Abstract
For mitral valve pathologies like mitral stenosis and mitral regurgitation various treatment options are currently available. However, in patients who have previously operated on mitral valves only a few options are applicable. In this case report, we provide detailed insight into transcatheter mitral valve-in-valve implant in a patient with a degenerated surgically implanted mitral bioprosthetic valve.
Introduction
Modern mitral valve surgery includes various conventional, minimally invasive, and endovascular techniques for the treatment of mitral valve pathologies.1, 2 The goal of reconstructive mitral valve surgery is to preserve the patient’s native valve. If one cannot reconstruct the native valve, a mechanical or bioprosthetic mitral valve replacement (MVR) is usually performed. 2 Despite the fact that bioprosthetic valves were primarily considered for older patients, improvements in their durability and the lower risk of thromboembolic complications have broadened the range of patients towards younger age groups. However, the durability of bioprosthetic mitral valves remains limited. Structural valve degeneration can be observed in 10%–15% of patients with a conventional MVR after 10 years. 3
The treatment of a degenerated mitral valve prosthesis remains a clinical challenge associated with significant mortality and morbidity. Outcomes from conventional redo MVR operations suggest in-hospital mortality ranging from 0.8% to more than 10% and high incidences of major adverse events of up to 40%. 4 In this constellation, valve-in-valve transcatheter mitral valve replacement (ViV TMVR) has been increasingly performed in patients with high surgical risk. The introduction of ViV TMVR has significantly dropped the number of in-hospital deaths, the length of hospital stay, and the risk of complications compared to conventional redo-MVR surgery. 5 Therefore, the implementation of ViV TMVR in patients with structural valve deterioration is of major clinical interest.
Case Report
A 61-year-old lady with recurrent heart failure admissions had a history of rheumatic heart disease severe mitral stenosis with mitral regurgitation and severe aortic stenosis which eventually required the surgical implantation of a bioprosthetic mitral valve (Mitral tissue valve 25 mm, St. Jude Medical, Inc., St. Paul, MN, USA) and aortic valve (Aortic tissue valve 19 mm, St. Jude Medical, Inc.) in 2013. Echocardiographic evaluation revealed structural degeneration of the bioprosthetic mitral valve leading to severe mitral stenosis (dPmean/max 22/32 mmHg). The left atrium was dilated. The left ventricular ejection fraction was above 55%; moderate tricuspid valve insufficiency (TRmax 78 mmHg +CVP) with a normal aortic bioprosthetic valve. Relevant comorbidities included paroxysmal atrial fibrillation while the patient was on oral anticoagulant. The patient had a high perioperative risk profile (STS PROM score 10.523%; EuroSCORE II 15.25%).
Therapy Decision Making
Due to our patient’s past medical history which included previous cardiac surgery with recurrent heart failure, and high risk for redo-MVR, computed tomography (CT) scanning was performed for preoperative planning of the ViV TMVR procedure.
Preoperative Planning of the Procedure
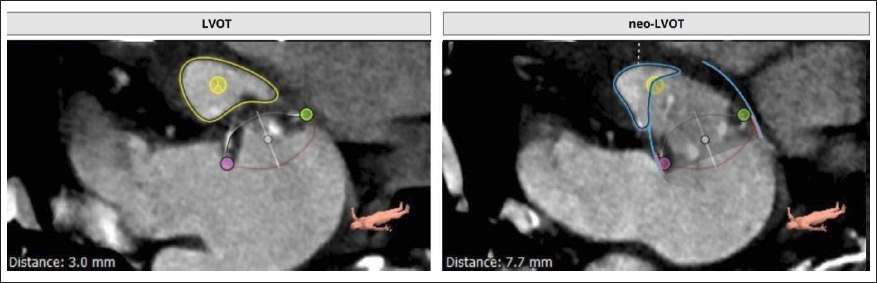
The 3 Mensio structural heart module software (Pie Medical imaging, Bilthoven, The Netherlands) was used for preoperative 3-dimensional CT reconstruction and measurements of the true internal diameter of the bioprosthetic mitral valve and neo-left ventricular outflow tract (neo-LVOT). Preoperative planning of the neo-LVOT for a TMVR-VIV is crucial due to potential outflow tract obstruction through the prosthesis. The estimated neo-LVOT area was 192 mm2, which was above the safety cut-off value of 177 mm2 (original area surface 285.6 mm2, area blocked by the prosthesis 33%) (Figure 1). 6
Simulation of LVOT (Surface Area 285.6 mm2) Estimation of the Neo-LVOT Surface Area (192 mm2) on Multislice Spiral Computed Tomography (MSCT) in Systole Using the Three Mensio Structural Heart Module Software (Pie Medical Imaging, Bilthoven, The Netherlands).
Crossing the Mitral Valve with a Guidewire and Anchoring the Wire
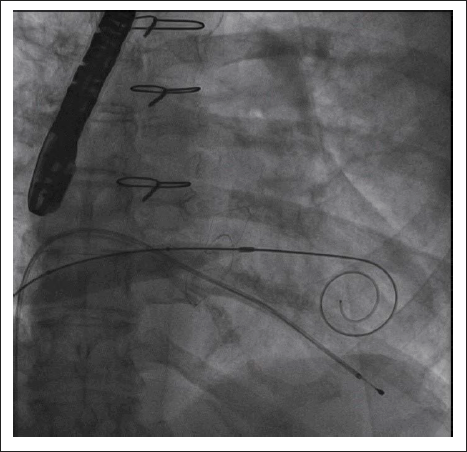
The 8.5 Fr steerable Agilis sheath (Abbott, St. Paul, MN, USA) was passed through the ASD into the left atrium. A 0.035-inch PTFE wire was advanced through the mitral valve prosthesis into the left ventricle. The wire was then replaced by stiff Safari wire (Boston Scientific Corp., Marlborough, MA, USA) using a wire-anchoring technique.
Dilating the Interatrial Septum and Placing the Python Sheath
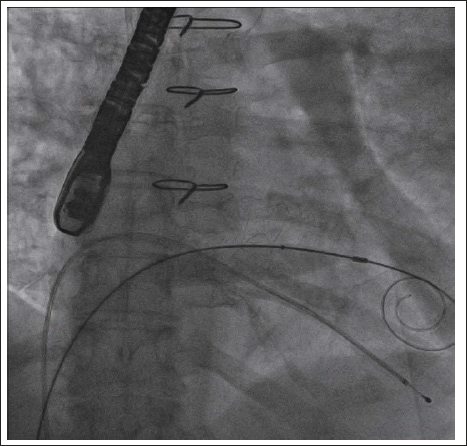
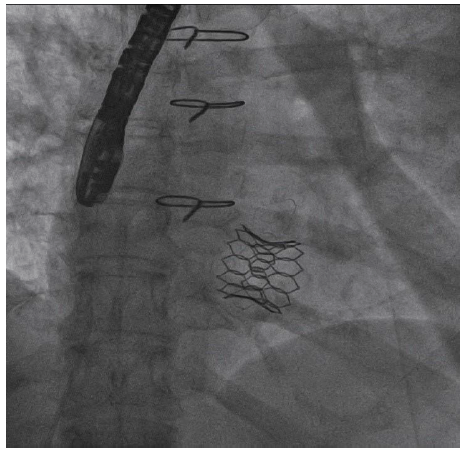
The interatrial septum was dilated using 12-mm standard percutaneous transluminal angioplasty balloon in order to achieve the safe passage of the Navigator/MyVal-Octacor delivery catheter system (Meril Lifesciences) through the interatrial septum (Figures 2 and 3). The previously placed 6 Fr introducer in the right femoral vein was exchanged with a 14 Fr python sheath (Meril Lifesciences).
Fluoroscopy Showing Balloon Dilation of the Interatrial Septum with a 12 mm Balloon over the Safari Wire That Has Been Advanced Through Interatrial Septum, Across the Bioprosthetic Mitral Valve, and into the Left Ventricular Apex.
Fluoroscopy Showing Same Septostomy Balloon Passed Through the Mitral Valve to Ensure Easy Passage.
Crossing the Septum with the Delivery System
The Navigator delivery system with the MyVal-Octacor 23-mm valve (size of the valve is selected based on the true internal diameter of the bioprosthetic mitral valve from CT measurements) is inserted through the Python sheath and advanced through the septum into the mitral bioprosthesis.
Positioning the Valve Prosthesis
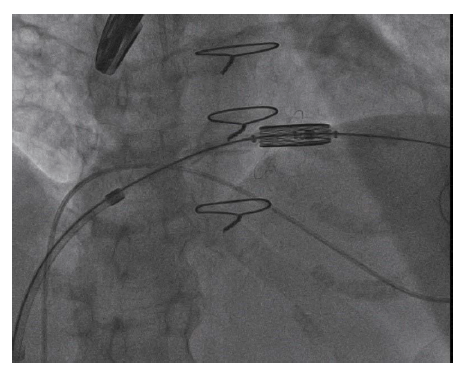
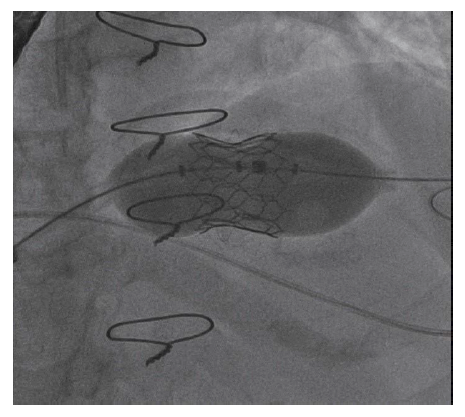
Under fluoroscopy guidance, the transcatheter valve was positioned in the mitral bioprosthesis (Figure 4). The atrial portion of the valve prosthesis was maximally 1–2 mm cranial to the sewing cuff of the surgical valve.
Fluoroscopy Showing Positioning of the Transcatheter Valve within the Ring of the Old Bioprosthetic Mitral Valve.
Deploying the Valve Prosthesis
After confirmation of correct positioning and alignment of the percutaneous valve, the valve deployment was performed in apnea and rapid pacing with 200 bpm (Figure 5). The Myval-Octacor valve was deployed using a slow balloon inflation technique. Immediately after full expansion, the balloon was completely deflated, rapid pacing was stopped, and ventilation was restarted.
Fluoroscopy Showing Balloon Inflation and Deployment of the Transcatheter Valve Inside the Bioprosthetic Mitral Valve.
Evaluating the Echocardiogram
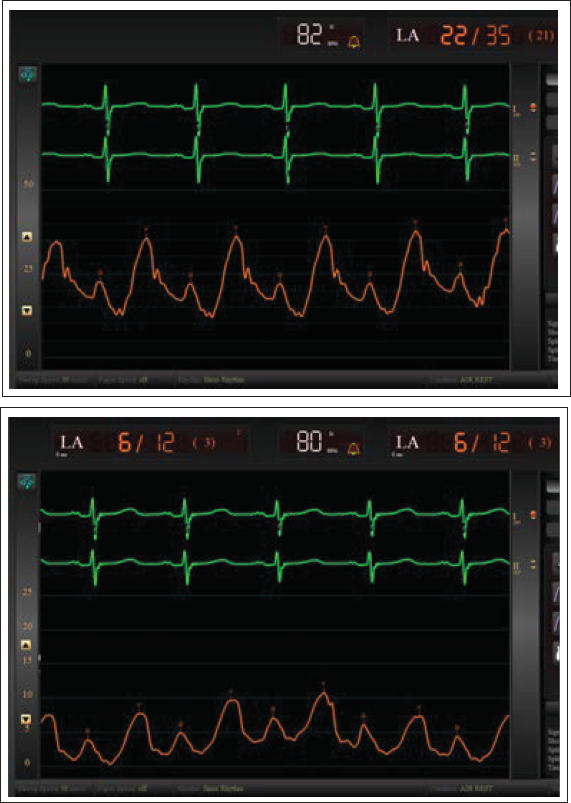
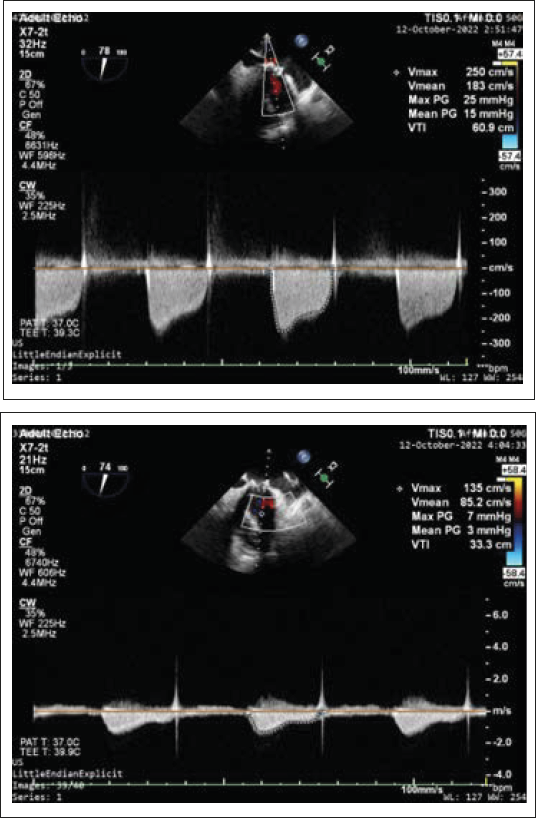
The initial fluoroscopic (Figure 6) and hemodynamic assessment confirmed a stable position and functioning of the valve with trivial mitral regurgitation and no PVL, and the mean left atrial pressure decreased (from 21 to 3 mmHg) (Figure 7). TEE showed reduction in mean mitral valve gradient from 15 to 3 mmhg (Figure 8). No hemodynamically relevant obstruction of the LVOT was observed (mean pressure gradient in the LVOT was 9 mmHg).
Final Fluoro Image Showing MyVal Octacor Transcatheter Valve in Mitral Position.
Pre and Post Procedure Pressure Tracings Showing Left Atrial Mean Pressure Reduced from 21 mmHg to 3 mmHg.
Pre and Post Procedure TEE Showing Reduction of Mean Mitral Valve Gradient from 15 mmHg to 3 mmHg.
Outcome
Post-procedure patient was transferred to the Cardiovascular intensive care unit (CICU) and extubated within two hours and transferred to the normal ward on postoperative day 2. Postoperative transthoracic echocardiography of the mitral valve showed a mean pressure gradient of 5 mmHg. The postoperative course was uneventful, so the patient was discharged home on postoperative day 4 in an improved physical condition.
Discussion
ViV TMVR represents an attractive alternative to redo-surgical MVR in patients with a degenerated bioprosthesis and is associated with lower mortality, periprocedural morbidity, and resource use compared to redo-surgery. 5 Preoperative decision-making and planning play key roles in achieving a successful ViV TMVR.
Assessment of the surgical prosthesis type and left ventricular anatomy are of considerable importance for the patient’s eligibility for ViV TMVR. The preoperative 3-dimensional (3D)-CT reconstruction and specially dedicated software tools for ViV TMVR provide optimal planning of the procedure and avoidance of specific pitfalls.
In contrast to transcatheter aortic valve replacement, the percutaneous valve prosthesis in the mitral position is exposed to higher pressure gradients during ventricular contraction and has a smaller anchoring area. 1 To achieve adequate anchoring and avoid delayed valve migration into the left atrium, sufficient oversizing and correct positioning have to be addressed in the preoperative planning. In the case of balloon-expandable valves, the atrial portion of the valve prosthesis should not exceed 30% of its total height. 1
LVOT obstruction is an unavoidable consequence of ViV TMVR and should be preoperatively measured with special software tools. The remaining neo-LVOT area should be at least 1.7 cm2. 6 The degree of LVOT obstruction depends not only on the implanted depth of the transcatheter prosthesis but also on the anatomical features of the left ventricle, such as the angle between the aortic and mitral valve planes and the interventricular septal thickening.
Atherosclerotic debris during surgical or catheter manipulations of the calcified valves and deteriorated valve prosthesis can lead to postoperative neurological complications. The use of cerebral protection devices during valvular surgery or interventions remains a point of considerable debate; however, they can be considered in high-risk patients with severe calcifications and atherosclerotic plaques. 7
Alternative access sites can be considered for a ViV TMVR implant, with comparable outcomes. ViV TMVR can be performed surgically through the left ventricular apex or the left atrium, using a left-lateral mini-thoracotomy. A percutaneous transseptal implant via a jugular vein has also been described.1, 8
Additionally, TMVR can be performed as a valve-in-ring (ViR) approach. However, this technique is more challenging and is associated with lower procedural success rates. The annuloplasty rings usually have a complex 3D ellipsoid form and are sometimes incomplete or have a rigid structure, which represents mechanical difficulties for proper anchoring. The semirigid annuloplasty rings are amenable to circular deformation, which can be utilized to overcome the shape mismatch when implanting the ViR. 9 Finally, yet importantly, annuloplasty rings are often larger than the available transcatheter valves. These issues lead to a higher risk of PVLs and may require an additional ViV TMVR on top of the ViR.1, 6, 8
TMVR can also be performed in native valves in case of degenerative mitral stenosis. Severe calcification of the mitral valve annulus can lead to life-threatening complications during the conventional replacement, including an annulus rupture, an iatrogenic ventricular septal defect, PVL, and injuries to the circumflex coronary artery. In this setting, TMVR can be considered a less invasive alternative in high-risk patients, but at the same time, meticulous planning is necessary to achieve secure anchoring and prevent relevant PVLs.1, 6, 8
Conclusion
ViV TMVR represents a feasible therapeutic approach for patients with structural deterioration of a biological mitral valve prosthesis and can be successfully performed as an alternative to conventional redo surgery in high-risk patients. Endovascular cerebral protection devices might be considered during ViV TMVR. Preoperative planning should include 3D-CT reconstructions and precise measurements of the future position of the prosthesis.
Footnotes
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Considerations
Institutional Ethics Committee approved prior to publication of case report. A written informed consent taken from the patient.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Patient Approval
Patient consent obtained for publication of this case report.
