Abstract
Left ventricular noncompaction cardiomyopathy (LVNC) is a type of primary genetic cardiomyopathy, which occurs during embryogenesis by the arrest in the ventricular myocardium compaction. LVNC is characterized by prominent wall trabeculations and intertrabecular recesses that communicate with the ventricular cavity. There are 2 types of cardiomyopathy: the first one is associated with other primary cardiac structural abnormalities like malfunctional cardiac valves as mentioned in the case report below and the second type is in which there are no other associated cardiac structural abnormalities also called isolated LV noncompaction cardiomyopathy. We report an association of severe rheumatic mitral valve disease and LV noncompaction with significantly reduced ejection fraction which is rare.
Case Presentation
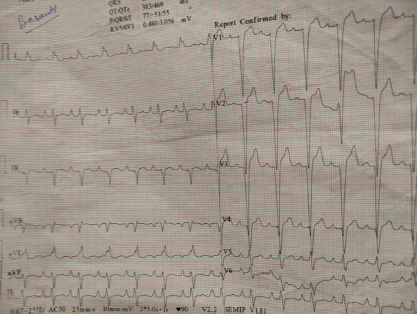
A 36-year-old man presented to the hospital with dyspnea on exertion for the past 3 months. He had no complaints of chest pain, palpitations, or syncope. No past history of hypertension or diabetes was present. He had a history of fever with joint pain 20 years back which subsided with oral medications. Family history did not reveal any history of sudden cardiac death or cardiomyopathy. Physical examination revealed bilateral crackles in the lower-lung fields and edema of lower extremity. Neurologic findings were unremarkable. An electrocardiogram showed complete Left bundle branch block with right atrial enlargement (Figure 1). The results of routine biochemical tests were within normal limits although his B-type natriuretic peptide level was 1,860.
ECG Showing Left Bundle Branch Block Pattern and Right Atrial Enlargement.
Investigations
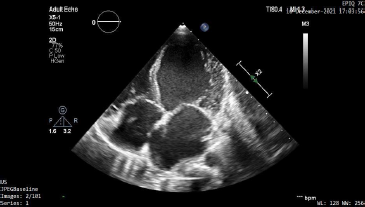
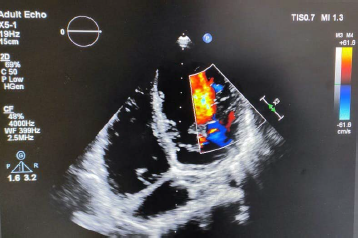
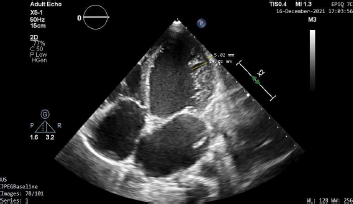
A transthoracic two-dimensional echocardiogram showed a dilated left ventricular (LV) cavity with global hypokinesia (Figure 2) and an ejection fraction of 35%. Perfusion of the intertrabecular spaces from the ventricular cavity was present at end-diastole on color-doppler echocardiography (Figure 3). The diagnosis of LV noncompaction was confirmed with the Stöllberger criteria 1 which showed >3 trabeculations protruding from the LV wall apical to the papillary muscles, perfused intertrabecular spaces, and a 2-layered myocardium with the noncompacted layer to compacted myocardium ratio being >2:1 in end-systole (Figure 4). Moderate mitral valve regurgitation and severe tricuspid valve regurgitation were seen with elevated pulmonary arterial systolic pressure of 36 mmHg. Severe mitral valve calcification with doming of the anterior mitral valve leaflet was seen in the parasternal long axis view (Figure 5). Mitral valve area was shown to be 0.4 cm2 by planimetry (Figure 6). Subsequently, coronary angiogram was done which showed normal epicardial arteries thus effectively ruling out ischemic causes of LV dysfunction. These findings confirmed the presence of heart failure secondary to LV noncompaction and presence of chronic rheumatic heart disease.
Apical 4 Chamber View Showing Dilated Left Ventricular Cavity With Global Hypokinesia.
Apical 4 Chamber View Showing the Characteristic Flow of Blood Into the Noncompacted LV Myocardium.
Apical 4 Chamber View Showing the Ratio Between Noncompacted to Compacted LV Myocardium to Be 2.79 Which Conforms the Presence of Left Ventricular Noncompaction.
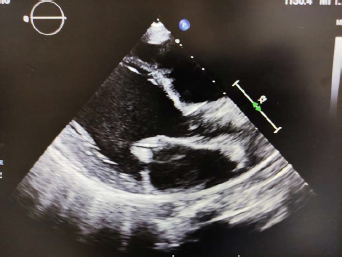
Parasternal Long Axis View Showing Severe Calcification and Doming of Anterior Mitral Valve Leaflet Leading to Restricted Opening of the Mitral Valve.
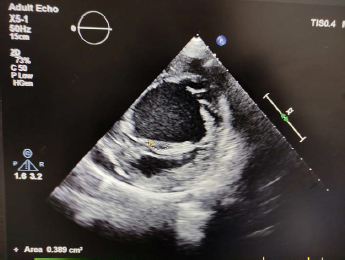
Parasternal Short Axis View Showing Mitral Valve Area to Be 0.4 cm2 by Planimetry
Outcome and Follow-Up
The patient was treated medically with beta blockers, ACE inhibitors, loop diuretic, digoxin, spironolactone, and was stable at follow-up. We did not initiate oral anticoagulation since no conclusive evidence of atrial fibrillation or prior cardioembolic event 2 was found as suggested by some studies.
Discussion
Left ventricular noncompaction cardiomyopathy (LVNC) was defined as a type of genetic cardiomyopathy by the American Heart Association in 2006 3 and described for the first time by Grant. 4 The main mechanism by which noncompaction occurs is said to be the ceasing of the ventricular muscular growth which is usually seen in the fifth and eighth week of gestational age. 5
Jansen et al 6 proposed that it has been hypothesized that hypertrabeculation was due to a defect in mammalian cardiogenesis and not due to failure of myocardial compaction. It was proposed that in view of the histological and magnetic resonance imaging findings, the trabeculations of embryos or ectotherms was not similar to the evolutionary and developmental models of normal and hypertrabeculated hearts in LVNC. Recently, immunohistochemical studies showed that the change from a noncompacted to a compacted myocardium 7 was due to growth in the compact zone of the embryonal myocardium and intermediate “hybrid zone.”
Currently, the diagnostic criteria for LVNC which is most widely accepted in adults relies on cardiac imaging which shows a 2-layered myocardial wall consisting of the noncompacted and the compacted layers and the ratio between them exceeding 2.1 as proposed by Jenni et al. 8 However, the existence of LVNC is considered as a morphologic trait shared by many other cardiomyopathies and to consider it as a distinct cardiomyopathy is under debate. 9
Echocardiography also estimates other parameters apart from measurements of noncompacted and compacted myocardial thickness, such as global LV function in ejection fraction and measures of ventricular size and volume. Increased mortality and adverse prognosis 10 have been consistently linked with reduced global LV function or LVEF. Though the association between the 2 conditions appears more as a coincidence, they impact patient management and prognosis by posing additional challenges due to gross myocardial dysfunction.
Learning Points
The hitherto rare association between chronic rheumatic heart disease and left ventricular (LV) noncompaction. The prevalence of adult LVNC ranges from 0.01% to 0.27% and the association of LVNC with valvular heart defects is even rarer.
The presence of 2 conditions concurrently, one causing systolic and the other causing diastolic heart failure, presents a therapeutic and diagnostic challenge and a multitude of life-threatening complications.
The importance of routine follow-up and echocardiographic assessment at timely intervals for patients with LV dysfunction cannot be understated as the prognosis of the patient worsens with further lowering of the ejection fraction.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
