Abstract
Background:
Green light photobiomodulation (GPBM, 500–565 nm) promotes tissue healing, reduces inflammation, and alleviates pain, with emerging potential in bone repair. This review evaluates GPBM’s therapeutic effects on bone-related cells (proliferation, differentiation, migration), inflammation, vascularization, and neuronal responses.
Methods:
We assessed the influence of different GPBM parameters, possible underlying molecular mechanisms, and prospects for in vivo applications based on available studies.
Results:
GPBM acts through mitochondria, opsins, and transient receptor potential vanilloid (TRPV) channels. The most effective irradiation method uses low irradiance (5.3–100 mW/cm2) with a single exposure of ∼5 J/cm2. Excessively low irradiance is ineffective, while excessively high irradiance may cause inhibitory effects. GPBM enhances bone-related cell functions, suppresses inflammation, and promotes vascular and neuronal responses.
Conclusions:
GPBM shows promise for bone repair, but further research is needed to optimize parameters, clarify molecular mechanisms, and standardize clinical trial protocols to ensure reliable in vivo outcomes.
Introduction
Bone repair is typically divided into four stages: the initial inflammatory response, cartilage tissue healing, hard bone tissue healing, and bone remodeling, which requires the synergistic action of bone-related cells (such as osteoblasts), vascular cells, and inflammatory cells. 1 Research has also indicated that the nervous system strongly affects bone repair. 2 Traditional bone repair methods primarily consist of bone grafting, including autologous bone grafts, allogeneic bone grafts, xenogeneic bone grafts, and biomaterial implants. 3 However, these methods have long been challenged by prolonged repair times, a low density of newly formed bone, high risk of infection, and poor biocompatibility. Photobiomodulation (PBM) is an innovative therapy that promotes bone repair through external irradiation and enhances the repair process when combined with bone grafting techniques. PBM has demonstrated its ability to overcome the limitations of traditional bone repair methods with immense therapeutic potential.
Since the discovery of PBM by Mester et al. in 1967, red (625–740 nm) and near-infrared light (740–1000 nm) with deep tissue penetration has been widely applied to promote human tissue healing, reduce inflammation, and produce analgesic effects.4,5 Numerous studies have shown that PBM at wavelengths of 635–980 nm can regulate osteoblast differentiation and proliferation, promote osteoclast formation, enhance vascular epithelial cell proliferation and migration, and inhibit osteocyte apoptosis through mechanisms involving cytochrome c oxidase (CCO), transient receptor potential (TRP) channels, hemoglobin, myoglobin, water, opsins, flavins, and cytochrome P450, thereby promoting bone repair. 6 In contrast, the use of shorter wavelength green (500–565 nm) and blue light (450–485 nm) received little attention in early studies regarding bone repair due to the limited tissue penetration depth of these wavelengths. Asagai et al. demonstrated that in children with cerebral palsy and incomplete fractures, light-emitting diode (LED) irradiation (with peak wavelengths ranging from 446 to 660 nm) increased bone density and femur cortical bone thickness, improving bone age, which suggested that blue and green light may be beneficial for human bone repair. 7 However, this study used mixed wavelengths of light, and therefore, the specific effects and mechanisms of green and blue light on bone repair were not clear.
Bone repair is closely associated with bone-related cellular differentiation (osteogenic differentiation and neurogenic differentiation), proliferation, migration, inflammation, and vascular regeneration. Over the past decade, green light has demonstrated significant advantages in in vitro studies related to these aspects of bone repair.1,2 Its safety and penetration depth are superior to those of blue light; further, blue light, with a wavelength close to that of ultraviolet light, is prone to generating excessive reactive oxygen species (ROS) in target tissue.5,8,9 Prayitno et al. were the first to discover that green light exposure increased bone mass in broilers, outperforming other light wavelengths. 10 Higuchi et al. found that visible light promoted the osteogenic differentiation of human amniotic fluid-derived mesenchymal stem cells, and that green light showed the best effect. 11 In the same year, Fushimi et al. discovered that green photobiomodulation (GPBM) promoted vascular regeneration in a mouse wound healing study. 12 Anwer et al. reported for the first time that GPBM enhanced the proliferation of human adipose-derived stem cells (hADSCs). 13 Ong et al. were the first to show that GPBM facilitated the migration of human orbital fat stem cells (hOFSCs). 14 Catao et al. found that GPBM exerted an anti-inflammatory effect during the wound healing process in rats. 15 Dehghani-Soltani et al. discovered for the first time that GPBM promoted the neurogenic differentiation of human umbilical cord-derived mesenchymal cells (hUCMs). 16 However, due to the limited penetration of green light, early studies mainly focused on superficial tissues, with few investigations extending into the deeper bone structures. It was not until 2020 that Jiang et al. overcame the limited green light penetration depth by using biodegradable optical waveguide technology, thus successfully applying green light to bone tissue repair. 17 These studies indicated the immense potential of green light in bone repair and new approaches for future clinical research.
This review focuses on the complex sequential process of bone repair by reviewing the role of green light in bone-related cellular differentiation (osteogenic differentiation and neurogenic differentiation), proliferation, migration, inflammation, and vascular regeneration; the effects of different GPBM parameters; the underlying molecular mechanisms; and implications for future research. This review presents three innovative aspects. (1)This review focuses on the effects of green light on bone regeneration-related processes. In addition to including studies related to GPBM and cellular migration, proliferation, and differentiation associated with bone, it also encompasses literature concerning tissue regeneration involved in bone healing, such as inflammation, vascular regeneration, and neurogenic differentiation. (2)This review incorporates the most recent research on the effects of green light on bone regeneration to date. (3) The PBM parameters were standardized by consolidating terms like “Spot area,” “Spot size,” and “Spot diameter” into a single unified term, “Spot area,” enabling consistent comparison across studies. This review synthesizes all currently published literature on green light-based interventions in bone tissue repair, systematically juxtaposing and elucidating the correlations between optical parameters and their biological effects. It standardizes heterogeneous terminologies in the field, thereby mitigating the conceptual ambiguities surrounding “bidirectional effects” among researchers and facilitating efficient retrieval of parametric data. Collectively, these efforts contribute substantial theoretical and practical insights to advance both fundamental research and clinical translation of green light applications in bone tissue repair.
Materials and Methods
Methods of searching articles
Web of Science, PubMed, and Scopus databases were searched for articles related to the effects of green light PBM on bone repair. This primarily included studies on green light PBM related to cell migration, proliferation, and differentiation associated with bone, and on inflammation, immunity, vascular regeneration, and neural applications. The following keywords were combined using Boolean operations to formulate the search strategy:
All fields AND PBM or photobiomodulation or PBMT or “photobiomodulation therapy” or LLLT or “low level light therapy” or “low intensity laser” or “low level laser” or “low intensity light therapy” or “low intensity laser therapy.”
All fields AND green and light or green and laser or “visible light” or “visible laser” or 500 nm or 505 nm or 520 nm or 532 nm or 540 nm or 550 nm or 560 nm.
All fields AND bone or osteoblast or osteoclast or osteoprogenitors or fractures or chondrocytes or “stem cell” or vasculature or “blood vessel” or inflammation or immunity or macrophages or granulocytes or lymphocytes or monocytes or nerves or neuronal or neurology or neuroscience.
Topic NOT PDT or “photodynamic therapy.”
Method for selecting articles
The selected articles were research articles written in English, and excluded other article types, such as reviews and conference reports. Moreover, the selected articles related directly and indirectly to green light PBM and bone, including GPBM and osteoblasts and differentiation of stem cells into osteoblasts; GPBM and proliferation, migration, and differentiation of stem cells, such as mesenchymal stem cells (MSCs) and GPBM, and inflammatory responses and vascular regeneration. The article retrieval strategy is summarized in Fig. 1.
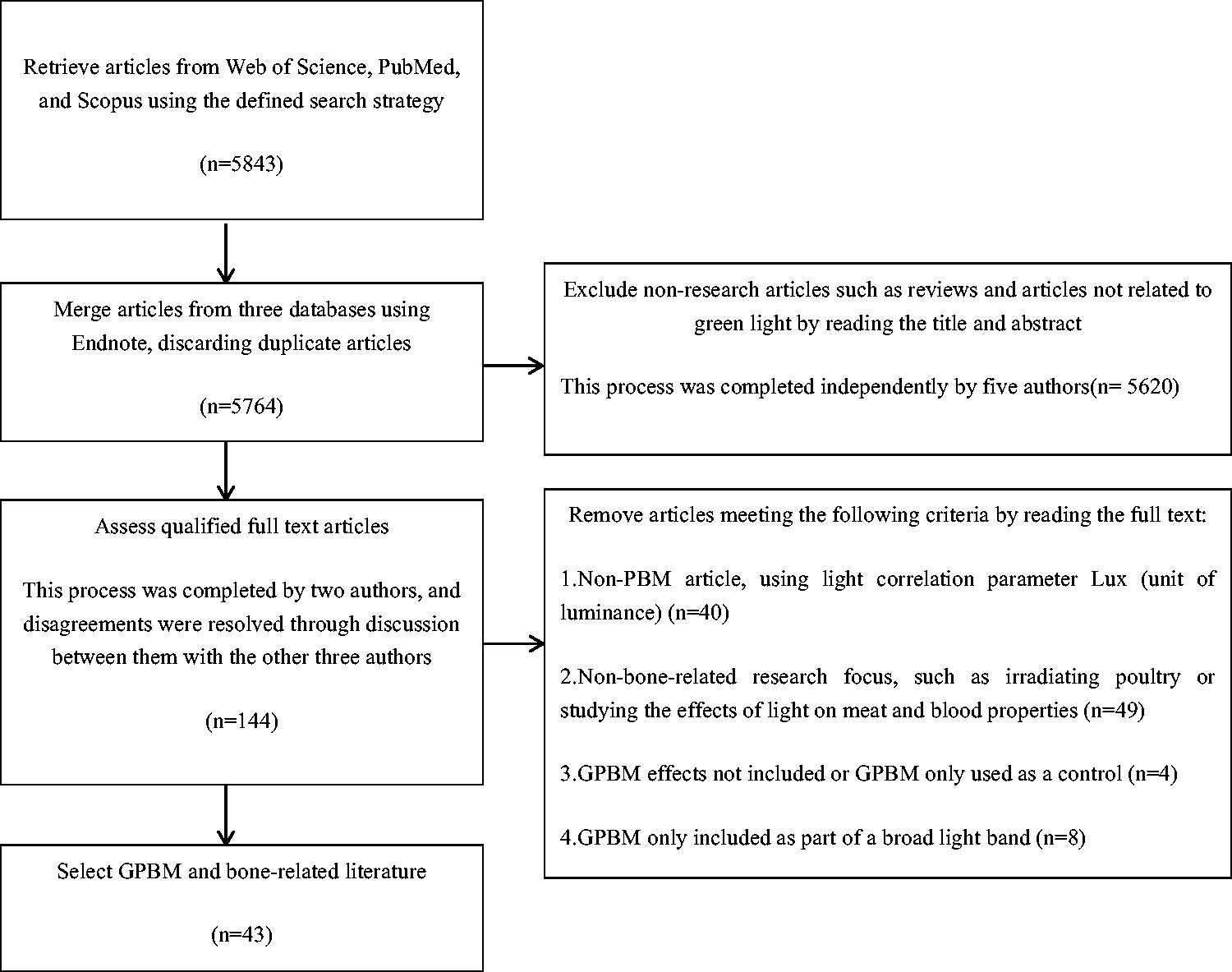
Flowchart illustrating the strategy for identifying relevant articles on the effects of green light PBM in bone repair. PBM, photobiomodulation.
Results
Screening results
Bone repair and GPBM are directly correlated
A total of 20 qualifying research articles were ultimately selected that investigated the effects of GPBM on the differentiation, proliferation, and migration of bone-related cells and stem cells with the ability to differentiate into bone-related cells. Among these, 14 articles pertained to the impact of GPBM on cell proliferation, 6 articles addressed the influence of GPBM on cell migration, 10 articles examined the effects of GPBM on the differentiation of cells into bone-related cells, and 1 article investigated the optimal wavelength for promoting stem cell differentiation (Table 1). The findings of these studies showed that the effects of GPBM on stem cell proliferation exhibited considerable variability, but relatively consistently promoted cell migration and differentiation.
The Effects of Green Light Photobiomodulation on Cell Proliferation, Migration, and Bone-Related Differentiation (Numbers Indicate Number of Articles)

Bone repair and GPBM are indirectly related
Additionally, a total of 10 research articles related to GPBM and vascular regeneration were identified, which primarily demonstrated promoting effects (Table 2). Seven research articles focusing on GPBM and inflammation were identified, which predominantly reported the anti-inflammatory properties of GPBM (Table 2). There were seven research articles reporting neural applications of GPBM, which primarily evaluated GPBM effects on stem cell neurodifferentiation and dendritic growth (Table 2).
The Effects of Green Light Photobiomodulation on Cell Angiogenesis, Inflammation, and Neuronal Development (Numbers Indicate Number of Articles)

Unifying definitions and PBM parameters
During our article review, we observed considerable diversity in the parameters used to characterize PBM, which posed a minor challenge when interpreting and comparing the articles. Consequently, in order to accurately compare the findings of each study, we standardized the expressions used for PBM parameters (Table 3). The parameters were subsequently uniformly expressed as radiant exposure (J/cm2), irradiance (mW/cm2), and spot area (cm2), with the spot diameter derived from the spot area.
Standardized Parameters and Their Associated Expressions

The first line contains standardized terminology. The terms in the column corresponding to the first line are those that have appeared in the literature, which have been compiled and will uniformly represent using the terminology from the first line.
The following formulas were used to convert between parameters:
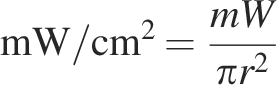
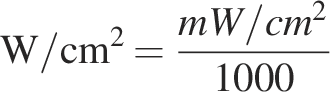
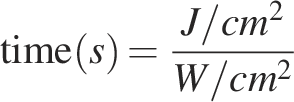
PBM parameters, assessment times, relevant research directions, and associated conclusions of the studies
After reading the full text of the selected research articles, we extracted author names, light source, dose, time period of the related index assessment, study type, conclusion, and other relevant information. After standardizing and unifying the parameters according to the method in the “Screening Results” section, we compiled the data shown in Tables 4 and Tables 5. Because many articles compare GPBM with PBM of other wavelengths, we also included other wavelengths used in the studies.
Summary of the Literature Evaluating the Effects of Green Light Photobiomodulation on Cells Related to Bone Differentiation, Proliferation, and Migration
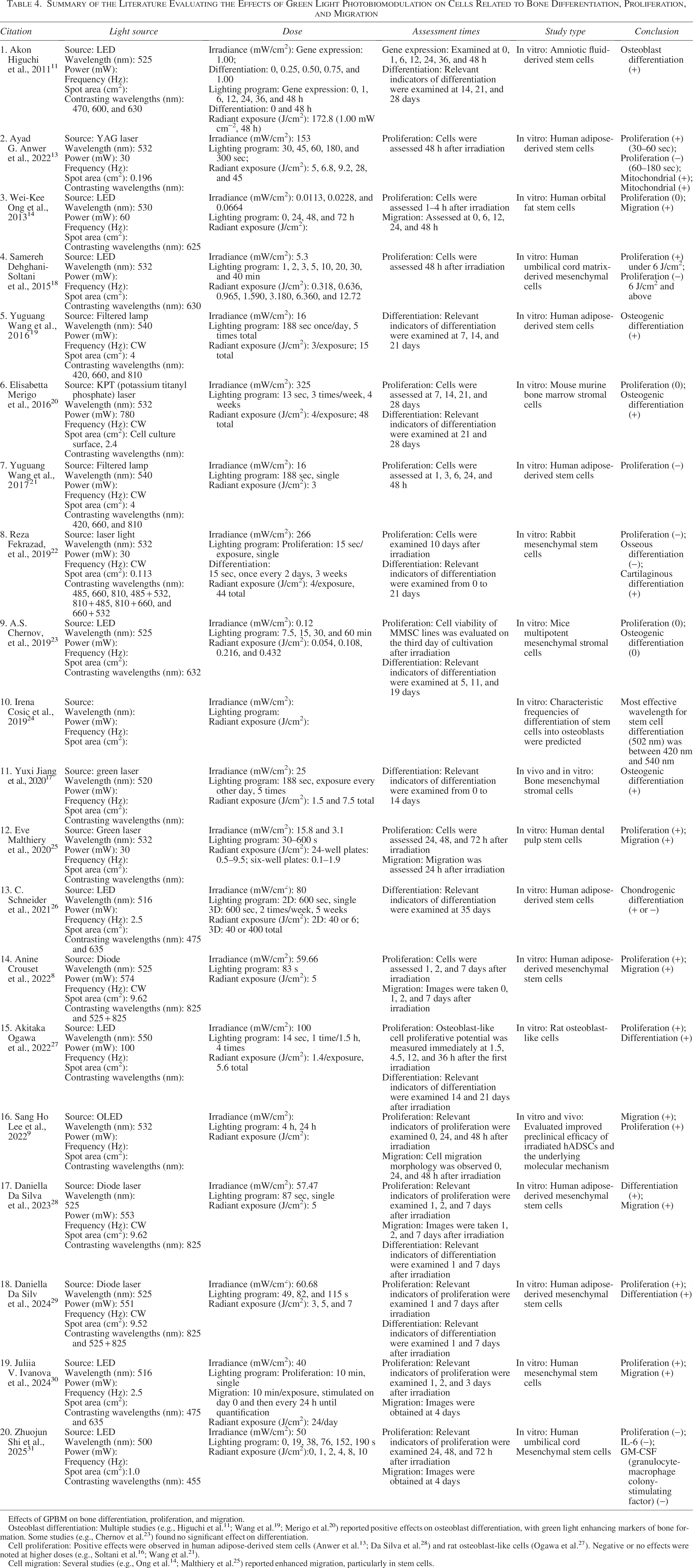
Effects of GPBM on bone differentiation, proliferation, and migration.
Osteoblast differentiation: Multiple studies (e.g., Higuchi et al. 11 ; Wang et al. 19 ; Merigo et al. 20 ) reported positive effects on osteoblast differentiation, with green light enhancing markers of bone formation. Some studies (e.g., Chernov et al. 23 ) found no significant effect on differentiation.
Summary of the Literature Evaluating the Effects of Green Light Photobiomodulation on Cell Angiogenesis, Inflammation, and Neuronal Development
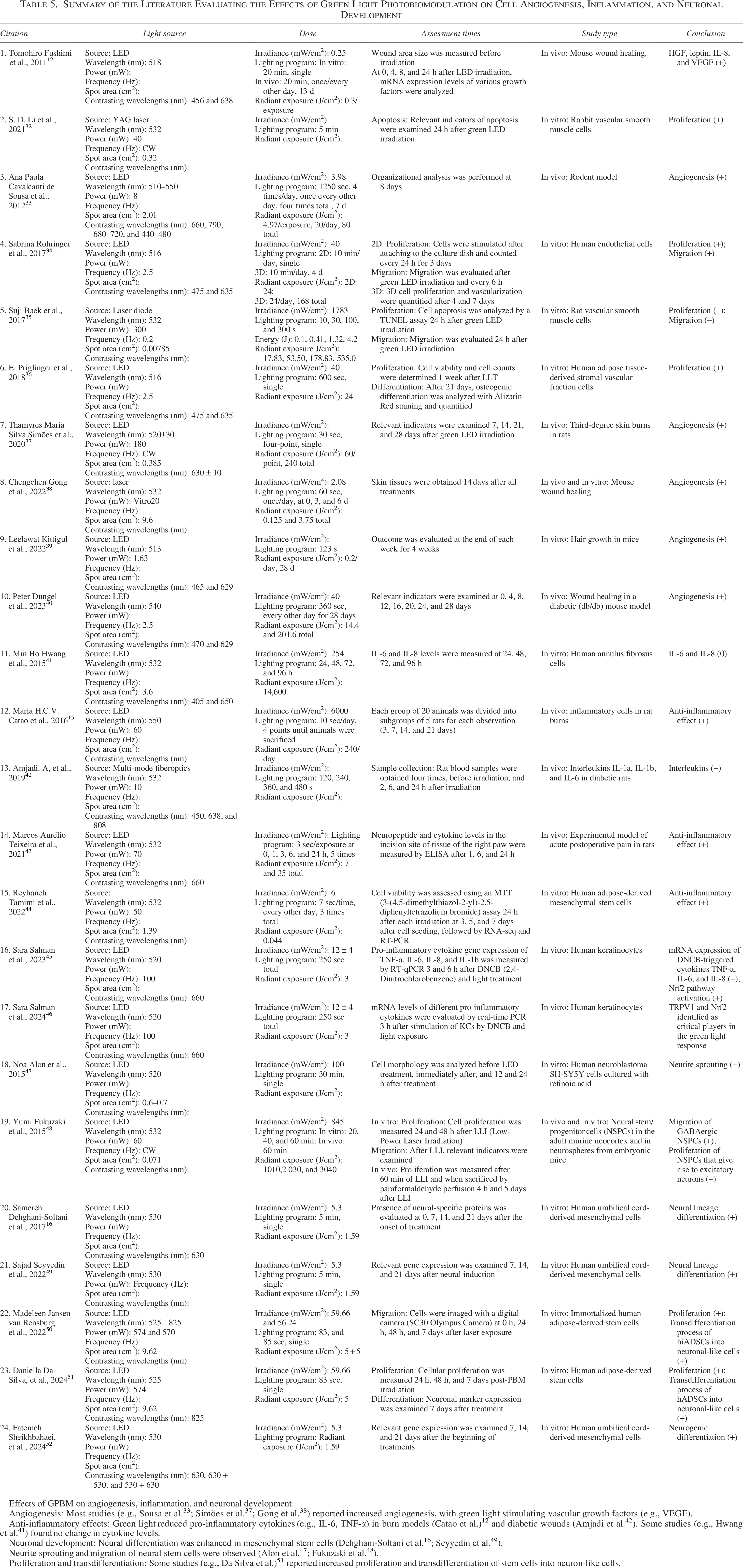
Effects of GPBM on angiogenesis, inflammation, and neuronal development.
Angiogenesis: Most studies (e.g., Sousa et al. 33 ; Simões et al. 37 ; Gong et al. 38 ) reported increased angiogenesis, with green light stimulating vascular growth factors (e.g., VEGF).
Anti-inflammatory effects: Green light reduced pro-inflammatory cytokines (e.g., IL-6, TNF-α) in burn models (Catao et al.) 12 and diabetic wounds (Amjadi et al. 42 ). Some studies (e.g., Hwang et al. 41 ) found no change in cytokine levels.
Neuronal development: Neural differentiation was enhanced in mesenchymal stem cells (Dehghani-Soltani et al. 16 ; Seyyedin et al. 49 ).
Neurite sprouting and migration of neural stem cells were observed (Alon et al. 47 ; Fukuzaki et al. 48 ).
Proliferation and transdifferentiation: Some studies (e.g., Da Silva et al.) 51 reported increased proliferation and transdifferentiation of stem cells into neuron-like cells.
Discussion
Effects of different GPBM parameters
The effects of PBM on biological organisms are closely related to the specific PBM parameters, as different parameter settings can lead to varying biological effects. Radiation exposure produces a powerful biphasic effect, whereby low-dose radiant exposure exerts a stimulatory effect on cells, while high-dose radiant exposure exerts an inhibitory effect, with factors such as irradiance and exposure time strongly influencing therapeutic outcomes; even with the same radiation exposure level, different irradiance and exposure times can produce different effects.53,54 The frequency of exposure has been shown to affect bone regeneration, with multiple exposures shown to be more effective than a single exposure. 55 There are also differences in therapeutic effects produced by continuous light compared to pulsed light. 56 Therefore, whether different parameters promote, inhibit, or show no significant effects on bone-related cellular differentiation (osteogenic differentiation and neurogenic differentiation), proliferation, migration, inflammation, and vascular regeneration are discussed.
In studies investigating the promoting effects of PBM on osteogenic differentiation, most research used short-duration irradiation. High-irradiance studies typically used a single exposure, while low-irradiance studies generally used multiple exposures. However, both approaches shared a commonality in that the radiant exposure (J/cm2) for each exposure was in the single-digit range.17,19,20,27–29 Only one study applied irradiation using long-duration and low irradiance (1 mW/cm2, 1–48 h). 11 Of the studies in which PBM did not affect osteogenic differentiation, one study used extremely low irradiance (0.12 mW/cm2) and varied the radiation exposure (0.054, 0.108, 0.216, 0.432 J/cm2); this failed to reach the activation threshold of the cells, and thus did not influence osteogenic differentiation. 23 Interestingly, the parameter values that inhibited osteogenic-related differentiation in one study were very similar to those that promoted differentiation in another study.20,22 This discrepancy may be attributed to the different cell types used: the former study used rabbit-derived MSCs, while the latter used mouse bone marrow-derived MSCs. In two studies focused on the differentiation of stem cells into chondrocytes, one concluded that PBM promoted chondrogenic differentiation. 22 While the other found that the effect of GPBM on chondrogenesis was highly donor-dependent, with varying effects observed under the same parameters when using different hADSC donors. 26 Due to the limited number of related studies and their inconsistent conclusions, it is challenging to delineate any consistent effects of PBM parameter values on osteogenic or chondrogenic differentiation.
When a bone fracture or injury occurs, stem cells become activated and begin to proliferate. By proliferating, stem cells produce more daughter cells, which provide the necessary foundation for bone repair. In studies related to stem cell proliferation, most PBM parameter values affected proliferation in a manner similar to those of the osteogenic differentiation studies. However, in one study that used a radiant exposure of 24 J/cm2, a promoting effect on stem cell proliferation was also observed. 30 This may have been due to the use of low-irradiance, long-duration irradiation (40 mW/cm2, 10 min) or the use of pulsed light. Previous research indicated that pulsed light was more effective than continuous light for promoting cell proliferation. 56 Among the reviewed articles, nine studies used pulsed light, 15 used continuous light, and 18 did not specify the light frequency, making direct comparisons of the effects of pulsed and continuous light on stem cell proliferation difficult. Additionally, one study used high irradiance short-duration irradiation (266 mW/cm2, 15 sec), which produced an inhibitory effect on stem cell proliferation. 22 However, a different study using even higher irradiance (325 mW/cm2, 13 sec) found no significant impact on proliferation. 20 This may represent a biphasic effect, in which excessively high irradiance saturated or even negated the promoting effects of low-intensity PBM, and therefore produced no effect or even inhibited stem cell proliferation. 53 The variability in results may also be related to the types of cells used. In neural differentiation studies, low irradiance, short-duration irradiation promoted the differentiation of stem cells into neurons and promoted dendritic growth, consistent with the effects of GPBM in osteogenic differentiation.16,47,49–52 However, high long-duration irradiation (845 mW/cm2 for 20, 40, or 60 min) also promoted the proliferation and migration of neural stem/progenitor cells (NSPCs), both in vitro and in vivo. 48 This finding contradicted the traditional biphasic effect theory and may be due to the strong light tolerance of NSPCs.
In studies on the migration of stem cells, two noteworthy differences were observed: first, extremely low-irradiance, long-duration irradiation (0.0113, 0.0228, and 0.0664 mW/cm2 for 0, 24, 48, and 72 h) promoted OFSCs migration, yet it did not significantly affect proliferation. 14 Second, at higher irradiance levels (845 mW/cm2 for 20, 40, and 60 min), light continued to enhance neural stem cells (NSCs) migration. 48 This suggested that stem cell migration may exhibit a strong adaptability to light exposure, with a positive response across a broad range of irradiation conditions; it is also possible that the differences were due to the type of stem cells used (OFSCs or NSCs).
The effect of GPBM on vascular-related cells was also reviewed. Low irradiance (40 mW/cm2) generally promoted the activity of these cells [such as human endothelial cells (HUVECs) and human stromal vascular fraction (hSVF) cells].34,36 However, higher irradiance levels (125 and 1783 mW/cm2) tended to inhibit vascular-related cells [rabbit vascular smooth muscle cells (VSMCs) and rat VSMCs].32,35 This discrepancy may also be attributed to the different cell types used in the studies. In in vivo studies on angiogenesis, low irradiance consistently facilitated vascular formation.12,33,36–40 GPBM clearly promoted vascular regeneration; however, the specific pathways through which it influences specific cells remain unclear due to the limited research available.
The effects of PBM on inflammation were examined in two in vivo studies using high irradiance with short exposure times, while another in vivo study used a different approach involving fiber optic intravenous irradiation (70 mW). The results of these studies were consistent with three studies that used low-irradiance irradiation at the cellular level, all of which demonstrated an inhibitory effect on inflammation.15,42–46 Only one study using high irradiance with prolonged exposure (254 mW/cm2 for 24, 48, 72, and 96 h) did not show a significant impact on inflammation.41 It was speculated that this lack of effect may be due to excessively high irradiance and prolonged exposure times.
Serrage et al. found that the median radiant exposure that produced a positive promoting effect in GPBM-related studies was 4 J/cm2 (range: 0.00362–30 J/cm2), which aligned with the results of existing studies. 5 However, they did not conduct an in-depth analysis of the irradiance and exposure time. According to the biphasic dose response observed in low-level light therapy, low and moderate doses of laser irradiation promote cell proliferation, while high-intensity light may negate this effect or even produce an inhibitory response. 53 Therefore, a constant radiant exposure with low irradiance (0–100 mW/cm2) and an extended exposure time may be more effective. This approach may prevent relevant signaling pathways from being inactivated due to insufficient radiant exposure, while also avoiding suppressing the positive effects of GPBM caused by excessively high irradiance. Further, although multiple exposures may increase the total radiant exposure, this does not necessarily lead to negative responses and instead may enhance therapeutic effects, provided that the intervals between exposures are appropriately determined.
Use of combined light
Fekrazad et al. examined the effects of many light combinations on osteogenic and chrondrogenic gene expression; They found that the use of infrared light photobiomodulation (IRPBM) and red light photobiomodulation (RPBM) alone promoted the expression of osteogenic-related markers, while the use of a GPBM and infrared-red light photobiomodulation (IR-RPBM) combination, and infrared and blue light photobiomodulation (IR-BPBM) combined exhibited inhibitory effects; The IRPBM stimulated chondrogenic-related gene expression in MSCs the most, with IRPBM, GPBM, blue light photobiomodulation (BPBM), and the IR-BPBM combinations also promoting the expression of chondrogenic-related genes; The blue-green light photobiomodulation (B-GPBM) combination had the strongest effect on cell proliferation, whereas the use of GPBM alone inhibited the proliferation of MSCs; BPBM had no significant effect on proliferation, while the red-green light photobiomodulation (R-GPBM) combination promoted cell proliferation, but to a lesser extent than that of RPBM alone, possibly due to the inhibitory effect of GPBM. 22 Silva et al. discovered that the combined use of GPBM and near-infrared light photobiomodulation (NIRPBM) enhanced the differentiation of immortalized human adipose-derived MSCs (hADSCs) into early osteoblasts, with the near-infrared green light photobiomodulation (NIR-GPBM) combination showing the most significant pro-differentiation effect compared to the use of NIRPBM or GPBM alone. They also found that NIR-GPBM promoted the migration and neural differentiation of immortalized human adipose-derived stem cells (hiADSCs).29,51 Additionally, Crous et al. found that NIR-GPBM promoted cell proliferation, with effects superior to those of GPBM alone, but did not reach the proliferation effect achieved with NIRPBM alone. 8 Sheikhbahaei et al. found that alternating GPBM with RPBM effectively promotes hUCM neural differentiation, and the order of application also affected the outcomes. 52 These results indicated that by strategically combining multiple light sources, the limitations of a single light source can be overcome, and the advantages of each source can be leveraged to achieve optimal therapeutic effects, as reflected in previous broadband light studies.7,57
Bone repair-related GPBM mechanisms
PBM is thought to directly affect the electron transport chain located on the mitochondrial membrane, particularly CCO (complex IV), to promote adenosine triphosphate (ATP) synthesis and generate ROS.58,59 Serrage et al. summarized four signaling pathways mediated by ROS, the nuclear factor kappa-B (NF-κB) signaling pathway, transforming growth factor-β (TGF-β), nuclear factor erythroid 2-related factor 2(Nrf2) signaling pathway, and mitogen-activated protein kinases (MAPK) signaling pathway, including ERK (extracellular regulated protein kinases), p38 MAPK (p38 mitogen-activated protein kinases), and JNK (c-Jun N-terminal kinase) signaling pathway. 5
Opsins are G protein-coupled receptors that are activated by blue or green light, and therefore have generated strong interest in phototherapy research. 60 Researchers have suggested that the downstream targets of opsins may include TRP channels, especially the TRP vanilloid I (TRPVI) subtype (capsaicin receptor), which can be activated by light. Additionally, nitric oxide (NO) is proposed to photodissociate from CCO, based on the reversible inhibitory effect of NO on CCO. 5 It is noteworthy that CCO has also been shown to exhibit nitrite reductase activity, and therefore locally produces NO within the mitochondria. 61
Gonadotropin-inhibitory hormone(GnIH), an upstream regulator of the hypothalamic–pituitary–gonadal (HPG) axis, maintains bone homeostasis by inhibiting osteoclast activity via G protein-coupled receptor 147(GPR147)-mediated suppression of the PI3K/AKT, MAPK, NF-κB, and nuclear factor of activated T-cells, cytoplasmic 1 (NFATc1) signaling pathways. 62 Additionally, exposure to green light increases GnIH secretion, thereby exerting bone-protective effects. 62 This mechanism may partly explain how low-level laser therapy promotes bone regeneration.
The mechanism by which GPBM affects stem cell differentiation, proliferation, and migration
Higuch et al. demonstrated that visible blue light promoted osteogenic differentiation not through ROS induction, but via another mechanism, although the specific mechanism is undetermined. 11 Merigo et al. were the first to observe that potassium titanyl phosphate (KTP) enhanced the osteogenic differentiation of BMSCs. 20 Jiang further investigated the effects of GPBM on BMSCs and found that green light likely activated phospho-extracellular regulated protein kinases (P-ERK) and regulated the downstream osteogenic transcription factor Runt-related transcription factor 2 (RUNX2) to facilitate osteogenic differentiation. 17 The expression of the osteogenic genes Runx2, Osteocalcin (OCN), and Osterix (OSX) is regulated by intracellular Ca2+; GPBM increases intracellular Ca2+ by activating TRPV ion channels, which then decrease ATP levels and produce substantial amounts of ROS. 21 However, TRPVI may not be the primary (or indeed the only) light-gated ion channel in hADSCs. 19 Chernov found that GPBM decreased mitochondrial dehydrogenase activity in multipotent mesenchymal stromal cells (MMSCs). 23 Some studies indicated a direct correlation between the degree of differentiation and mitochondrial membrane potential (MMP), with undifferentiated cells showing no significant differences in MMP.63,64 Crous et al. demonstrated that the electron transport chain in hADSCs was stimulated by light-activated cytochrome C, leading to an increase in MMP that promoted hADSC differentiation. 8 Silva discovered that after GPBM treatment of hADSCs for a period of time, ATP resources shifted toward cell differentiation rather than proliferation, and GPBM did not inhibit proliferation.28,29
Anat Lipovsky et al. were the first to find that broadband visible light promoted rat MSC proliferation, and noted an increase in ROS and NO levels in cells post-PBM. 57 However, the effects of GPBM on human stem cells remained unclear. Anwer et al. were the first to study the effects of GPBM on human stem cells, and discovered that GPBM promoted hADSC proliferation by enhancing MMP, leading to an increase in ATP, and they speculated that short-duration, low-dose laser exposure produced trace amounts of ROS, thereby enhancing the mitochondrial membrane potential. 13 Crous et al. also noted this phenomenon and demonstrated that the increased intracellular ROS did not have harmful effects on cells. 8 Lee et al. also confirmed this. 9 Lee et al. observed no changes in Ca2+ levels in hADSCs under GPBM irradiation; however, green light stimulation upregulated the gene expression levels of Osteopontin3 (OPN3) and Osteopontin4 (OPN4), which only stimulated the expression of OPN4 protein, which increased intracellular ROS levels and subsequently promoted hADSC proliferation. 9 It was reported that moderate levels of ROS may upregulate cytokines such as vascular endothelial growth factor (VEGF), which then influence the proliferation, migration, and adhesion of MSCs. 9 In the research by Wang et al., GPBM was found to activate TRPV ion channels, increase intracellular Ca2+, and produce large amounts of ROS, leading to a decrease in MMP and a reduction in ATP, thereby inhibiting hADSC proliferation. 21 The main divergence in these studies appears to be related to ROS; on the one hand, low-dose, nonharmful ROS may promote proliferation through adverse effects on mitochondria, and on the other hand, large amounts of ROS generated by activating TRPV ion channels may inhibit proliferation. This discrepancy may relate to the PBM parameters and cell types used in the studies.
Ong was the first to investigate the effects of GPBM on stem cell migration, finding that retinal pigment epithelium-derived rhodopsin homolog (RRH) and OPN3 were two nonvisual opsins in hOFSCs that acted as photoreceptors for GPBM irradiation, activating the ERK/MAPK/p38 signaling pathway to promote hOFSC migration. 14
The mechanism by which GPBM affects angiogenesis
Rohringer et al. found that GPBM promoted HUVEC proliferation and migration, enhanced NO production in cocultures with ADSCs, and facilitated wound healing and angiogenesis by mediating interactions between angiogenesis, remodeling, and anti-angiogenic factors, such as serpin family F member 1 (Serpin F1). 34 Fushim et al. found that while both GPBM and RPBM stimulated mouse fibroblast cytokine secretion, only GPBM induced the production of leptin, interleukin-8 (IL-8), and VEGF, which are pivotal for angiogenesis and epithelialization. However, GPBM did not significantly affect CCO activity or ATP production in human fibroblasts, likely due to differences in the experimental conditions, such as radiation energy, exposure time, and cell types, and the presence of a specific light receptor for green LEDs. 12
VEGF and inducible nitric oxide synthase (iNOS) were confirmed to promote angiogenesis, and studies have suggested that 804-nm laser irradiation positively regulated the expression of VEGF and iNOS, thereby promoting angiogenesis. 65 Chen et al. found that a 632.5-nm helium-neon laser enhanced NO secretion from endothelial cells and increased endothelial nitric oxide synthase (eNOS) expression via the phosphoinositide 3-kinase/protein kinase B/endothelial nitric oxide synthase(PI3K/Akt/eNOS) pathway, thus promoting endothelial cell proliferation and angiogenesis. 66 Lim et al. pointed out that 635-nm LED light inhibited endogenous ROS production, increased VEGF expression, and subsequently activated endothelial cells through the ERK/MAPK signaling pathway to promote angiogenesis. 67
Further, Sousa et al. found that green LED light was more effective than other light sources for promoting angiogenesis, and that there were similarities in the angiogenesis-promoting mechanisms of green LED, red, and near-infrared light, although the related mechanisms still require further exploration. 33 Li et al. observed apoptotic body formation in VSMCs after 24 h of irradiation, suggesting that GPBM inhibited VSMC proliferation by promoting apoptosis, thereby potentially modulating the structural and functional properties of blood vessels by regulating the balance between VSMC growth and apoptosis. 32 Baek et al. further validated this conclusion by showing that GPBM irradiation effectively inhibited platelet-derived growth factor (PDGF-BB)-induced proliferation and migration of VSMCs, and proposed a mechanism in which the GPBM and PDGF-BB interaction promoted apoptosis in these cells; moreover, GPBM activated the p38 MAPK signaling pathway, inducing vascular apoptosis. 35
These research findings suggested that GPBM regulated the behavior of vascular smooth muscle cells by directly affecting cell proliferation and migration and modulating key apoptotic signaling pathways, potentially playing a role in vascular and bone repair.
Vascular cell apoptosis plays a crucial regulatory role in bone tissue repair, dynamically coordinating with different healing phases to facilitate bone regeneration and maturation by maintaining vascular homeostasis and optimizing vascular network structure. Specifically, it eliminates damaged cells to reduce inflammatory injury, modulates the balance between quality and quantity of angiogenesis, coordinates osteoblast–endothelial cell coupling, and participates in vascular remodeling during late repair. Enhanced vascular apoptosis is predominantly observed in two key stages: the early inflammatory phase (3–7 days postinjury), where apoptotic clearance of damaged endothelial cells paves the way for neovascularization, and the late remodeling phase (2–4 weeks postinjury), where increased apoptosis of vascular smooth muscle cells and endothelial cells prunes excessive vessels, promoting functional maturation of the vascular network. Aberrant vascular apoptosis (either excessive or insufficient) may lead to impaired bone healing.
The mechanism by which GPBM affects inflammation
Hwang et al. first investigated the effects of low-level laser therapy (LLLT) on a macrophage-mediated inflammatory response in adipocyte fibroblasts (AFs), which revealed that exposure to 405-nm low-level LED light significantly reduced IL-6 and IL-8 production in AFs stimulated by macrophage-conditioned medium derived from THP-1 cells, whereas exposure to LED light at 650 nm and 532 nm showed no significant effect on cytokine production. 41 However, other studies showed that GPBM exerted anti-inflammatory effects across various cell types and animal models. For example, research on hADSCs and mouse keratinocytes (KCs) indicated that GPBM produced anti-inflammatory effects by reducing inflammatory factor levels.44–46 Other animal studies, particularly those using rat models, have demonstrated anti-inflammatory properties of GPBM.15,42,43
Salman et al. systematically investigated the underlying anti-inflammatory mechanisms of PBM for the first time. They discovered that in KCs induced by 2,4-dinitrochlorobenzene, GPBM significantly inhibited the mRNA expression of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin-8 (IL-8); however, GPBM did not regulate the transcription of pro-inflammatory cytokines under unstimulated baseline conditions. 45 The study also highlighted the critical role of the nuclear factor erythroid 2-related factor 2 (Nrf2) signaling pathway in the anti-inflammatory effects of PBM. Nrf2 is an important transcription factor regulating oxidative stress and inflammatory responses by promoting antioxidant and anti-inflammatory reactions.68–70 Salman et al. proposed that GPBM primarily activated the Nrf2 pathway through TRPVI channels to suppress inflammatory responses, and that Nrf2 activation did not seem to regulate inflammation via the ROS pathway, thus providing new insights into the anti-inflammatory mechanisms of PBM.45,46
The mechanism by which GPBM affects neuronal development
Fukuzaki et al. found that GPBM increased the levels of pAkt and Akt, which have a crucial role in regulating cellular processes essential for neuronal development, including gene transcription, cell proliferation, and neuronal migration. 48 Therefore, GPBM influenced NSPCs by activating Akt-related signaling pathways. Seyyedin et al. discovered that GPBM induced differentiation of hUCM neurons by stimulating ROS generation, which in turn activated the phosphorylation of ERK 1/2, JNK, and p38. 49 Summarizing, GPBM significantly promoted neuronal differentiation and development by regulating multiple signaling pathways, particularly the Akt and MAPK pathways.
Summary
Rosenberg et al. discovered that the transmittance of visible light in the range of 420–580 nm affected cell maturation and metabolism, while visible light in the range of 560–650 nm influenced cell proliferation and cell death. 71 The articles reviewed here indicated that GPBM has effects on both proliferation and differentiation, primarily acting as a promoter. Thus, it is hypothesized that GPBM may operate through mechanisms similar to both red PBM and blue PBM. Wang et al. found that blue light (420 nm) and green light (540 nm) inhibited proliferation and promoted differentiation, and that this effect was linked to TRPVI.19,21 Silva proposed a potential mechanism of action by green light: Green light at a wavelength of 525 nm penetrates the cell membrane, activating downstream target photoreceptor (TRP) channels for photobiomodulation. After stimulation, TRP channels open, allowing Ca2+ to flow into the cytoplasm, which activates Ca2+/CaM-dependent protein kinase II (CAMKII) and triggers the phosphorylation of cAMP response element-binding protein (CREB) in the nucleus, leading to gene transcription, such as for cell differentiation. 28 Additionally, she proposed a mechanism for red light: Near-infrared light at a wavelength of 825 nm mediates photobiomodulation by penetrating the cell membrane and reaching the mitochondria. CCO, located within the mitochondria, absorbs infrared light and plays a role in the electron transport chain during ATP production. An increase in ATP content leads to enhanced gene transcription within the nucleus, increasing DNA and RNA synthesis and ultimately promoting cell proliferation. 28 Many studies have found that GPBM increased intracellular MMP, which in turn stimulates ATP synthesis.8,13,28 This mechanism is similar to that of photobiomodulation by near-infrared light at 825 nm, suggesting that GPBM may also exert its effects on cells through this mechanism.
We summarized the signaling pathways proposed by the reviewed articles, as presented in Fig. 2 and Fig. 3. Besides, the dashed portion in Figure 3 represents a hypothesized potential pathway. GPBM influences bone-related cellular differentiation (osteogenic differentiation and neurogenic differentiation), proliferation, migration, inflammation, and vascular regeneration through these mechanisms to subsequently promote bone repair.
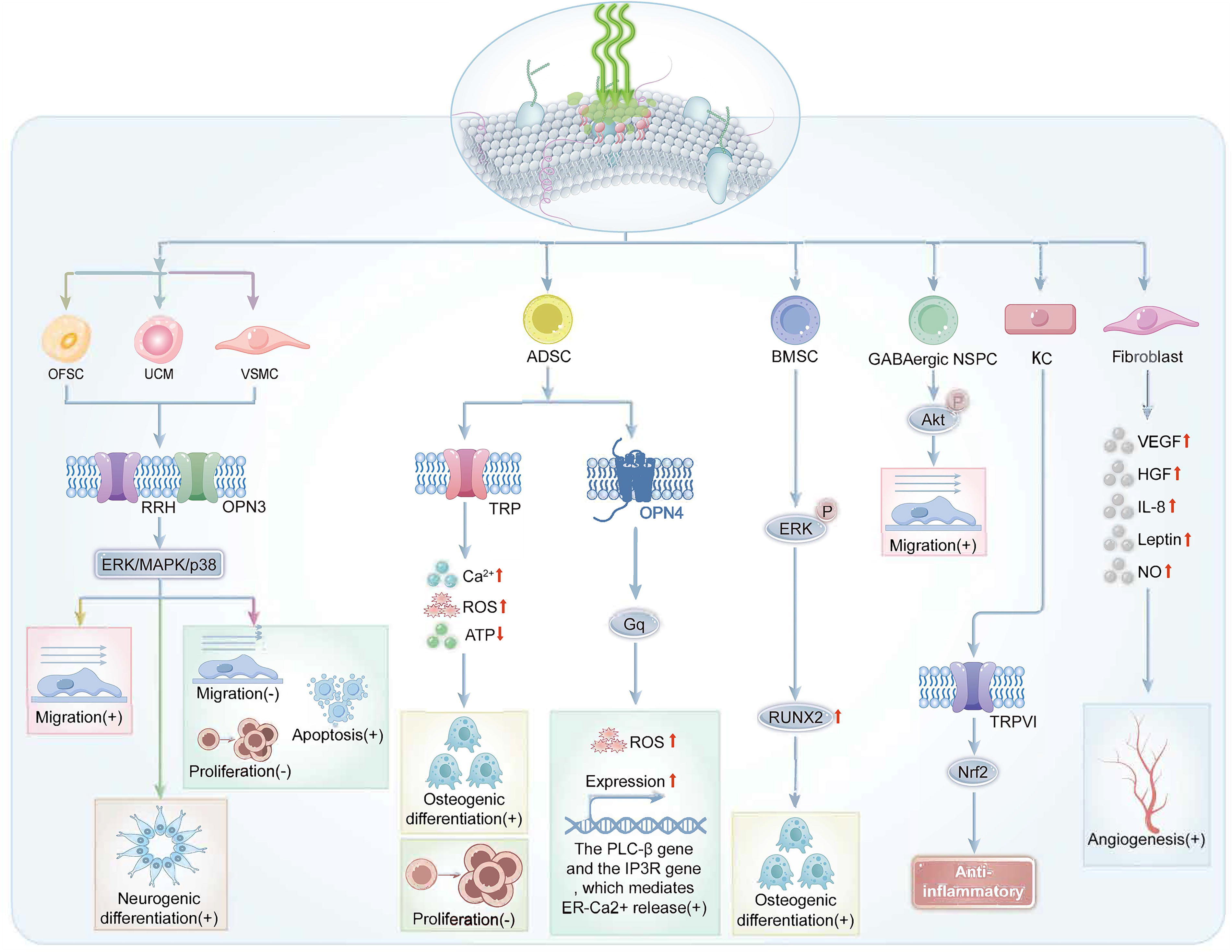
Proposed signaling pathways based on the reviewed literature. Green light irradiation of cells, such as orbital fat stem cell (OFSC), activates certain ion channels and signaling pathways, such as the opsin3 (OPN3) channel and the ERK/MAPK/p38 pathway. This activation promotes or inhibits the production of specific substances, such as reactive oxygen species (ROS), which ultimately regulate the cellular behavior. ADSC, adipose-derived mesenchymal stem cell; ATP, adenosine triphosphate; BMSC, bone marrow stromal cell; ER, endoplasmic reticulum; ERK, extracellular regulated protein kinases; GABAergic NSPC, gamma-aminobutyric acid-ergic neural stem/progenitor cell; Gq, Gq protein; HGF, hepatocyte growth factor; IL-8, interleukin 8; IP3R, inositol 1,4,5-trisphosphate receptor; KC, keratinocyte; MAPK, mitogen-activated protein kinases; Nrf2, nuclear factor erythroid 2-related factor 2; OPN4, opsin4; p38, p38 mitogen-activated protein kinases; PLC-β, phospholipase C-beta; RRH, retinal pigment epithelium-derived rhodopsin homolog; ROS, reactive oxygen species; RUNX2, Runt-related transcription factor 2; TRP, transient receptor potential; TRPV1, transient receptor potential vanilloid 1; UCM, umbilical cord-derived mesenchymal cell; VEGF, vascular endothelial growth factor; VSMC, vascular smooth muscle cell.
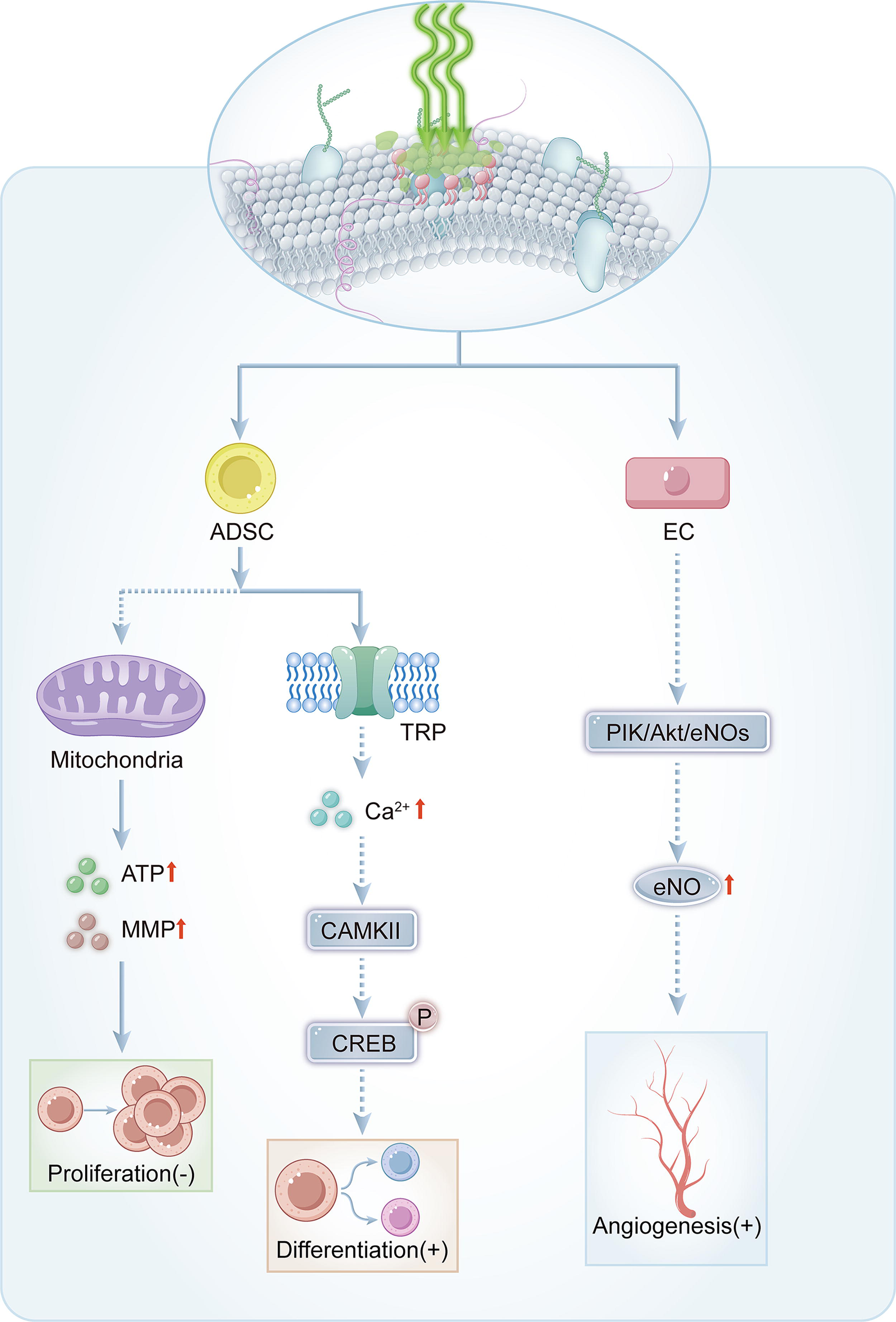
Comprehensive summary of signaling pathways for green light regulation of bone tissue repair. Green light irradiation of cells, such as adipose-derived stem cell (ADSC), may alter the mitochondrial membrane potential (MMP) or activate transient receptor potential (TRP) channels, thereby promoting or inhibiting the production of certain substances, such as Ca2+, which ultimately regulate the cellular behavior. The dashed portion represents a hypothesized potential pathway. CAMKII, Ca2+/CaM-dependent protein kinase II; CREB, cAMP response element-binding protein; EC, endothelial cell; PI3K/Akt/eNOS, phosphoinositide 3-kinase/protein kinase B/endothelial nitric oxide synthase.
Research status and prospects
Biomodulation has been widely applied in the treatment of diabetes, neurological disorders, skin diseases, complications after radiotherapy, and dentistry. 72 However, current research on GPBM’s effects on the proliferation, differentiation, and migration of osteogenic-related cells is predominantly limited to in vitro studies. Only a few studies have explored in vivo effects under 3D conditions. Merigo et al. found that green visible light from a potassium titanyl phosphate (KTiOPO4, KTP, 532 nm) laser promoted the osteogenic differentiation of mouse BMSCs, yet researchers have yet to investigate GPBM in vivo effects. 20 Jiang et al. used optical fibers as biodegradable implants to deliver green light to deep tissues, which facilitated bone defect regeneration in a rodent model and thereby demonstrated the feasibility of GPBM for bone repair. 17 Silva et al. used embedded ADSCs in a PBM 3D hydrogel matrix, highlighting potential innovative GPBM treatments for degenerative bone diseases. 29 Further, Rohringer et al. studied the effects of GPBM on HUVEC migration under 3D conditions, which contributed to cellular-level research on angiogenesis. 34 Schneider et al. examined the effect of GPBM on chondrogenic differentiation under 3D conditions. 26 Ivanova et al. investigated GPBM effects on hMSC proliferation and migration in 3D, although they did not use a medium. 30 Nonetheless, there is a scarcity of relevant in vivo studies, underscoring the need for in vivo research to leverage GPBM in bone repair.
Conclusion
This review explored research advancements using GPBM in bone repair in six areas directly and indirectly related to bone healing, including the proliferation, differentiation, and migration of bone-related cells, and inflammation, vascular regeneration, and neural development. The findings indicated that GPBM produced notable positive effects in all six areas. A synthesis of the selected articles identified that the optimal GPBM parameters for promoting stem cell proliferation and differentiation was low-irradiance (5.3–100 mW/cm2) with a single total radiant exposure of approximately 5 J/cm2. Irradiance levels that were too low (0.0113, 0.0228, 0.0664, and 0.12 mW/cm2) did not significantly affect cell proliferation, while excessively high irradiance levels (325 and 266 mW/cm2) also failed to promote the desired effects and even induced inhibitory responses. In stem cell migration, the range of parameters that promoted movement appeared to be broader. Several studies have observed the effects of GPBM on angiogenesis and inflammatory responses through various cellular and wound healing models; however, dedicated research on angiogenesis and inflammation modulation through GPBM remains insufficient, and the relevant mechanisms have not been clearly described. Therefore, future research should focus on elucidating the specific mechanisms by which GPBM operates in vivo, as well as further clarifying the parameters most suitable for stem cell proliferation, differentiation, and migration to achieve optimal bone repair outcomes. Additionally, there is an urgent need to enhance research on the application of GPBM in vascular regeneration and inflammation to explore its potential for bone repair, particularly regarding its feasibility in clinical applications.
Authors’ Contributions
W.B., J.Z., F.L., J.H., X.C., and Y.J. researched the literature. W.B. wrote the original draft. Y.J. reviewed and supervised the writing of the article. All authors have read and agreed to the published version of the article.
Footnotes
Acknowledgment
Author Disclosure Statement
The authors declare no conflicts of interest.
Funding Information
This work was supported by grants from the Guangdong Basic and Applied Basic Research Foundation (2021A1515110211) and the Guangdong Medical Science and Technology Research Foundation (B2023072).
