Abstract
Inferior vena cava agenesis (IVCA) is a rare entitiy wherein many patients remain clinically silent and are found incidentally on radiological imaging studiesas a result of the compensatory enlargements of many collateral veins to maintain venous return. However, IVCA may also be diagnosed following serious acute complications, particularly unprovoked proximal iliofemoral deep vein thromboses (DVT) in young individuals. Herein, we report a 25-year old postpartum female patient presented with acute swelling on the right lower extremity and diagnosed with IVCA following acute occlusive iliofemoral DVTs.We aimed to emphasize the importance of radiological evaluation and management of these patients with some considerations out of routine practice.
Keywords
Introduction
The embryonic development of inferior vena cava (IVC) is a complex process with regressions and anastomoses of embryonic veins. Anomalous embryologic development of the IVC and its tributaries are rare congenital abnormalities. It was first described by Abernethy in 1793 with demonstration of a congenital mesocaval shunt and continuation of IVC with azygos vein in a 10-month-old child. 1 Since then, many other IVC abnormalities have been defined and started to be more frequently realized with the widespread use of cross-sectional imaging, particularly computed tomography (CT). IVC agenesis (IVCA) is one of the rare anomalous development of IVC having a controversial pathophysiological mechanism in which defective development of the embryonic veins and intrauterine/perinatal IVC thrombosis are both suggested. 2 Many patients remain clinically silent but, it may also be diagnosed following its serious complications, most commonly unprovoked multiple deep vein thromboses (DVT) in young individuals, particularly in lower extremities (LEs). Herein, we aimed to present a young postpartum woman diagnosed with IVCA following intractable proximal DVT.
Case Presentation
A 25-year-old female patient admitted to the emergency department of a hospital with complaint of acute swelling on the right LE, 1 week after giving birth. On Doppler ultrasonography (US), acute DVT in the right LE was confirmed and she was discharged with conservative treatment including oral anticoagulants and compression stockings. After a week, due to the increased swelling and ongoing symptoms, she was admitted to our hospital for further evaluation. She was a nonsmoker and her past medical history regarding thrombophilia was unremarkable except being in the postpartum period. On the physical examination, the right LE was swollen, painful, and erythematous. Laboratory investigations revealed a normal hemogram and biochemical markers. In the coagulation screen, international normalized ratio (INR) was 2.19 (range: 0.15-1.2), prothrombin time (PT) was 25 s (range: 10-14), and activated partial thromboplastin time (APTT) was 41.5 s (range: 30-40) under the anticoagulation treatment which was started before 1 week. C-reactive protein was raised (43 mg/L, range: 0-10) and the D-dimer was found to be 1,009 ng/mL (normal range: 0-500). On Doppler US, she was found to have an extensive, occlusive right iliofemoral thromboses. The hepatic IVC was normal till the level of renal hilus but the lowermost segments could not be visualized under the bowel gas artefacts. On the left LE, there was no thrombosis. Pharmacologic catheter-directed thrombectomy with AngioJet Zelante DVT catheter was performed (Figure 1). Residual thrombosis was observed and an ordinary 5Fr Pigtail catheter was punctured at multiple spots along distal portion with a needle to open small infusion holes due to the lack of special infusion catheters (Figure 2). Overnight thrombolysis with 1 mg/h tissue plasminogen activator was performed and the next day control venography before catheter withdrawal revealed good flow and minimal residual thrombosis. However, it was realized that the catheter did not advance into the IVC. Without forcing, it was decided to try on the left side through the left popliteal vein (PV) but the same challenge was present (Figure 3). CT venography (CTV) of the abdomen and pelvis was ordered to evaluate the abnormality. Agenesis of the infrarenal segment of the IVC with normal suprarenal IVC segments was revealed (Figure 4). Patent renal veins and numerous dilated paravertebral collateral veins draining into the azygous/hemiazygous system were demonstrated (Figure 5). The confluence of renal veins was shown to be continues as the dilated azygous vein drains into the superior vena cava (SVC) by passing posterior to the diaphragm to enter the thorax. Both of the common iliac veins (CIVs) were also absent with thrombosed right external iliac and femoral veins (Figure 6). Her thrombophlia screen was negative. During hospitalization, she received low molecular weight heparin (enoxheparin, 60 mg twice daily) and warfarin (5 mg/day) in addition to wearing compression stockings. She was discharged when INR reached 2 but represented a fortnight later with a swollen and painful right LE. Doppler US confirmed the presence of recurrent occlusive right-sided iliofemoral thrombus extending all the way through the PV. The thromboses were reaspirated again with AngioJet Zelante DVT catheter but this time Clopdiogrel (75 mg/day) and acetylsalicylic acid (81 mg/day) were added to the warfarin therapy during hospitalization and compression stockings were not applied with the consideration of its possible worsening effect. Her edema regressed and she was discharged with acetylsalicylic acid (81 mg/day) and warfarin (5 mg/day). The INR at discharge was 2.5.
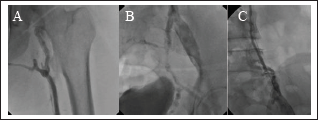
(A) Diagnostic venography of right extremity performed from popliteal vein sheath. Heavy thrombosis of femoral vein and absence of filling after iliac vein. (B) Heavy thrombosis of external and internal iliac veins of right lower extremity. (C) Heavy thrombosis of external, internal iliac veins and venous drainage after iliacs by a well developed collateral.
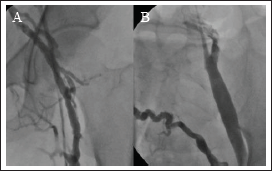
(A) Reduced thrombus burden after multiple passages of Angiojet Zelante aspiration catheter in deep femoral and iliac veins. (B) Minimal thrombus in main iliac and large collaterals and absence of vena cava visualization.
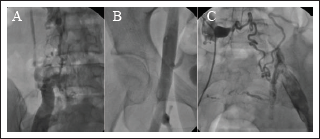
With consideration of chronic occlusion of right main iliac vein or IVCA, a diagnostic catheter was inserted also from the left PV in order to reach from left iliac vein and the absence of IVC was confirmed. (A) Left iliac venography showing absence of IVC and well-developed collaterals for iliac vein drainage. (B) Normal left iliofemoral veins. (C) A well-developed collateral from left iliac to right iliac vein.
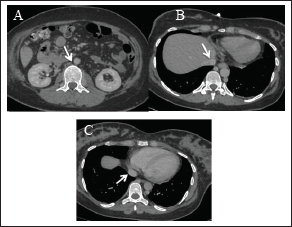
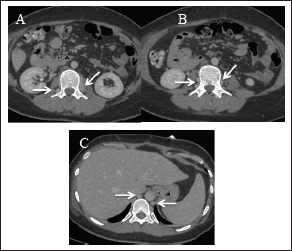
CTV of the abdomen and pelvis showing absence of the infrarenal segment of the IVC (A, arrow) with normal suprarenal IVC segments; hepatic IVC (B, arrow); and suprahepatic IVC (C, arrow).
CTV of the abdomen and pelvis showing numerous dilated paravertebral collateral veins (A, B, arrows) draining into the dilated azygous/hemiazygous veins (C, arrow).
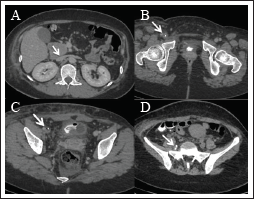
CTV of the abdomen and pelvis showing the confluence of the renal veins which continue as the dilated azygous vein (A, arrow). The thrombosed right femoral (B, arrow) and external iliac vein (C, arrow) are demonstrated. The catheter turn in the right EIV due to the inability to advance into the CIV is visible (D, arrow).
Discussion
Normal Anatomy
In the normal anatomy, venous return to the right atrium is provided by the SVC for the upper half of the body and by the IVC for the lower half of the body. The azygous/hemiazygous venous system connect the superior and inferior caval systems and provide an alternative pathway to the right atrium when there is an interruption in either of these pathways. The IVC runs on the right side of the vertebral column and the aorta, draining venous blood from the abdomen, pelvis, and lower limbs to the right atrium. It is formed by the confluence of 2 CIVs at the level of L5 vertebra. During its retroperitoneal course in the abdominal cavity, the inferior phrenic veins, the lumbar veins, right gonadal vein, renal veins, right suprarenal vein, and hepatic veins drain into the IVC before they traverse the diaphragm through the caval hiatus at T8 level and drain into the right atrium after a short intrathoracic course. On the left side, the gonadal and suprarenal veins drain into the left renal vein.
Embryology
In the embryological development of IVC, a complex process involving anastomoses and posterior regressions of primitive embryological veins, namely the vitelline vein and the paired posterior cardinal, subcardinal, and supracardinal veins, takes place at around 6 to 8 weeks of gestation. While the infrahepatic IVC develops from 3 paired embryonic veins, the infrarenal IVC develops from the posterior cardinal and supracardinal veins. The posterior cardinal veins are progressively replaced by the subcardinal and supracardinal veins in the abdomen, but persist in the pelvis to form distal most IVC and its bifurcation into the CIVs. In the thoracic region, the supracardinal veins give rise to the azygous and hemiazygous veins. 3
Inferior Vena Cava Agenesis
Congenital IVCA is one of the less prevalent malformations of IVC with an approximate incidence from 0.005% to 1%. 4 It can occur in isolation or can be associated with other malformations like cardiovascular abnormalities, biliary atresia, or polysplenia/asplenia syndrome. 5 It can be encountered either as the form of “the interruption of infrarenal segment with preservation of the suprarenal IVC” as in our case or “the interruption of the infrahepatic segment with azygous/hemiazygous continuation.” Absence of infrarenal IVC implies the failure of the posterior cardinal and supracardinal veins. In these cases, a normal suprarenal IVC is formed by the confluence of the renal veins. 3 Interruption of the infrahepatic segment of the IVC with azygos continuation implies the failure of the subcardinal, supracardinal, and posterior cardinal veins. In this case, the hepatic segment of IVC is either absent or hypoplastic. While the hepatic veins drain into the suprahepatic IVC, the confluence of renal veins continues as the dilated azygous vein (Figure 6A) which drains into the SVC by passing posterior to the diaphragm to enter the thorax. In both of these conditions, CIVs may also be absent and the external iliac veins (EIVs) and internal iliac veins (IIVs) join to form lumbar veins, as in our case or the CIVs may be formed and drain into the lumbar veins, which in turn drain into the dilated azygous-hemiazygous system via collateral paraspinal circulation to convey the venous blood into the right atrium. As a result, venous drainage of the kidneys and the LEs into the right atrium occurs by means of SVC and facilitated by the compensatory dilatation of extensive venous collaterals. In addition, superficial abdominal veins, the gonadal venous system, and the haemorrhoidal plexus veins may also be dilated to serve as a collateral pathway for venous return. Because of this extensive collateral venous pathway, many patients remain asymptomatic. However, the increased venous pressure and stasis as compared to the general population induces development of DVTs, particularly in the LEs and occasionally in the abdomiopelvic veins especially in the presence of secondary risk factors like prolonged immobilization, smoking, or oral contraceptive use. As a result, the most encountered presentation is unprovoked DVTs, usually bilateral proximal (iliofemoral) DVTs in young patients. 6 Much more rare than that, intraabdominal particularly renal vein thrombosis in these patients has also been reported in the literature. 7 In patients with IVCA, pulmonary embolism and post-thrombotic syndrome (PTS) are the main concerns after venous thrombosis. The extensive collateral development reduces the risk of pulmonary embolism in these patients but it is still probable. 8 On the other hand, PTS, a syndrome of chronic venous insufficiency due to valvular incompetence associated with an inflammatory response, is characterized with chronic venous reflux and chronic venous hypertension with varicose veins, limb swelling, pain, discoloration, and in severe cases, venous ulcerations is a frequent and disabling complication of DVTs. It is particularly more frequent and severe in patients with proximal iliofemoral, recurrent, and anatomically extensive ipsilateral DVTs, as in cases with IVCA. The other problem in these patients is kidney and liver transplantation surgeries. In the absence of infrahepatic IVC, liver transplantation is more commonly required in pediatric patients with associated biliary atresia occurring as a component of syndromic presentations. However, in adult cases with isolated absence of IVC, kidney and liver transplantations may also be required as in general population. In these cases, radiological evaluation for vascular abnormalities is very important to determine the surgical and interventional radiological approach accurately.
Radiological Imaging
In the radiological evaluation of the anomalies of the IVC, the gold standard is conventional venography. As a non-invasive imaging modality, intravenous (IV) contrast enhanced abdominopelvic CT scan is the method of choice, in which venous structures are easily identified at delayed venous phase imaging. On routine portal venous phase abdominopelvic CT scans (60-70 s after IV administration of contrast agent), the admixture artefact in the infrarenal/renal IVC caused by flow of enhanced blood from renal veins and non-pacified blood returning from lower limbs should be kept in mind to avoid misdiagnosis of this filling defect as a thrombus. Increasing the delay after contrast material injection from 70 to 90 s allows more uniform enhancement of the entire IVC. The use of US and Doppler US has a limited role due to the superimposed bowel gas in the abdomen in the evaluation of the infrahepatic IVC in adults. However, US plays an important role in the way of diagnosis by demonstrating the DVTs progressing through the iliac veins and making the abdominal imaging necessary for the evaluation. On Doppler US, when a proximal iliofemoral DVT is present, it is important to perform spectral analysis on the common femoral vein (CFV) of the contralateral asymptomatic side. Although the presence of phasic flow and good augmentation cannot exclude proximal obstruction, nonphasic or asymmetrical pattern of flow in the CFV during respiration suggests an involvement of a more proximal vein. 9 On CTV, in addition to absent IVC segments, CIVs may also be absent, as in our cases. The EIVs and IIVs join to form enlarged ascending lumbar veins, which convey venous blood from the LEs to the azygos and hemiazygos system via anterior paravertebral collateral veins. On a frontal chest radiograph, the dilated azygous veins may be seen as widened right paratracheal/paraspinal stripe and mistaken as a mass lesion similar to the prominent collateral lumbar veins that can be mistaken for paraspinal masses or adenopathies. If the IV contrast of CT is contraindicated, magnetic resonance imaging may be used to show flow voids or flow-associated enhancement in order to differentiate the anomalous collateral venous structures from retroperitoneal masses or adenopathy.
Management
The treatment of IVCA is difficult and not standardized. In incidentally found asymptomatic and noncomplicated patients, IVCA requires no treatment. In order to prevent development of thrombosis in these patients, the risk factors of hypercoagulability like smoking, OC use and long-time immobilization should be avoided. In cases presenting with acute DVT, the decision of endovascular mechanical thrombectomy and/or thrombolysis should be made on a case-by-case basis. Before anything else, the possibility of IVCA should be kept in mind particularly in young patients with proximal iliofemoral unprovoked DVTs and appropriate diagnostic imagings with CTV or magnetic resonance venography should be performed. Urgent thrombolysis in the acute DVT is indicated to prevent end-organ complications. Comparable procedural success was reported with the use of adjunctive endovascular thrombectomy to augment pharmacologic catheter-directed DVT thrombolysis and can be performed after a careful evaluation of bleeding risk in selected patients with extensive DVTs to reduce the thrombosis burden which in turn reduces both the required thrombolytic dose and the risk of recurrence. 10 Oral anticoagulation is recommended but it is controversial that whether lifelong anticoagulation should be performed or a shorter duration of anticoagulation (3-6 months) should be selected if there are no additional risk factors for thrombophilia. 11 Concurrent use of anti-thromboembolic compression stockings is recommended in the acute setting but after DVT, its continuation to prevent DVT recurrence and PTS is not advocated.12, 13 However, also in the acute setting the use of compression stockings may not be advocated with possible worsening effect as in our case in whom the recurrent DVT occurred despite being on therapeutic anticoagulation. In our patient, we thought that compression stockings caused more distal stasis in our patient due to the ineffective proximal venous drainage and we excluded it from the treatment, which improved the situation in in addition to more aggressive anticoagulation therapy. However, due to the risk of bleeding, it was decided to continue with a target INR between 2 and 3 with acetylsalicylic acid.
Conclusion
IVCA may present with serious acute complications. Especially, in young patients presenting particularly with proximal iliofemoral DVTs, the possibility of developmental anomalies of IVC should be kept in mind to precisely manage the situation. Moreover, knowledge of the IVCA is important even in the asymptomatic patients to avoid misdiagnoses during imaging studies and for appropriate preoperative surgical and interventional radiological preparations.
Learning Points
During venous interventions, IVCA should be remembered before attempting aggressive wires and maneuvers for chronic total venous occlusion passage.
Contralateral popliteal puncture and CT imaging at that time clarifies anatomy and should not be hesitated.
In venous interventions, thrombectomy and catheter thrombolysis can be complemented for an optimal result.
Compression stockings may have a worse effect when acute DVT is associated with IVCA.
Footnotes
Author Contributions
Concept: Berrin Erok, Nu Nu Win; Design: Berrin Erok, Ayhan Olcay; Supervision: Berrin Erok, Ayhan Olcay, Analysis: Berrin Erok, Nu Nu Win, Elidor Agolli Ayhan Olcay; Literature search: Berrin Erok, Elidor Agolli; Writing: Berrin Erok; Critical revision: Berrin Erok, Ayhan Olcay.
Availability of Data and Materials
Resource scanning was done in PubMed with appropriate keywords.
Declaration of Conflicting Interests
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from the patient.
