Abstract
Background
Epidural spinal cord stimulation (EES) is a promising intervention for motor rehabilitation after spinal cord injury (SCI), but the extent and trajectory of motor recovery remain unclear.
Objective
This phase II trial evaluates the acquisition of voluntary movements in paraplegic patients (ASIA A or B) following SCI, assessed by the Fugl-Meyer Lower Extremity (FMA-LE) score and electromyography (EMG).
Methods
This interim analysis includes five patients implanted with EES and followed for 12 months. The primary outcome was motor recovery, measured by FMA-LE and EMG. Secondary outcomes included balance (Berg Balance Scale), spasticity (Modified Ashworth Scale), pain, autonomic functions, mood, quality of life (WHO-QOL), and safety. Adverse events were monitored.
Results
The FMA-LE score improved from 36 ± 9 (SD) to 55 ± 2 at 3 months (P < 0.05), 59 ± 2 at 6 months (P < 0.05), and 64 ± 4 at 12 months (P < 0.05). EMG confirmed increased voluntary activation. Balance and spasticity improved, while pain and autonomic functions remained unchanged. Motor gains plateaued after 5 months, reaching 68% above baseline. No serious adverse events occurred, though minor complications included transient nociceptive pain and a self-resolving pressure ulcer.
Conclusions
These findings support the role of EES in facilitating early motor recovery in SCI patients, consistent with prior studies. However, the plateau effect suggests a limit to long-term gains. Future research should explore strategies to sustain improvements, including regenerative therapies or optimized neuromodulation protocols.
Introduction
Currently, spinal cord injury (SCI) is one of the leading causes of premature morbidity and mortality worldwide. An estimated 27 million individuals are living with paralysis due to spinal cord injuries globally, with approximately 1 million new cases reported each year (incidence rate of 0.93, 95% CI = 0.78-1.16) and age-standardized incidence rates of 13 (11-16) per 100,000 inhabitants, predominantly affecting young and economically active individuals. Furthermore, SCI accounts for 9.5 million years of life with disability (YLD), imposing a significant socioeconomic burden on modern society. 1
A critical goal for patients with paralysis resulting from spinal cord injury is the ability to maintain an upright position or walk. 2 While physical exercise and rehabilitation therapies currently represent the standard treatment, most approaches fall short of achieving these objectives. Recent research has focused on developing technologies that promote orthostasis and ambulation by reorganizing neural motor pathways. Examples include exoskeleton for lower limbs under central control (brain-machine interfaces), 3 cellular or bio-matrix transplants,4,5 and spinal cord neuromodulation therapies. Furthermore, there is renewed hope with the advent of epidural electrical stimulation (EES) for gait recovery.6,7
Neuromodulation, defined as changes in neural activity induced by a directed external stimulus, has proven beneficial in various areas of neuroscience, including deep brain stimulation for Parkinson’s disease and epilepsy. 8 Technological advancements have facilitated the development of diverse neuromodulation techniques, with electrical stimulation demonstrating efficacy in treating multiple conditions. 9 Pioneering human studies by Harkema et al, 6 Angeli et al, 7 and Wagner et al. 10 Have provided compelling proof-of-concept, demonstrating that EES can reactivate dormant spinal circuits and facilitate voluntary movement, standing, and even over-ground walking in individuals with motor-complete paralysis. Clinical studies have also indicated that optimizing stimulation parameters on standard neuromodulation devices can lead to significant motor improvements in patients with spinal cord injuries. 11
While these seminal works have established the feasibility of EES, a comprehensive understanding of the time course and maximum achievable gains and their temporal stabilization (plateau effect) of motor recovery with EES is crucial for optimizing rehabilitation protocols and managing patient expectations. Previous studies have largely focused on demonstrating the possibility of recovery, 12 but a quantitative framework for predicting the trajectory and limitations of functional gains has been lacking.
This study addresses these critical gaps by investigating the efficacy of a standard, commercially available EES system (Medtronic Specify 5/6/5 electrode, Activa RC/Intellis generator) in promoting motor recovery in severely paralyzed patients (ASIA A and B) after chronic SCI. Crucially, we employ mathematical modeling (Gompertz 3-parameter model) to precisely characterize the temporal trajectory of motor improvement and identify the point at which functional gains tend to stabilize, or plateau. By providing an interim analysis of a phase I/II clinical trial, this study aims to quantify the extent of motor recovery, characterize the maximal potential of these gains, and identify predictive factors of response. Our findings contribute directly to the evidence base necessary for future regulatory considerations and the broader clinical translation of this promising therapy, particularly in contexts where access to highly specialized devices may be limited.
Material and Methods
Study Design and Ethics
This is a phase II early exploratory prospective trial with a within-subject design conducted at Hospital das Clínicas, Medical School, University of São Paulo, Brazil. The study received approval from the local Ethics Committee (no. 30702520.6.0000.0068) and is registered on clinicaltrials.gov under the number NCT06847295. The study was initiated on January 28, 2022, with an anticipated completion date of January 10, 2027. We plan to enroll 10 patients in total; to date, five patients have been implanted and are reported here. The study is investigator-initiated, and is being sponsored by Medtronic (Minneapolis, Minnesota, USA), which provided the devices to our public institution but did not participate in data collection or analysis. All study participants have been informed about the aims of the study and the potential adverse effects, and all provided written informed consent prior to enrollment.
The primary objective is to assess the acquisition of voluntary movements measured by the Fugl-Meyer Lower Extremity 13 score and electromyography (EMG), in patients with complete motor impairment (ASIA A or B) following spinal cord injury. Fugl-Meyer is a reliable and widely recognized tool for evaluating motor recovery, particularly in the context of stroke rehabilitation, but it has also been adapted for use with SCI patients. The scale assesses motor function in various domains, including lower limb movements, balance, sensation, and joint function. Although not designed specifically for spinal cord injuries, it is a useful instrument in measuring recovery in SCI patients, especially in those with incomplete injuries or those undergoing rehabilitation.14,15
Secondary objectives include evaluating changes in balance, movement kinematics, spasticity, pain, vegetative functions (bowel and bladder), mood, quality of life, and safety. Safety assessments were systematically conducted to monitor potential adverse events, including infection, exacerbation of pain (measured by pain scores), pressure ulcers, and other device-related complications.
We conducted a power analysis in JMP to verify the accomplishment of the primary goal (exact Agresti-Coull method, two-sided, null proportion = 0, alpha = 0.05). The analysis resulted in a power of 85% to detect 3 or more responsive patients in a sample size of 10 participants (considering any functional gain in EMG).
Inclusion Criteria
• Adult patients, male and female, aged between 18 and 50 years • Stable spinal cord injury, at least 6 months post-injury • ASIA A or B score, with inability to stand or walk • Injury location between T1 and T10 • Intact segmental reflexes below the injury level • Signed informed consent and the ability to understand its content • Good and stable clinical health conditions
Exclusion Criteria
• Current use of electronic devices such as a baclofen pump, cardiac pacemaker, etc. • Presence of significant comorbidities that may increase the risk of morbidity/mortality from device implantation surgery (eg, cancer, chronic infection, coagulopathy) • MRI demonstrating complete spinal cord transection • Clinically significant mental illness • Voluntary movements present during EMG testing in bilateral lower limbs • Presence of pressure ulcers • Severe depression (Beck Depression Index >19)
Surgical Procedure
Patients were positioned in the prone position, and the implantation level was marked using X-ray fluoroscopy. Subdermal and intramuscular needles were inserted for electromyographic recordings in the following muscles: iliopsoas, vastus lateralis, rectus femoris, anterior tibialis, semitendinosus, gastrocnemius, soleus, long extensor of the hallux, biceps femoris, and gluteus maximus. Following this, standard degermation, asepsis, and antisepsis protocols were performed. A skin incision of approximately 5 cm was made between the L1 and L2 vertebrae, and dissection of the subcutaneous tissue and paravertebral muscles was conducted in preparation for interlaminar fenestration at the L1/2 level. The electrode lead (Medtronic Specify 5/6/5) was initially placed at the D12 level under fluoroscopy. Sufficient time was allocated to electrophysiologically verify the functional level of the implant and its symmetry, based on the evoked EMG activity in the target muscles. Repositioning of the electrode was performed as needed. Finally, the internal pulse generator (IPG; Medtronic Intellis) was implanted in the subcutaneous space below the iliac crest on the back. Patients were discharged after 3 to 5 days, and daily epidural stimulation sessions commenced on the 15th postoperative day.
Rehabilitation Program under Epidural Electrical Stimulation
During the early stages of the rehabilitation program, one physician (MP) and one biomedical technician (LA) configure the epidural stimulation device with motor-specific routines, including left and right hip flexion/extension, left and right knee flexion/extension, left and right foot dorsiflexion and plantarflexion, standing, treadmill walking, cycling exercises, and bladder/bowel management. Given the dynamic nature of impedance changes over time, these settings are continuously adjusted, including electrode selection, frequency, amplitude and intensity, on a daily basis. These daily adjustments are guided by real-time electromyography (EMG) feedback, patient perception of motor responses, and observed motor performance during rehabilitation sessions, ensuring optimal and personalized stimulation.
In the initial phase, isolated muscle groups are strengthened through targeted recruitment, which refers to precisely adjusting EES parameters to activate specific muscles (eg, quadriceps, hamstrings, tibialis anterior) to facilitate isolated movements (eg, hip flexion/extension, knee flexion/extension, dorsiflexion/plantarflexion) using increasing weights. This approach aims to restore and strengthen connections for individual muscle control before progressing to more complex movements. The use of increasing weights reflects the application of the progressive overload principle, essential for motor learning and muscle strengthening. This phased approach, from isolated to multi-segmental training, is a cornerstone of effective neurorehabilitation.
In subsequent phases, more complex, multi-segmental movements are introduced, such as weight-bearing activities like standing with parallel bars (with sustained lower limb extension), treadmill walking, and exercises on an ergometer bicycle. Participants receive comprehensive instructions for their home-based stimulation program and are asked to comply with 2 to 4 h of epidural stimulation per day. During each follow-up visit, the spinal cord stimulation (SCS) settings are personalized, with up to 12 individualized configurations. Additionally, participants are provided with a paper calendar outlining the prescribed settings for one hour of training, five times per week, at our facility.
Clinical Assessments
Patients were assessed at baseline and every 3 months, with a follow-up period of 12 months following the implantation of the epidural electrode, using the scales described below. • Fugl-Meyer Assessment Lower Extremity (FMA-LE)
13
: A comprehensive evaluation of motor function, balance, sensation, and joint function specifically for the lower extremities following neurological impairments. • Berg Balance Scale (BBS)
16
: A 14-item scale that measures balance in patients through various functional tasks, providing a reliable assessment of fall risk. • NINDS Scale for Tendon Reflex Assessment
17
: A standardized scale to assess deep tendon reflexes, providing insights into the neurological function of patients. • Modified Ashworth Scale
18
: A clinical scale used to measure spasticity by evaluating the resistance during passive movement of a limb. • Brief Pain Inventory (BPI)
19
: A questionnaire designed to evaluate pain severity and its impact on daily functioning. It helps in assessing both the intensity of pain and the extent to which pain interferes with activities. • Pain Disability Index (PDI)
20
: A scale that measures the impact of pain on daily activities and overall functioning, consisting of 7 items related to different aspects of life. • DN4 Questionnaire for Neuropathic Pain
21
: A brief questionnaire consisting of 10 items to aid in diagnosing neuropathic pain based on symptoms and physical examination findings. • Beck Depression Index (BDI)
22
: A self-report inventory that measures the presence and severity of depressive symptoms. It consists of 21 multiple-choice items. • World Health Organization Quality-of-Life Questionnaire (WHOQOL-BREF)
23
: A 26-item questionnaire that assesses quality of life across 4 domains: physical health, psychological health, social relationships, and environment. • Neurogenic Bladder Symptom Score (NBSS)
24
: A scale that assesses the severity of urinary symptoms in patients with neurogenic bladder dysfunction. It includes questions related to frequency, urgency, and incontinence. • Neurogenic Bowel Dysfunction Score (NBD)
25
: A tool for evaluating the severity of bowel dysfunction in patients with neurogenic conditions, focusing on symptoms such as constipation and incontinence. • Braden Scale for Predicting Pressure Ulcer Risk
26
: An assessment tool that evaluates the risk of developing pressure ulcers based on 6 criteria: sensory perception, moisture, activity, mobility, nutrition, and friction/shear.
Instrumentalized Diagnosis
Surface EMG was obtained before surgery and every 3 months throughout the study observation period, to assess brain motor control. To this end, we used an 8-channel system (Trigno Avanti Research +, Delsys, Natick, MA, USA). Sensors were configured with a sampling rate of 2 kHz and without initial filtering. Muscle signals from the iliopsoas, vastus lateralis, rectus femoris, anterior tibialis, semitendinosus, gastrocnemius, soleus, long extensor of the hallux, biceps femoris, and gluteus maximus muscles were acquired. Integrated accelerometers and gyroscopes within the surface electrodes were used to capture kinematic data (acceleration and angular velocity) in conjunction with muscle activity, allowing for correlation between muscle activation and movement patterns.
The signals were pre-processed with a 30-500 Hz band-pass filter and digitized at 2 kHz. Signals were processed offline in Matlab (Matlab R_2019a). Next, we applied Fast Fourier Transform (FFT), Wavelet Transform, and Root Mean Square (RMS) calculation, with the objective of extracting relevant information from the signals. The Fast Fourier Transform (FFT) was used to extract spectral components of the signal and identify dominant frequencies associated with muscle activity. The Wavelet Transform allowed for time-frequency analysis, enabling the detection of transient patterns in the signal. The Root Mean Square (RMS) was calculated as a measure of the average magnitude of the signal in the time domain, frequently used as an index of muscle activation.
Statistical Analysis
Statistical analyses were performed in JMP v17.0 software (SAS Institute, Cary, NC, USA). Each patient’s scores were plotted over a 12-month follow-up period. Linear regression was initially applied to the clinical scores to categorize patients as either showing clinical improvement (indicated by a positive or negative slope of the regression line, depending on the clinical score) or having a stable condition (indicated by a horizontal line within the confidence interval). Clinical assessments are represented as points on scatter plots, accompanied by the corresponding regression line and 95% confidence interval. The F-ratio was reported to assess the significance of the overall model, where the F-statistic is the ratio of the mean sum of squares for regression to the mean sum of squares for error (residuals). A higher F-ratio indicates a better model fit for explaining data variability. Additionally, analysis of variance (ANOVA) was used to compute the probability of P>F.
We then focused on motor scores using the Fugl-Meyer scale for lower limbs to predict motor improvement over time. A generalized linear model was applied to examine the evolution of motor scores. Based on a visual inspection of data dispersion, various models were tested, including Gompertz 3-parameter (3P), Logistic 3P, Probit 3P, Logistic 4P, Gompertz 4P, Probit 4P, and Weibull Growth. The following parameters were used to compare model performance: • AICc (Akaike Information Criterion, corrected for small sample sizes)
27
a measure of model fit, with the lowest AICc indicating the best-fitting model. • AICc weight: the probability that a particular model is the best among those compared, with a weight closest to one indicating the best fit. • BIC (Bayesian Information Criterion): a likelihood-based measure of model fit, with lower BIC values indicating a better fit. • SSE (Sum of Squared Errors): a measure of the total deviation of predicted values from the actual values. • MSE (Mean Squared Error): the average of squared errors, assessing the model’s prediction accuracy. • RMSE (Root Mean Squared Error): the square root of MSE, estimating the standard deviation of random errors. • R-squared: the proportion of variance in the response variable explained by the model; higher values closer to one indicate a better fit.
Finally, electromyographic (EMG) and kinematic data were obtained directly from the recording software and processed offline in MATLAB. Five-minute epochs were analyzed in both “ON” and “OFF” conditions during cycling exercises, with means and standard deviations calculated for each epoch. These were compared using Student’s t-tests, assuming normal distribution of the data.
Results
Patients Demographics
Patient’s Demographics and Clinical Characteristics

Clinical Assessments
Overall, the five patients in this study demonstrated improvements in motor scores (Fugl-Meyer Assessment, F ratio = 25.9, P < 0.0001), spasticity (Modified Ashworth Scale, F ratio = 5.2, P < 0.05), and balance (Berg Balance Scale, F ratio = 18.6, P < 0.001) (see Figure 1). The most significant recovery was observed in motor scores, where the mean baseline FMA was 36 ± 9 (SD), increasing to 55 ± 2 after 3 months of training (P < 0.05, Wilcoxon), 59 ± 2 at 6 months (P < 0.05), 60 ± 4 at 9 months (P < 0.05), and 64 ± 4 at 12 months (P < 0.05). This represented a 78% improvement relative to baseline values. Clinical Progression of Five Patients Over a 12-Month Follow-Up Period Circles Represent the Clinical Measurements. Linear Regression was Applied to Classify Patients as Either Improving (Indicated by a Positive or Negative Slope, Depending on the Clinical Score) or Stable (Indicated by a Horizontal Line Within the Confidence Interval). The First Three Scores Indicate Clinical Improvements in the Fugl-Meyer Motor Score for the Lower Limbs; A, Berg Balance Scale; B, and Ashworth Spasticity Scale; C. All Three Models Were Statistically Significant: Fugl-Meyer, F Ratio = 25.9, P < 0.0001; Berg, F Ratio = 18.6, P < 0.001; and Ashworth, F Ratio = 5.2, P < 0.05. For the Measurements Shown in Red, No Statistical Significance was Achieved, Indicating that the Model was No Better than a Random Distribution (Horizontal Line). A Trend Towards Improvement was Observed for the WHO Quality of Life (WHO-QoL) Score (D) and Beck Depression Scale (E), although Statistical Significance was Not Reached. This May Change With an Increase in Sample Size, as Variance is Expected to Decrease. Clinical Stability was Observed in Deep Tendon Reflexes (NINDS, F), Pain (Pain Disability Index, G; Brief Pain Inventory, H; and Neuropathic Pain Questionnaire DN4, I), Pressure Ulcer Risk (Braden Scale, J), and Autonomic Functions (Neurogenic Bladder Symptom Score, K; and Neurogenic Bowel Dysfunction Score, L)
Regarding mood, as measured by the Beck Depression Inventory, and quality of life, as assessed by the WHO Quality of Life (WHO-QoL) questionnaire, a trend toward improvement was observed, though neither reached statistical significance. We anticipate that these measures may achieve significance with an increased sample size as the trial continues, given the generally positive feedback from participants. Notably, baseline Beck Depression scores were below the threshold for moderate or severe depression (≥19), which served as an exclusion criterion in this study.
Scores for deep tendon reflexes (NINDS), pain (Brief Pain Inventory, Pain Disability Index, and Neuropathic Pain Questionnaire DN4), pressure ulcer risk (Braden Scale), and autonomic functions (Neurogenic Bowel Dysfunction Score and Neurogenic Bladder Symptom Score) remained stable throughout the study. For all these measures, the ANOVA for the full model did not reach significance (P > 0.05).
Another notable finding was the patient’s ability to voluntarily move their legs even when the stimulation was turned off, suggesting the induction of plasticity. All patients were able to achieve at least some degree of voluntary movement without stimulation. This phenomenon was observed between the 4th and 8th months of follow-up. In the illustrative case presented in Figure 2, electromyography (EMG) activity during stationary cycling with stimulation (ON, left panels) is compared to activity without stimulation (OFF, right panels). Unprocessed EMG signals showed lower amplitudes in the quadriceps muscles (B and D) in the OFF condition, significantly reduced burst activity (F), and slower acceleration and angular velocity cycles, as indicated by longer cycle periods (H and J). Quantification of these kinematic parameters is shown in Figure 3, where performance was surprisingly similar across some axes, though it was associated with significantly reduced muscle power in the OFF condition. Specifically, EMG amplitudes were 62% lower (C), and power spectral density was reduced by 60% (D). Kinematic Analysis During Stationary Cycling Graphs on the Left Display Measurements During the on Condition, while Those on the Right Show the OFF Condition. All Graphs Show the Same 30-Second Exercise Period. Panels a and B Present the Raw EMG Signals From the Vastus Lateralis Muscle Over 30 s (Amplitude in Volts). In Panels C and D, the EMG Signals Have Been Processed Using the Root Mean Square (RMS) Method. Panels E and F Illustrate EMG Burst Activity, where Bursts Were Defined by a Peak Amplitude of at Least 1.0 mV and a Duration of at Least 0.4 Seconds. Panels G and H Show Acceleration Values (in g) for the Three-Dimensional Axes (X, Y, and Z). Panels I and J Display Angular Velocity (in Degrees per Second) Along the Same Three-Dimensional Axes Mean and Standard Deviation Values of the Kinematic Parameters Shown in Figure 2 Were Recorded Over a 30-Second Period at a Sampling Rate of 2 kHz and are Presented as bar Graphs for the on (Red) and OFF (Blue) Conditions. Panel a Shows Acceleration (g), Panel B Displays Angular Velocity (degrees/second), and Panel C Illustrates the Amplitude of the Unfiltered EMG Signals. In Panel D, the Power Spectral Density is Plotted for Both Conditions; Notably, the OFF Condition Demonstrates Lower Power and Higher Frequencies Compared to the on Condition

Next, we aimed to predict motor responsiveness during the follow-up period. To achieve this, we applied a generalized linear model to identify the best predictor. Several models were systematically tested (see comparison data in Figure 4D). The model that best described the motor outcomes was the Gompertz 3-parameter (3P) model, which is commonly used for time series data (see the corresponding equation in Figure 4B). Interestingly, the model predicts a rapid improvement in motor scores during the first 2 months of stimulation, with a rate of 5 points per month. However, as the model also predicts, this improvement is finite, with a plateau reached by the 5th month at a Fugl-Meyer Assessment (FMA) score of 59, representing a 68.5% improvement from baseline. Prediction of Motor Improvement Using Linear Modelling. We Employed a Generalized Linear Model to Make Inverse Predictions of Motor Improvement Based on Fugl-Meyer Scores for the Lower Limbs. First, We Tested Several Linear Functions for Their Fit to the data: Gompertz 3-Parameter (3P), Logistic 3P, Probit 3P, Logistic 4P, Gompertz 4P, Probit 4P, and Weibull Growth Models. The Fitting Curves are Shown in Color in Panel a, where Circles Represent all Clinical Assessments During the Follow-Up Period. Panel B Presents the Equation for the Best-Fitting Model, Gompertz 3P, With the Parameters: a (Asymptote), b (Growth Rate), and c (Inflection Point). This Model Allows for the Estimation of Motor Scores at Specific Time Points During Follow-Up (C)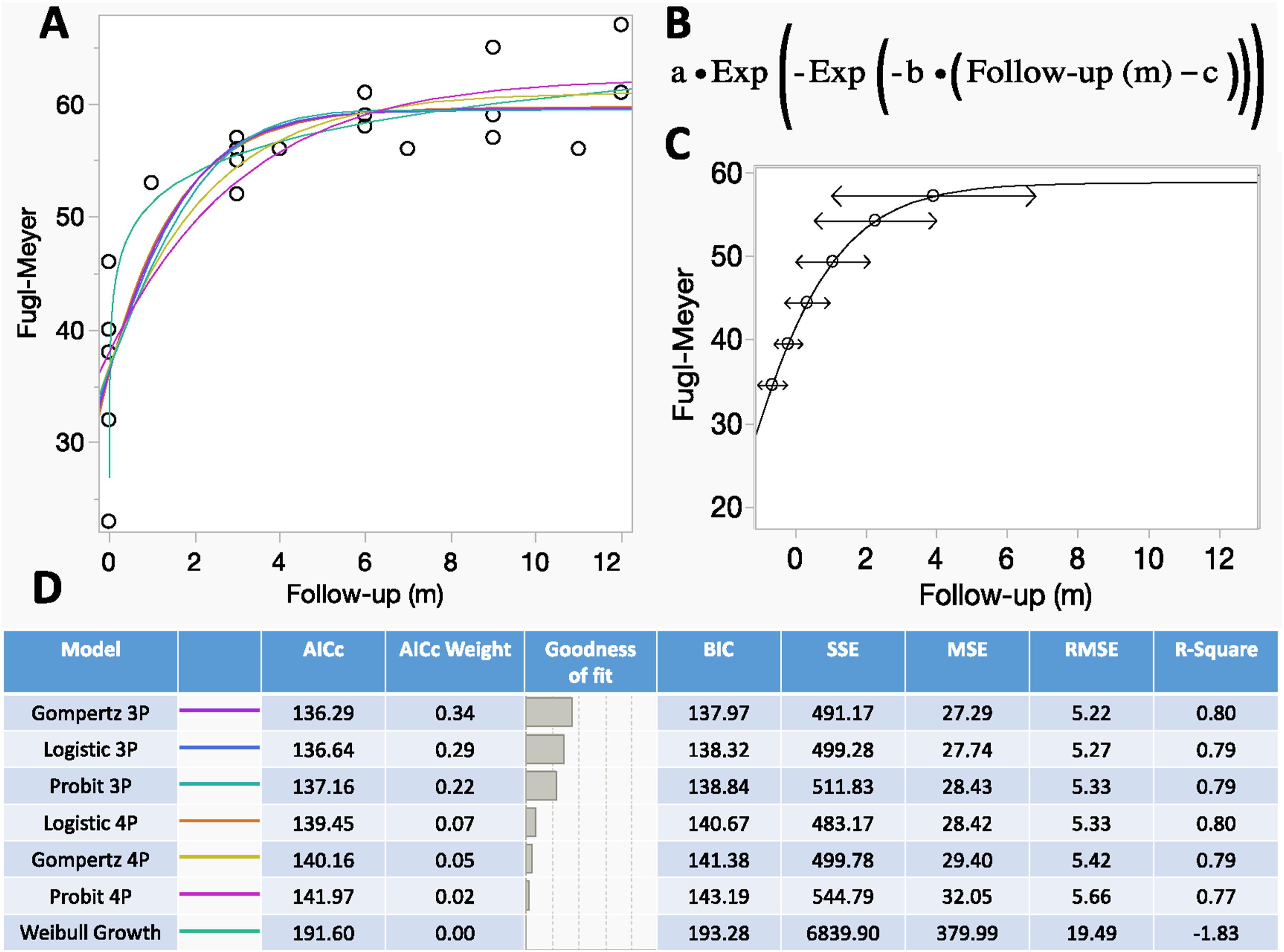
This level of improvement is sufficient to transform a patient with severe motor impairment (FMA 35)—who typically requires assistance for most activities of daily living and is dependent on a wheelchair for mobility—into a patient with moderate motor impairment. Such a patient would have partial motor control in the lower body, be capable of performing some functional movements, and be able to engage in self-care activities. Mobility would be supported by assistive devices in controlled environments (eg, at home) and for short distances.
According to the model, the predicted mean Fugl-Meyer score at the start of training was 35. The model predicts a significant improvement during the first 2 months, with an increase of 5 points per month. However, this improvement is finite, and a plateau is reached at a mean Fugl-Meyer score of 59, by the 5th month of training. Figure 4D provides a detailed comparison of the model parameters, ranked by goodness of fit (see methods for more details).
In terms of safety, no infections related to the spinal cord stimulation system were observed among the five patients. The only complications reported were transient peaks of nociceptive pain following training in 2 patients, which were managed with appropriate analgesia, and a low-grade skin injury associated with the use of weight-bearing support equipment on the treadmill. This skin injury was promptly treated and resolved without further complications. These adverse events did not affect the continuation of treatment and were managed without the need for additional surgical intervention.
Discussion
The treatment of paraplegia following spinal cord injury (SCI) has progressed substantially over the past decade, especially with the development of epidural electrical stimulation (EES), though the underlying mechanisms remain incompletely understood. This study provides an interim analysis of a phase II trial, offering quantitative insights into the time course and extent of motor recovery, the phenomenon of stimulation-induced neuroplasticity, and the safety profile of EES using a conventional device in severely paralyzed patients. Our findings contribute to a deeper understanding of EES efficacy and its clinical implications.
Key Findings and Clinical Implications
Our study makes significant contributions by providing quantitative data on motor recovery and its trajectory in severely paralyzed SCI patients undergoing EES with a conventional device. The observed improvements in FMA-LE scores were substantial, increasing from a mean baseline of 36 ± 9 (SD) to 64 ± 4 at 12 months, representing a 78% improvement relative to baseline values. The application of a Gompertz 3-parameter mathematical model revealed a rapid improvement in motor scores during the first 2 months of stimulation, at a rate of 5 points per month.
However, this improvement was finite, reaching a plateau around the fifth month. This plateau, while indicating a limit to the current intervention’s capacity, is a crucial predictive marker. It suggests that intensive rehabilitation efforts may be most beneficial within the first 6 months, followed by a maintenance program, and highlights the need to explore combined therapeutic approaches to potentially overcome this ceiling. Potential reasons for this plateau include the limits of neural reorganization achievable with the current stimulation parameters, patient fatigue, biomechanical limitations, or the extent of remaining tissue damage within the spinal cord.
A particularly notable finding was the emergence of voluntary movement without stimulation in all patients, observed between the 4th and 8th months of follow-up. This phenomenon is a direct evidence of stimulation-induced neuroplasticity, indicating a fundamental reorganization of spinal neural circuits rather than merely a transient facilitation. The ability to move voluntarily without continuous stimulation implies a more autonomous and potentially durable recovery, with significant implications for patient independence and quality of life. This genuine neurological recovery suggests that EES actively reorganizes or strengthens dormant or weakened neural pathways, allowing the brain to regain some level of voluntary control over spinal circuits even in the absence of external stimuli.
Beyond motor recovery, our study also revealed significant clinical benefits in other domains. Spasticity, as measured by the Modified Ashworth Scale, significantly decreased (F ratio = 5.2, P < 0.05). This reduction is clinically important, as spasticity can severely impair functional recovery and quality of life. We propose that this decrease may be attributed to the reintegration of deafferented intrinsic spinal neurons into functional networks due to neuromodulation, potentially leading to reduced gamma hyperactivity and, consequently, less spasticity. Similarly, balance, assessed by the Berg Balance Scale, showed significant improvement (F ratio = 18.6, P < 0.001), which is crucial for orthostasis and ambulation.
The safety profile of the EES procedure in our cohort was favorable. No serious adverse events were observed, and minor complications, such as transient peaks of nociceptive pain following training in 2 patients and a low-grade skin injury associated with the use of weight-bearing support equipment, were manageable and did not affect treatment continuity. These findings reinforce the feasibility and safety of this approach.
Pre-clinical Studies and Mechanisms of EES-Induced Neuroplasticity
The observed motor recovery and neuroplasticity in our patients are supported by a growing understanding of the neurophysiological mechanisms underlying EES. Pre-clinical studies, particularly in rodent models, have been instrumental in elucidating how EES promotes motor recovery. Research suggests that motor efferent fiber activation induced by the exogenous current from epidural stimulation depends on afferent firing rate of neuromuscular spindle fibers, particularly types Ia and II. 28 Previous studies have indicated that afferent input from spindles correlates with bursts of electromyographic (EMG) activity, which aligns with the synchronization of alpha-gamma motor responses.29,30 It is proposed that the motor responses observed in hindlimbs during EES are primarily mediated by thick myelinated fibers involved in spindle-muscle sensory feedback.31-33 These spindle afferents likely activate central pattern generator (CPG) centers in the spinal cord,34-37 which interact with descending signals to increase spinal neuron excitability.38,39 This facilitation is crucial, as SCI often leads to a suppression of excitability in circuits below the lesion, rendering them inactive or dysfunctional.
Beyond transient electrophysiological modulation, recent animal studies have demonstrated that EES is capable of inducing significant structural changes within the injured spinal cord. A recent study by our team, published by Angelin et al (2024), 40 investigated EES efficacy and underlying neurophysiological mechanisms in a rodent model of SCI. This study revealed a significant increase in the mean neural cell count and synaptophysin signal intensity (a marker of synapses) in stimulated animals. Conversely, Caspase 3 signal intensity, an indicator of apoptosis, 41 was significantly lower in stimulated animals, approaching levels observed in control groups. These findings suggest a regenerative and protective effect of EES, indicating that stimulation not only activates circuits but also promotes neuronal survival and the formation of new synaptic connections, contributing to the complex reorganization of the spinal cord. These cytoarchitectural changes likely underpin the electrophysiological plasticity and functional recovery observed clinically and experimentally, including the voluntary movements without stimulation seen in our cohort.
Comparison with Existing Literature
Our findings are consistent with pioneering human studies that established the proof-of-concept for EES in SCI rehabilitation. Harkema et al (2011), 6 Angeli et al (2018), 7 and Wagner et al (2018) 10 demonstrated that EES could reactivate dormant spinal circuits, facilitating voluntary movement, standing, and even over-ground walking in individuals with motor-complete paralysis. A systematic review by Chalif et al (2024), 12 encompassing 64 studies and 306 patients, further corroborates significant improvements in motor function with EES, reporting that 44% of patients achieved assisted or independent stepping or standing.
While our study demonstrates substantial improvements in FMA-LE scores and the remarkable emergence of voluntary movement without stimulation, it is important to contextualize the level of functional recovery achieved. For instance, studies by Gorgey et al,42,43 who utilized single-lead percutaneous electrodes, reported significant gains in trunk control and standing, or motor control, but did not consistently report independent ambulation. In contrast, our participants achieved partial motor control and mobility supported by assistive devices in controlled environments. This difference in reported functional outcomes (eg, independent ambulation vs assisted mobility) highlights the heterogeneity in patient populations, injury characteristics, and rehabilitation paradigms across studies. Our work, utilizing a conventional and widely available EES device, emphasizes the translational potential of existing technology to achieve clinically meaningful gains, even if not always leading to full independent ambulation. This approach offers a practical and accessible pathway for improving quality of life for a broader range of patients, by transforming severe impairment into moderate, and enabling assisted mobility.
Limitations of the Present Study
Despite the promising results, our study has limitations that warrant consideration. Firstly, the demographic homogeneity of our patient cohort, consisting solely of young male individuals with injuries sustained less than 3 years prior, limits generalization of our findings to more diverse populations. Nevertheless, this demographic profile is consistent with the epidemiology of traumatic spinal cord injury in our hospital, located in São Paulo, Brazil, where the majority of victims are young males involved in traffic accidents, especially motorcycle accidents. Future research should aim to include more heterogeneous cohorts to validate the efficacy of EES across a broader spectrum of individuals with SCI.
Secondly, our study did not include pre-operative measurements of bone density and muscle mass. These data are currently being collected from newly enrolled patients and will be reported in due course.
Thirdly, despite the promising motor outcomes, improvements in sensory function and autonomic responses, such as neurogenic bowel and bladder dysfunction, remained limited in our study. This is in contrast to some literature reporting positive effects. 44 This absence of significant improvement may be attributed to the small sample size and the specific EES parameters optimized primarily for motor recovery. This highlights a significant challenge in EES therapy: optimization for one function (eg, motor) may not automatically optimize for others (eg, autonomic). This suggests that distinct stimulation parameters, electrode placements, or even different stimulation modalities may be necessary to achieve comprehensive improvements across multiple systems, pointing to the need for highly personalized and potentially multimodal stimulation strategies. Furthermore, the inclusion of complementary investigative modalities, such as urodynamic examinations, could inform targeted rehabilitation strategies aimed at improving autonomic function.
Conclusions
This study reinforces that epidural electrical stimulation (EES) is a safe and effective strategy for promoting motor recovery in patients with chronic spinal cord injury. Our novel application of a Gompertz mathematical model provides a quantitative framework, demonstrating a rapid improvement in motor scores during the initial 2 months, followed by a plateau around the fifth month. This predictable trajectory is crucial for optimizing rehabilitation strategies and orient patient’s expectations, suggesting that intensive interventions are most beneficial early on, and highlighting the need for combined therapies to potentially overcome this ceiling.
Furthermore, a particularly notable finding was the emergence of voluntary movement without stimulation, observed between the 4th and 8th months of follow-up. This is direct evidence of EES-induced neuroplasticity, indicating a genuine functional reorganization of spinal circuits that leads to more autonomous and potentially durable recovery. Beyond motor gains, our findings also show significant reductions in spasticity and improvements in balance, both contributing to a better quality of life.
Finally, this interim analysis, utilizing a conventional and widely available EES device, presents valuable evidence in favor of the translational potential of EES in the treatment of patients with spinal cord injury.
Footnotes
Acknowledgements
We would like to thank all participants for their invaluable contribution to this study.
Ethical Considerations
The study received approval from the local Ethics Committee of Hospital das Clínicas, Medical School, University of São Paulo (no 30702520.6.0000.0068).
Consent to Participate
All study participants were informed about the aims of the study and the potential adverse effects, and all provided written informed consent prior to enrollment.
Author Contributions
All authors contributed to the study design, data acquisition, analysis, or interpretation, and critically reviewed the manuscript. The CRediT (Contributor Roles Taxonomy) information detailing each author’s specific contributions was provided in the online submission system.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was investigator-initiated. Medtronic (Minneapolis, Minnesota, USA) provided the epidural stimulation devices to our public institution but did not participate in data collection or analysis.
Declaration of Conflicting Interests
The authors declared absence of any conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
