Abstract
Background
Paracetamol is one of the popular causes of liver injury primarily mediated by oxidative stress and increased inflammation within the liver. Bersama abyssinica has active ingredients with antioxidant activities and is traditionally used to treat liver diseases. However, its effects have not been confirmed by in vivo studies.
Objective
This study was aimed at evaluating the hepatoprotective effect of a methanolic extract of Bersama abyssinica leaf on paracetamol-induced hepatotoxicity in Swiss albino mice.
Methods
The study was conducted on thirty male Swiss albino mice, randomly grouped into six groups of five mice each; normal, negative, and positive control groups received 1 mL/kg of distilled water, paracetamol 250 mg/kg only, and silymarin (100 mg/kg) orally with paracetamol, respectively. While treatment groups I, II, and III received 200, 400, and 600 mg/kg of the Bersama abyssinica leaf methanolic extract with paracetamol 250 mg/kg, respectively. At the end of the experiment, the mice were sacrificed after being anesthetized, and blood samples were collected via cardiac puncture, and the liver was removed for histopathological investigation. Data were entered into Epi Data version 3.1 and exported to SPSS version 25 for further analysis using a one-way ANOVA.
Results
The negative control group showed higher levels of ALT, AST, ALP, GGT, and bilirubin compared to the normal control. However, significant hepatoprotective effects were noted in the treatment and positive control groups, as evidenced by the substantially reduced levels of these liver biomarkers towards the normal range. The significantly lower levels of indirect bilirubin were seen among treatment groups II and III as compared to group II. Abnormal liver architectures were observed in the negative control group; however, these changes were reversed following administration of the leaf extract.
Conclusion
The study found that the methanolic leaf extract of Bersama abyssinica exhibited hepatoprotective effects against paracetamol-induced liver injury.
Introduction
The liver is an organ that is more exposed to drug-induced damage due to its playing a major role in the metabolism and detoxification of a variety of drugs. Liver disease is a serious health problem that is responsible for 2 million deaths per year worldwide, according to the report by the World Health Organization (WHO).1,2 Most of the drugs are metabolized in the liver and are one of the factors resulting in the liver disease. 3 Paracetamol (PCM) is the widely used analgesic drug that can result in liver damage, which is one of the leading causes of poisoning worldwide, especially when overdose or long-duration intake is commonly associated with liver damage.4,5 Paracetamol–induced liver damage accounts for 39% of all cases of acute hepatic injury and 20% of the liver transplant cases in the USA.4,6-9 The initial indicators of PCM-induced hepatotoxicity are characterized by elevated levels in liver function tests, including aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), and bilirubin.10-12
Normal therapeutic dose of PCM 90% of it is metabolized and converted into a nontoxic form by phase II conjugating enzymes and finally excreted with the urine. The rest of PCM is metabolized through cytochrome P450 enzymes (CYP450) and converted into N-acetyl-p-benzoquinone imine (NAPQI). However, in the case of overdose, intake of PCM and metabolism through CYP450 cause the accumulation of the toxic molecule NAPQI. Antioxidant glutathione (GSH) binds and detoxifies oxidant NAPQI. This leads to the depletion of GSH and increases the generation and accumulation of reactive oxygen species (ROS), resulting in oxidative stress (OS). OS causes covalent joining of sulfhydryl groups of amino acid residues in cellular proteins, as well as mitochondrial proteins. The resulting mitochondrial dysfunction ultimately causes liver cell necrosis. Hepatocyte necrosis changes the permeability of hepatocytes and increases leakage of cellular enzymes to the blood.13-19
Current treatments for liver diseases are often ineffective and frequently associated with undesirable side effects and high costs. 20 Consequently, there is a critical need to explore viable and effective alternatives with therapeutic or protective potential. Recently, natural products have attracted considerable interest due to their affordability and favorable safety profiles. 21
Bersama abyssinica (Melianthaceae) belongs to the genus Bersama and comprises four other species, which are Bersama engleriana, Bersama swynnertonii, Bersama swinnyi, and Bersama yangambiensis. 22 It is distributed in the Democratic Republic of Congo, Tanzania, Mozambique, Zambia, Zimbabwe, Angola, Nigeria, Ethiopia, Kenya, Sudan, and Uganda.23,24 Bersama abyssinica Fresen grows in several parts of Ethiopia and is locally recognized as Azamir (Amharic) and Lolchissa (Afaan Oromo). 25 Traditionally, B. abyssinica has various treatment purposes, such as cancer, 26 rheumatism, 27 ascariasis, 28 microbial infections, 29 tonsillitis, 30 roundworm, 31 liver and kidney diseases. 32 In addition, several scientific studies have demonstrated that it has in vivo anti-diarrheal, 33 anti-malarial, 34 anti-hyperglycemic, anti-hyperlipidemic, 35 antipyretic, 36 and wound-healing 37 activities.
In Ethiopia, an ethnobotanical survey conducted in the Ari and Male communities showed that the bark and buds of Bersama abyssinica are used for treating liver diseases. 38 The Sidama people of Dale District in Southern Ethiopia use the bark by powdering, dissolving, boiling, and consuming small amounts to treat liver disease. 39 The leaves of Bersama abyssinica also contain secondary metabolites such as flavonoids, alkaloids, terpenoids, phenols, and steroids.40,41 These active ingredients are thought to play a major role in the reduction of inflammation and oxidative stress-induced liver damage via detoxification of free radicals and enhancing the body’s antioxidant scavenging system. 42 It prevents liver cell membrane lipid peroxidation and necrosis to liver tissue mainly through its ability to inhibit the formation of OS within the liver and liver mitochondrial dysfunction. Medicinal plants that exhibit antioxidant, anti-inflammatory, and antihyperlipidemic properties also demonstrate protective effects on the liver, particularly in conditions like liver fibrosis and non-alcoholic fatty liver disease (NAFLD).43-45 According to findings from earlier studies, Bersama abyssinica demonstrated antioxidant, anti-inflammatory, antidiabetic, and antihyperlipidemic activities.23,46,47 These results support the notion that the extract of Bersama abyssinica may possess hepatoprotective properties. Nevertheless, to the best of our knowledge, no previous study on hepatoprotective activities of Bersama abyssinica had been reported. Therefore, the current study aimed to explore the hepatoprotective activities of the methanolic leaf extract of Bersama abyssinica against paracetamol-induced hepatotoxicity in Swiss albino mice.
Methods and Materials
Study Area and Period
Phytochemical screening and extraction of Bersama abyssinica Fresen leaves were conducted at the Biochemistry Laboratory, Organic and Inorganic Chemistry Postgraduate Laboratory of Jimma University, and biochemical and histopathological investigations were done at the Jimma University Medical Center. The extract of Bersama abyssinica Fresen leaf was administered to mice to evaluate its hepatoprotective effects from December 2 to 16, 2021, at the Jimma University Tropical and Infectious Disease Research Center in Sokoru, Jimma, Ethiopia.
Study Design
A post-test control group experimental study design was conducted on a mouse model to assess the hepatoprotective effect of Bersama abyssinica Fresen leaf methanolic extract.
Chemicals and Reagents
Chemicals and reagents used in this study included methanol (assay (GC) > 99.9%), ethanol, acetone (ACA 280223), chloroform (EMSURE© ASC ISO), petroleum ether, sulfuric acid (Laboratory use 98%), hydrochloric acid, iodine, sodium hydroxide, potassium iodide, ammonia, copper sulfate, silymarin, PCM, distilled water, normal saline, 10% buffered formalin (Brand-Microxpress 500 mL, histological fixative), paraffin wax, ketamine, xylazine, hemato-xylin (Harris) and eosin, liver function test reagents.
Apparatus and Instruments
The types of equipment that were used for this study were: Whatman filter paper no.1, Cotton, electric grinder, Test tube, Dropper, Flasks (2L, 5L, 0.5 L), Buchner funnel, Rotary evaporator (Buchi Rota Vapor, Switzerland), Beaker (10 mL, 100 mL, 800 mL), Blunt needle, ice box, Stainless Steel and Plastic Cage, Oral gavage needle (24G), Syringe, Centrifuge (Gemmy industrial corp.,Taiwan), Micropipettes (Human Pipette) (20 μL, 20-200 μL, 1000 μL), Eppendorf tubes, Electrical balance (A&D company, Limited Tokyo Japan), Refrigerator (Beko), Open tissue processor (LeicaTP 1020, Germany), Tissue embedder (Tissue-Tek), Mold (tissue cap), Wax dispenser (Electrothermal Model: MH8523, China), ribbon Microtome (Leica Model: TP 1020, Germany), Slides, Light microscopy (Olympus CX21FS1, Philippines), Oven (Gallen Kamp), Desiccator, Fully automated serum analyzer (Cobas R-6000), Tissue cassettes, and automated built-in digital photo camera (Camera KRUSS optronic Germany 3.0 MP USB 2.0).
Plant Collection and Preparation
Leaves of Bersama abyssinica Fresen were collected from Jiren Mountain, near Jimma town in the Oromia region of Ethiopia. The plant was identified and authenticated by Melaku Wondafrash, a plant taxonomist at the National Herbarium of Addis Ababa University, and assigned the voucher number SM-001. The plant leaf was deposited at the herbarium for further reference. After collection, the leaves of Bersama abyssinica Fresen were washed with tap water to remove dust and debris, and then air-dried in a shaded area at room temperature.
Plant Extraction
The extraction was done in the Organic Chemistry Postgraduate Laboratory at Jimma University. After the leaves were dried subjected into coarse powder using an electric grinder and 250 grams of the powdered material were weighed using a digital balance and soaked in 2.5 liters of methanol (GC grade, >99%) in a 1:10 ratio. The mixture was kept in a flask for 72 hours and mechanically shaken three times daily for three consecutive days. This extraction process was repeated until the plant residue no longer produced any coloration, ensuring maximize yield. The extracts was then filtered using Whatman filter paper No. 1 Methanol was removed from the filtrate using a rotary evaporator (Model-Laborota 4000 and Brand-Heidolph) at 90 rotations per minute at 50°C, followed by further concentration using a water bath at 40°C. To eliminate any residual methanol, the extract was dried in an oven at 40°C. A total of 31.226 grams of crude extract was obtained, packed in airtight glass bottles with appropriate labeling, and stored in a refrigerator at 4°C until further use. The percentage yield of the extract was calculated by using the following formula.
Phytochemical Screening of the Plant
Testing for alkaloids, flavonoids, phenols, steroids, quinones, saponins, tannins, and glycosides of the Bersama abyssinica Fresen leaf was done using different solvent systems. Different solvent systems: methanol, ethanol, hydro-methanol, hydro-ethanol, acetone, petroleum ether, and chloroform were used. Tests for phytochemicals were done using the following procedures obtained from previous studies.48-50
The phytochemical analyses were performed using the following standard methods:
Test for alkaloid (Mayer’s test): The plant extract was mixed with a few drops of Mayer’s reagent and looked for white creamy precipitation, which showed the presence of alkaloids. 48
Test for quinones (Borntrager’s test): 1g of plant powder was dissolved in 20 mL of chloroform in a test tube, and the mixture was placed in a steam bath for 5 minutes. The mixture was then filtered (while hot) and allowed to cool. Equal parts of the filtrate and 10% ammonia solution were mixed and shaken well, and the presence of a pink color on the upper parts of the aqueous layer indicated anthraquinones. 51
Test for steroids: The presence of steroids was determined using chloroform and concentrated sulfuric acid (H2SO4) (2 mL of chloroform + H2SO4 + 5 mL crude extract → red colour layer). 49
Test for phenolic compounds (Ferric chloride test): 50 mg of plant extract was dissolved in 5 mL of distilled water, a few drops of ferric chloride solution (5% neutral) were added, and the dark green color product was examined. 52
Test for tannins: half a gram of plant extract and bromine water (10 mL) were mixed, and the decolorization of bromine water indicated the presence of tannins.35,53
Test for saponins: 50 mg of the plant extract was dissolved in 20 mL of distilled water. The mixture was shaken for 15 minutes in a cylinder, and the presence of saponins was indicated by the formation of a 2 cm layer of foam. 54
Test for flavonoids: The Shinoda test or alkaline reagents test was used to detect the presence of flavonoids (Mg2+ ribbon + concentrated HCL + crude extract → pink color or 2% of NaOH + crude extract → gives a deep yellow color +2 drops of diluted acid → colorless).55,56
Salkowski’s test was used to check for the presence of glycoside components (2 mL of H2SO4 + plant crude extract → reddish-brown color). 56
Oral Acute Toxicity Test
An acute oral toxicity test was performed on five female Swiss albino mice aged 6 to 8 weeks and weighing between 25 and 36 grams, were randomly selected and housed in a laboratory cage for a week to acclimatize to laboratory conditions prior to the beginning of the study to determine the median lethal dose (LD50) of the Bersama abyssinica leaf extract, following Organization for Economic Co-operation and Development (OECD) guideline number 425.57,58 During this acclimatization period, the mice were maintained under a 12-hour light/dark cycle at room temperature, with free access to standard food pellets and tap water, and fasted thereafter for three hours. 59 After the body weights of the mice were recorded, 2000 mg/kg of the extract was given via oral gavage. The mouse was closely observed during the first 30 minutes, for 24 hours, and then daily for 14 days after dosing for behavioral changes and signs of toxicity, including tremors, convulsions, salivation, lethargy, and coma.
Grouping of Experimental Animals and Dosing
Adult male Swiss albino mice weighing 25-36 grams, which are healthy, were used in this study. The PCM dose used to induce hepatic toxicity was adopted from the previous study. 60
Grouping of Experimental Animals and Treatment Protocol for a Period of 14 Days
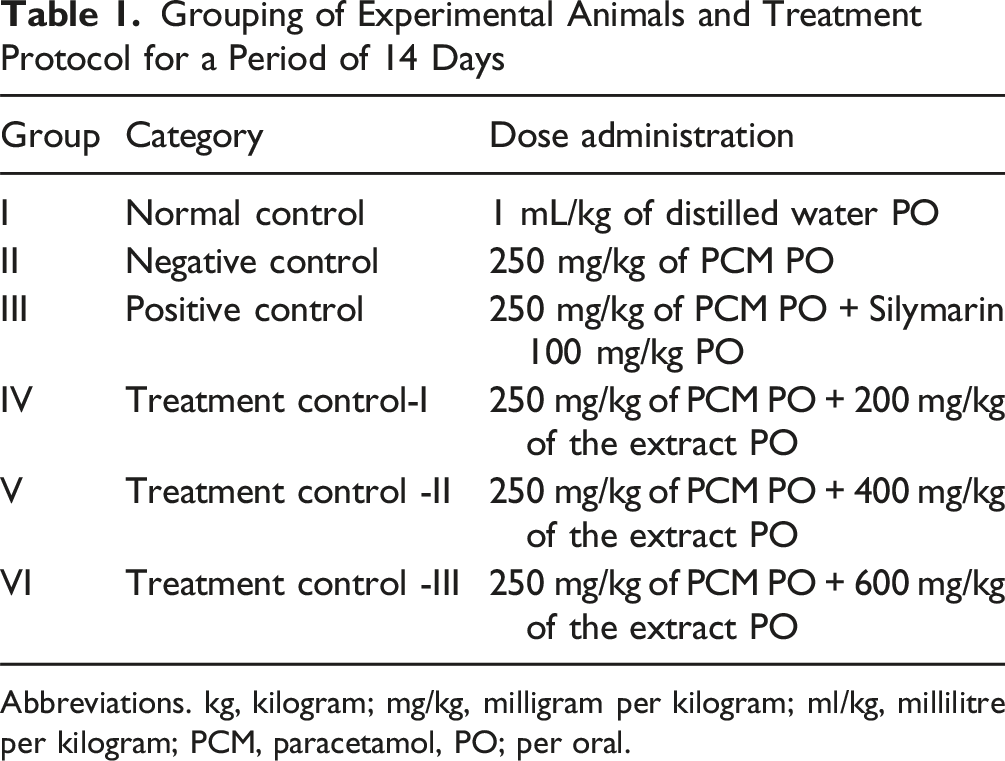
Abbreviations. kg, kilogram; mg/kg, milligram per kilogram; ml/kg, millilitre per kilogram; PCM, paracetamol, PO; per oral.
Group I (normal control) received 1 mL/kg of distilled water per oral (PO) for 14 consecutive days.
Group II (negative control) received PCM 250 mg/kg PO suspended in normal saline for 14 consecutive days.
Group III (positive control) received 100 mg/kg Silymarin PO and 250 mg/kg PCM 3 hours after the administration of Silymarin for 14 consecutive days.
Group IV (treatment control-I) received 200 mg/kg of Bersama abyssinica leaf extract PO for 14 consecutive days and 250 mg/kg PCM.
Group V (treatment control-II) received 400 mg/kg of Bersama abyssinica leaf extract PO for 14 consecutive days and 250 mg/kg PCM.
Group VI (treatment control-III) received 600 mg/kg of Bersama abyssinica leaf extract PO for 14 consecutive days and 250 mg/kg PCM.
All mice found in groups IV-VI were treated with PCM (250 mg/kg body weight) 3 hours following the 14th day administration of the extract.
Blood Collection and Serum Preparation from Mice
At the end of the experimental period, 24 hours after the last dose was given, all the experimental animals were fasted overnight and anesthetized with a combination of ketamine and xylazine (intramuscular, 500 mg/10 mL (50 mg/ml)), and approximately 2-2.5 mL of blood was collected from each mouse via cardiac puncture using the diaphragmatic approach,61,62 which was performed by inserting a 1-inch, 22-gauge needle attached to a 3 mL (3 cc) syringe through either notch. The collected blood samples were kept in an Eppendorf tube for 30 minutes at room temperature and centrifuged at 3000 rpm for 10 minutes to completely separate the serum from the formed element. Then, the serum was separated using a micropipette into a tube and placed in an icebox (4-8°C) until it was analyzed.
Biochemical Analysis
Serum samples were analyzed for liver function tests (ALT, AST, GGT, and ALP) using a fully automated serum analyzer (Cobas R 6000), following the principles and procedures outlined in the manufacturer’s manual. The serum bilirubin levels were also determined using commercial diagnostic kits. 63
Histopathological Study
At the end of the 2-week experimental period, the mice were sacrificed, and liver tissues were carefully harvested from each animal. The tissues were excised using sterile scissors and handled with blunt forceps to minimize damage. Each tissue sample was fixed in 10% neutral buffered formalin for 12 hours. Following fixation, the tissues were taken out of the preservative and washed with ethanol at concentrations of 70%, 80%, 95%, and 100%. After that, xylene solutions were used to remove ethanol from the tissue and replace this ethanol with a fluid that is readily miscible with paraffin wax, which enhances the tissue’s ability to embed easily with the wax to form tissue blocks. Then after, the tissue block was sectioned into 4-5 µm thickness by precision knives (microtome), and that section was immersed in a water bath at 45°C. The unfolded section was then taken and dried by putting it in an oven at 56°C for 20 minutes. Then, this dried section was stained with hematoxylin and eosin (H&E) stain after the removal of paraffin wax by using ethanol with a descending concentration and examined by using a light microscope under 400X magnification. Images were taken using a microscope camera, and pictures were read and interpreted by histologists and pathologists. 64
Data Analysis Procedures
Data were entered into EpiData software version 3.1, checked for completeness and accuracy, and then exported to SPSS version 25.0 software for analyses. Results were presented by tables and figures and expressed as mean ± SEM. Statistical analysis were performed using one-way analysis of variance (ANOVA), followed by a post hoc Tukey test with multiple comparisons. A P-value of <0.05 was considered statistically significant.
Results
Acute Oral Toxicity Test
Throughout the followup period none of the tested mice dosed the leaf extract of Bersama abyssinica Fresen at 200 mg/kg show signs of toxicity and died. So that the LD50 for the leaf extract of Bersama abyssinica Fresen was considered to be greater than 2000 mg/kg body weight. Therefore, 200, 400 and 600 mg/kg of the Bersama abyssinica Fresen leaf extract dosages were chosen for our current experimental study.23,65
Phytochemical Screening Test
The Result of Phytochemical Screening of Bersama abyssinica Fresen

Note. (−): Absent, (+): Low concentration, (++): Moderate concentration, (+++): High concentration.
Effect of Bersama abyssinica Fresen Leaf Methanolic Extract on Liver Enzyme Parameters in Paracetamol-Induced Mice
Effect of Bersama abyssinica Fresen Leaf Methanolic Extract on Liver Enzyme Parameters in Paracetamol Challenged Mice

Abbreviations. ALP; Alkaline phosphatase, ALT; Alanine amino transferase, AST; Aspartate amino transferase, GGT; gamma-glutamyl transferase, IU/L; International unit per liter.
Note. The results were expressed as mean ± SEM.
asignificant difference as compared to normal control.
bsignificant difference as compared to negative control.
The Levels of Direct, Indirect and Total Bilirubin
The current study also found that the mice treated with the PCM alone developed hepatotoxicity as seen by the increased level of indirect and total bilirubin when compared to the normal control (P < 0.05). However, administration of the leaf methanolic extract significantly reduced these increased levels compared with PCM-only-treated mice (P < 0.05) (Figure 1). Regarding the serum levels of direct bilirubin, insignificant results were found across all the groups. The serum levels of direct, indirect and total bilirubin among the groups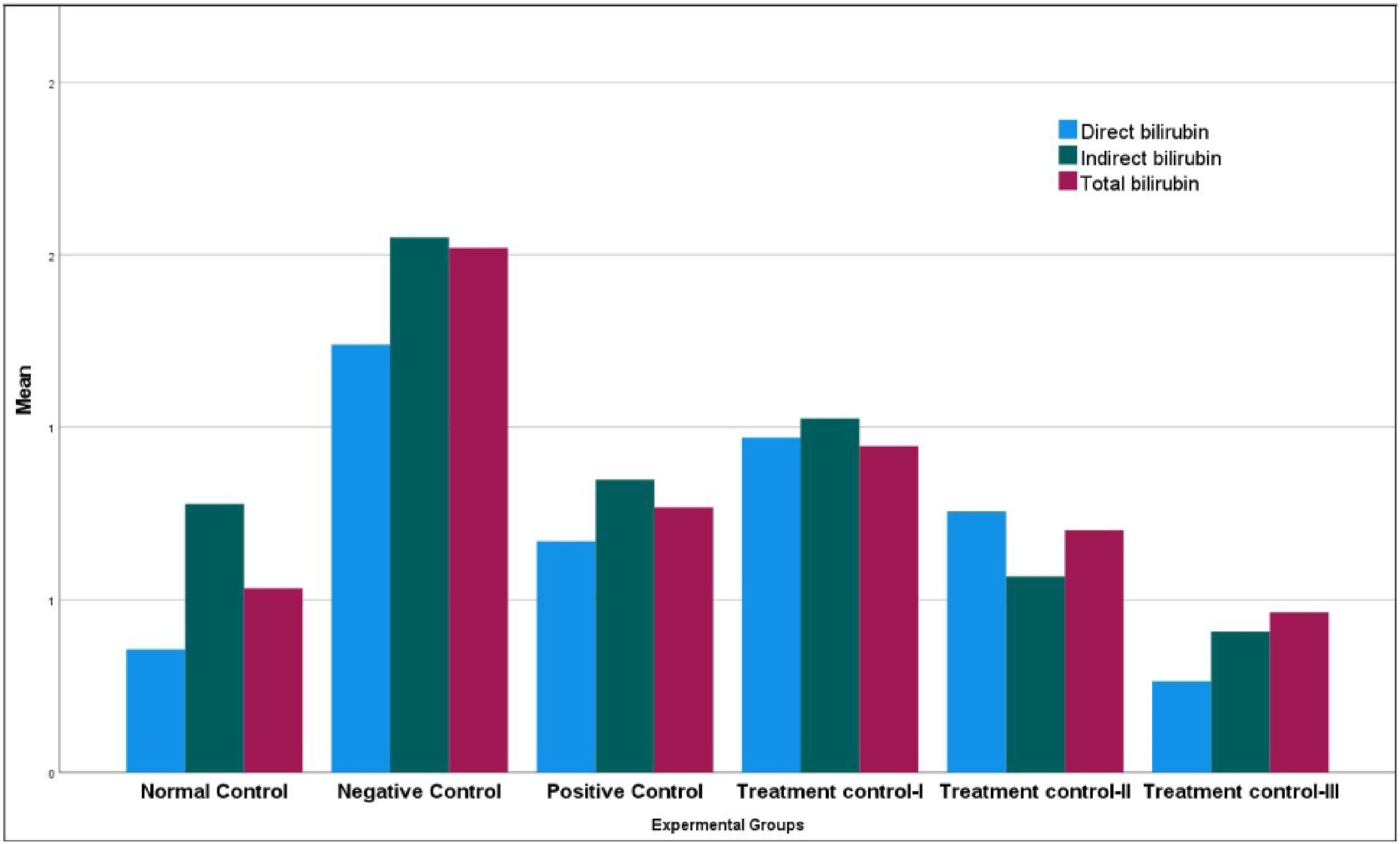
Effect of Bersama abyssinica Fresen Leaf Methanolic Extract on Liver Tissue Histopathology
The microscopic examination of the liver tissue of the normal control group indicates normal hepatic architecture with a normal central vein and normal sections of liver cells. However, after intoxication by PCM, they lost their normal hepatic architecture and showed hepatic cholestasis, lobular and lymphocytic inflammation, necrosis, vacuolar degeneration, and sinusoidal dilation, as displayed by Figure 2. In contrast, the positive control group taking silymarin showed normal liver architectures. Similarly, the mice treated using methanolic extract of Bersama abyssinica Fresen leaf showed significantly regenerated the liver architecture induced by PCM, and a dose-dependent difference in regenerative capacity was observed between the treatment groups which is more in the treatment control-III. (The details are presented in Figure 2 below). Photomicrograph Liver tissue of the Swiss albino mice (at 400x)
Discussion
In the present study, we tried to assess the serum levels of ALT, AST, ALP, GGT, and bilirubin and examined the histopathology of the liver tissue. We found elevated levels of ALT, AST, ALP, GGT, and bilirubin in the serum and abnormal liver histopathological changes among the negative control group as compared to the normal control, which were reduced after administration of the methanolic leaf extract.
Excessive intakes of PCM cause an increase in the formation of the oxidant compound NAPQI because it is metabolized by CYP450 enzymes system, which detoxifies PCM into NAPQI, which is that again detoxified by binding with GSH, ultimately resulting in the reduction of GSH. 66 The reduction of GSH enhances the generation of ROS resulting in the formation of OS. OS once formed damages the hepatocyte membrane with lipid peroxidation, mitochondrial dysfunction, and necrosis, which causes the loss of functional integrity of hepatocytes. 67 These all cause an increase in the permeability of liver cells and leakage of cellular enzymes (ALT, AST, and ALP) and bilirubin towards the blood.13-19
According to the result of the current study, Bersama abyssinica Fresen leaf methanolic extract treatment at doses of 400 and 600 mg/kg showed a significantly lower (P < 0.05) level of ALT as compared to the negative control group, which showed that treatment of the mice with Bersama abyssinica Fresen leaf methanolic extract restored the serum level of liver enzymes to near normal. This result was in harmony with the results of the studies done on the hepatoprotective effects of Heliotropium strigosum and Acacia nilotica extract against PCM-induced hepatotoxicity.68,69 This observed hepatoprotective effect of Bersama abyssinica Fresen leaf extract might be due to the presence of anti-inflammatory and antioxidant secondary metabolites like alkaloids, tannins, flavonoids, saponins, and glycosides that restore the injured hepatocytes and elevate the liver enzymes.46,47
In the present study the significant reduction of the serum level of ALP was found among Groups III (only taken silymarin), V (400 mg/kg of extract dose) and VI (600 mg/kg of extract dose),as compared to the negative control group (P < 0.05). However, the mice treated by the extract at a dose of 200 mg/kg showed an insignificant reduction in ALP levels as compared to the mice treated only by PCM (group II). This finding was inconsistent with the previous studies.70-72 Despite the fact that the exact reasons for these variations are unknown, possible explanations might be due to the amount of inducing dose, duration of the experiment, and different experimental animals involved in the study.
In the current study the serum level of AST was also investigated, and it was found that the leaf methanolic extract of Bersama abyssinica Fresen treatment restored the elevated level of AST caused by PCM when compared with the negative control group, which is congruent with the finding of the previous studies.69,73,74 In addition, our study revealed that the serum level of GGT was significantly higher in the group of mice treated by PCM alone as compared to the normal control, and the significant reduction of this higher level was seen among groups V and IV mice. This was in harmony with a study result done in Egypt on the Hepatoprotective effects of artichoke leaf and rosemary extract against paracetamol-induced toxicity in albino Rats. 75 The reduction of AST and GGT was likely due to the ability of the extract to inhibit liver cell membrane lipid peroxidation and mitochondrial dysfunction caused by the formation of OS. 54
Likewise, it was seen from the results of this study that treatment of the mice by the extract’s gives the best protection to the liver due to the extract ability to significantly restore and reduce the PCM induced increased serum levels of indirect and total bilirubin, which is almost parallel to silmarin (P < 0.05). This result was in line with the study results done on methanol extract of Agave americana leaves on paracetamol-induced hepatotoxicity in Wistar albino rats (Hepatot) and the hepatoprotective effects of Malvects of Malva sylvestris L. against. 76 This could be explained by the antioxidant activity of the extract enhancing the liver cells’ ability to conjugate the bilirubin due to it protecting the liver cells from the damage resulting from ROS, which may lead to decreased conjugation and excretion of bilirubin that elevates bilirubin in the plasma.77,78
The phytochemical screening of the current study plant extract also showed the presence of saponins, flavonoid, tannin, and alkaloid that have hepatoprotective activities as a result of possessing antioxidant, and anti-inflammatory effects.78-81
Moreover, there were abnormal histopathological changes, such as cholestasis, necrosis, inflammation, sinusoidal dilation, and vacuolar degenerations observed in the liver sections of the mice intoxicated by PCM (negative control group), which were regenerated significantly after treatment with plant extract and Silmarin (group III). This is in agreement with the findings of earlier studies.60,82.72,74 These abnormal histopathological changes of the liver tissue caused by PCM were possibly due to the PCM-mediated accumulation of the toxic metabolite NAPQI, which is responsible for GSH reduction and acute centrilobular hepatic necrosis resulting from OS. The possible reasons for improved liver architecture seen after Bersama abyssinica Fresen leaf extract administration could be due to the antioxidant and anti-inflammatory effects of flavonoids, alkaloids, and glycosides found in it.51,83
Even though the present study tested the hepatoprotective effect of the plant extract, it is not without limitations: we were unable to include all parts of Bersama abyssinica Fresen and identify the standard antioxidant marker compounds from the extract. This study does not investigate the molecular mechanism of action through which the extract protects the liver, and also does not investigate and measured phytochemicals quantitatively.
Conclusion
From the current study it is concluded that the methanolic leaf extracts of Bersama abyssinica Fresen showed hepatoprotective activity similar to silymarin in a dose-dependent manner due to the restoration of the higher levels of liver function tests as well as the regeneration of abnormal histopathological changes of the liver tissue caused by PCM. So, the current study finding might provide scientific justification for the social claim in the use of Bersama abyssinica Fresen. Further experimental research is needed on the hepatoprotective effects of Bersama abyssinica Fresen leaf extract using different solvents. Additionally, we are eager to recommend that researchers conduct further studies on the quantitative phytochemicals, antioxidant enzymes (reduced Glutathione (GSH), Superoxide Dismutase (SOD), or Catalase (CAT)), and oxidative stress markers (Malondialdehyde (MDA)) assessment in liver tissue. Conducting studies on the different parts of Bersama abyssinica Fresen other than the leaf is also recomended. Moreover, we love to recommend doing further studies to determine the molecular mechanisms of how the extract protects the liver and its long-term toxicity to confirm whether it is safe for humans. 59
Footnotes
Acknowledgements
The authors would love to acknowledge the Biochemistry Unit for giving us a chance to work on this study and the Organic and Inorganic Chemistry Postgraduate Laboratory of Jimma University for providing their laboratory space to us, as well as the data collectors at Jimma University and the Tropical and Infectious Disease Research Center for their unreserved contributions.
Ethical Consideration
This study was conducted after getting an ethical clearance letter from the institutional review board of Jimma University with a reference number of IHRPGD/10/21. A support letter for the Jimma University Tropical and Infectious Disease Research Center was taken from the Department of Biomedical Sciences at Jimma University. All procedures on mice were carried out in accordance with the NIH Guidelines for Care and Use of Laboratory Animals to protect the rights of experimental animals and to avoid pain and injury.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.

