Abstract
Introduction:
We aimed to report the clinical evaluation results of gait training with the Honda Walking Assist Device® (HWAT) in a patient with spinal cord injury (SCI).
Patients and Methods:
A 63-year-old male with SCI (grade D on the American Spinal Injury Association Impairment Scale) underwent 20 HWAT sessions over 4 weeks. The self-selected walking speed (SWS), mean step length, cadence, 6-minute walking test (6MWT), Walking Index for SCI score, SCI Functional Ambulation Inventory gait score, American Spinal Injury Association Impairment Scale grade, neurological level, upper and lower extremity motor scores, modified Ashworth Scale, Penn Spasm Frequency Scale, and Spinal Cord Independence Measure version III were measured on admission, at the start of HWAT, at 2 and 4 weeks post-HWAT, and at discharge. Three-dimensional kinematic gait analysis and electromyographic assessments were performed before and after HWAT.
Results:
The patient safely completed 20 HWAT sessions. We found improvements above the clinically meaningful difference in SWS and 6MWT as well as increased hip extension, ankle plantar- and dorsi-flexion range of motion and increased hip extensor, abductor, adductor, and ankle plantar flexor muscle activity.
Discussion:
The SWS improved more markedly during the HWAT intervention, exceeding the minimal clinically important difference (0.10 to 0.15 m/s) in walking speed for people with SCI until discharge. Moreover, the 6MWT results at 2 weeks after the start of HWAT exceeded the cutoff value (472.5 m) for community ambulation and remained at a similar value at discharge.
Conclusion:
The walking distance (6MWT) and the walking speed (SWS) both demonstrated clinically important improvements following 20 treatment sessions which included HWAT.
Introduction
The incidence of non-traumatic spinal cord injuries (NTSCI) is reported to be higher than that of traumatic SCI (TSCI); the former are expected to further increase as the population ages. 1 Therefore, patients with NTSCI require significant clinical attention, especially those who need to regain their walking ability in order to perform activities of daily living independently. Previous studies showed promising results of robot-assisted gait training, a form of walking training for persons with SCI currently subject to clinical trials, as a therapeutic intervention that improves the walking ability to a meaningful degree. 2 -6 One such device, the Lokomat® (Hocoma, Volketswil, Switzerland), has been used for patients with severe (American Spinal Injury Association Impairment Scale [AIS] grade A or B) to relatively mild (AIS grade C or D) SCI. 7 Using such a device makes it possible to perform repeated walking training by assisting the lower limbs; moreover, depending on the device, additional weight-bearing training can be undertaken as needed. However, according to a recent meta-analysis, there is currently no firm evidence that the effects of robot-assisted gait training, including Lokomat, are superior to ground walking training or other forms of physiotherapy. 5 Since individuals with SCI who have higher lower limb function (such as AIS grade D) can walk with lighter assistance or supervision, 8 such individuals spend most of the physiotherapy session performing gait training. 9 It remains questionable whether it is necessary to use a device such as the Lokomat, which provides enough passive assistance to perform walking training even in patients with complete or motor-complete SCI (AIS grades A–B) and high lower limb function (capable of performing walking training with lighter assistance or supervision). When rehabilitating patients with central nervous system diseases, the voluntary effort of the patient to move is crucial for recovery 10,11 ; physical assistance and support by the robot should therefore be adjusted as needed to ensure that patients do not lack the opportunity to exert voluntary efforts. 12 -14
The Honda Walking Assist Device® (HWA; Honda Motor Co., Ltd., Tokyo, Japan) is a wearable robotic walking training device. The hip joint actuator assists the left-right symmetry and the movement of the hip joint during walking by supporting the alternating movements of hip flexion and extension. Angle and torque sensors built into the hip actuator monitor the hip angle and assist torque production. The hip joint assistive torque exerted by the actuator is calculated in real time by an algorithm that adjusts to approximate normal walking. The simple structure and light weight of the HWA make it easy to be handled by a single physiotherapist.
Although walking training with the HWA (HWAT) has previously been shown to improve gait-related parameters in clinical reports and trials in patients with stroke, 15 -17 amputated lower extremities, 18 and total hip arthroplasty, 19 there are no reports on patients with SCI. The same physiological factors and rehabilitation strategies as those of stroke patients are considered to be present in the process of recovering gait function in patients with SCI. 13 With reference to the interventions of these previous studies, this study aimed to evaluate the changes in walking function, lower limb function, gait kinematics, and muscle activity during walking in a patient with SCI before and after HWAT.
Patients and Methods
A 63-year-old male retired police officer (height: 163 cm; weight: 76.0 kg) who was diagnosed with ossification of the yellow ligament in the thoracolumbar spine developed lower limb weakness and difficulty in walking, and was urgently admitted to an acute care hospital. Magnetic resonance imaging revealed spinal cord compression due to ossification of the yellow ligament at the T2 level; a T2 laminectomy was performed 56 days after admission. After surgery, the patient underwent rehabilitation and started walking using a wheeled walker. He was transferred to our hospital 124 days after admission to continue rehabilitation. A complete clinical assessment was performed (Table 1). His chief complaint was the inability to walk. The patient started physiotherapy and occupational therapy on the day after being transferred to our hospital. Physiotherapy was conducted for 60–120 min per day, 6 days a week, and included lower limb strength training, stretching, overground walking training indoors or outdoors with a walker or cane, treadmill training with and without body weight support, and stair climbing training. The training program was implemented by selecting and combining different exercises as needed. Occupational therapy was conducted for 40–120 min per day, 6 days a week, and included upper limb ergometer training in a standing position, shoulder stretching, indoors or outdoors walking, and stair climbing practice. These physiotherapy and occupational therapy sessions were implemented according to the patient’s fatigue status. The patient was highly motivated and was allowed to start walking using a caster walker as self-training in the ward 139 days after the first admission. Two-hundred- and 1-days post-SCI, the patient walked 1.2 km outdoors slowly with a cane in each hand under close therapist supervision. However, the patient wished to start HWAT to further improve his walking ability.
Assessment From Admission to Discharge.

This study was conducted in accordance with the World Medical Association’s Declaration of Helsinki and was approved by the Ethics Committee of the Ibaraki Prefectural University of Health Sciences (approval number: e192). Written informed consent was obtained from the patient for publication and the use of accompanying images in this case study.
We referred to interventions from 2 previous randomized controlled trials that examined the effects of HWAT on patients with stroke. One trial performed 10 sessions of 20 minutes HWAT and showed an improvement in HWAT walking speed. 15 In the other trial, 18 sessions of 30-minute HWAT were conducted, and improvements in walking speed and walking duration were shown after HWAT. 17 Accordingly, our patient underwent each HWAT session for a maximum net walking time of 20 minutes excluding rest time, once a day, 5 times a week for 4 weeks (20 sessions in total). An intermediate outcome measurement was performed after 10 sessions. The HWAT is illustrated in Figure 1. The torque assistance was adjusted by the physiotherapist at rest at the start of each session based on patient complaints and visual motion analysis to ensure that walking was as comfortable as possible. The patient underwent HWAT during physiotherapy using a cane as a replacement to the walking training that was previously performed during this period. Occupational therapy was conducted as described above.
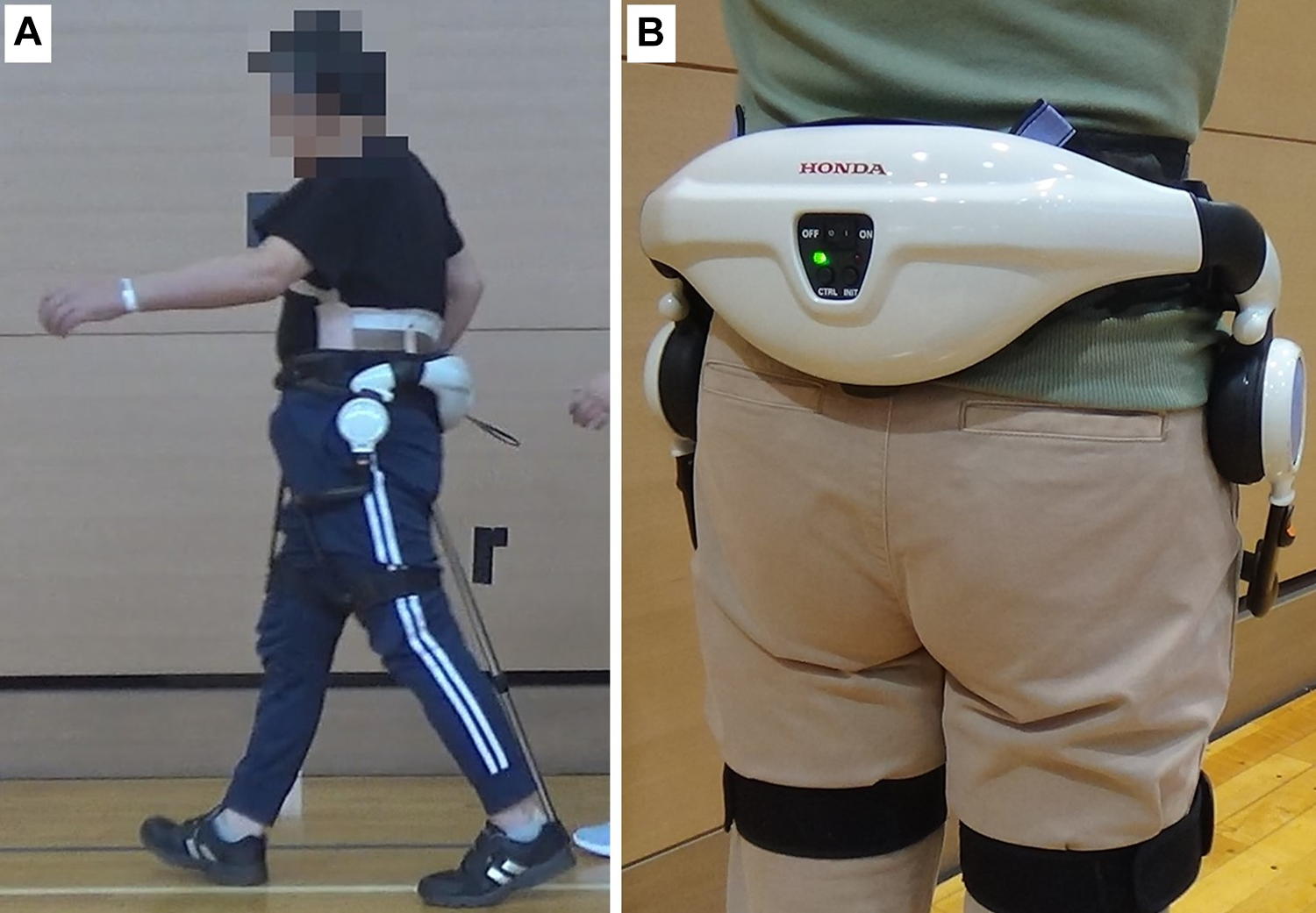
(a) Training with Honda Walking Assist Device® and (b) the Honda Walking Assist Device®.
Self-selected walking speed (SWS); mean step length at SWS; cadence at SWS; 6-minute walking test (6MWT); Walking Index for Spinal Cord Injury (WISCI-II) score; SCI Functional Ambulation Inventory (SCI-FAI) gait score for observational gait assessment; AIS grade; neurological level; upper and lower extremity motor scores; modified Ashworth Scale for passive resistance due to spasticity; Penn Spasm Frequency Scale for self-assessed muscle spasm frequency and severity; and activities of daily living mobility subscale of the Spinal Cord Independence Measure version III (SCIM-III) score for mobility in the room, toilet, indoors, and outdoors were all assessed on admission, at discharge, at the start of HWAT, and at 2 and 4 weeks after the start of HWAT (after 10 and 20 sessions of HWAT) (Table 1). Adverse events such as pain and skin problems (redness or scarring due to contact with the equipment) during HWAT were carefully observed during each session. For SWS testing, the time required to cover the intermediate 10 m of a total distance of 16 m was measured; the patient was allowed 3 m for acceleration and deceleration. Parameters were measured twice and the faster time was used. To calculate step length and cadence at SWS, the number of steps during SWS testing was counted.
To analyze joint angles and muscle activity during gait at SWS, gait analysis was performed using 3-dimensional kinematic gait analysis and electromyography (EMG) at the start of HWAT and 4 weeks after. Lower limb kinematic data were recorded using 8 infrared cameras at a sampling rate of 100 Hz (Vicon Motion Systems; Oxford Metrics, Oxford, UK). A widely used conventional marker set model (Plug-In Gait Model; Oxford Metrics, Oxford, UK) was used to calculate the joint angles after segment definition. Sagittal joint angles were calculated for the hip, knee, and ankle. Kinematic data of the left leg (weaker side) were measured and analyzed.
EMG sensors were placed on the left gluteus maximus, gluteus medius, tensor fascia lata, adductor longus (ADD), rectus femoris, vastus medialis, vastus lateralis, biceps femoris, semitendinosus, tibialis anterior, medial head of the gastrocnemius (MG), and soleus. The appropriate positions for electrode placement on each muscle were determined according to the Surface EMG for the Non-Invasive Assessment of Muscles project. 20 Only the position of the ADD electrode was adapted; the electrode was placed along the muscle at the proximal third of the distance between the pubic symphysis and the adductor tubercle. 21 The EMG activity was recorded using a wireless surface electrode with a sampling rate of 2000 Hz and bandpass filtering of 20–450 Hz (Trigno Lab; Delsys Inc., Boston, MA). EMG data recorded during gait were rectified and the root mean square value of 500-ms windows was calculated and expressed as a percentage of maximum voluntary contraction (MVC). MVC was obtained from the root mean square value of a window containing the 1.5 seconds before and after the peak of muscle activity (3 seconds in total) during a 5-second contraction for each muscle. MVC measurements were performed by experienced therapists using an isometric contraction task based on manual muscle testing after practicing the task with the patient. A walking cycle was defined from a left heel contact to the next left heel contact. Trimming was performed for 10 walking cycles and the time of 1 walking cycle (from 0 to 100 points) was standardized using cubic spline interpolation; the data of the standardized 10 walking cycles were averaged and EMG data were processed using MATLAB R2018a (MathWorks Inc., Natick, MA).
Results
The patient completed 20 sessions of gait training; no adverse events occurred during HWAT. SWS showed an improvement of +0.31 m/s during HWAT (Table 1). Additionally, the SWS improvement was greater during the 4-week HWAT intervention than during the period from admission to the start of HWAT (Figure 2). The improvement in SWS during the HWAT intervention was greater in the first (+0.20 m/s) than in the last 2 weeks (+0.10 m/s) (Figure 2). A slight decrease in SWS was observed 2 weeks after the completion of HWAT (at the time of discharge); however, the SWS remained higher than at the start of HWAT (+0.22 m/s). Cadence increased during the HWAT intervention in a similar manner to SWS. Step length increased during the first 2 weeks of the HWAT intervention and was stable during the last 2 weeks. The distance achieved in the 6MWT increased during the first 2 weeks of the HWAT intervention (+66.1 m) and remained higher at the completion of the HWAT intervention than at the start (+37.9 m); this distance increased again at discharge (+58.1 m). WISCI-II remained unchanged throughout the HWAT period.

Changes in self-selected walking speed, step length, and cadence.
The upper extremity motor score increased by 1 point in the right C8 myotome (finger flexors) during the HWAT period. The right lower extremity motor score was increased by 2 points, 1 point each for the L2 (hip flexors) and S1 myotomes (ankle plantar flexors). The left lower extremity motor score increased by 2 points in total, 1 point each for the L5 (long toe extensors) and S1 (ankle plantar flexors) myotomes during the HWAT period. No significant changes in AIS, motor or sensory neurological level, modified Ashworth Scale, and Penn Spasm Frequency Scale were observed during the HWAT intervention. During HWAT, an improvement of 10° was observed in the passive hip extension range of motion on the right side. At discharge, this improvement had reached 15° on both sides.
The mobility subscale on the SCIM-III increased by 2 points during the HWAT period. The specific area in which an improvement was observed was the transfer between the wheelchair and floor. This improvement meant that help was no longer needed when the patient performed transfers between the floor and wheelchair. At discharge, the SCIM-III score showed a 2-point improvement in outdoor walking. This improvement meant that walking over 100 m with a cane outdoors no longer required supervision.
Changes in the average lower limb joint angles during the 10 walking cycles at SWS from the start to the end of HWAT are illustrated in Figure 3. In the hip joint, a large difference was observed between the pre- and post-HWAT angles at the time of maximum extension, which occurs near the moment of transition from the terminal stance phase to the swing phase (at about 50% of the gait cycle), and at the maximum flexion in the mid-swing phase (at about 90% of the gait cycle). At the knee joint, 5–10° of flexion were maintained throughout the terminal stance phase (at 30 to 50% of the gait cycle) after the HWAT period, and an increase was observed in the maximum flexion angle in the mid-swing phase (at about 75% of the gait cycle). In the ankle joint, there was a greater dorsiflexion angle from the initial heel contact to the end of the terminal stance (0 to 50% of the gait cycle) following the HWAT period. In particular, at the end of the stance phase (at about 50% of the gait cycle), the maximum dorsiflexion angle increased by approximately 10°. Furthermore, after HWAT, the ankle plantarflexion angle increased at the start of the swing phase (at about 65% of the gait cycle). Moreover, there was a greater dorsiflexion angle during load preparation in the terminal swing phase (at about 90 to 100% of the gait cycle).

Left lower limb kinematic data at the self-selected walking speed. These graphs show the averaged angle for 10 cycles (solid line), the standard deviation (light-color-filled area), and the averaged muscle activity as %MVC during the 10 walking cycles. Red line: start of HWAT; blue line: end of HWAT (4 weeks).
Changes in EMG activity (%MVC) are shown in Figure 4. The gluteus maximus, gluteus medius, ADD, MG, and soleus showed increased activity during the loading response phase, the first half of the subsequent mid-stance phase (about 0 to 20% of the gait cycle), and close to the end of the terminal swing phase (about 90 to 100% of the gait cycle). The rectus femoris, vastus lateralis, biceps femoris, and semitendinosus showed decreased muscle activity during the same phase.

Muscle activity (%MVC) during the self-selected walking speed at the commencement and completion of HWAT. These graphs show the averaged %MVC of 10 walking cycles from a left heel contact to the next left heel contact. Gmax, gluteus maximus; Gmed, gluteus medius; TFL, tensor fascia lata; ADD, hip adductor; RF, rectus femoris; VM, vastus medialis; VL, vastus lateralis; BF, biceps femoris; ST, semitendinosus; TA, tibialis anterior; MG, medial head of the gastrocnemius; SOL, soleus.
Discussion
The SWS improved more markedly after 10 and 20 sessions after the HWAT intervention, exceeding the minimal clinically important difference (0.10 to 0.15 m/s) in walking speed for people with SCI 22 until discharge. Moreover, the 6MWT results at 2 weeks after the start of HWAT exceeded the cutoff value (472.5 m) for community ambulation 23 and remained at a similar value at discharge. For this patient, this improvement can be significant in promoting the patient’s community activities.
The post-inpatient rehabilitation SWS and 6MWT outcomes in patients with NTSCI have been reported to be similar to those in patients with TSCI 24 ; walking speed has also been associated with the performance of activities of daily living, as evaluated by the SCIM-III score. 25 Walking speed in individuals with SCI was almost unchanged or slightly reduced between 6 and 12 months after injury, according to results of recent studies. 26 -28 A similar pattern has been found for the 6MWT, lower extremity motor score, and WISCI-II. 26 The patient in our study started HWAT more than 7 months after the NTSCI, which indicates that the time for significant recovery was approaching its limit. Nevertheless, the improvement in SWS (evidenced by an increased slope) during HWAT was observed. The results of a randomized controlled trial by Buesing et al. about an HWAT intervention in patients with stroke demonstrated the potential of HWAT to provide a better or equivalent improvement in gait function than conventional high-intensity physiotherapy. 16 One of the most important benefits of applying robots in rehabilitation is the provision of intensive training 12 ; however, rigorous clinical trials are needed to confirm whether the robotic training performed in this study leads to better outcomes than standard rehabilitation for people with SCI.
All muscle activity patterns indicated that there were no major changes before or after HWAT. Furthermore, with the exception of the increased activity of the MG and soleus during the early stance phase, there were no major deviations from the normal gait pattern before the implementation of HWAT. 29 The lack of changes in the gait pattern before and after HWAT, as evaluated by EMG activity, is consistent with the lack of changes in WISCI-II and SCI-FAI scores, which are indicators related to the gait pattern. As for the changes in %MVC in each muscle, the results indicate that the increased activity of the gluteus maximus, gluteus medius, ADD, MG, and soleus during the early stance phase may generally be associated with increased gait speed (i.e., increased gait propulsion). 29 These increases in muscle activity and gait speed were consistent with increases in hip extension angle and ankle dorsiflexion angle in the late stance phase. 29
Although the gait pattern was already acceptable before HWAT, the results showed an increase in gait speed with a further increase in the angle of hip extension after HWAT. Similar improvements have been reported in previous studies using HWAT for post-stroke 15 -17 and total hip arthroplasty 18 rehabilitation, and the authors note that symmetrical hip flexion and extension assistance during HWAT may have improved gait speed. A faster walking speed with greater hip extension is thought to promote gait improvements in patients with SCI. 13 It is considered that this case shows similar results to those of previous studies.
This study has some limitations. First, this was a single-case study with no controls, which means the results cannot be used to draw generalized conclusions about the effectiveness of the methods used considering the study design. Moreover, the patient was only followed up for about 2 weeks after HWAT. Based on the results of this case report, it is necessary to examine the feasibility, safety, and effects of HWAT in a larger sample and to perform future comparative studies. The severity of the case, timing of intervention, and the training protocol (dose, frequency, and duration of the session) should also be considered in future studies.
Conclusion
The 6MWT and SWS both demonstrated clinically important improvements following 20 treatment sessions which included HWAT.
Footnotes
Abbreviations
Acknowledgments
We would like to thank all staff of the Physical Therapy Department at Ibaraki Prefectural University of Health Sciences Hospital, represented by Kazuhide Tomita, the manager of Department of Rehabilitation, Tomoyuki Matsuda, the manager of the Department of Physical Therapy, and Keiko Miyamoto, the robotic technical assistant of the Department of Physical Therapy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Honda Motor Corporation lent us the HWA free of charge for this research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Project Research (1655) from the Ibaraki Prefectural University of Health Sciences.
