Abstract
Introduction:
The World Health Organization in 1971 first classified fibro-osseous lesions (FOLs) as cementum forming tumors of jaws as ossifying fibroma (OF), cement-osseous dysplasia, and fibrous dysplasia. Various theories have been put forward regarding the origin of cemento-ossifying fibroma (COF) but current interests are oriented toward traumatic and developmental etiologies. The COFs are a slow-growing osteoexpansile tumor which grows to a considerable size. The tumor usually arises from tooth bearing and periodontal ligament region basically of odontogenic origin.
Background:
An inordinate, bony hard swelling, present united in both the jaws showing radiopaque-radiolucent mass, corticated margins, with expansion of buccal as well as lingual cortex gives a delusional image for the provisional diagnosis for surgeon, particularly to decide the surgical approach. These traits showing similarity with FOLs, more particularly cemento-OF, owing to its odontogenic or periodontal origin.
Case Report:
In this case, a 42-year-old female presented with abovementioned features was first taken for biopsy and then planned for simultaneous surgical resection of pathology for both jaws, from an intraoral approach giving a scar-free result. Preclusion of immediate complications was dealt with reconstruction of mandible with 2.5-mm titanium reconstruction plate.
Conclusion:
The diagnosis of cement-OF is based on clinical-radiographic-histopathologic evaluation. If remain untreated, these tumor can grow up to an average size of 80 mm, therefore, demanding an early surgical intervention. In our case, intraoral approach leads to almost no residual fibrosed tissue. The patient is further being planned for rehabilitation with either free fibula graft or 3-dimensional mandibular prosthesis.
Keywords
Introduction
The World Health Organization (WHO) in 2017 introduced a new classification on head and neck tumors which included not only tumors and cysts but also various other modifications in its fourth edition. The concept of “fibro-osseous lesions” (FOL) was also included in the new classification which is broadly recognized as cement-osseous dysplasia (COD), fibrous dysplasia (FD), cemento-ossifying fibroma (COF), respectively. 1 The old term ossifying fibroma (OF) is now being replaced as COF in view of making a differential diagnosis and also the fact that the tumor usually arises from tooth bearing and periodontal ligament (PDL) region basically of odontogenic origin. A specific code was allotted to COF which is designated as ICD-O-9274/0 in the 4th edition of WHO classification. The acronym COF applies only to OFs excluding its juvenile counterpart such as juvenile-ossifying fibromas (JOF) trabecular and psammomatoid types. The COFs are rarely encapsulated or well-demarcated lesions made up of fibrous tissue with varying amounts of mineralized tissues such as bone, cementum, or both. They are most commonly seen in women than men usually in the fourth or fifth decades affecting the mandibular body region predominantly and they account for about 70% of benign FOLs in the head and neck region. 1 This case report portrays a giant cement-OF of mandible which was managed by segmental resection by intraoral scar-free approach.
Case Presentation
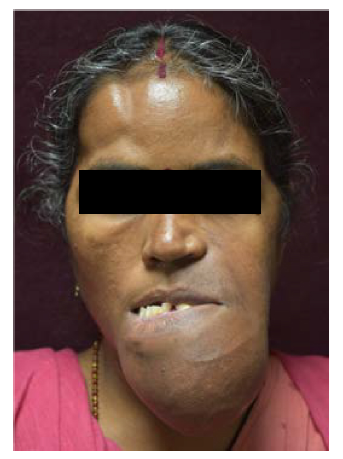
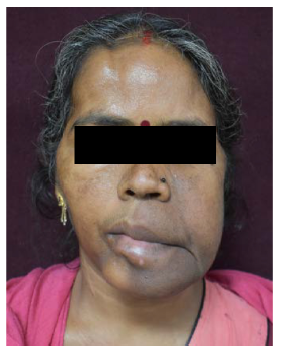
A 42-year-old female patient reported to the department with the chief complaint of swelling in both the jaws and difficulty in eating for 20 years (Figure 1). The swelling gradually increased in size and developed to the present size. On extraoral examination, there was gross facial asymmetry over middle and lower third of face more on the left side. The swelling over midface was about 6×7 cm and extends superior-inferiorly from left preauricular region to angle of mandible and anteroposteriorly extends from left preauricular region to commissure of mouth. The swelling over lower third of face extends from left angle of mandible to right side body of mandible with dimensions 12×7 cm anterioposteriorly. Superioinferiorly swelling extends from angle of mouth to lower border of hyoid bone. The skin over swelling is erythematous and stretched with no signs of any pus discharge. On palpation, the swelling is firm in consistency, nontender, and no complaint of paranesthesia over lips and skin. Intraorally, all teeth are present and displaced within the tumor in second, third, and fourth quadrants. In maxilla, the swelling extends from midline to 26 region obliterating the labial and buccal vestibule, respectively. In mandible, the swelling extends from 36 to 46 region obliterating labial, buccal, and lingual vestibules. The dimensions of swelling in mandible and maxilla were 11×5 cm and 6×4 cm, respectively. The swelling is firm in consistency, nontender, no pus discharge, and bleeding on provocation.
Radiographic Evaluation
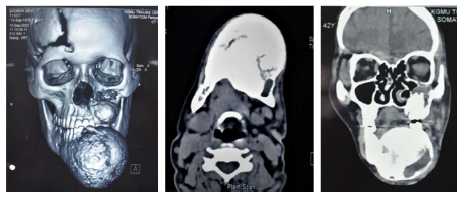
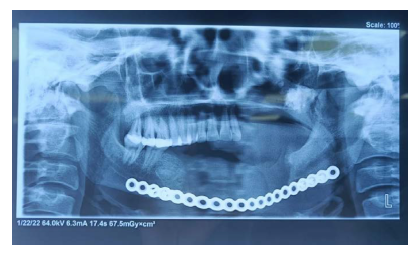
Noncontrast computed tomography (CT) was done which revealed 2 well-defined radiopaque expansile lesions which were present in the left maxilla and across the midline of mandible, respectively (Figure 2).
Mandible
The lesion in mandible extends across the midline from 45 to 38 mesiodistally, buccolingually and superior-inferiorly lesion extends across the entire width and height of mandible causing expansion of both buccal and lingual cortices. The lesion is round in shape with corticated margins. The internal structure is in-homogenously radiopaque with dense radiopaque masses presenting some amount of amorphous calcifications. The lesion along with bicortical expansion shows buccal drifting of 32,33,34,35.
Maxilla
The lesion in maxilla extends mesiodistally from 21 to entire length of maxilla. Superiorly, it extends from nasal floor in anterior part and the sinus floor in posterior part displacing them superiorly and the crestal bone inferiorly. Buccolingually, the lesion extends along the entire length of alveolus causing expansion of cortices. The lesion is irregular in shape with corticated margins. Internally, the lesion is in-homogeneously radiopaque with multiple amorphous calcifications. The radiographic appearance was conclusive of fibro-osseous lesion (cement-OF).
Histopathological Evaluation
Preoperative Evaluation
An incisional biopsy was done from both the maxilla and mandible which showed abundant cellular stroma with variable-shaped cells, and many small round ossicles are seen embedded in the cellular fibrous stroma as well as within bony trabeculae. Osteoblastic rimming is noted along with a few endothelial-lined blood vessels seen with extravasated red blood cells suggestive of OF.
Postoperative Evaluation
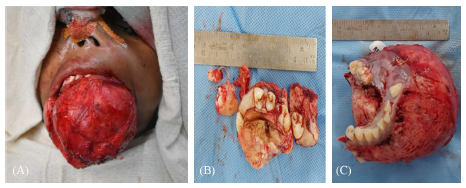
In maxilla, complete resection of lesion was done measuring 5.5×3.0×3.0 cm with attached upper canine, premolar, and molars, whereas in mandible sample size measuring 8.5×7.0×5.0 cm with multiple teeth from third and fourth quadrants were sent for histopathological evaluation (H/E) showing proliferation of round-to-spindle cells with bony trabeculae seen embedded in a cellular fibrous stroma which is rimmed by osteoblasts along with a fair number of multinucleated giant cells, and small oval to spindle mononuclear cells are noted. The histopathological diagnosis is consistent with cement-OF (Figures 3, 4, and 5).
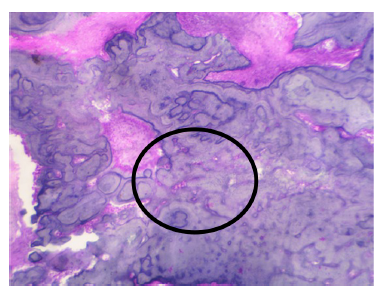
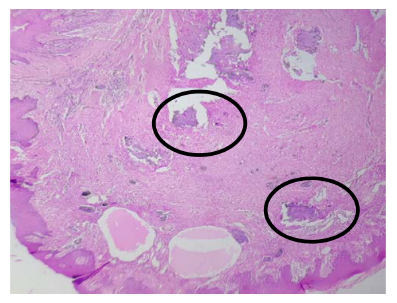
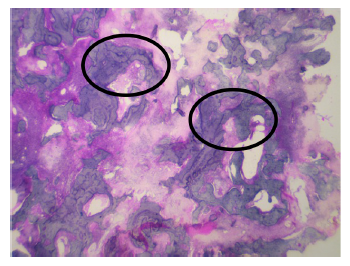
There is a famous saying by late Charles Waldron that “In absence of good clinical and radiologic information a pathologist can only state that a given biopsy is consistent with a FOL.” So the final diagnosis is made on the basis of correlations between clinical, radiological, and H/E, respectively.
Diagnosis and Treatment
On correlating clinical, radiographic, and histopathological findings, a final diagnosis of cemento-OF of maxilla and mandible was made (Figure 6). Complete segmental resection of mandible from molar to molar region and partial maxillectomy of the lesion and reconstruction was done with 2.5-mm Indian titanium reconstruction plate done under general anesthesia by an intraoral vestibular approach. Postoperative healing was satisfactory with minimal cosmetic deformity noted on subsequent follow-ups. (Figures 7 and 8) The patient was followed up for a period of 6 months.
Discussion
Almost a century and a quarter ago, Menzel in 1872 gave a detailed description of a variant of OF of mandible calling it COF. 2 Since then, various authors reported such occurrences in craniofacial bones and called them by different names owing to the difference in clinical, histological, and radiographic evaluations which were later known to be called “fibro-osseous lesions” (FOL) of craniofacial bones. The WHO in 1971 first classified FOLs as cementum forming tumors of jaws as OF, COD, and FD. The former 3 editions in 1972,1992, and 2005 paved a way for present nomenclature and terminology. 1 Various theories have been put forward regarding the origin of COF but current interests are oriented toward traumatic and developmental etiologies.3, 4 Bernier et al postulated that COF may arise from fibrosis and inflammation of the periapical region leading to irritation of PDL and resulting COF. 5 Cakir et al explained that some nasopharyngeal COF originated from embryonic nests, respectively. 6 The COF may also be present in sphenoid, ethmoid, parietal, and temporal bones suggesting the presence of ectopic periodontal membrane tissue. Gollin et al did some cytogenic and cario-typing analysis where his research showed mutation in G protein in chromosome 13 along with 3 translocations responsible for it. 7
The COF is broadly classified as central and peripheral type, where the latter affects predominantly the soft tissues of the jaw and is known by various names such as ossifying fibroepithelial polyp, peripheral fibroma with calcifications, and calcifying fibroblastic granuloma, respectively. 2
The COF most commonly affects mandible and maxilla and is most frequently seen in women in the third and fourth decades of their lives as reported in this case. The ratio is in the range of 2:17 to 5:111. 2 The lesion when it occurs in the juvenile age group is usually aggressive in nature with more vascularity at a pathological level and needs aggressive surgical intervention. 8 The COFs are a slow-growing osteoexpansile tumor that grows to a considerable size averaging 80 mm in Asian populations. Classically, this tumor appears ovoid in shape owing to a centrifugal pattern of growth, that is, growth from the center toward periphery. 9 Swelling is the most common symptom and it attains a huge size with time.
Radiographically on CT images, they appear as a single well-defined radiolucent area with diffuse radiopacities and thick sclerotic rim showing peripheral osteocondensation. 10 Eversole et al described 2 major patterns such as unilocular and multilocular radiolucent patterns with scattered radiopaque foci. 11 The size of the tumor is directly proportional to the amount of mineralized tissue deposits like cementum or bone slowing converting a radiolucent lesion to radiopaque one with time, thus contributing to the phenomenon of “maturization.” This term is a misnomer and doesn’t favor neoplasias, as neoplasm continues to grow and does not mature biologically. Marcia de Andrade et al in their case series showed that 62.5% of cases were mixed lesions. 12 Waldron and Giansanti showed that 26% were lytic, 12% were diffuse and homogenous, and 63% were mixed lesions.13, 14 Contrastingly, a study by Titinichi et al showed 49% of lesions were radiopaque, 34% were mixed, and only 15.9 % are radiolucent. 15 Whereas the panoramic study showed predominantly radiolucent lesions. A systematic review by Macdonald-Jankowski of 64 reports showed that 84% cases showed buccolingual expansion of the cortices and almost half of the cases showed downward displacement of the lower border of mandible and 90% cases in maxilla involved the maxillary antrum. 10 Based on osteoblastic rimming at periphery, Barberi et al in their study showed 40% of cases were without osteoblastic rim, 45% with osteoblastic rimming, and 15% of cases have diffuse ill-defined borders. 16 Tejinder Kaur et al in their case series showed that 87.5% of cases demonstrate ground glass opacification with divergent root or resorption and mixed radiolucent-radiopaque picture. Provisional diagnosis in our case was given based on clinical and radiological features. 17 The differential diagnosis was cement-osseous dysplasia, calcifying epithelial odontogenic tumor (CEOT), calcifying odontogenic cyst, cementoblastoma, and Paget’s disease.
According to Muller and Slootweg’s concepts of marginal definition, a lesion with a transitional zone of <1 mm is considered to be well defined. This helps to easily differentiate COF and FD as the latter has ill-defined margins and is also easily distinguishable from other carcinomas and sarcomas. It is even more difficult when the lesion contains impacted teeth as it is also seen in calcifying odontogenic cysts (Gorlin cysts) and CEOT, respectively. The lesion may also resemble when it occurs in proximity to roots (cementoblastoma) or when it involves more than 1 jaw as in florid-osseous dysplasias. The COF can be easily differentiated from odontomas as they show multiple tooth like structures, unlike COF which shows multiple radiopaque foci. The focal osseous dysplasia when occurs in its intermediate stage could become a potential differential diagnosis except for poor differentiated radiographic margins. Serum alkaline phosphatase levels and an incisional biopsy is required to differentiate it from osteosarcoma and Paget’s disease. 18 So, the final diagnosis heavily relies on histopathological examination.
Histologically, the COF shows increased cellular stroma with irregular calcified fragments embedded in the matrix depicting deposition at varying time periods. Although it is difficult to differentiate between osteoid and cementum, histologically the basophilic staining and osteoblastic rimming on hematoxylin-eosin stained sections helps COF to differentiate it from FD which lacks it. In FD, peritrabecular clefting is an important diagnostic feature. Immunochemical studies by Endo et al showed that the COF shows high immune reactivity for keratin sulphate whereas FD shows positivity for chondroitin-4-sulphate.
The COF is known to coexist in patients with end-stage renal disease or can transform into an entire new lesion. The hyperparathyroidism-jaw tumor syndrome is in which patients suffer from familial parathyroid adenomas, multiple OFs of jaws, Wilms tumor, and renal cysts. The HRPT-2 gene mutation in this syndrome inactivates fibromin protein which has antiproliferative properties. 19 The studies by Trent et al, Wojno and Mccarthy, and Padwa et al showed that aneurysmal bone cyst (ABC) is associated with or secondarily gets transformed from other FOLs 21%, 12%, 22%, respectively, by a phenomenon called “hemorrhagic blowout of the lesion.” 20 This shows COF can get transformed into ABC secondarily giving the name ABC plus. In our case, there was no such transformation but intraoperative surgical excision was difficult as the lesion was highly vascular and completely excised by an intraoral approach.
The COF is usually treated by conservative enucleation and curettage or radical surgery. As COF is well circumscribed and separated from the bone, it can be treated by conservative surgical excision. The criteria are enucleation and curettage for small lesions and lesions with no definitive radiolucent margins at the periphery. The marginal resection with bone grafts for lesions where lower border was spared and complete segmental resection and reconstruction with vascularized or nonvascularized bone grafts done for giant lesions with classic bowing of lower border are recommended. In our case, surgical resection was facilitated by an intraoral approach owing to the simultaneous expansion of the jaw along with soft tissues which contributed to the delivery of the lesion without an external scar. The reconstruction was done with 2.5 mm Indian titanium reconstruction plates and the patient was followed up for a period of 8 months. Tapas et al in their study showed a 10.1% recurrence rate after 25 months of follow-up. Eversole and Liu in their study showed recurrence rates of 28% and 27.2%, respectively, after conservative enucleation and curettage of the lesion with 10 years follow-up. 21
Conclusion
The COF is a benign tumor of jaws affecting most commonly women in their mid-30s and 40s and can attain a humongous size if left untreated. The most common etiology is trauma leading to stimulation of mesenchymal cells of PDL and ectopic PDL tissue and appears as round or ovoid nontender swelling clinically owing to its centrifugal growth. Radiographically, usually appears as a mixed radiolucent-radiopaque lesion at the time of presentation. The final diagnosis is made on the basis of clinical-historadiographic evaluation. The treatment is conservative resection if the lesion is small without continuity defect or complete resection of the lesion and primary reconstruction with 2.5 mm reconstruction plate or with vascularized bone grafts either primarily or secondarily after a minimum follow-up of 5 years. The lesion rarely involves orbital floor or causes blindness but causes deviation of the nasal septum, displacement of dentition, and mass effect on vital structures posterior to maxilla. When it involves mandible, it might cause posterior displacement of the tongue or will compress the trachea causing difficulty in breathing. The tumor is radioresistant and doesn’t respond to any chemotherapeutic agents. The recurrence rate is very low after good surgical clean-up. The delivery of a gigantiform COF often requires an external incision, unlike rarely in our case which is exceptional.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
