Abstract
Background:
Obstetric brachial plexus palsy (OBPP) is a condition impairing limb function caused by birth injury. In 20 to 30% of cases, severe OBPP can cause life constraints in feeding, grooming, and clothing tasks.
Objective:
The present study, using voxel- and surface-based morphometry (VBM and SBM), examined the brain structure of pediatric OBPP patients to better understand the effects of this peripheral motor deficit on early brain development.
Methods:
Thirty-six T1-weighted images of 18 patients (2-17 years old, mean age = 11.3, 8 females) and 18 healthy controls (2-17 years old, mean age = 10.1, 8 females) were collected for this study. MRI data were processed and analyzed using the Statistical Parametric Mapping 12 (SPM12) toolbox. The custom pediatric tissue probability map was created with the CerebroMatic (COM) toolbox. The results were considered significant if they survived whole-brain family-wise error correction (P < .05).
Results:
We have found differences in grey matter volumes in the bilateral anterior hippocampus (left P < .001 and right P = .01) and left cerebellum exterior (Crus I) (P < .001). We have also found differences in cortical thickness in the bilateral parahippocampal gyri (left P = .001 and right P = .005) and right orbitofrontal cortex (OFC) (P < .001).
Conclusions:
These structural differences might be linked to the altered environmental adaptation that children with OBPP face due to their primary motor deficit. Our findings hint at a complex interplay between motor capabilities, brain structure development, and cognitive functions. However, more research combining neuroimaging, behavioral, cognitive, and clinical data is needed to support stronger conclusions on this subject.
Keywords
Introduction
Obstetric brachial plexus palsy (OBPP) is a perinatal condition originating from brachial plexus traction during difficult delivery and resulting in damage of the descending motor projections and sensory input and to the paralysis of 1 of the upper limbs. 1 The disease’s prevalence is 2.9 per 1000 births, with about half of the patients spontaneously restoring their motor function. 2 The OBPP is commonly classified into 3 groups: upper Erb’s (C5-C6 roots injured), extended Erb’s (C5-C7 roots injured), and total palsy (C5-Th1 roots injured). 3 OBPP can cause shoulder and elbow paralysis, cosmetic deformity, chronic pain, and lifestyle constraints in daily tasks. In contrast, severe OBPP can additionally cause persistent hand dysfunction and skeletal deformity in 20 to 30% of patients.4,5
Even after rehabilitation and muscle reinnervation, some patients still exhibit restrained motor function of the affected upper limb, 6 lack of automatic swing during running or walking, and the lack of balancing compensatory arm movements. These persistent clinical manifestations in patients with almost full neurological recovery have been known as “developmental apraxia”—maladaptation of central motor programming due to poor afferent input during a critical time window of brain development. 7
Even in adults, peripheral nerve injury has been reported to produce radical changes in the brain’s organization. 8 Previous studies with brachial plexus injury (BPI) patients have reported distinctive patterns of brain remodeling after trauma. At the cortical activity level, it has been shown that BPI patients have reduced inter-hemispheric functional connectivity in the sensorimotor areas, both during motor task performance and at rest.9-11 Multiple studies deploying resting state fMRI on adult BPI patients also indicate post-traumatic cortical plasticity.12-14
Since the injury leading to OBPP occurs perinatally, its effects can be even more robust due to the plastic nature of the developing brain. The rapid transformations undergone by the brain during early childhood might also explain why OBPP patients display better recovery rates than their adult counterparts with BPI. However, more attention should be paid to the central outcomes of neonatal brachial plexus nerve ripping compared to similar injuries occurring later in life. Previous studies with OBPP populations have indicated changes in functional brain activity, including deactivation of the supplementary motor area, 15 increased activity in the bilateral premotor area, and 16 reduced activation of the sensorimotor network on the side contralateral to the injured limb. 16 As for the structural changes concerned, the literature is still scarce, without any consensus on the consequences of this condition: some studies have reported that OBPP patients display bilateral lower volumes of the corpus callosum 16 and primary motor areas, 17 while others have found no structural brain differences between OBPP patients and healthy controls. 18
The evidence reviewed above indicates patterns of brain remodeling in patients following an injury-related motor impairment. But what transformations does an immature brain undergo following a perinatal peripheral trauma? Since the injury severely debilitates the ability to use an upper limb since birth, we hypothesized that OBPP patients would have lower grey matter volume and reduced cortical thickness, especially in brain regions involved in sensory and motor functions. We have addressed our hypothesis by conducting an observational case-control study.
OBPP is a unique condition, both by the time of its occurrence (perinatal, not congenital) and by the clinical prognosis (about half of spontaneous recovery cases). By studying it, we can perhaps better understand how a motor disability can affect early brain development.
Methods
Participants
Thirty-two OBPP patients were initially included in the clinical sample. They were first scanned on a 3.0T MRI machine at the Turner National Medical Research Center for Children’s Orthopedics and Trauma Surgery (Saint-Petersburg, Russian Federation). Then, they were assessed by a neurologist and orthopedist. A neurologist determined the root level of injury post OBPP using electromyography. The exclusion criteria were the presence of additional neurological or psychiatric disorders and poor-quality MRI data. Fourteen OBPP patients had to be excluded from the final sample due to additional neurological and psychiatric conditions and the quality of MRI recordings (movement artifacts).
Eighteen healthy controls were collected after the clinical sample was defined to match the patients by gender and age. Participants in the control group were scanned on a 3.0T MRI machine at the National Research Institute of Emergency Children’s Surgery and Traumatology (Moscow, Russian Federation). Similarly to the patients, they were assessed by a neurologist and orthopedist. Only healthy participants with no history of neurological, psychiatric, or orthopedic disorders were included in the control group. Thus, 36 children participated in this study: 18 children with OBPP and 18 age- and gender-matched healthy controls. All healthy participants were right-handed. The hand dominance in the clinical group varied depending on the side of the palsy. All patients in the OBPP group had symptoms related to the disease but no other significant neurological or orthopedic impairments. All OBPP patients received conservative treatment prior to the date of MR scanning.
Ethical considerations
This study complies with the Declaration of Helsinki and was performed according to ethics committee approval. This study was approved by the HSE Committee on Interuniversity Surveys and Ethical Assessment of Empirical Research (Ethics Code: No. 19–3) on 9 December 2019. The neuroimaging and clinical data of patients was collected before the research began. All children’s legal guardians provided written informed consent before enrolment in the study.
The Mallet score
Additionally, orthopedists from the Turner Research Center gave us the Mallet scores for each patient. The patients were asked to follow instructions and perform different movements, while the youngest patient copied his mothers’ movements. The Mallet score is a classification used to assess shoulder abduction before and after surgical procedures in OBPP treatment. It quantifies the extent of external shoulder rotation, with 1 representing a severely restricted shoulder and 5 representing a normal one. 7 The Mallet classification also has 5 subscores for various shoulder movements: abduction, external rotation, hand-to-neck, hand-on-spine, and hand-to-mouth. The maximum attainable score under this classification is 25.
MRI data collection
T1-weighted 3D images were collected via 2 Philips Ingenia Elition X 3.0T magnetic resonance scanners at the Turner National Medical Research Center for Children’s Orthopedics and Trauma Surgery and at the National Research Institute of Emergency Children’s Surgery and Traumatology. The following parameters were used for MRI data acquisition: voxel size = 1.0 × 1.0 × 1.0 mm3, repetition time = 8.5 ms, echo time = 3.7ms, 180 slices, slice thickness = 1 mm, acquisition matrix = 256 × 256, field of view = 256 mm × 256 mm, flip angle = 8°.
MRI data preprocessing
All MRI data were processed using MATLAB R2019a software. Image preprocessing was conducted based on the CAT12 toolbox (http://dbm.neuro.uni-jena.de/cat12/), an extension of the Statistical Parametric Mapping 12 toolbox (SPM12; http://fil.ion.ucl.ac.uk/spm/). The VBM analysis based on the CAT12 toolbox was performed with the following pipeline for each participant. Firstly, the T1-weighted structural images were manually re-oriented to ensure the origin was falling on the anterior commissure-posterior commissure line. Secondly, images were segmented into grey matter (GM), white matter (WM), and cerebrospinal fluid (CSF) maps. The segmented data were registered to a custom pediatric tissue probability map created with the CerebroMatic (COM) toolbox. 19 Specifically, the COM toolbox can be used to generate custom tissue probability maps by matching sample demographics to parameters that influence brain structure using a flexible non-parametric approach: multivariate adaptive regression splines. In the next step, extracted GM maps were spatially registered to the custom pediatric template created in the COM toolbox. Then, GM images were modulated with Jacobian determinants from the normalization procedure to preserve regional volumes. The modulated normalized non-linear images were checked for sample homogeneity, and no outliers were detected. The GM images were smoothed with an 8 mm full-width at half-maximum smoothing kernel. The voxel size of processed images was 1 mm × 1 mm × 1 mm. The total intracranial volume (TIV) was calculated within CAT12 for each participant as follows: TIV = grey-matter volume + white-matter volume + CSF volume.
Additionally, we conducted SBM to investigate differences in cortical thickness between the 2 groups. The automated method in CAT12 allowed us to achieve central surface reconstructions and cortical thickness measurements in 1 step; subsequently, topological defects of cortical surface mesh were repaired with a spherical harmonic method. 20 A recommended 15-mm FWHM isotropic Gaussian kernel was used for cortical thickness data. By applying both VBM and SBM workflows, we aimed to improve the accuracy of detecting morphological differences.
Statistical analysis
Voxel-wise group comparisons of the smoothed GM volumes were performed using two-sample t-tests within SPM12. TIV and age were set as covariates to account for differences in individual brain size. All neuroimaging results were considered significant if they survived whole-brain family-wise error (FWE) correction (FWE P < .05). The same analysis pipeline was followed for the cortical data.
Results
Participants
The final experimental sample included 36 participants: 18 children with OBPP (2-17 years old, mean age = 11.3, 8 females) and 18 age- and gender-matched healthy controls (2-17 years old, mean age = 10.1, 8 females). Six OBPP patients had upper Erb’s palsy, ten patients had extended Erb’s palsy, and 2 patients had total flaccid palsy of the injured limb (C5-Th1 roots injured). Twelve OBPP patients had undergone surgical treatment, including tendon transfer, neurolysis, invasive nerve stimulation, shoulder derotation osteotomy, and derotation osteotomy of the radius. The large age gap between patients (and, therefore, healthy controls) is explained by the time their parents discovered the possibility of treatment at the Turner Research Institute. Clinical and demographical profiles of participants are shown in Table 1.
Profiles of participants.
a. Clinical profiles of patients with Obstetric Brachial Plexus Palsy (OBPP).

All patients had limitation of shoulder movements. For estimation of shoulder movements we use Mallet scale (min 5, max 25 points).
b. Demographical profiles of healthy controls.

Voxel-based morphometry
VBM analyses returned significant differences in the group in 3 voxel clusters, with OBPP patients displaying lower s than controls. Cluster peaks were located in the left (k = 1365, P = .002 FWE-corrected) and right anterior hippocampus (k = 517, P = .0002 FWE-corrected), as well as in the left cerebellum exterior (Crus I) (k = 1535, P = .01 FWE-corrected; see Table 2, Figure 1). The OBPP group had no regions displaying greater grey matter volume (GMV).
Grey matter differences between patients and healthy controls detected by voxel-based morphometry (VBM) analysis with MNI coordinates of peak levels.

The P values are corrected for family-wise error (PFWE-corr).

The areas with a significant volumetric difference revealed by voxel-based morphometry (VBM) analysis. The volume of the bilateral hippocampus and left cerebellum exterior was decreased in patients compared to healthy controls (P < .05, family-wise error [FWE]-corrected).
Surface-based morphometry
Children with OBPP showed significant cortical thinning in the right anterior orbital gyrus (k = 486, P = .000003 FWE-corrected) compared to controls. In addition, SBM analyses returned smaller but significant clusters of bilateral parahippocampal areas (right parahippocampal area k = 38, P = .005 FWE-corrected, left parahippocampal area k = 62, P = .001) that were thinner in patients compared to HC (see Table 3, Figure 2). No cortical regions with greater thickness than controls were found in the OBPP group.
Cortical thickness differences between patients and healthy controls detected by surface-based morphometry (SBM) with MNI coordinates of peak levels.
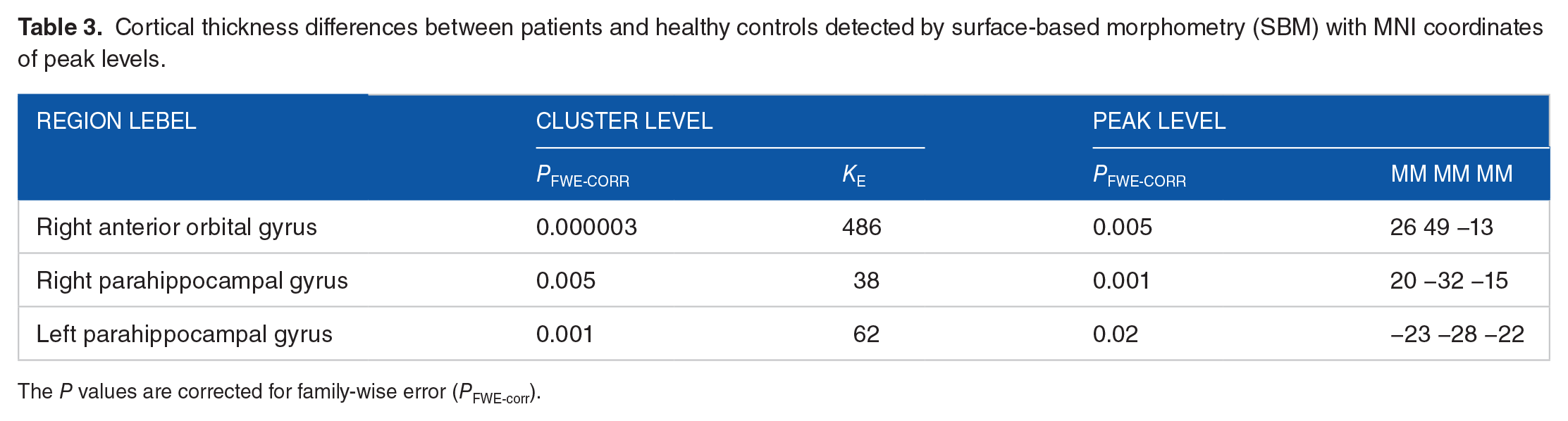
The P values are corrected for family-wise error (PFWE-corr).

The cortical areas with a significant thickness difference revealed by surface-based morphometry (SBM) analysis. The thickness of the right anterior orbital gyrus and bilateral parahippocampal areas was decreased in patients compared to healthy controls (P < .05, family-wise error [FWE]-corrected).
Discussion
In the present study, we aimed to evaluate the brain structure of children with OBPP through the combination of VBM and SBM analyses of structural MRI data. We hypothesized that OBPP may lead to central structural changes at subcortical and cortical levels, especially in regions involved in sensorimotor functions.
Our results confirmed the hypothesis that brain structural alterations are present in children with OBPP. Generally, while comparing children with OBPP to healthy controls, we found brain tissue differences in several brain regions: the bilateral anterior hippocampi, the bilateral parahippocampal gyri, the left cerebellum (Crus I) and the right anterior orbitofrontal gyrus. Below, we provide some possible interpretations of our findings for each particular structure and suggest some prospects for future investigations.
Hippocampus and parahippocampal gyrus
We found significant group-level differences in hippocampal GMV and thickness of parahippocampal areas, bilaterally: OBPP patients with upper limb dysfunction showed reduced volume and thickness compared to healthy controls. A complex, plastic, and vulnerable brain structure, the hippocampus is targeted by various neurodegenerative, psychiatric, and cardiovascular diseases.21-23 The peripheral nerve injury is, however, less often connected with hippocampal alterations, though some studies with rodents report abnormalities in hippocampal functioning related to persistent peripheral pain,24-26 which is prevalent among OBPP patients. 27 Although not the most obvious candidate to suffer from motor-debilitating peripheral nerve injury, such as OBPP, the bilateral hippocampi and the cortical areas surrounding them were indeed highlighted by both VBM and SBM analyses.
Two main long-standing viewpoints have been on the dominant hippocampal function: mediating declarative memory and facilitating spatial navigation. The first view was derived from 1950’s lesion studies, where the patients with lesioned hippocampus exhibited a decline in conscious, explicit memory.28,29 The second view, stating the critical role of the hippocampus in creating mental maps of our physical surroundings, was primarily informed by the discovery of hippocampal place cells (those that actively fire when an animal enters a familiar corner of a maze) in the 1970’s. 30 More recently, the hippocampus has also been involved in encoding temporal, 31 situational, 32 and even social 33 information. Thus, an effort has been made to bridge different theories of hippocampal function by conceptualizing the hippocampus and parahippocampal cortex as an organizing hub of informational inputs within a multidimensional cognitive map of various contexts.34,35 This encompassing concept speaks directly to our findings, that is, underdeveloped hippocampal and parahippocampal regions in OBPP patients. These patients struggle with everyday tasks (eg, grooming or feeding) and autonomously discover their surroundings—as they are usually accompanied and assisted by caregivers. This might have restricted their motor learning abilities (since they can typically operate only with one hand), limited social contacts (their disability makes social connections difficult, and patients also spend a considerable amount of time in hospitals), and in some cases is accompanied with persistent pain in the brachial plexus. All these factors might contribute to the pronounced structural differences in the hippocampus compared to healthy individuals.
It is also important to reiterate that the bilateral differences in hippocampal GMV were found precisely in its anterior part. As Zeidman and Maguire 36 state in their review, a consistent body of research indicates that this portion of the hippocampus critically contributes to imagination, visual scene perception, and episodic memory. The latter function was previously found deficient amongst OBPP pediatric patients.37,38 In addition, early delay in language—a function associated with lower hippocampal volume 39 —has been reported in toddlers with OBPP. 40 Here, we can speculate that a loop connecting OBPP-related motor impairment, cognitive delay and hippocampal development might exist and explain our results. However, further research combining neuroimaging and behavioral data is needed to support more robust conclusions. Finally, this structure was underdeveloped among children with OBPP in our previous study. 41 However, since the study sample included patients with additional psychiatric disorders and the analysis was not conducted using customized pediatric tissue probability maps, we cannot claim the consistency of our findings.
Cerebellum
The pronounced GM differences found in the left cerebellum exterior (Crus I) are generally consistent with the role played by the cerebellum in motor coordination and learning processes. Since the patients who participated in our study suffered from motor impairment of the affected upper limb, hindered cerebellar development was if not expected but at least not surprising. Classically, the cerebellum was considered to anticipate the expected outcome of motor commands to refine future movements.42,43 However, in the past decades, it has been suggested that the cerebellum plays a much more complex and multifunctional role than simply refining motor actions.44,45 For instance, in a recent review Zhang et al 46 summarized the evidence for an involvement of the cerebellum in visuospatial processes, attention, executive functions, language, and working memory. Importantly, in light of our results, the activation and the volume of Crus I—the portion of the cerebellum returned by our VBM analysis as having significantly lower GMV in OBPP patients—were found to be related to working memory performance by various recent studies.47-49
In addition, the cerebellum was found to be functionally lateralized, contralaterally to the cerebrum: the right cerebellar hemisphere is predominantly involved in verbal performance, whereas the left one is mainly engaged in visuospatial process.50-52 As our results indicate lower GMV in the left hemisphere, we can speculate that there might be an interplay between the OBPP-related motor impairment, cerebellar structural underdevelopment and visuospatial deficits. A further investigation of OBPP patients’ visuospatial abilities via tasks such as mental rotation and shape copying should be conducted to support any further conclusions on this subject.
Orbitofrontal cortex
Our SBM analysis has also pointed to a portion of the right orbitofrontal cortex (OFC) being significantly thinner among children with OBPP, whose daily life is limited by disability. The OFC, a critical brain region for emotional processing, is among the last to fully develop in humans and is thus vulnerable to the effects of early stress. 53 Beyond visible motor impairment, OBPP pediatric patients live with a great deal of constraints: they are unable to eat, dress, and play with their peers autonomously, and their childhood is far from that of unimpaired children. Though we did not assess their mental state, we believe that participants from our clinical group might be exposed to constant stress originating from their complex medical condition and worsened by the reality arising from it. Previous studies with OBPP patients support this assumption, reporting a greater risk of psychiatric disorders among patients and their mothers, 54 disturbed body image and self-image, 55 lower self-esteem and social difficulties. 56 In the light of psychosocial problems that OBPP patients face, consistent evidence coming from cross-sectional and longitudinal studies states that early-life life stress exposure is connected to reduced OFC thickness, precisely in the right hemisphere.57-59 Some studies suggest further connections, relating early stress and OFC thinning to long-term depressive syndromes, 60 anxiety, 61 and impaired social cognition. 62 Studies conducted with adults have also widely suggested A connection between interpersonal cognition and the OFC. Specifically, patients suffering from OFC lesions have consistently shown intact intellectual abilities but declining social cognition.63-65 Other studies have also found associations between OFC thickness 66 and activation 67 and social cognitive competence. Hence, we can hypothesize that OFC structural abnormalities, if not leveled out, might have prolonged effects on the OBPP patients’ social lives in adulthood. Once again, as with previously discussed results, we advocate for further investigations of the three-way relationship between OBPP-related motor impairment, OFC thinning, and potential effects on cognition to shed light on the matter.
Strengths and limitations
The main strength of this study is the advancement of OBPP knowledge beyond motor impairments. Our results revealed that the structural underdevelopment of brain structures is crucial for higher cognitive functions in OBPP patients. In addition, a wide range of studies has confirmed structural and functional cerebral reorganization after brachial plexus injuries in adults.68-70 Thus, this study may also serve practical rehabilitative purposes, becoming a basis for cognitive interventions designed to set back maladjusted neurostructural changes in pediatric OBPP patients and adults with similar peripheral injuries.
There are indeed some drawbacks to this study. The lack of a broader clinical assessment of OBPP patients prevented us from directly testing the cognitive and neuropsychological effects of their condition and linking it to their brain morphology. Moreover, due to the heterogeneous nature of the patient sample in terms of the specificity of their diagnosis (ie, lesion severity), we could not examine the finer aspects of cerebral outcomes of peripheral injury. However, further research combining neuroimaging and clinical and cognitive testing is required.
Conclusion
The present investigation suggests that the peripheral injury suffered by OBPP patients at birth might have pronounced effects on their brain structure. Furthermore, our findings, revealing structural underdevelopment of the hippocampus, left cerebellum exterior (Crus I) and right anterior OFC in children with OBPP, might hint at possible cognitive deficits related to OBPP. However, since the regions returned by our VBM and SBM analyses are involved in various cognitive functions (eg, memory, learning, visuospatial abilities, social cognition, and motor functions), we cannot predict precisely which processes might be targeted by the disease in question. Indeed, more research combining neuroimaging and behavioral data is needed to understand better the relationship between neurostructural and cognitive patterns of children with OBPP. Still, our results can enrich rehabilitation strategies for OBPP patients. Taking into account the plastic nature of children’s brains, potential OBPP-related deficits might be minimized using multifaceted cognitive training integrated into conventional motor recovery programs.
Footnotes
Author Contributions
D.K.V. Kadieva — data collection and processing, analysis, preparation of the text of the article. F.Gallo — editing of the article, literature review. M.A.Ulanov — editing of the article, literature review. Alina Hodorovkaya — MRI collection. O.E. Agranovich — surgical treatment of the patient, assistance with collecting MRI data; V.V. Moiseeva — editing of the article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is an output of a research project implemented as part of the Basic Research Program at the National Research University Higher School of Economics (HSE University) and was carried out using HSE Automated system of non-invasive brain stimulation with the possibility of synchronous registration of brain activity and registration of eye movements.
