Abstract
Abstract
The pandemic caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is far from over as new strains are emerging all over the world. Selenium as a micronutrient is important for immunity and also has anti-viral activity.
Objective
The study evaluated the activity of a Selenium enriched garlic powder (SeGP or SelenoForce®) against SARS-CoV-2 viral replication in vitro and explored its possible mechanism of action.
Methods
The anti-SARS-CoV-2 activity assay was carried out in Vero E6 cells in vitro. Human lung carcinoma A549 cells were used to study the antioxidant activity, expression of angiotensin converting enzyme (ACE), transmembrane protease, serine 2 (TMPRSS2) and the activity of proprotein convertase, and furin. Anti-inflammatory activity was evaluated in lipopolysaccharide-activated RAW 264.7 cells.
Results
SeGP inhibited the replication of SARS-CoV-2 in Vero E6 cells with an IC50 of 19.59 μg/ml. It exhibited significant antioxidant activity in vitro with IC50 value determined as 43.45 μg/ml. The Selenium enriched product inhibited the expression of ACE and TMPRSS2 and also showed inhibition of furin protease activity. In the presence of SeGP, the secretion of nitric oxide, interleukin −6 and TNF-α were reduced in activated RAW 264.7 macrophages.
Conclusion
The results of the study suggest that Selenium enriched garlic powder could inhibit SARS-CoV-2 multiplication in vitro, reduce oxidative stress and inflammatory mediators suggesting that it could be developed as an effective supplement or adjunct therapy to combat viral infections.
Introduction
Infectious viral diseases pose a massive threat to public health and remain as one of the major causes of death all over the world. Viruses are called “acellular parasites of cellular hosts” as they utilize the host cellular mechanisms for propagation and replication which make it difficult to destroy them without affecting the host cell. 1 Immunization with effective vaccines has led to the eradication of few viral diseases like polio and smallpox and is effective to combat other viral diseases including, hepatitis virus, herpes simplex virus (HSV), respiratory syncytial virus (RSV), and influenza virus. 2 However, the emergence, resurgence and spreading of animal viruses have become serious public concern in the past few decades despite the tremendous progress in human medicine. The scenario got worsened when the world population was adversely affected by novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), responsible for the pandemic, Coronavirus disease-2019 (COVID-19). SARS-CoV-2 infection primarily induces pro-inflammatory cytokines (eg, IL-1 and IL- 6) and lung inflammation. It ultimately affects the normal functioning of vital organs enhancing comorbidities and mortality. 3 Till now, the SARS-CoV-2 virus has infected around 7 billion people and caused over 6 million deaths around the world (https://www.worldometers.info/coronavirus).
In addition to vaccines, drugs also become life-saver at the time of the pandemic. A treatment strategy combining the pharmaceutical drugs and herbal medicines as an adjunct therapy was found effective in managing the COVID-19 infection. 4 Natural products, especially plants are a rich source of pharmacologically active substances and become excellent candidates for drug discovery due to their chemical diversity and safety. Several clinical studies provide evidence for the efficacy and safety of herbal extracts and phytochemicals as potential immune boosters to defend against SARS-CoV-2 infection.5-10 Additionally, novel formulations of natural products were found to be targeting multiple signaling pathways of SARS-CoV-2 infection and were suggested as supportive therapy to hasten recovery and reduce severe morbidity and mortality in patients. 11 An understanding of the molecular mechanisms of viral invasion and replication will enable us to design anti-viral drugs from natural products targeting the different stages of the viral replication cycle. Inhibitors of angiotensin-converting enzyme 2 (ACE2), transmembrane protease, serine 2 (TMPRSS2) and furin, the protease that activates the spike protein of SARS-CoV-2 virus were found to be effective in reducing viral load which make it a potent therapeutic approach for treatment of SARS-CoV-2 infection. 12 Further, antioxidant and anti-inflammatory activities are beneficial in preventing the secondary complication of the infection.13,14
Micronutrients like vitamins and essential minerals are crucial in every stages of the immune response. Micronutrient deficiencies were found associated with immunosuppression and increased susceptibility to infections. 15 Selenium (Se), an essential micronutrient has a long history of lowering the severity of viral infections.16,17 The source and chemical form of Selenium in food are important factors affecting its bioavailability in humans. Generally, organic Selenium compounds are more bioavailable than inorganic Selenium compounds and Selenium from plant sources shows better bioavailability than that of animal sources.18,19 The physiological functions of Selenium are mainly executed by selenoproteins that are crucial for maintaining the redox balance in cells, and controling other functions such as cell proliferation, differentiation, apoptosis, cytokine expression, and anti-viral activities.20,21 Selenium is known to lower the susceptibility and severity of viral infections and consequently, its deficiency impacts immune response to viruses, expression of viral proteins and antioxidant response.22-24 Selenium status was reported to have a positive correlation with the survival of patients with COVID-19 compared with non-survivors suggesting that Selenium supplementation may be helpful in reducing the comorbidities associated with COVID-19.25-27
Garlic or Allium sativum L. is popular as a functional food and a traditional medicine since ancient time. It contains organosulfur (allicin and alliin) compounds and flavonoids like quercetin which are found to be responsible for anti-viral activities via immune modulation.
3
Selenium enriched garlic powder (SeGP) is manufactured using a proprietary soil-less culture technique containing a unique composition of organoSelenium compounds. In this process, garlic bulbs are naturally enriched with selenoamino acids, L-methyl selenocysteine (MSC) and
Materials
SeGP (marketed as SelenoForce®) was obtained from Sami-Sabinsa Group Limited. Dulbecco’s modified minimal essential medium (DMEM) from Life Technologies (CA, United States) and fetal bovine serum (FBS) from GIBCO/Invitrogen (Carlsbad, CA, United States), Trizol reagent® from Invitrogen (Carlsbad, CA, United States), Revert-aid First Strand cDNA synthesis kit from Thermo Fisher Scientific and ELISA kits from R&D Systems (Minneapolis, MN, United States) were used in the study. pERTKR-AMC (L-pyroglutamyl-Arg-Thr-Lys-Arg-7-amino-4-methylcoumarin) was purchased from R&D Systems (Minneapolis, MN, United States).
Methodology
Cell Culture
In vitro experiments in Vero E6 cells (derived from the kidney of African green monkey) were carried out at the Institute of Life Sciences, Bhubaneswar, an autonomous institute of the Department of Biotechnology, Govt. of India.
Human lung carcinoma A549 and RAW 264.7 macrophage cells were procured from the National Center for Cell Sciences (NCCS), Pune, India and maintained at 37°C in a 5% CO2 atmosphere, in complete Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% of fetal bovine serum (FBS) and 40 μg/ml gentamycin.
Preparation of SeGP for in vitro Experiments
SeGP was dissolved in dimethyl sulphoxide (DMSO) at a concentration of 10 mg/mL, sonicated in a water bath sonicator at room temperature for 5 min and centrifuged at 2000 r/min for 5 min to remove the insoluble particles from garlic bulb. The stock was used for the preparation of different dilutions in culture media or buffers for in vitro experiments. The in vitro activities were studied at 50, 25, 12.5 and 6.2 μg/ml of SeGP which correspond to approximately 50, 25, 12.5 and 6.5 ng/mL of selenoamino acids respectively.
In vitro Anti-SARS-CoV-2 Activity
In vitro anti-SARS-CoV-2 activity assay was carried out in Vero E6 cells (derived from the kidney of an African green monkey) at the Institute of Life Sciences (ILS) Bhubaneswar, an autonomous institute of the Department of Biotechnology, Govt. of India, with established platforms for drug screening for viral diseases. SARS-CoV-2 strain ILS-03 (Accession no. EPI_ISL_1196305) successfully adapted and isolated in Vero cells at ILS, Bhubaneswar was used for this study. No animals/humans were used in the study.
Cytotoxicity Assay
Briefly, Vero E6 cells were seeded in the 96 well plate at 80% confluency and treated with various concentrations of SeGP. For toxicity determination, minimum of 3 concentrations were used. Cytotoxicity was determined 24 h post-treatment using (3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide) assay (MTT assay). 100 μl of MTT solution (5 mg/mL) was added to 96 wells and incubated for 2-3 h at 37°C. After incubation, 100 μL of DMSO was added to each well to solubilize the formazan crystals, under shaking conditions. The absorbance was recorded at 570 nm. Each concentration was assayed in triplicates and the percentage cell viability was calculated with respect to vehicle control.
Anti SARS-CoV-2 Activity Assay
Vero E6 cells seeded in 96-well plates at 80% confluency were infected with SARS-CoV-2 isolate at multiplicity of infection (MOI) of 0.1 for 2 h. Subsequently, the inoculum was aspirated and fresh media containing different concentrations of the SeGP was added to the cells. The supernatant of 24 h post-infection was subjected to viral RNA isolation followed by qRT-PCR for determining the SARS-CoV-2 viral load in the cells (cell associated) and culture supernatants (released virus particles). The qRT-PCR was performed using primers specific for the viral spike, nucleocapsid and ORF1a for both released and cell-associated virus. Percentage reduction of viral loads in cells and culture supernatants was plotted in comparison to vehicle treated controls. Remdesivir was used as a positive control.
In vitro Assays in Human Lung Carcinoma A549 Cells and RAW 264.7 Cells
Cytotoxicity/Selection of Nontoxic Concentrations for Cell Assays
Human lung carcinoma A549 cells and RAW 264.7 cells (5 × 104 cells/well) were exposed to varying concentrations of SeGP for 24 h. Cytotoxicity/cell viability of SeGP was analyzed by using MTT colorimetric assay according to the standard method described previously. 28
H2O2 Induced Reactive Oxygen Species Scavenging Activity
Human lung carcinoma A549 cells were seeded at 5 × 104 cells/well in 96 well black plates and allowed to grow as a monolayer for 24 hours. Cells were exposed to 25 mM hydrogen peroxide (H2O2) with or without the test sample for 4 hours in DMEM supplemented with 2% FBS. Freshly prepared dichlorodihydrofluorescein diacetate (DCFH-DA) reagent was added to all the wells (1 μg/well). The plate was incubated at 37°C for 30 min and fluorescence was recorded at 485:520, Ex:Em wavelength in microplate reader (FLUOstar Optima, BMG Labtech, Germany). 29
Furin Inhibition Assay
Human lung carcinoma A549 cells were seeded at 5 × 104 cells/well in 96 well black plates and allowed to grow as a monolayer for 24 h. Cells were treated with samples for 24 h in DMEM containing 2% FBS. Wells with only DMEM +2% FBS media served as control. After 24 h of incubation, media was replaced with 25 μM fluorogenic peptide substrate, pERTKR-AMC (L-pyroglutamyl-Arg-Thr-Lys-Arg-7-amino-4-methylcoumarin) as substrate in 25 mM Tris buffer (pH-9, 1 mM CaCl2, 0.5% (w/v) Brij-35, 0.1% BSA) for 1 h. The fluorescence was recorded at 380:460, Ex:Em wavelength in microplate reader (FLUOstar Optima, BMG Labtech, Germany). Furin activity was calculated in comparison to control.
ACE-2 and TMPRSS2 Inhibition Assay
List of Primer Sequences.

Anti-Inflammatory Assay
RAW 264.7 cells (0.5 × 105 cells/well) were seeded into 96 well cell culture plates in growth media. The cells were stimulated with 100 ng/mL lipopolysaccharides (LPS) along with different concentrations of sample and incubated for 24 h at 37°C, in a CO2 (5%) incubator. The wells without LPS (100 ng/mL) served as control. The supernatants were collected to detect the levels of inflammatory markers. The inducible nitric oxide (NO) level is detected based on the Griess reaction. Briefly, 50 μL of cell culture medium was mixed with 50 μL of Griess reagent I [1% (w/v) sulfanilamide in 5% (v/v) phosphoric acid] and 50 μL Griess reagent II [0.1% (w/v) N-(1-Naphthyl) ethylenediamine dihydrochloride], incubated at room temperature for 10 min, and then, the absorbance was taken at 540 nm. The inflammatory markers like tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) were quantified using ELISA kits as per the manufacturer’s instruction (R & D Systems, Minneapolis, MN, United States).
Statistical Analyses
All data are presented as the mean ± standard deviation (SD) of the triplicates. P values were determined by one-way analysis of variance (ANOVA) using GraphPad prism software version 10.0.2 (GraphPad Software, Inc, La Jolla, CA, USA). Significance was defined at 5% level.
Results
Selenium Compounds Present in SeGP
Garlic powder, naturally enriched with Selenium contained selenoamino acids and dipeptides as major Selenium compounds (Figure 1). Selenoamino acids and dipeptides present in SeGP.
SeGP Showed Anti-SARS-CoV-2 Activity in vitro
Anti-SARS-CoV2 Activity of SeGP in Vero Cells.

Cytotoxicity/Viability Assay in Human Lung Carcinoma A549 and RAW 264.7 Macrophages
Non-cytotoxic concentrations of SeGP towards different cell lines, human lung carcinoma A549 and RAW 264.7 macrophages were detected after 24 h of exposure followed by MTT assay. Both the cells showed significant cytotoxic response only at 100 μg/ml of SeGP (Figure 2) and the noncytotoxic concentrations of 12.5, 25 and 50 μg/ml of SeGP were used for subsequent studies. Cytotoxicity of SeGP in A549 and RAW264.7 cells by MTT assay.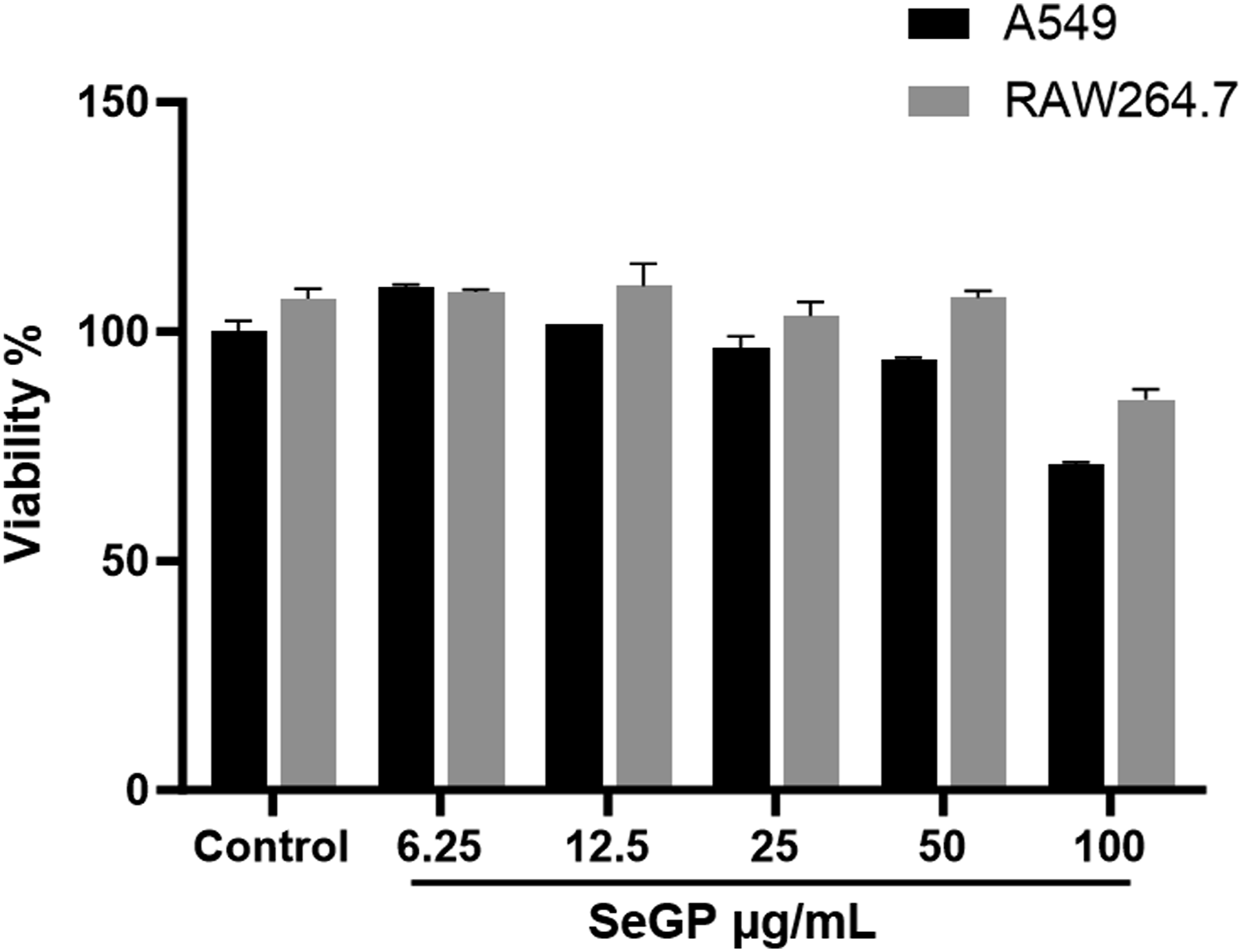
SeGP Showed Significant Inhbition of Furin Activity in Human Lung Carcinoma A549 Cells
Human furin inhibition activity of SeGP was measured using fluorogenic peptide pERTKR-AMC as substrate. Furin hydrolyzes the Arg-AMC amide bond and releases 7-amino-4-methylcoumarin (AMC), a highly fluorescent group detectable with the fluorescence reader. SeGP dose-dependently reduced the activity of furin compared to the normal lung carcinoma cells. At concentrations of 12.5, 25 and 50 μg/ml, SeGP, inhibited the activity of furin by 7 %, 10 % and 14% respectively compared to the normal cells and the changes were found statistically significant. The treatment with 6.25 μg/ml of SeGP did not induce a significant change in furin activity (Figure 3). Effect of SeGP on furin activity. Data are expressed as mean ± SD. *P < 0.05 and **P < 0.005 vs control cells.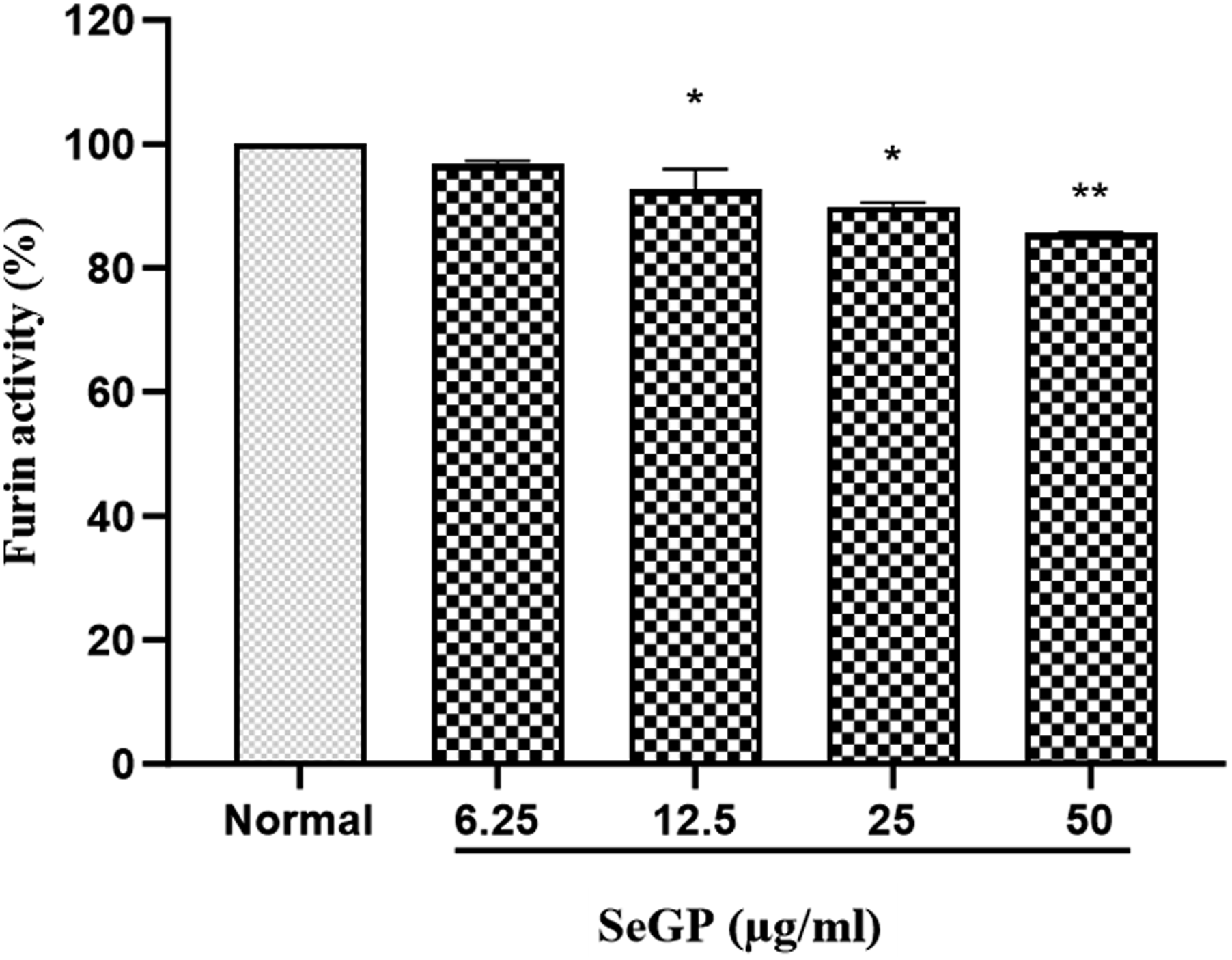
SeGP Down Regulated the Expression of ACE2 and TMPRSS2 in Human Lung Carcinoma A549 Cells
SeGP treatment reduced the expression of ACE2 and TMPRSS2 in human lung carcinoma A549 cells. At a concentration of 12.5 μg/ml the relative mRNA of ACE2 and TMPRSS2 expression were reduced by 24.4% and 20.7% respectively compared to the untreated control (Figure 4). Relative expression of ACE2 and TMPRSS2 genes in SeGP (12.5 μg/ml) treated cells compared to untreated control.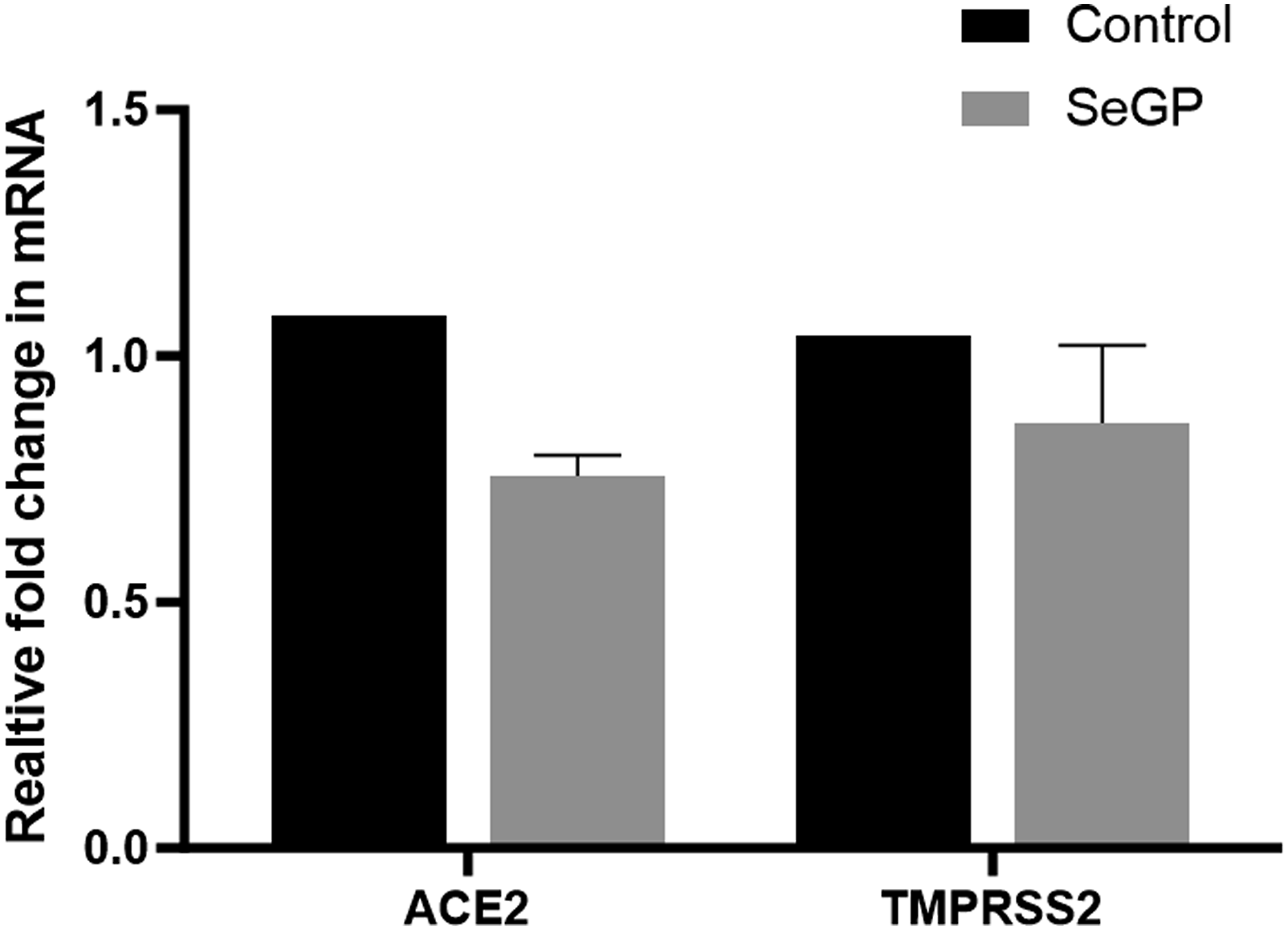
SeGP Reduced Oxidative Stress in Human Lung Carcinoma A549 Cells
The antioxidant activity of SeGP was observed in H2O2-induced ROS in lung carcinoma A549 cells using fluorescent probe 2′, 7′-dichlorodihydrofluorescein (DCFH). SeGP at concentrations of 12.5, 25 and 50 μg/ml reduced ROS levels to 72.7% (P < 0.05), 61.3% (P < 0.005) and 49.3% (P < 0.0005) respectively whereas treatment with 6.25 μg/ml did not show any significant reduction in ROS level (96%) compared to the lung cells induced with H2O2 (Figure 5). Thus, the sample showed a dose-dependent reduction in ROS levels with an IC50 value of 43.45 μg/ml. Effect of SeGP on H2O2-induced reactive oxygen species (ROS) generation in A549 cells. Data are expressed as mean ± SD. #P < 0.0001 vs Control, and *P < 0.05, **P < 0.005 and ***P < 0.0005 vs H2O2 treated cells.
SeGP Showed Significant Anti-Inflammatory Activities in RAW 264.7 Macrophages
The inducible secretion of NO by the activated macrophages were detected using Griess reaction. The treatment with SeGP showed a significant reduction in the concentration of NO dose dependently indicating its anti-inflammatory activity. Nitric oxide production was reduced from 26.48 μM to 21.5 μM (18.8% reduction) when LPS induced RAW 264.7 macrophages were treated with SeGP at a concentration of 50 μg/ml (Figure 6A). Effect of SeGP on inflammation. (A) NO, (B) TNF-α and (C) IL-6. Data are expressed as mean ± SD. *P < 0.05, **P < 0.005 and **P < 0.0005 vs LPS induced cells.
The levels of inflammatory markers in the supernatant of RAW 264.7 macrophages activated with LPS were detected by ELISA. It was found that the secretions of inflammatory cytokines TNF-α and IL-6 were inhibited in a dose-dependent manner by SeGP treatment in LPS activated RAW 264.7 macrophage cells. Treatment with LPS induced the secretion of 24 476 ± 1506 pg/mL of TNF-α. At the concentrations of 12.5, 25 and 50 μg/ml, SeGP reduced the TNF-α in culture supernatants to 20 722 ± 355 pg/mL (84.6%), 20 231 ± 48 pg/mL (82.7%) and 16 908 ± 1147 pg/mL (69.1%) respectively (Figure 6B). The IL-6 levels were significantly reduced from 1995 ± 225 pg/mL to 1146 ± 288 pg/mL (57.4%), 911 ± 11.5 pg/mL (45.6%) and 540 ± 41.4 pg/mL (27.1%) at 12.5, 25 and 50 μg/ml respectively (Figure 6C).
Discussion
Selenium, an important micronutrient, is incorporated into selenoproteins, which are indispensable for the normal functioning of our body. Selenium is known to lower the susceptibility and severity of viral infections and consequently, its deficiency impacts immune response to viruses, expression of viral proteins and antioxidant response.22-24 Several preclinical and clinical studies suggested that selenoproteins are involved in reducing oxidative stress, inflammation, ferroptosis and endoplasmic reticulum (ER) stress associated with viral infections including COVID-19.26,27,30 Hence, Selenium supplementation is suggested to be beneficial in severely diseased and Selenium-deficient COVID-19 patients. 31
However, excessive as well as deficient consumption of Selenium induces health risks. The World Health Organization (WHO) recommends a Selenium intake level of 55 μg/day for adults, with the tolerable upper limit set at 400 μg/day and plasma/blood levels in the general population lie around 100 μg Selenium/l.32,33 SeGP is a novel extract of organoselenium enriched extract of garlic, containing approximately 1000 μg of Selenium per gram of the extract. The dose of this supplement is fixed at not more than 100 mg/day equivalent to 100 μg of organoselenium. In the present sudy, we demonstrate the anti-viral, antioxidant and anti-inflammatory activities of SeGP in vitro.
As per the recent report by the WHO, there is a resurgence of SARS-CoV-2 infection globally, with a 52% increase in new cases during the 28-day period of 20 November to 17 December 2023 as compared to the previous 28-day period. 34 Hence, COVID-19 is still a concern due to its emerging variants, breakthrough infections and a general relaxation restrictions. Understanding the mechanisms of Coronavirus infections are crucial in developing new treatment strategies to attenuate the impact of the disease.
SARS-CoV-2 entry into the host cells is mediated by the binding of the viral spike protein to ACE2 and the activation of this spike protein by the proprotein convertases. 35 The capacity of SARS-CoV-2 to infect the cells is mediated by 2 indispensable cleaving steps of the viral spike protein. This protein is cleaved into S1 and S2 subunits, by the TMPRSS2 protease. 36 The S1 domain is responsible for receptor binding and the S2 domain initiates the membrane fusion. 37 Following endocytosis of the virus, lysosomal proteases further cleave the S2 subunit to release the hydrophobic fusion peptide to fuse with the cell membrane. Furin, is a proprotein convertase, located in the trans-Golgi network which cleaves precursor proteins with specific motifs (The first and fourth amino acids at the N terminus of the substrate cleavage site must be arginine “Arg-X-X-Arg ↓”) to produce mature proteins with biological activity. 38 One of the reasons for high infectivity of SARS-CoV-2 was proposed to be due to the presence of redundant furin cleavage site in its spike protein. 39 The reduced expressions of furin, ACE-2 and TMPRSS2 in lung cells and other tissues could be exploited by anti-viral compounds to ameliorate the spread of the virus. We observed that SeGP could inhibit the expression of all these proteins in vitro. These findings are consistent with earlier studies demonstrating the anti-viral properties of plant extracts and Selenium derivatives. Resveratrol, was reported to reduce the expression of nucleocapsid (N) protein essential for MERS-CoV replication in Vero cells. 40 Several phytochemicals have been shown to interfere with SARS-CoV-2 viral entry and replication proteins by docking studies. 41 Ebselen, a synthetic organoSelenium drug molecule and its derivative were shown to inhibit the SARS-CoV-2 main protease (Mpro), critical for viral replication. 42
SARS-CoV-2 infection involves complex interactions between the viruses and target cells where oxidative stress plays a crucial role. ROS induces tissue damage, thrombosis, and red blood cell dysfunction, contributing to the severity of SARS-CoV-2 infection. 43 SARS-CoV-2 infected patients have reported that excessive level of ROS is associated with severity of illness which promotes a cascade of biological events to drive host pathological responses.44,45 Increased oxidative stress significantly reduced anti-viral host responses and increased virus-induced inflammation leading to multi-organ damage. 46 Hence, the compounds with antioxidant activity are expected be an active drug candidate in COVID-19 therapy. SeGP was effective in reducing oxidative stress in H2O2 induced A549 cells with an IC50 value of 43.45 μg/ml.
Although hyper-inflammation is a key characteristic of SARS-CoV-2 infection, no therapy has succeeded to reduce the exacerbated immune response associated with severe SARS-CoV-2 infection.47,48 Hence apart from preventing viral entry, attenuation of SARS-CoV-2 -induced hyper-inflammation also remains as a prime focus in the therapeutic modality of SARS-CoV-2 infection. The anti-inflammatory potential of SeGP was evaluated in activated macrophages in vitro. Nitric oxide (NO) serves as a pro-inflammatory mediator that induces several downstream inflammatory pathways 49 which was reduced significantly by SeGP treatment. SeGP also significantly reduced the levels of TNF-α and IL-6 in the culture supernatant of activated macrophages. Several studies have suggested that elevated levels of IL-6 are associated with cytokine storm and longer hospital stays. 50 In a systematic review and meta-analyses, including 1798 patients, patients with COVID-19 were found to have elevated levels of IL- 6 in serum. 51 Thus, a reduction in IL-6 may have multiple benefits in patients with viral infection. Downregulation of these inflammatory components, consequently, reduces the severity and progression of the disease suggesting that SeGP could be a beneficial supplement to be used as an adjunct therapy for viral infections.
The molecular basis for this antiviral effect still needs to be completely elucidated. Further, the product also contained Alliin (S-allyl cysteine sulfoxide), at 0.04% w/w. Alliin is known to have antioxidant activity, which could have also influenced the overall activity of SeGP, which needs to be independently evaluated in future studies. However, it may be conceivable that the antiviral activity of SeGP may be mediated by multiple pathways viz inhibiting furin, ACE2 receptor and TMPRSS2. SeGP is also effective as an antioxidant and anti-inflammatory extract, which are beneficial in controlling the comorbidities associated with SARS-CoV-2 infection.
Conclusions
In conclusion, the results of this study demonstrate that Selenium enriched garlic powder inhibited SARS-CoV-2 multiplication in vitro, probably by interfering with the virus and host cell interactions. Further the Selenium product was also effective in reducing oxidative stress and inflammatory mediators, which will augment the effect of the product as an adjunct therapy for viral infection. Further studies are warranted to enumerate the detailed pathways of viral replication influenced by the product and to evaluate the efficacy in clinical settings.
Footnotes
Acknowledgments
The authors thank the Institute of Life Sciences, Bhubaneswar, Odisha, India for their help in studies with antiviral activity against SARS-CoV 2 virus. The help from the staff of Department of biotechnology- Institute of Life Sciences BIOINCUBATOR (An Autonomous Institute of Department of Biotechnology, Government of India),is gratefully acknowledged.
Author Contributions
Conceptualization, Muhammed Majeed and Kalyanam Nagabhushanam; Formal analysis, Lincy Lawrence and Lakshmi Mundkur; Investigation, Lincy Lawrence and Priji Prakasan; Methodology, Lincy Lawrence and Lakshmi Mundkur; Project administration, Kalyanam Nagabhushanam; Resources, Muhammed Majeed; Supervision, Lakshmi Mundkur; Writing – original draft, Lincy Lawrence and Priji Prakasan; Writing – review & editing, Muhammed Majeed, Kalyanam Nagabhushanam, and Lakshmi Mundkur. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All the authors are employees of Sami-Sabinsa Group Limited or Sabinsa Corporation, which markets SelenoForce®.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patents
The following patents are granted for SelenoForce. WO2007067600A2, NZ568233A, EP06839082A, PT06839082T.
Data Availability Statement
All data associated with the study are available in the manuscript. Any additional data may be provided on request to the corresponding author.
