Abstract
Background. Paralysis of the upper limbs from spinal cord injury results in an enormous loss of independence in an individual’s daily life. Meaningful improvement in hand function is rare after 1 year of tetraparesis. Therapeutic developments that result in even modest gains in hand volitional function will significantly affect the quality of life for patients afflicted with high cervical injury. The ability to neuromodulate the lumbosacral spinal circuitry via epidural stimulation in regaining postural function and volitional control of the legs has been recently shown. A key question is whether a similar neuromodulatory strategy can be used to improve volitional motor control of the upper limbs, that is, performance of motor tasks considered to be less “automatic” than posture and locomotion. In this study, the effects of cervical epidural stimulation on hand function are characterized in subjects with chronic cervical cord injury. Objective. Herein we show that epidural stimulation can be applied to the chronic injured human cervical spinal cord to promote volitional hand function. Methods and Results. Two subjects implanted with a cervical epidural electrode array demonstrated improved hand strength (approximately 3-fold) and volitional hand control in the presence of epidural stimulation. Conclusions. The present data are sufficient to suggest that hand motor function in individuals with chronic tetraplegia can be improved with cervical cord neuromodulation and thus should be comprehensively explored as a possible clinical intervention.
Introduction
More than 2.5 million people currently live with chronic spinal cord injury (SCI) with more than 130 000 newly afflicted individuals each year worldwide. 1 Investigational therapies aimed at restoring locomotor function, including pharmacologic agents, 2 intense motor retraining, 3 and epidural spinal cord stimulation 4 (ES) have yielded promising results in the laboratory setting, especially in quadrupedal animal models. More recently, ES at the lumbosacral region in chronic, motor-complete paraplegic SCI subjects resulted in improved lower-limb volitional control as well as postural function.5,6 It remains to be determined if ES can similarly restore lost volitional upper-limb motor function after cervical cord injury in humans, especially in light of a recent preclinical study. 7 For tetraplegic patients, even partial restoration of upper-limb volitional function would greatly enhance the quality of life 8 and thus remains an important clinical goal. Few therapeutic directions exist for reanimating a paralyzed upper extremity following chronic tetraplegia. Among them, functional electrical stimulation 9 strategies are the most developed, where muscles are activated via stimulation of peripheral nerves, either via surface 10 or implanted 11 electrodes. We hypothesize that cervical premotor circuits in chronic SCI subjects are also amenable to epidural electrical neuromodulation for promoting volitional hand control, as has been shown for the lumbosacral locomotor circuits. While reduction of spasticity with ES has been demonstrated previously in patients with high cervical injury,12,13 to our knowledge, volitional hand function has not been investigated in the context of ES-based interventions. In this study, we investigated the therapeutic potential of cervical ES on voluntary hand function in 2 chronic, cervical SCI subjects. Voluntary hand function, including maximum grip strength and hand dexterity tasks were assessed before, during, and after application of ES.
Materials and Methods
The University of California, Los Angeles Institutional Review Board approved all procedures. The effects of cervical ES on volitional hand strength and control were investigated in 2 chronic American Spinal Injury Association (ASIA) Impairment Scale (AIS) B subjects with neurological injury levels at C5 (subject 1) and C6 (subject 2) (Figure 1, A1 and A2). Both subjects suffer from medically refractory chronic pain conditions, limited to the shoulders and proximal upper extremities, but not hands. The criteria for ES were based on its usage for the treatment of pain, as approved by the Food and Drug Administration. One author, D.C.L., a board-certified neurosurgeon, and 2 other physicians confirmed the indication for implantation. Informed consent was obtained from each subject prior to enrollment in the study.
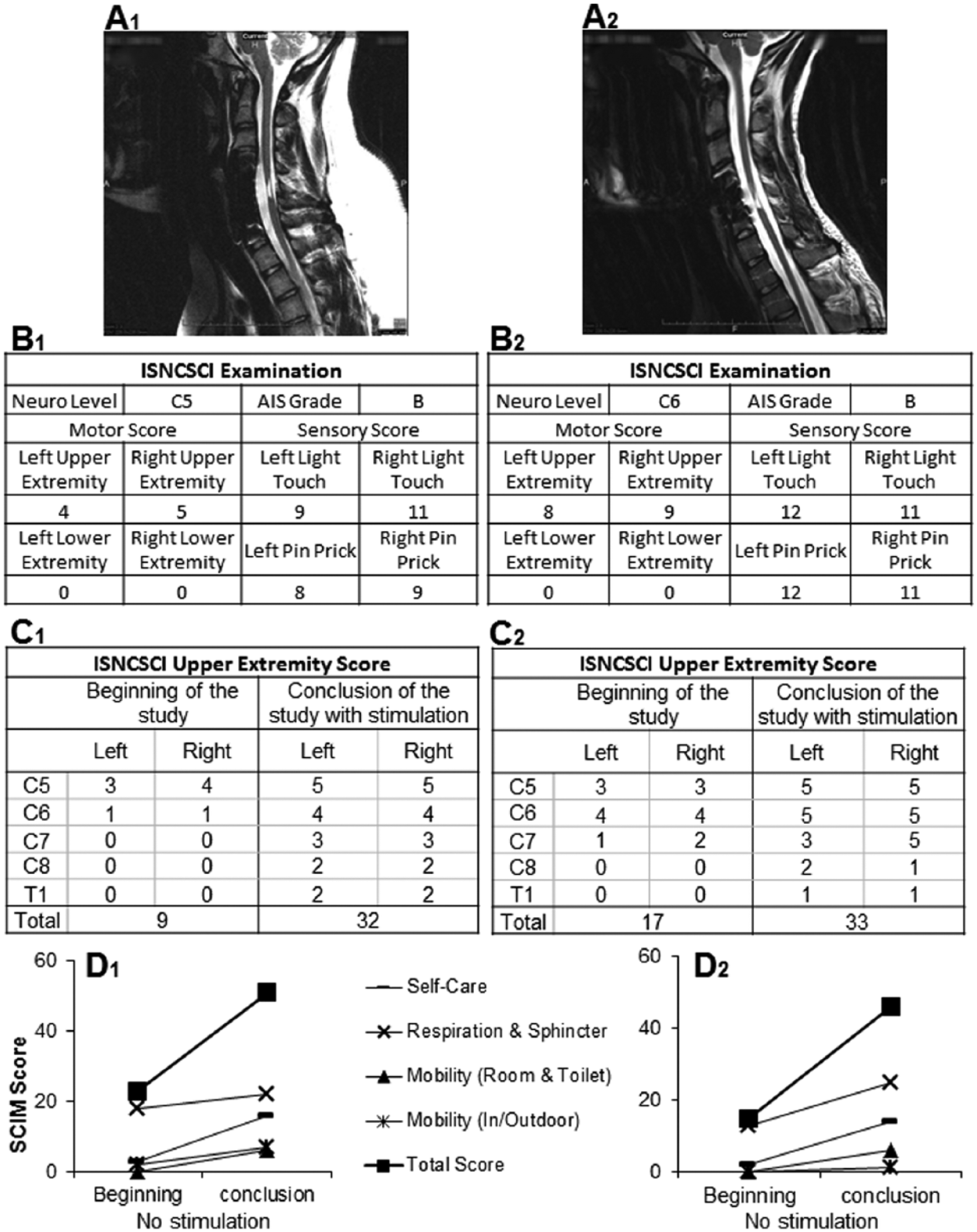
The location of the cervical spinal cord injury for (A) subjects is shown on sagittal T2 magnetic resonance imaging (MRI) to be at approximately the C5 spinal level. The injury location exhibits a high intensity T2 signal corresponding to a glial scar formation. Spinal cord tissue distal and proximal to the injury locus has a normal appearance without evidence of post-traumatic syrinx formation. (B) International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI) examination of subjects before and after intervention. ISNCSCI scores were assessed at the start of the study without stimulation and at the conclusion of the study with stimulation. (C) Clinically significant increases of 23 and 16 points for the upper extremity motor score were observed for subjects 1 and 2, respectively. The minimal clinically detectable change has been reported to be 1 36 for this motor score. The spinal cord independence measure (SCIM), version 3, was used to assess the activities of daily living by in both subjects at the beginning and conclusion of the study. (D) Improvement in all categories was observed in both subjects. An overall improvement of 28 and 31 points, respectively, were recorded. The scores represent the subject’s state at home in a non-stimulated condition.
Injury Assessment
International Standards for Neurological Classification for Spinal Cord Injury (ISNCSCI) was assessed in both subjects (Figure 1, B1 and B2). In addition, electrodiagnostics in the form of motor evoked potentials (MEPs) and somatosensory evoked potentials (SSEPs) were conducted that revealed a motor impairment at or below C6 (for details, see Supplementary Materials and Methods).
Timeline
Baseline upper-limb motor function was examined in subjects 1 and 2 for 4 months prior to temporary electrode implantation (ie, lead-in phase). Subject 1 underwent a temporary (7 days) implant trial, followed by a permanent implant placement and was subsequently tested weekly for eight weeks. During the temporary implant phase, subject 1 did not undergo formal functional testing. Subject 2 was tested for 7 days with a temporary implant and is currently awaiting a permanent implant. The data presented in this study from subject 1 were collected exclusively during the permanent implant phase, whereas data for subject 2 were collected with a temporary implant. For both subjects, pain improvement occurs almost immediately within 15 minutes of initial stimulation prior to these experiments. Furthermore, the stimulation parameters for pain control are different with respect to electrode location, frequency, and amplitude. ES as used in this study for improvement of motor performance had no impact on pain and was only delivered to the subjects during testing sessions. Both subjects who participated in this study enrolled in a two-year study with no established endpoint for termination.
Electrophysiological Testing
Temporary implants consisted of two 16-contact percutaneous epidural leads (Linear Lead, Boston Scientific, Valencia, CA) spanning C4-T1 for both Subjects 1 and 2. After seven days of confirmed efficacy in the treatment of pain, one Boston Scientific Artisan (Valencia, CA) 16-electrode epidural array, encompassing C5-T1, and one Boston Scientific Precision Plus Spinal Cord Stimulator (Valencia, CA) were permanently implanted into Subject 1. During the experiments, stimulation amplitudes ranged between 0.1 and 10.0 mA, frequencies ranged between 2 and 40 Hz, and pulse width was at 210 µs. The range of ES parameters used was wider than those employed in previous studies of ES for spasticity reduction.12,13
Different bipolar electrode combinations, starting from the rostral end of the electrode array to the caudal end, were tested using various stimulation parameters to identify electrode pairs that produced the greatest hand motor responses. The efficacy of the electrode combinations was determined via spinal evoked potentials in response to biphasic stimulation based on the following criteria: (1) minimum stimulation strength at which motor evoked responses were observed and the peak amplitudes of evoked responses in the biceps brachii, triceps brachii, brachioradialis, extensor digitorum, flexor digitorum, thenar eminence, and hypothenar eminence with increasing stimulation strengths (see Figure S2 in Supplementary Material) and (2) the electromyography (EMG) amplitudes of each of these same muscles with a continuous train (frequency range of 20 Hz) stimuli. Based on these results, electrode pair −13+14 was selected for subject 1 and electrode pair −22+26 for subject 2. Both locations of stimulation are caudal to site of injury.
During the implanted phase (Figure 3A), both subjects were tested in the same manner: In each session, maximum grip strength and ability to control the grip were tested before, during, and after application of ES. ES was delivered to each subject for up to 60 minutes during the middle third of each 180-minute testing session. Stimulation frequency and intensity were varied to select for the most effective stimulation parameters for each subject that improved force, accuracy, and response time (Figure 3). Because of potential fatigue bias, each graph in Figure 3 was obtained during separate days of testing and was duplicated during a different testing session (ie, rested condition). The results were similar during these 2 sessions except less fatigue symptoms were observed when tested on a separate day. Spinally evoked responses from the arm, forearm, and hand muscles to consecutive pulses (2, 5, 10, 20, and 30 Hz) throughout a maximum handgrip effort were obtained from a representative hand contraction with different stimulation frequencies during voluntary neuromuscular resting and active states (Figure 4). Evoked response profiles also were identified in other contractions at different levels of perceived forces (Figure 5).
Motor Performance Metrics
Voluntary motor control performance was assessed during each session with several tasks employing a handgrip force measurement device14,15 in conjunction with a computer program (Figure S1). This device measures displacement against interchangeable springs with a range of spring constants. Each task reflects a different aspect of voluntary hand control, including maximum handgrip force, grip force perception, voluntary hand control, repetitive hand contraction, relaxation speed, and response time. Motor assessments were conducted on the more functional arm postinjury regardless of the preinjury dominant side. Tenodesis grip was discouraged and was restricted by a wrist brace during testing.
Measurement of maximal voluntary forces was conducted by asking (ie, verbal command) the subject to contract and relax the hand against the springs to maximally displace the handgrip (Figures 2A, 2B, and 3C). Subjects relied on their own perception of their performance without any feedback cues. Each contraction lasted for approximately 3 to 5 seconds, repeated 3 times over a period of 60 seconds. Grip force perception was evaluated by asking the subject to produce different grip force levels relative to the subject’s perceived maximal grip force (Figures 2 and 3).
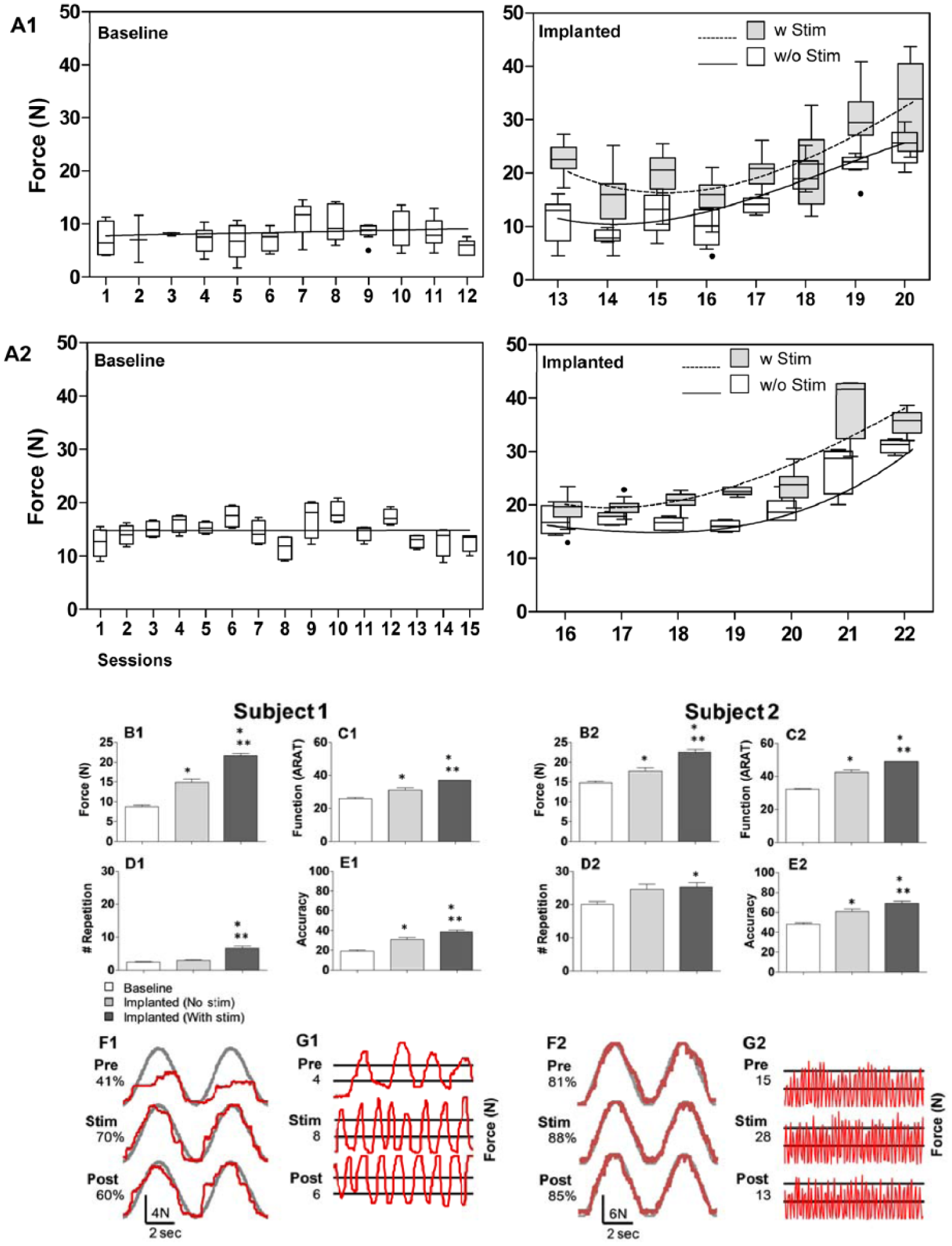
Subjects 1 and 2 were assessed for hand function over a period of at least 12 weeks prior to implantation. (A) A session-by-session comparison of the grip force generated by subjects shows the stability of baseline performance (A, left panel), and the gradual improvement of grip force in sessions with and without stimulation following implantation for both subjects (A, right panel). X-axis shows each testing session, all of which were conducted weekly except for the implanted phase in A2, in which the sessions were conducted daily. Boxplots of maximum contraction force by session for both subjects. Lines reflect best linear fit across baseline sessions and third order polynomial fits across implanted sessions with and without stimulation. Stimulation parameters varied in each session with electrode pairs of −13+14 (subject 1) and −22+26 (subject 2) unless stated otherwise. (B) Average peak forces as measured by handgrip device. (C) The Action Research Arm Test (ARAT) was used to assess hand function in everyday tasks. (D, E) Hand endurance and control were assessed by a repetition and accuracy task, respectively. (F) Representative sinusoidal traces of accuracy task with scores are displayed on the left. Note that F1 and F2 are scaled differently. (G) Representative traces of a rapid oscillatory task over 10 seconds with the number of threshold crossings are displayed on the left. For all assessments shown (B-E), scores from all examinations in each situation (Baseline, Implanted (No stim), Implanted (With stim)) were averaged to demonstrate the overall effects. * and ** indicate significant differences from Baseline and Implanted (No stim), respectively, at P < .05.
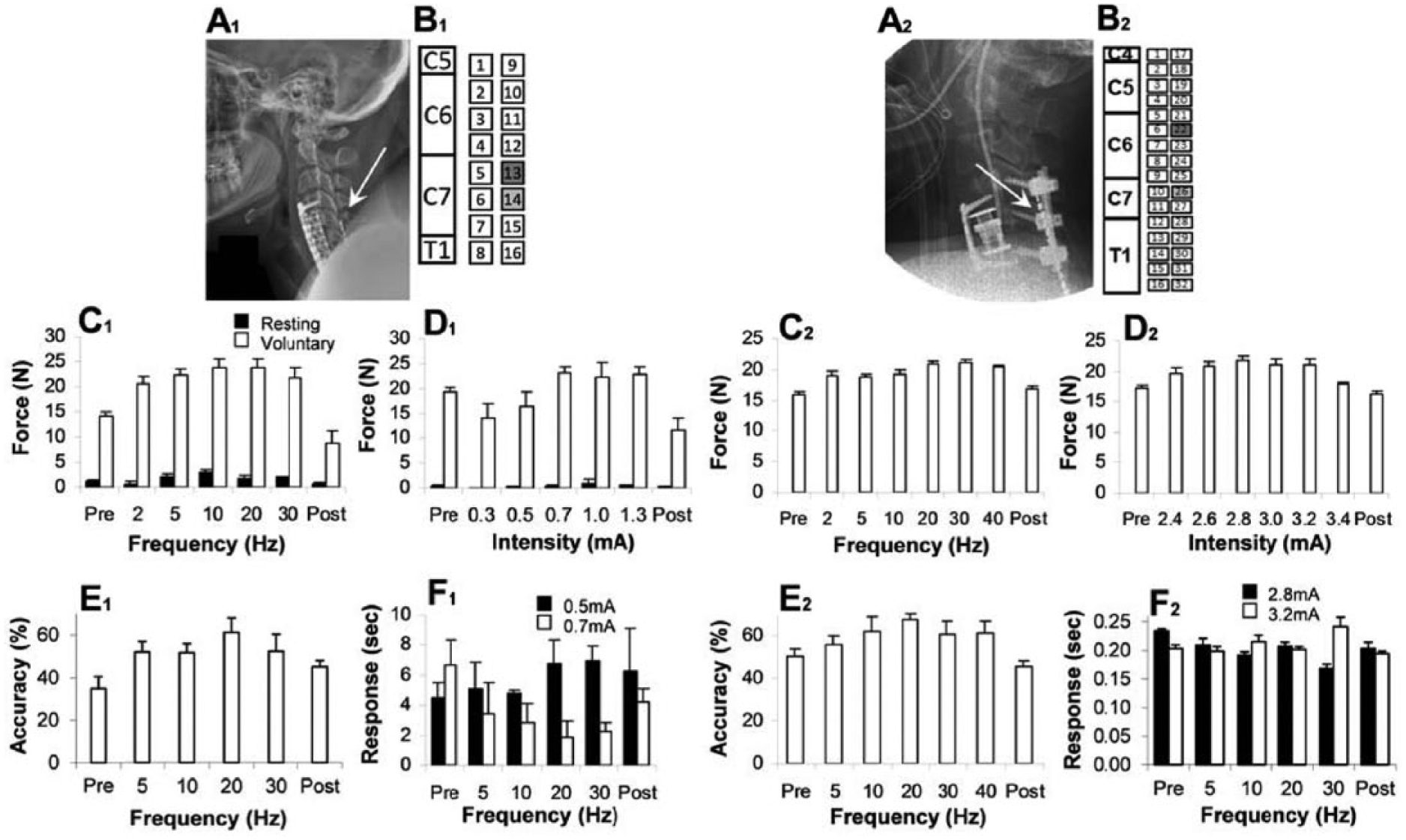
(A1) Subject 1 was implanted with one 16-channel cervical epidural stimulation paddle array spanning spinal cord levels C5-T1. (B1) All stimulation was conducted at electrodes 13 and 14 (shaded in the schematic diagram). (A2) Subject 2 was implanted with 2 parallel, temporary, percutaneous linear 16-channel cervical epidural stimulation electrodes spanning spinal cord levels C4-T1. (B2) All stimulation was conducted at electrodes 22 and 26. Stimulation locations were selected based on optimized evoked responses recorded for different electrode pairs. Grip strength (maximum voluntary contraction) was assessed over different frequencies (C) and intensities (D) of stimulation. Force output from the device was collected with the subject at rest (Baseline) and during voluntary contraction (Voluntary). Note that there was no observed tonic contraction during baseline phase for subject 2 (C2, D2). (C) Tests of different frequencies were conducted at a constant intensity of 1.0 mA for subject 1 and at 2.8 mA for subject 2. (D) Tests of different stimulation intensities were conducted at a constant frequency of 20 Hz for both subjects. (E) To assess hand control, the ability to accurately follow a targeted sine wave was assessed with several stimulation frequencies applied during the test (5, 10, 20, and 30 Hz) during each test session, which was preceded (Pre) and followed (Post) by testing without stimulation. (F) Time to actuate the handgrip device (Response) was assessed at different stimulation frequencies and intensities. For both subjects and all assessments, the mean values (±SEM [standard error of the mean], 3 trials for each condition) are shown.
A computer program was used to assess voluntary hand control by having the subject trace a sinusoidal wave (0.15 Hz) that appeared on the computer screen by squeezing to move the cursor upward, and releasing to move the cursor downward (Figure 2, F1 and F2). Tracing accuracy was determined as the percentage of points that fell within a circular window around the projected sinusoidal pattern at a 15-Hz sampling rate. 16 Similarly, repetitive hand contraction frequency was measured by having the subject contract and release the hand as fast as possible above and below 2 lines on a computer screen that were preset at 10% and 50% of the subject’s maximum voluntary contraction (Figure 2, G1 and G2). The number of completed contraction-release sequences crossing both lines over a period of 10 seconds was measured.
During all handgrip assessments, EMG activity was recorded via surface electrodes (2DT2, MultiBioSensors, El Paso, TX) placed unilaterally on the paraspinal muscles (near the electrode pair), deltoid, biceps brachii, triceps brachii, brachioradialis, extensor digitorum, flexor digitorum, thenar eminence, and hypothenar eminence. EMG signals were differentially amplified (10 Hz to 5 kHz) (T50 Myopac Wireless System, Konigsberg Instruments, Pasadena, CA) and digitized at 10 kHz using a 16-channel A/D board (BNC 2115, National Instruments, Austin, TX) with customized LabVIEW software (National Instruments, Austin, TX, USA).
In addition, clinical scores including the Action Research Arm Test (ARAT), 17 Spinal Cord Independence Measure (SCIM),18,19 and ISNCSCI with upper extremity motor score (UEMS), 20 were assessed prior to and throughout the study interventions (Figure 1B-D). ARAT is a test of upper extremity fine motor function in categories of grasp, grip, pinch, and gross movement. This test was selected because of its focus on arm motor ability and has been previously applied in the SCI setting.21 -23 SCIM is an interview tool that assesses categories relevant to the subject’s independence, that is, self-care, respiration/sphincter management, and mobility. ISNCSCI is a standardized clinical assessment guideline for classifying the SCI neurological level and extent of the injury. 24 UEMS is a test of motor strength of the upper extremity muscles. Two staff members independently assessed the subjects with these clinical measures during each study phase, and provided a consensus measure. The ARAT examination was performed at least 5 times during the lead-in phase and 3 times during the implanted phase.
Blinding
Subjects perceived a nonpainful hand-tingling sensation during stimulation, but were blinded to the exact stimulation parameters as were the research staff members conducting the handgrip tests. The staff responsible for the control of the stimulator was not blinded as the stimulation parameters were manipulated during each session: However, this member did not interact with the subject and each staff member was instructed to follow the same script when administering the various tasks regardless of the stimulation paradigm.
Statistical Analyses
Performance metrics (grip force, repetition, accuracy, ARAT score, ISNCSCI, SCIM, UEMS) were averaged across all observations in a given phase for each subject. Using these values, means for each outcome measure over the three phases were compared using a repeated measure analysis of variance model. The Tukey correction was used for post hoc comparisons under this model. The repeated measure linear mixed model takes into account the correlation across time for the same subjects and 5 measures were used in our analysis. Quantile-quantile plots of the residual errors confirmed that the data followed a normal distribution. Values are reported as mean ± standard error of the mean (SEM). A 2-sided P < .05 was considered significant.
Results
Assessment of Baseline Functional Capacity
The baseline stability of upper extremity function was assessed over 4 months during which the subjects were tested and trained weekly with the handgrip device (Figure 2, A1 and A2). In both subjects, there were no consistent improvements during this “lead-in” phase that could be attributed to training in the absence of any ES. Baseline electrophysiological testing revealed motor impairment at or below C6 for subject 1, with preserved but impaired sensory conduction for the right leg but not for the other limbs: for subject 2, motor impairment at or below C6, with normal sensation of the arms and impaired but partially preserved sensory sensation of the legs. Baseline clinical measures are as follows: ISNCSCI UEMS of 9 and 17 (Figure 1, C1 and C2), SCIM score of 22 and 18 (Figure 1, D1 and D2), and ARAT score of 23 and 31 (Figure 2, C1 and C2) for subjects 1 and 2, respectively. These results are consistent with severely cervical injured individuals.
Improvements in Grip Strength and Control With ES
Average maximum grip force increased cumulatively over multiple sessions with ES following implantation in both subjects (Figure 2, A1, A2, B1, and B2). There was a gradual increase in the force generated following repeated exposure to ES, even in the absence of stimulation, exceeding baseline performance prior to stimulation exposure. This phenomenon was observed in both subjects (Figure 2, A1 and A2). An increased average number of squeeze-release repetitions (Figure 2, D1 and D2) and sinusoidal tracking accuracy scores (Figure 2, E1 and E2) in both subjects also indicated that hand control, including hand opening and closing, improved with ES exposure.
In the initial sessions after subject 1’s permanent implantation, a series of motor tests were conducted to compare the effects of different stimulation parameters on performance. It became apparent that different combinations of frequency and amperage had dramatic yet varying effects on voluntary grip strength. Maximum force values were observed at 10 and 20 Hz. A frequency of 20 Hz, at 0.7, 1.0, and 1.3 mA, was chosen for further investigation, due to less tonic contraction and more overall voluntary hand control at this frequency of stimulation (Figure 3, C1 and D1). The accuracy of following a targeted sine wave was highest at 20 Hz (Figure 3, E1), and the response time in actuating the handgrip was shorter at 0.7 mA than at 0.5 mA for most frequencies (Figure 3, F1). Similar tests were repeated with subject 2 following his temporary implantation again demonstrating that force output (Figure 3, C2 and D2), accuracy performance (Figure 3, E2), and response time (Figure 3, F2) varied with the frequency–current intensity combination used. While the forces recorded throughout this study were considerably lower than those of uninjured subjects (~400 N), 25 they were substantially larger than either subjects’ baseline values (Figure 3, C1, C2, D1, and D2).
Corresponding Increase in EMG Activity
The effects of ES on EMG activity from multiple upper extremity muscles in response to a range of stimulation sites, intensities, and frequencies were studied. Continuous stimulation of subject 1 was associated with a substantial increase in maximal voluntary force and a corresponding increase in EMG amplitudes recorded at the hypothenar and thenar eminences (compare Figure 4, A1 and B1). When stimulation was initiated during rest, there were no detectable potentials evoked by the stimulus pulse except at the hypothenar and thenar eminences (Figure 4, B1, C1, and D1, blue). When a voluntary effort was initiated, synchronized evoked potentials were observed in all muscles studied, even as force was declining (Figure 4, B1 and D1, green). Interestingly, the force generated was largely independent of the magnitude of the synchronized responses. For example, the amplitude of the potentials was unrelated to the force generated when electrical stimulation was delivered at 2, 5, 10, 20, or 30 Hz and 1.0 mA (Figure 4, C1, red). The integrated EMG amplitude for the flexor digitorum (FD) in synchrony with the stimulus pulses (Figure 4, E1, green squares) was much less than that of the asynchronous EMG (Figure 4, E1, red triangles). Figure 4E1 demonstrates how the early synchronized responses (ER only EMG) and the later asynchronous responses (non-ER EMG) relate to the force generated in a given contraction. The early synchronous activity in Figure 4E1 (green squares, ER only EMG) corresponds to less than 50% of the total EMG signal in subject 1, and therefore, a larger component of the force produced corresponds to later asynchronous EMG activity (red triangles, non-ER EMG). These observations also were confirmed in Subject 2 (Figure 4, A2, C2, and E2) where later asynchronous EMG activity was associated with greater force generation.
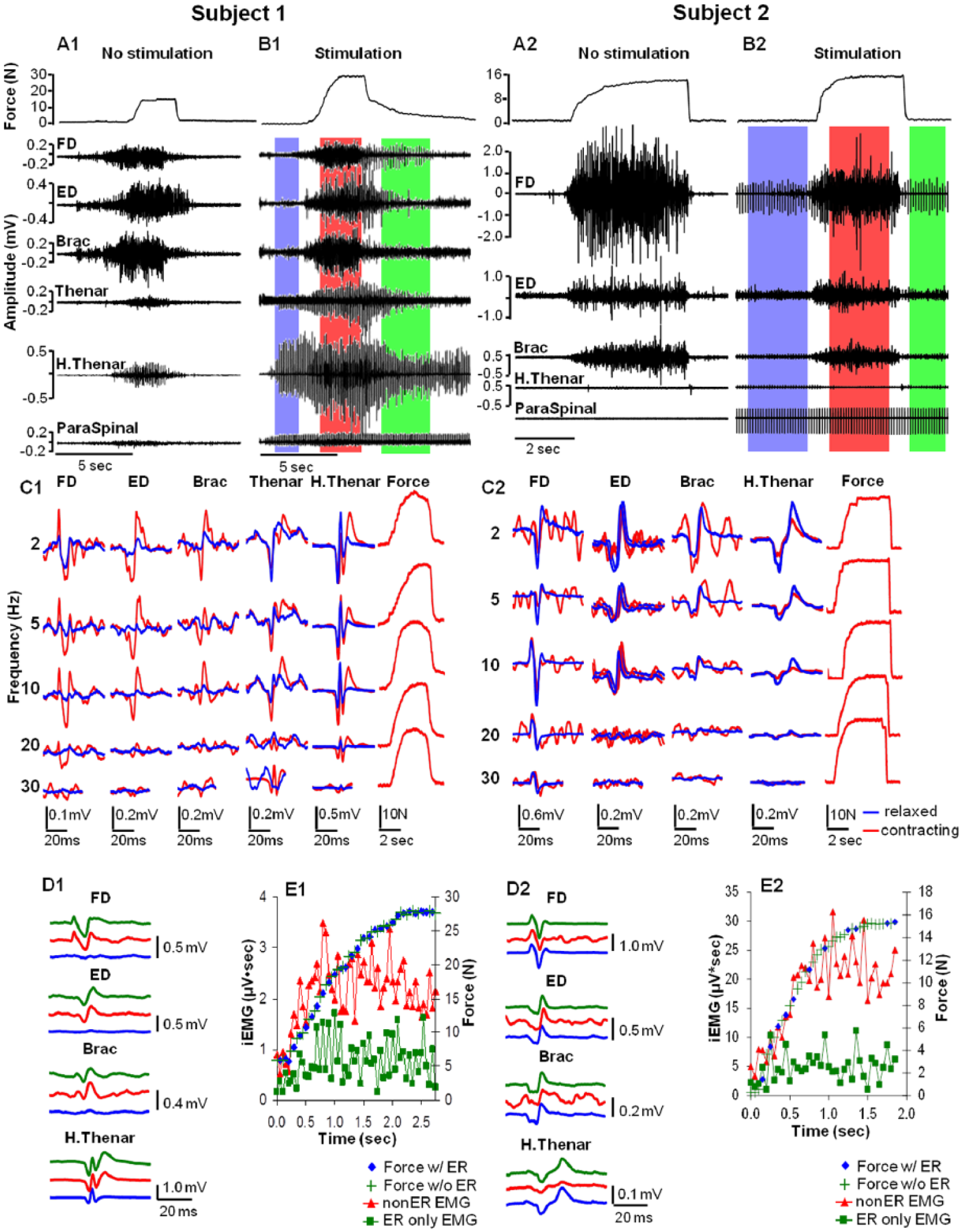
(A, B) Handgrip force and electromyography (EMG) during a maximum handgrip performed without (A) and with (B) epidural spinal cord stimulation (ES) of the cervical spinal cord (electrodes −13+14, 10 Hz, 1.0 mA for subject 1 and electrodes −22+26, 10 Hz, 5.5 mA for subject 2). The performance with ES is segmented into 3 distinct phases of activity: the initial stimulation phase without any voluntary effort (blue shaded area), the voluntary contraction phase (red shaded area), and the relaxation phase of the voluntary effort (green shaded area). (C) The effect of stimulation frequency on the average evoked potentials and force patterns for three seconds during the initial stimulation phase was determined both before any voluntary effort (blue traces) and during the voluntary contraction phase (red traces) in each muscle. (D) Evoked potentials were collected during the three phases of activity. More than 20 potentials evoked in each muscle were averaged. (E) The rising pattern of force along with the 2 corresponding components of the total FD iEMG signal (µV·s). Pulse synchronized EMG at 10 Hz (red triangles) versus the remainder of the total EMG signal (ie, nonsynchronized potentials, green squares) during the initial phase of the contraction is shown. The presence (blue diamonds) or absence (green plus signs) of the lower synchronized signal was not correlated with the force generated during a given time bin. ER, evoked response; FD, flexor digitorum; ED, extensor digitorum; Brac, brachioradialis; H. Thenar, hypothenar; iEMG, integrated EMG.
Maintenance of Grip Force With ES
The effects of ES during volitional hand contractions were evaluated. The subjects were asked to perform 7 consecutive pulls on the handgrip device at each of 3 distinct percentages of effort, all based subjectively by the subject on the initial maximum effort deemed to be “100%.” Without stimulation, the force levels at the lowest percentage effort (10%) were sustainable, but the force levels at the higher percentage efforts (25% and 75%) declined in both subjects during consecutive contractions reflecting some combination of neural and/or muscular fatigue (Figure 5, A1 and A2). While in a fatigue state, the experiment was repeated with increasing stimulation intensity (Figure 5, C1 and C2). For subject 1, there were no marked differences in the subject’s ability to estimate a target force over a wide range of stimulation intensities at the lowest percent effort (Figure 5, C1). During the series of higher percent efforts, fatigue was evident at moderate stimulation intensities, but this was overcome with higher stimulation currents. The experiments shown in Figure 5A1 and C1 occurred within a 15-minute period on one testing day. To assess the effects of potential neuromuscular fatigue with repeated tests within the same day, the 2 tests were repeated with 24 hours of rest between the 2 sequences of 7 contractions (Figure 5). In this instance, compensation in motor unit recruitment was not evident as the subject was able to maintain similar forces throughout the different percent efforts. This set of experiments was repeated with subject 2, although he was able to maintain force levels more successfully at all efforts with stimulation (in Figure 5, compare C2 and E2).
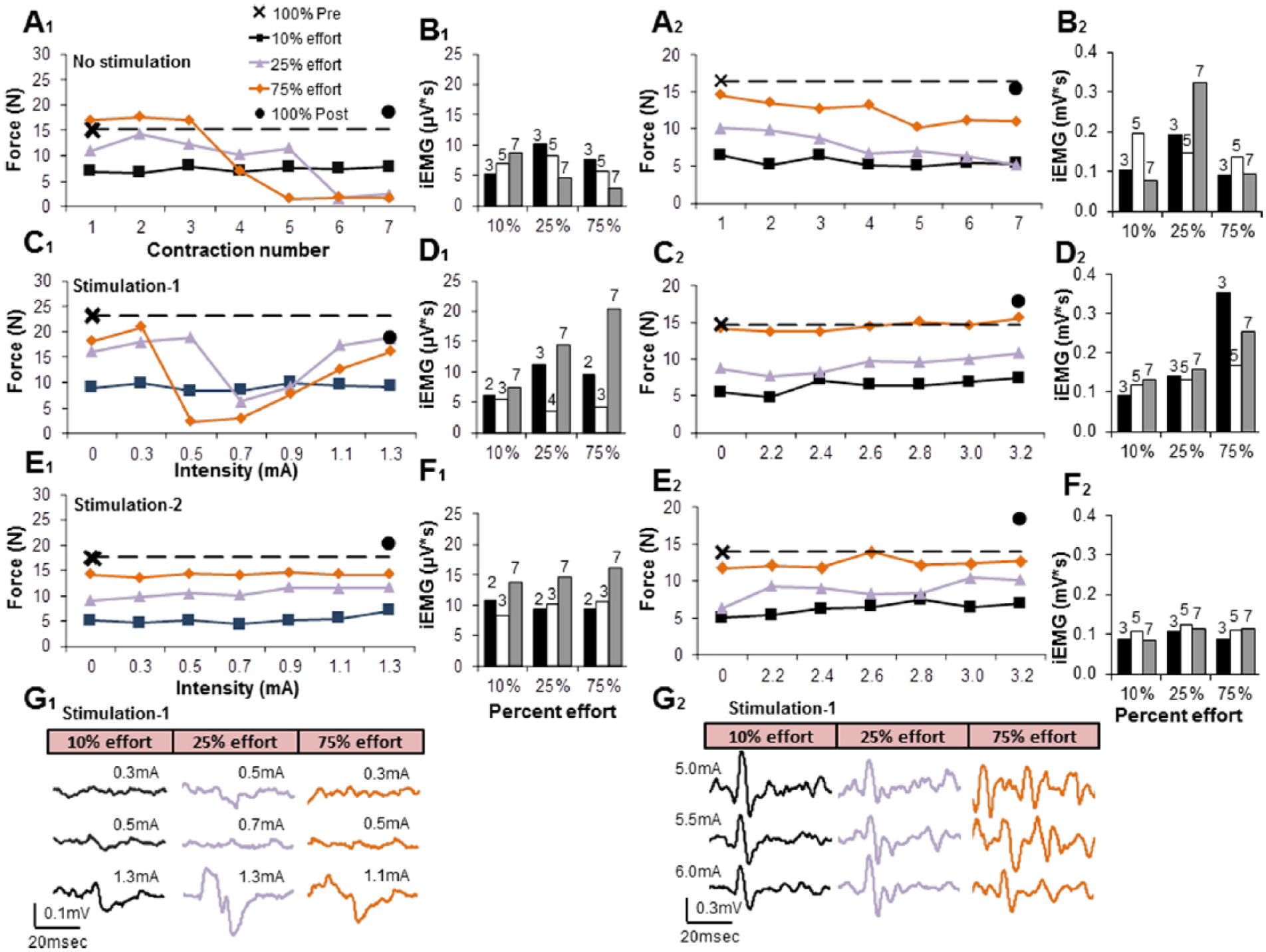
(A) Handgrip force measurements consisted of a maximum contraction at the beginning of testing (100% Pre) followed by 7 consecutive contractions labeled on the x-axis at 10 (black), 25 (purple), 75 (orange) % effort, and a maximum contraction at the end (100% Post). The subjects were given five seconds of rest between each contraction, and five minutes of rest between different efforts without stimulation. Maximal forces (100% Pre and 100% Post stimulation) were conducted after a 5-minute rest interval. (C) Fifteen minutes after the completion of A, the same series of contractions at different % maximum efforts were repeated in the presence of increasing intensities of stimulation. (E) The experiments with stimulation were repeated on another day (without the No Stimulation series) to assess possible fatigue effects. (B, D, F) The flexor digitorum integrated EMG (iEMG) for 3 specific contractions is shown, with the number above the bar referring to the contraction number (1-7) corresponding to the 7 consecutive contractions at different stimulation strengths shown in A, C, and E, respectively. (G) The patterns of evoked potentials at selected percent efforts and at different strengths of stimulation when there was modest (25% effort) or substantial (75% effort) fatigue and when there was no apparent fatigue (10% effort) are shown, corresponding to C and the iEMG in D. While no responses were visible consistently either before or during the fatigued state, synchronized responses were recorded when subject 1 appeared to recover from fatigue when stimulated at a higher intensity (G1). Subject 2 was asked to continue the series of contractions shown in C2 up to a maximum intensity of 6.0 mA and no indications of fatigue were noted based on the amplitudes of evoked potentials. Subject 2 did not exhibit fatigue (E2 vs C2) and synchronized responses were recorded at all intensities (G2). The stimulation parameters for subject 1 were an electrode pattern of −13+14, 20 Hz, and 0 to 1.3 mA. The stimulation parameters for subject 2 were an electrode pattern of −22+26, 20 Hz, and 0 to 6.0 mA.
Improved Clinical Scores With ES
The overall time course of recovery is demonstrated in Figure 2. For subject 1 (Figure 2, A1), immediate improvement was seen with stimulation for each session (No stim vs With stim). There was a session-to-session long-lasting improvement in grip force observed over 8 testing sessions or 8 weeks. Similarly, subject 2 (Figure 2 A2) demonstrated immediate and long-lasting improvements, seen over 7 testing sessions or 1 week. The 20% (6.0 points) increase in ARAT score seen in both subjects is above the 5.7 points responsiveness threshold established for this instrument. 26 ES also resulted in some long-lasting effects in improved self-care and mobility due to improved hand and arm function. The subjects’ self-care was affected in all subcategories (feeding, bathing, dressing, and grooming), and mobility in bed and transfers. This is reflected in the improved ISNCSCI upper extremity motor scores (Figure 1). There were no issues with patient tolerability or adverse events associated with this study.
Discussion
Although ES has been successfully applied to lumbosacral spinal circuits with substantial motor improvements,5,6 it was unclear if such approaches would be translatable to the cervical spinal circuits responsible for volitional motor control of the upper limbs after quadriplegia. To our knowledge, this is the first demonstration of ES modulating the physiological state of the injured cervical cord to improve volitional hand control and grip strength in chronic cervical SCI human subjects. In both subjects, there were immediate improvements in maximal hand strength and control with a wide range of ES parameters within a single test session. In addition, repeated test sessions resulted in sustained long-term improvements akin to a training phenomenon. These results are notable given that previous studies have shown little recovery with a single application of an intervention acutely or from repeated practices in severely paralyzed chronic (more than 12 months) cervical SCI subjects.27,28 Both subjects were more than 18 months beyond their initial injuries, and baseline motor function was unchanged over 4 months of preimplantation testing in each subject. The present results suggest that ES facilitates the recruitment of viable, but previously nonparticipating, cervical interneuronal networks projecting to motor pools, similar to what was observed with ES at the lumbosacral region. 5
Our observations that the evoked potentials generated by ES during a voluntary effort were not synchronized solely to each evoked stimulation pulse suggest that the ES effects were largely mediated via interneuronal networks rather than direct stimulation of motor neurons 29 or primary afferents of the dorsal roots.30,31 We propose that ES increases the probability of randomized miniature excitatory potentials generation (thereby shifting network excitability closer to motor threshold) in a wide population of continuously varying networks of interneurons in response to each stimulation pulse. Therefore, the more optimal stimulation parameters seem to be those that enable the supraspinal input to spinal interneuron networks and motoneurons, which results in relatively normal EMG bursting patterns. 5 As such, we suggest that the underlying concept of ES is most consistent with the concept of “enabling” the individual to produce and control movement (by modulating the spinal cord’s intrinsic circuitry) rather than “inducing” it with stimulation (directly activating the motoneurons or motor nerve roots). Further evidence of the enabling concept is that the subject chooses whether and when to volitionally generate different movement patterns in the presence of continuous stimulation.
The positive improvements to ES in both subjects in terms of strength, control, and action latency, were similar even though the subjects have different injuries, the operations undergone to address these injuries, baseline function, and combinations of stimulation leads used. As a consequence, some functional differences (eg, grip strength, accuracy, endurance, and response time) between subjects might be expected along with dissimilarities in their responses to ES (Figure 3). For example, subject 1 had a prolonged voluntary response time (in seconds) as compared with subject 2 (in fractions of a second). Despite intersubject anatomical and physiological differences, both subjects responded positively to ES in all functional parameters measured. Long-lasting improved hand and arm function were observed in both subjects and resulted in an increased ability for self-care (see Figure 1C, ISNCSCI scores, before and after). Although the results from this study do not address the type or proportion of chronic SCI subjects that would similarly respond in a positive manner, the results provide a proof-of-principle for ES-based upper extremity functional restoration in chronic SCI subjects.
There are 2 main limitations to this study. First, this study is only a proof-of-concept based on 2 SCI subjects. The lack of a separate control group and the inability to truly blind subjects to the stimulation intervention warrants some caution. However, the functional improvements observed within as well as across multiple stimulation sessions and, given the severity and chronicity of paralysis, is unlikely attributable to a placebo effect or some subtle bias in the experimental design. This viewpoint is reinforced by the results seen with previous experiments in 1 subject 6 on lower-limb function, which was followed by comparable results in 3 subjects treated similarly. 5 The positive observations reported herein with cervical ES along with our recent experiences with lumbosacral spinal ES suggests a potentially significant clinical impact and merits further investigation. Second, the tested parameters (electrode combinations, frequency, intensity, pulse width) are a fraction of the variables that could be examined to achieve greater hand and upper-limb function.
This study extends the general concept of neuromodulation, or electrical enabling motor control (eEmc) as a strategy to regain motor function. 4 Previous application of eEmc include lumbosacral ES in 4 subjects with chronic, complete paralysis, who recovered the ability to stand independently as well as regain significant levels of lower limb voluntary control with stimulation and motor training.5,6 We also have observed the recovery of some voluntary lower-limb control after complete paralysis in 5 subjects after transcutaneous lumbosacral spinal cord stimulation combined with motor training, 32 which was further enhanced by pharmacological neuromodulation. Similar results were seen with one other paralyzed subject receiving transcutaneous stimulation while being trained in an exoskeleton device. 33 Our results together with these previous findings suggest that (1) there is a high probability of residual anatomical, but nonfunctional connectivity, across many “complete” spinal cord lesions; (2) the intrinsic spinal neural circuitry caudal to a spinal cord lesion can be modulated to reenable previously nonfunctional but viable supraspinal connections across the lesion; and (3) the physiological state of the spinal networks can be pharmacologically modulated, electrically modulated transcutaneously or via implanted epidural electrodes, even at low levels of prolonged direct current stimulation, 34 as well as via electromagnetic stimulation. 35 With the increasingly recognized plasticity within spinal and supraspinal neuronal networks, the challenge going forward is how to effectively integrate these technical neuromodulation strategies given the intrinsic sensorimotor processing within spinal networks to regain functional recovery after a SCI.
Footnotes
Acknowledgements
We thank Dr David McArthur for statistical support, Dr James Leiter for critical review of the paper, and Ms Naomi Gonzalez for logistical support.
Authors’ Note
Morteza Modaber, MD, and Nicholas AuYong, MD, PhD, contributed equally to this study.
Declaration of Conflicting Interests
The authors declared the potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCL, VRE, RRR, and YG researchers on the study team hold shareholder interest in NeuroRecovery Technologies. VRE is president and chair of company’s board of directors. DCL, VRE, RRR, and YG hold certain inventorship rights on intellectual property licensed by the regents of the University of California to NeuroRecovery Technologies and its subsidiaries.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was made possible by a Research Initiation Grant from the UCLA Department of Neurosurgery Board of Advisors, generous support through the J. Yang & Family Foundation, and the NIH: EB15521 and R01EB007615 grants, funded by NIBIB, NINDS, and NICD. The research described was conducted in the UCLA Clinical and Translational Research Center (CTRC), which was supported by NIH/National Center for Advancing Translational Science (NCATS) UCLA CTSI Grant Number UL1TR000124. D.C.L. is a 1999 Paul & Daisy Soros New American Fellow. Partial support for data analysis and interpretation of the results was provided to VRE and YG from the Russian Science Foundation (RSF grant No. 14-45-00024).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
