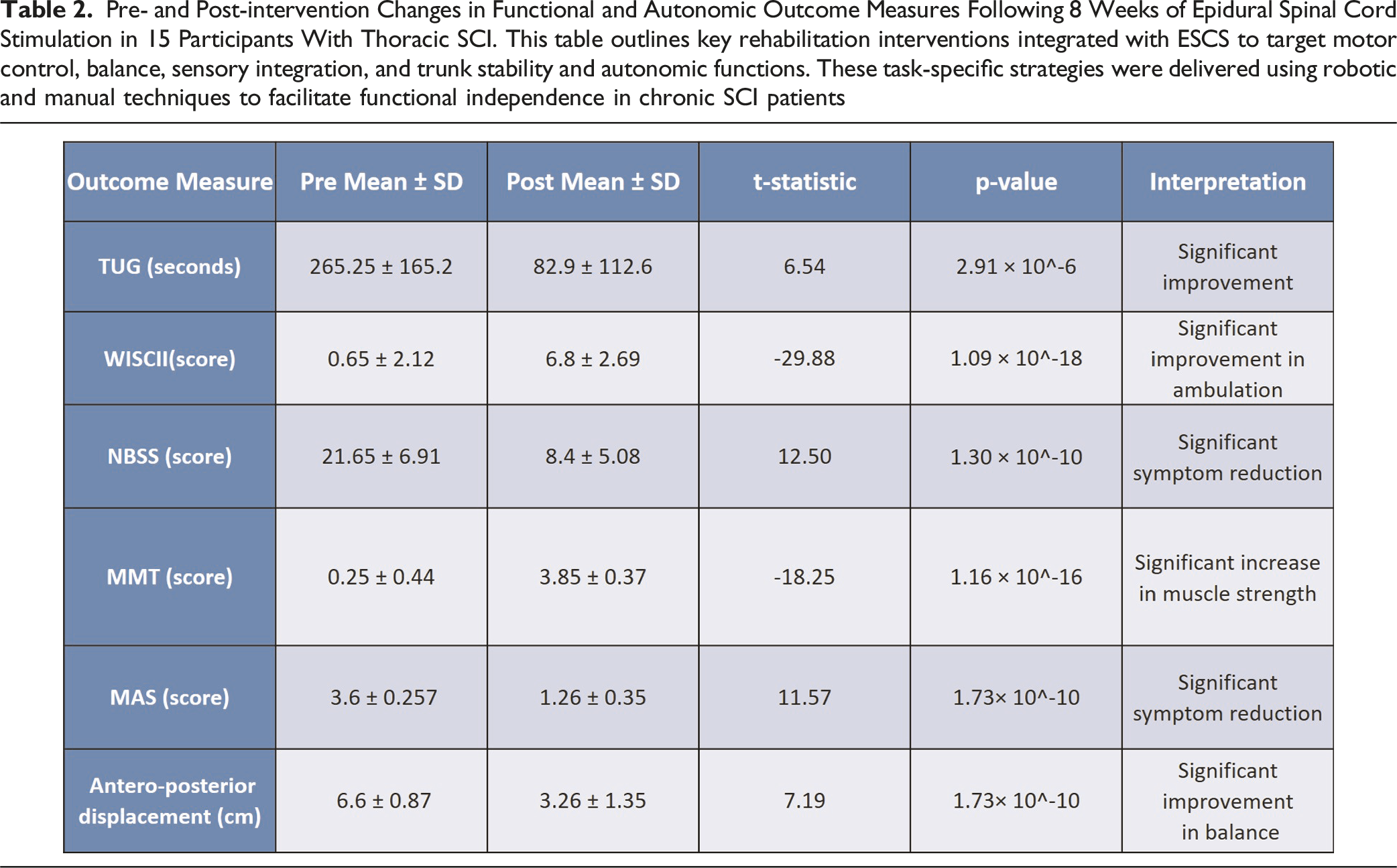
Pre- and Post-intervention Changes in Functional and Autonomic Outcome Measures Following 8 Weeks of Epidural Spinal Cord Stimulation in 15 Participants With Thoracic SCI. This table outlines key rehabilitation interventions integrated with ESCS to target motor control, balance, sensory integration, and trunk stability and autonomic functions. These task-specific strategies were delivered using robotic and manual techniques to facilitate functional independence in chronic SCI patients
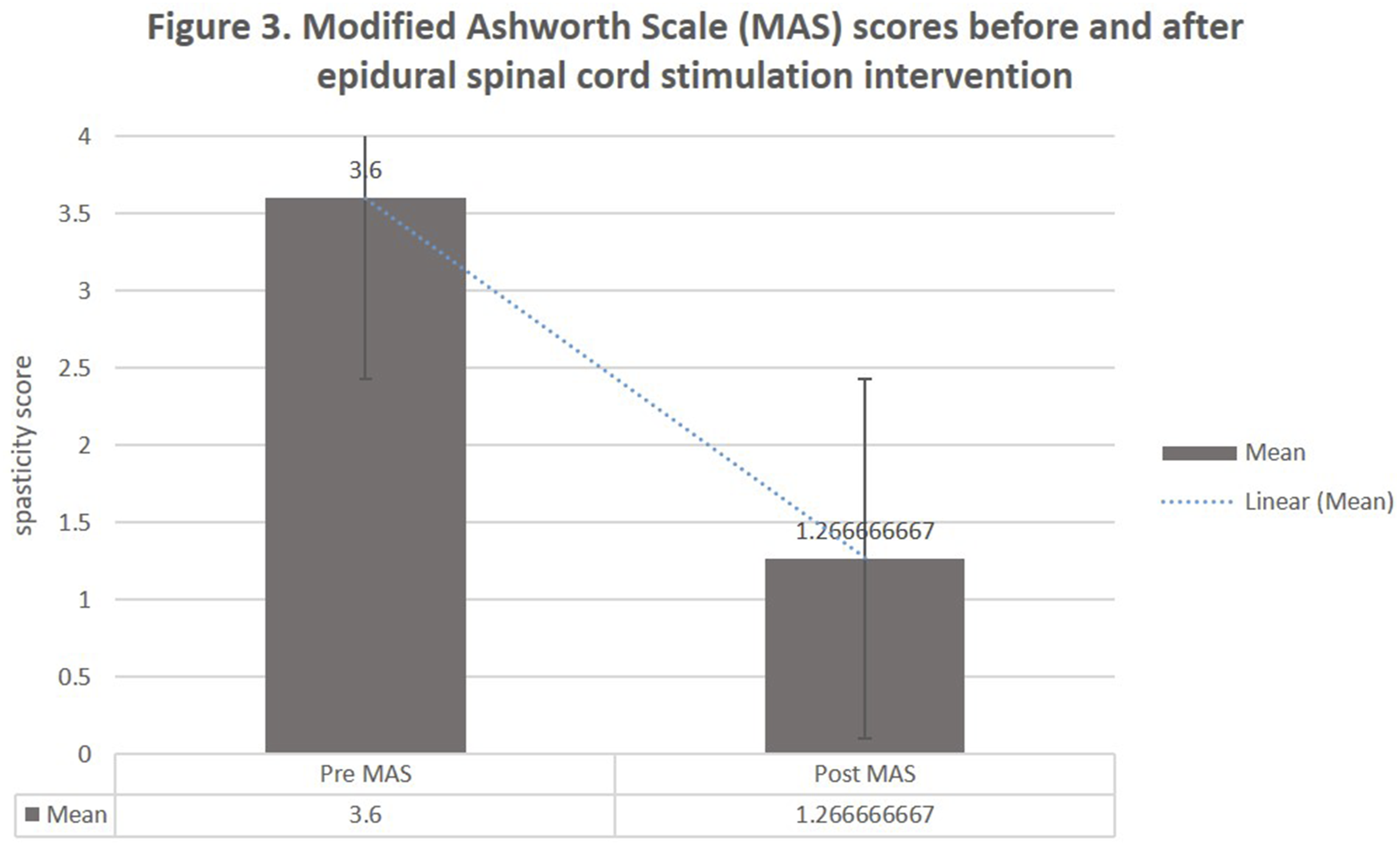
Change in Modified Ashworth Scale (MAS) Scores
MAS scores decreased from 3.6 ± 0.257 to 1.26 ± 0.35 post-intervention (t = 11.57, P = 1.73 × 10-10), reflecting a significant reduction in spasticity, likely due to neuromodulation-induced changes in spinal excitability. Figures 9 and 10. Participant Performing Balance Training on Tyromotion Robotic Balance Platform. Photograph Showing a Study Participant With Chronic Thoracic Spinal Cord Injury Engaged in Robotic Balance Training Using the Tyromotion Balance Board System. This Equipment was Used in Combination With Epidural Spinal Cord Stimulation and Physiotherapy as Part of the Eight-Week Intervention Protocol to Improve Postural Stability and Functional Balance Modified Ashworth Scale (MAS) Scores Before and After Epidural Spinal Cord Stimulation Intervention. Comparison of Spasticity Levels Measured Using the Modified Ashworth Scale (0-4 Scale, where 0 = no Increase in Muscle Tone and 4 = Affected Part Rigid in Flexion or Extension) in Fifteen Participants With Chronic Thoracic Spinal Cord Injury. Data Presented as Individual Participant Scores at Baseline (Pre-intervention) and Following Eight Weeks of Daily Epidural Spinal Cord Stimulation Combined With Physiotherapy and Robotic Balance Training (Post-intervention). Lower Scores Indicate Reduced Spasticity. Statistical Significance Determined Using Paired t-Test (*P < 0.05)
Discussion
The present study demonstrates that a structured physiotherapy protocol targeting mobility, balance, bladder symptoms, and lower limb strength can produce significant improvements in individuals with neuromotor impairments. The substantial reduction in TUG time reflects enhanced mobility and reduced fall risk. This aligns with existing literature emphasizing the effectiveness of gait and balance training in neurorehabilitation. The combination of epidural spinal cord stimulation (ESCS) with Tyromotion-assisted neurorehabilitation yielded significant improvements in voluntary motor function, functional mobility, and postural control in individuals with chronic spinal cord injury (SCI). The marked enhancement in Timed Up and Go (TUG) performance, along with gains in manual muscle strength and Walking Index for Spinal Cord Injury II (WISCII) scores, indicate meaningful restoration of motor output and trunk coordination. These outcomes reflect increased central excitability and improved neuromuscular recruitment, likely mediated by ESCS-facilitated activation of the spinal networks below the level of injury.
This finding aligns with previous research showing enhanced lower extremity motor function with Epidural stimulation in spinal cord injury14,18. The significant improvement in TUG performance suggests that Epidural stimulation may facilitate activation of spinal locomotor networks, potentially through increased excitability of motor neurons and interneurons below the level of injury.15,16
The improved mean WISCI II scores post-intervention reflect a notable enhancement in ambulatory function among participants. This upward trend indicates increased walking capability, reduced dependence on assistive devices, and improved lower limb motor coordination following ESCS combined with targeted neurorehabilitation. Enhanced sitting balance is a critical component of functional independence and may facilitate transfer abilities and wheelchair skills in individuals with SCI. These improvements in postural control may be attributed to the activation of trunk muscles and enhanced proprioceptive feedback during stimulation. 17
AP Displacement Improves More due to several factors Trunk and lower limb motor control ESCS often enhances extensor control and volitional leg movement, which primarily contributes to AP postural adjustments (forward–backward balance).
Task alignment Most rehabilitation tasks (e.g., sit-to-stand, walking, stepping) challenge and train AP stability more than ML.
Spinal circuitry engagement ESCS targets lumbar spinal networks responsible for stepping and standing, which are more relevant to sagittal (AP) balance than frontal (ML) control. A particularly notable finding was the greater recovery in anteroposterior (AP) balance control compared to mediolateral (ML) stability. This directional specificity may be explained by the preferential engagement of extensor muscle synergies and postural reflexes along the sagittal plane during weight-bearing and stepping tasks. Enhanced AP control is clinically significant, as it directly impacts sit-to-stand transitions, gait initiation, and dynamic balance—key determinants of functional independence. 25
Sensorimotor integration Proprioceptive feedback during gait and static balance rehabilitation is more AP-dominant, especially with robotic or visual cues. 22
Additionally, the Tyromotion Balance System’s focus on core activation and trunk stability indirectly supports improved bladder and bowel function. By strengthening abdominal and pelvic muscles through balance training, SCI patients may experience improved control over their autonomic functions. Clinical reports suggest that, post-EES, the use of balance therapy can further optimize the outcomes of bladder and bowel function, with some patients reporting a reduction in the need for catheterization or other invasive interventions.
The improvements in bladder function observed in this study may be attributed to several mechanisms 1 : activation of autonomic circuits controlling bladder function;20,23 enhanced sensory awareness of bladder fullness; and 3 improved coordination between bladder contraction and sphincter relaxation. These findings support previous research suggesting that spinal cord stimulation can modulate autonomic functions in addition to motor control 21 .
The observed improvements in walking and balance are likely due to the reactivation of CPGs. ESCS enhances excitability in spinal networks, allowing for coordinated motor output despite a lack of direct cortical input. 10 Additionally, ESCS may facilitate plasticity at the synaptic level, promoting adaptive reorganization in response to stimulation23,24.
Bladder function improvements suggest that ESCS modulates spinal reflex circuits governing urination. The restoration of bladder control could result from increased excitability of neural pathways regulating the detrusor muscle, leading to improved urinary function and reduced incontinence episodes25,27.
Improvements in Postural Control and Weight Distribution
Postural control and symmetrical weight distribution are essential for standing and balance in SCI patients, and these are often impaired due to the loss of motor control. The Tyromotion Balance System addresses these challenges by providing real-time feedback on posture and weight distribution, enabling patients to adjust their movements and improve balance. This is particularly beneficial for patients with thoracic SCI, who may struggle with trunk stability and lower limb control.24.
The system’s ability to encourage symmetrical weight distribution is crucial for reducing compensatory movements that can lead to further musculoskeletal issues. By training patients to distribute their weight more evenly, the Tyromotion system helps improve postural alignment, reduces the risk of falls, and enhances overall functional mobility. This has significant implications for daily activities, as better postural control translates into greater independence in tasks such as transferring, standing, and walking.
The improvement in WISCII scores indicates better trunk control and sitting balance, which are critical for functional independence. Previous studies have suggested that trunk stability training significantly contributes to improved postural alignment and safety during transfers and ADLs (Activities of Daily Living).
Several Aspects of our protocol May Have Mediated These Favorable Results
Stimulator parameters assigned uniquely to each subject, Mixed stimulation with functional workouts (standing, stepping, sitting), Autonomic Functions Selective Stimulation, The intervention period was long enough (8 weeks) to facilitate neural adaptations and Sufficient intervention duration (8 weeks) to allow for neural adaptation. These findings suggest that ESCS should be considered as part of a comprehensive rehabilitation approach for individuals with thoracic SCI. However, it is important to note that ESCS is an invasive intervention requiring surgical implantation, and careful patient selection is essential.
Bladder function, measured through NBSS, showed a marked decrease in symptoms post-intervention. This suggests the intervention’s effectiveness in addressing autonomic dysfunctions commonly observed in neurological conditions. Improvements may be attributed to neuromodulatory exercises and patient education on bladder management techniques. The use of epidural stimulation has shown promise in improving autonomic functions, including neurogenic bladder and bowel control, which are often compromised in patients with thoracic SCI. The enhanced stimulation of neural pathways involved in autonomic regulation may reduce the severity of neurogenic bladder symptoms, such as incontinence or retention. 25
The rise in MMT scores confirms enhanced strength in knee extensors, which is vital for standing, walking, and maintaining posture. Strength gains are likely due to task-specific resistance training and motor facilitation exercises administered during the protocol.
Overall, these results underscore the synergistic effect of combining ESCS with task-specific, sensorimotor-enriched rehabilitation. The stimulation appears to prime the spinal circuitry, enhancing its responsiveness to afferent inputs during training. This process likely drives activity-dependent plasticity at both spinal and supraspinal levels, contributing to the observed functional gains. 26
Lastly, Spasticity Reduction Post-Epidural Stimulation
Spasticity is a common secondary complication following SCI, characterized by involuntary muscle contractions, stiffness, and exaggerated reflexes. Post-EES, many patients report a reduction in spasticity due to the reactivation of inhibitory pathways within the spinal cord. The Tyromotion Balance System enhances this effect by promoting active engagement of muscles, encouraging smooth, controlled movements that mitigate spastic episodes. 23
By integrating functional tasks into balance exercises, the Tyromotion system trains patients to control their movements more precisely, reducing spasticity in the lower limbs. This not only contributes to greater comfort but also enhances the overall quality of life by allowing patients to participate more fully in rehabilitation activities and daily living tasks.
The observed reduction in Modified Ashworth Scale (MAS) scores suggests a concurrent decrease in lower limb spasticity, supporting the hypothesis that neuromodulatory interventions may normalize abnormal reflex activity and facilitate more coordinated voluntary movement. In addition, improvements in neurogenic bladder symptom scores
These results underscore the potential of holistic, integrated physiotherapy approaches in neurorehabilitation settings. However, limitations include the absence of a control group, small sample size, and short follow-up duration. Future randomized controlled trials with larger cohorts are recommended. 19
Limitations and Future Research Opportunities
This study has several limitations that should be addressed in future research:
Limited generalizability (n = 0). 1. The absence of a control group makes it challenging to separate the impacts of ESCS from those of intensive rehabilitation alone. 2. There was no long-term follow-up to evaluate the sustainability of improvements. 3. The study focused on individuals with thoracic injuries (T6-T10) and may not generalize to other injury levels.
Conclusions
This study demonstrates that an 8-week program combining epidural spinal cord stimulation (ESCS) with task-specific physiotherapy and robotic balance training produces significant improvements across multiple functional domains in individuals with chronic thoracic spinal cord injury. Participants showed marked enhancements in walking ability (WISCI-II), sitting balance (FIST), functional mobility (TUG), postural stability (reduced anterior-posterior sway), lower limb strength (MMT), bladder function (NBSS), and spasticity reduction (MAS).
The therapeutic efficacy of ESCS likely stems from its ability to activate dormant spinal sensorimotor circuits and enhance afferent input processing, facilitating neuroplastic adaptations at both spinal and cortical levels. By stimulating dorsal root afferents, ESCS improves proprioceptive feedback and promotes reweighting of sensory inputs, enabling more effective integration of vestibular, somatosensory, and visual information for postural control. The concurrent rehabilitation training capitalizes on this enhanced neural excitability, promoting motor learning and functional recovery through repetitive, task-specific practice.
The comprehensive improvements observed across diverse outcome measures underscore ESCS as a potent therapeutic intervention with broad rehabilitative potential for spinal cord injury. These findings provide compelling evidence for the clinical translation of neuromodulation technologies and support the integration of ESCS protocols into standard rehabilitation practices. Future research should focus on optimizing stimulation parameters, determining ideal candidate selection criteria, and investigating long-term retention of functional gains to further establish ESCS as a cornerstone therapy for improving quality of life in the spinal cord injury population.
Limitations and Future Scope
The sample size is relatively small. However, this randomized controlled trial was conducted as an initial clinical study with the primary objective of evaluating efficacy and feasibility. In the current study phase, a control group was not included, as the primary objective was to evaluate feasibility and preliminary efficacy of the combined ESCS and Tyromotion-assisted rehabilitation approach. We agree that including a group receiving only rehabilitation training would provide valuable insights. Future studies should include a control group and long-term follow-up to validate these findings.
Footnotes
Author Note
All authors read and approved the final manuscript and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work were appropriately investigated and resolved.
Acknowledgement
The authors express their sincere gratitude to Dr Shahiduz Zafar, Dr Pritam Majumdar, and Dr Shagun Agarwal for their invaluable guidance and support throughout the research process. The authors also acknowledge the contributions of Serensa Health, Gurugram, India, Medipark Centre, Azerbaijan, and Galgotias University, whose support was instrumental in conducting this study. Special thanks are extended to Dr Mukul for facilitating the clinical aspect of the study and for providing essential resources, as well as to Serensa Health Centre and NeuraXis care and research center, NHI hospital for offering technical assistance. The authors are also grateful to the anonymous reviewers whose insightful feedback significantly enhanced the clarity and quality of this manuscript.
Ethical Considerations
We affirm that we have obtained Ethics Committee approval for this study. Ethics review and approval Letter from Galgotias University School of Ethics committee Ref no. SEC/PT/04/24 for granting approval for this study. We also acknowledge the Clinical Trial Registry-India for registering this study with CTRI/2024/03/064055. We understand and accept the journal’s policies regarding copyright, authorship, and ethical compliance.
Consent to Participate
The study was conducted according to the guidelines of the Declaration of Helsinki and written informed consent was obtained from each subject. The center research protocol for this study was approved by the Galgotias University School of Ethics committee (Ref no. SEC/PT/04/24) and to the Clinical Trial Registry-India for registering this study (CTRI/2024/03/064055).
Consent for Publication
All study participants provided consent for publication.
Authors’ Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data and materials, Supporting data is available can be obtained upon reasonable request first auther and corresponding author.
Disclosure
This study utilized the Boston Scientific 32-contact epidural stimulation system for mapping and neuromodulation procedures. The device was used solely for clinical and research purposes in accordance with institutional ethical guidelines. Boston Scientific was not involved in the study design, data collection, analysis, or interpretation of results.
Appendix
The following abbreviations are used in this manuscript.