Abstract
Recent reports indicated that human isolated CD271+ bone marrow mesenchymal stromal cells (BM-MSCs) have a greater expansion and potential for multipotent differentiation including chondrogenesis than classical plastic adherent (PA) BM-MSCs in vitro. Therefore, we set up a hypothesis that CD271+ MSCs may have a greater chondrogenic potential than PA-MSCs in vitro and in vivo. We investigated the superiority of CD271+ MSCs on chondrogenesis using in vitro expansion and pellet culture system and in vivo rat model of cartilage defect when compared to PA-MSCs. In the in vitro study, CD271+ MSCs showed higher expansion potential and produced larger pellets with higher expressions of chondrogenic genes when compared to the control groups. During the culture, CD271 expression decreased, which resulted in decreased chondrogenesis. In the in vivo study, immunohistochemical staining demonstrated differentiated human chondrocytes identified as double-stained cells with human-specific collagen type 2 and human leukocyte antigen-ABC in CD271+ and PA groups. The number of double-stained cells was significantly higher in the CD271+ group than PA group. Real-time RT-PCR analysis of tissue RNA isolated from the chondral defect site for human-specific chondrogenic markers demonstrated a significantly higher expression in CD271+ group than PA group. Macroscopic examination of chondral defect sites at week 8 revealed glossy white and well-integrated repaired tissues in the CD271+ and PA groups, but not in the PBS group. The average histological score in the CD271+ group was significantly greater than in the other groups. Apoptosis analysis at the cell transplanted site with TUNEL staining showed that the CD271+ group had significantly fewer apoptotic chondrocytes compared with the PA group. These results indicate that CD271+ MSCs have a greater chondrogenic potential than PA-MSCs in both in vitro and in vivo conditions.
Introduction
It is well known that the capacity of articular cartilage for self-repair is limited because of low mitotic activity, the absence of a vessel and nerve supply, and the immobility of articular chondrocytes. Joint surface defects that exceed a critical size heal poorly and usually lead to osteoarthritis. Many researchers have designed clinical methods and proposed various experimental approaches. In addition to the classical technique such as bone marrow stimulation and osteochondral autograft/allograft, autologous cell transplantation has recently been considered as a promising new approach. Chondrocytes (5,11,23,40) and mesenchymal stromal cells (MSCs) (21,27,44,45) are commonly used for cell transplantation therapy for cartilage repair. The chondrocyte is a natural and logical choice for cartilage repair applications, but the use of autologous chondrocytes for cartilage repair raises several major issues such as morbidity at the donor site, cell number on harvest, and the loss of chondrocyte markers in culture (2,12). According to Caplan, both chondrocytes and MSCs implanted into osteochondral defects in rabbits did generate cartilage, but a subchondral plate was observed only when MSCs were used (6). Yan et al. also reported that repaired tissues treated with bone marrow (BM) MSCs appeared to have better cell arrangement, subchondral bone remodeling, and integration with surrounding cartilage than repaired tissues generated by chondrocyte implantation (46). Therefore, BM-MSCs can be considered as one of the most promising strategies for cartilage regeneration. Furthermore, BM-MSCs transplanted into nonimmunocompromised animals undergo site-specific differentiation and persist in the long term (30). Recently, it has been reported that human MSCs modulate allogeneic immune cell responses (14,24,29). Thus, these cells are considered to be suitable for transplantation into various tissues.
MSCs are considered as a notable cell population because of their pluripotency, high expansion potential, and their ability to maintain their pluripotency (17,25,35), so much effort has been focused on engineering cartilage with these stem cells. BM is considered to be the main reservoir of MSCs (42), and many attempts have been made to isolate and find the most specific antigen for these stem cells (8,13,20,34,41). Additionally, recent reports have indicated that BM mononuclear cells (MNCs) isolated by cluster of differentiation 271 (CD271; low-affinity nerve growth factor receptor; LNGFR) positivity, which is one of the specific markers for MSCs, show a one to three log greater expansion than nonisolated BM-MSCs in culture and a greater capacity to differentiate to adipocytes, osteoblasts, and chondrocytes than the general plastic adherent BM-MSCs (PA-MSCs) in vitro (19,37). These previous reports suggest that BM-MSCs isolated by CD271+ antigen may produce greater results than general PA-MSCs.
Based on these findings, we hypothesized that CD271+ BM-MSCs (CD271+ MSCs) may have a greater capacity for cartilage repair than PA-MSCs. In the present study, we investigated in vitro and in vivo the chondrogenic potential of CD271+ MSCs and compared them to the potential of PA-MSCs.
Materials and Methods
Isolation and Culture of CD271+ and PA-MSCs
We isolated CD271+ MNCs from the BM-MNCs taken from a healthy woman (28 years old, African American) (Lonza, Walkersville, MD, USA) by the AutoMACS system (Miltenyi Biotec. Auburn, CA, USA) using the MSC Research Tool Box-CD271 (Miltenyi Biotec). The number of CD271+ cells in the isolated and the nonisolated cell fraction were determined by fluorescence-activated cell sorting (FACS) analysis using a CD271-specific monoclonal antibody (Miltenyi Biotec). The CD271+ MNCs were plated at 1×105 cells/well of a six-well plate (BD Biosciences, San Diego, CA, USA) with standard medium [a-minimum essential medium (MEM), Gibco BRL, Invitrogen Japan K.K., Tokyo, Japan] containing 10% fetal bovine serum (FBS) (Vitromex, San Antonio, TX, USA), 2 mM l-glutamine (Gibco BRL), 100 units/ml penicillin (Gibco BRL), and 100 μg/ml streptomycin (Gibco BRL) and incubated at 37°C with 5% humidified CO2. Four to 5 days after initiating incubation, the culture medium was changed twice weekly. Approximately 3–4 weeks later, the adherent cells were harvested with 0.25% trypsin/1 mM ethylenediaminetetraacetic acid (EDTA) solution (Gibco-BRL) and passaged into new dishes for further expansion. Cells from passages 1 to 3 were collected as CD271+ MSCs and used for flow cytometry, expansion assay, pellet culture, and in vivo transplantation. As a cell control group, nonisolated BM-MNCs were cultured under the same conditions as the CD271+ MNCs and collected as PA-MSCs after the culture (Fig. 1A).
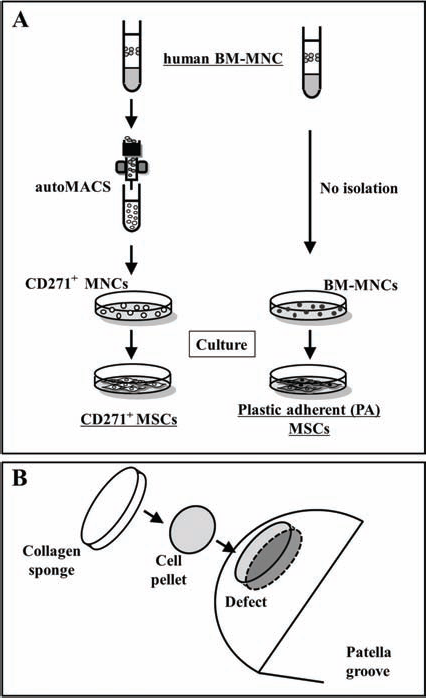
Schematic of cell preparation and in vivo model. (A) Preparation of CD271+ MSCs and PA-MSCs. CD271+ mononuclear cells (MNCs) were isolated from bone marrow-derived mononuclear cells (BM-MNCs) using the autoMACS (magnetic-activated cell sorting). The CD271+ MNCs and nonisolated BM-MNCs were cultured with standard medium for 3 weeks to obtain both types of mesenchymal stromal cells (MSCs). One to three passage cells were collected as CD271+ MSCs and plastic adherent (PA)-MSCs and used for each examination. (B) Cell transplantation method. Firstly, 5×105 cells of each MSC group were centrifuged at 300×g for 10 min as preparation for transplantation. Then the rats received local administration of the MSC pellet on the chondral defect site, which was then covered with an atelocollagen sponge.
Flow Cytometry Studies and Monoclonal Antibodies
Regular flow cytometric profiles were analyzed with a FACSCalibur analyzer and CELLQuest software [Becton Dickinson (BD) Immunocytometry Systems, Mountain View, CA, USA]. The instrument was aligned and calibrated daily using a four-color mixture of calibrate beads (BD Biosciences, San Jose, CA, USA) with FACS Comp software (BD Bioscience). Dead cells were excluded from the plots by propidium iodide (PI) staining (Sigma Co., St. Louis, MO, USA). All cells used in this study were washed twice with Hank's balanced salt solution (HBSS) (Gibco BRL) containing 3% FBS, incubated with 10 μl of FcR Blocking Reagent (Miltenyi Biotec) to increase the specificity of monoclonal antibodies for 20 min at 4°C, and incubated with the monoclonal antibodies for 30 min at 4°C. The stained cells were washed three times with phosphate-buffered saline (PBS) (Gibco BRL) containing 3% FBS and resuspended in 0.5 ml of HBSS, 3% FCS, PI, and analyzed by FACScan Caliber flow cytometer (BD Pharmingen, Franklin Lakes, NJ, USA). The following monoclonal anti-human antibodies were used to characterize the CD271+ cell population: CD271-fluorescein isothiocyanate (FITC; Miltenyi Biotec), CD34-FITC (BD Pharmingen), CD45-FITC (BD Pharmingen), CD133-allophycocyanine (APC; BD Pharmingen), CD29-PE (BD Pharmingen), CD31-FITC (BD Pharmingen), CD105-FITC (BD Pharmingen), CD44-phycoerythrin (PE; BD Pharmingen), CD166-PE (BD Pharmingen), STRO-1-FITC (BD Pharmingen), IgG1-FITC isotype controls (BD Pharmingen), and IgG1-APC isotype controls (BD Pharmingen).
In Vitro Expansion Ability
To examine the in vitro cell expansion ability, CD271+ MSCs and PA-MSCs (n = 5) were plated in 60-cm2 dishes (BD Biosciences), and routine cell expansion was performed every 14 days. At each passage, cells were replated to a density of 1×105 cells/dish. The number of population doublings (PDs) for each subculturing was calculated as the {log10(NH) - log10(N1)}/log10(2) (3). NH is the harvested cell number, and N1 is the plated cell number.
Pellet Culture
To investigate chondrogenic ability, 5×105 cells were placed in a 15-ml polypropylene tube (BD Biosciences) and centrifuged at 300×g for 10 min to form a pellet. Then the pellet was cultured in chondrogenic medium: high-glucose Dulbecco's modified Eagle medium (DMEM) (Gibco BRL) supplemented with 10−7 M dexamethasone (Sigma), 10 ng/ml transforming growth factor-β3 (R&D Systems, Minneapolis, MN, USA), 50 μg/ml ascorbate-2-phosphate (Sigma), 40 μg/ml praline (Sigma), 100 μg/ml pyruvate (Sigma), and 50 mg/ml ITS + Premix [6.25 μg/ml insulin, 6.25 μg/ml transferrin, 6.25 ng/ml selenious acid, 1.25 mg/ml bovine serum albumin (BSA), and 5.35 mg/ml linoleic acid] (Sigma) for 3 weeks. This study design consisted of two groups: CD271+ MSCs and PA-MSCs groups (n = 5 in each group).
Femoral Chondral Defect Model and Local Transplantation of CD271+ MSCs and PA-MSCs
Female athymic nude rats (F344/N Jcl rnu/rnu, CLEA Japan, Inc., Tokyo, Japan) aged 8–12 weeks and weighing 150–170 g were used in this study. The rats were fed a standard maintenance diet and provided water freely. The institutional animal care and use committees of RIKEN Center for Developmental Biology approved all animal procedures including human cell transplantation. A reproducible model of full thickness articular cartilage defect (1.5×1.5×1.5 mm) in the trochlear groove of each femur was created in nude rats with a microdrill (26,28,47). To make the pellets of MSCs for transplantation, 5×105 cells of each group were placed in a 15-ml polypropylene tube and centrifuged at 300×g for 10 min. Then the rats received local administration of the following cell pellet on the chondral defect site which was covered with a 1.5-cm-diameter atelocollagen sponge (KOKEN, Tokyo, Japan) immediately after cell transplantation; CD271+ MSCs (CD271+ group), PA-MSCs (PA group), and sponge alone [no cell (NC) group] (Fig. 1B) (n = 20 in each group).
RT-PCR Analysis of RNA Isolated From Pellets
Total RNA was obtained from pellets after CD271+ MSCs and PA-MSCs cultured for 3 weeks in chondrogenic induction medium using Trizol (Life Technologies, Gaithersburg, MD, USA) according to the manufacturer's instructions. The first-strand cDNA was synthesized using the RNA LA PCR Kit Ver 1.1 (Takara Bio. Inc., Otsu, Japan), amplified by Taq DNA polymerase (Advantage-GC cDNA PCR Kit, Clontech, Mountain View, CA, USA and AmpliTaq Gold DNA polymerase, Applied Biosystems, Foster City, CA, USA). PCR was performed using a PCR thermocycler (MJ Research PTC-225, St. Bruno, Quebec, Canada). The human-specific collagen type 2 (hCOL2), human-specific SOX9, and human-specific glyceraldehyde-3-phosphate dehydrogenase (hGAPDH) were amplified by Taq DNA polymerase (Advantage-GC cDNA PCR Kit, Clontech) at the following conditions: 35 cycles of 30-s initial denaturation at 94°C, annealing at 56°C for 1 min, and 30 s of extension at 72°C according to the manufacturer's instructions. Subsequently, PCR products were visualized in 1.5% ethidium bromide (Bio-Rad Laboratories, Tokyo, Japan) stained agarose gels (Takara Bio, Shiga, Japan). Human chondrocytes (Lonza) were used for positive control for human-specific chondrocyte-related genes.
Primers
To avoid interspecies cross-reactivity of the primer pairs between human and rat genes, we designed the following human-specific primers using Oligo software (Takara Bio. Inc., Otsu, Japan). All primer pairs did not show cross-reactivity to rat genes (data not shown).
hCOL2 (390 bp): sense AAT GAG GCC ACA ACA AAC ATC ACA and antisense CTG TCC TTC TTA ACC TCC GCA CAG C; hSOX9 (368 bp): sense ATC GAT CAG TGG AAC TTT GCC TAT T and antisense GTG GCA TTT GAG ATT TGA TAG A; hGAPDH (320 bp): sense GTG CCA GCC TCG TCT CAT AGA and antisense CGC CAG TAG ACT CCA CGA CAT.
Real-Time RT-PCR Analysis of Collagen Type 2 and SOX9
Total RNA was obtained from the rat tissues within the chondral defect at week 4 as described for RT-PCR analysis (n = 5 in each group). After the first-strand cDNA was synthesized, the converted cDNA (2 μl) samples were amplified in triplicate by real-time PCR (ABI PRISM 7700, Applied Biosystems) in a final volume of 20 μl using SYBR Green Master Mix reagent (Applied Biosystems). Melting curve analysis was performed to ensure that only a single product was amplified using Dissociation Curves software (Applied Biosystems). Specificity of the reactions was confirmed by 2.0% agarose gel electrophoresis. Results were obtained using sequence detection software (ABI PRISM 7700) and evaluated using Microsoft Excel (Redmond, WA, USA).
Primers
hSOX9 (hSOX9): sense ATG TTT GGC CTG AAG CAG AGA and antisense GGC GGT ACA GGT CGA GCA TAT A; hCollagen type 2 (hCOL2): sense AAG CAA GTA GCG CCA ATC T and antisense GGA AGT AGG GTG CCA TAA CAC; hAggrecan (hAGG): sense ACT GGC GAG CAC TGT AAC and antisense TCT TGG GCA TTG TTG TTG AC; rGAPDH (rGAP): sense CTG ATG CCC CCA TGT TCG TC and antisense CAC CCT GTT GCT GTA GCC AAA TTC G.
Tissue Harvesting
Rats were euthanized with an overdose of ketamine and xylazine. Bilateral femurs were harvested and quickly embedded in an optimal cutting temperature (OCT) compound (Miles Scientific, Elkhardt, IN, USA), snap frozen in liquid nitrogen, and stored at −80°C for histochemical and immunohistochemical staining as described below. Rat femurs in OCT blocks were sectioned, and 6-μm serial sections were mounted on saline-coated glass slides (Matsunami Glass Ind., Ltd., Osaka, Japan) and air dried for 1 h before being fixed with 4.0% paraformaldehyde (Sigma) at 4°C for 5 min and stained immediately.
Immunohistochemical Staining
To detect the chondral restoration by transplanted human cells in the rat tissue at week 4, immunohistochemistry was performed with the following human-specific antibodies: human leukocyte antigen (HLA)-ABC (BD Pharmingen) to detect the various human cell lineage and hCOL2 (BD Pharmingen) to detect human cell-derived chondrocytes. Staining specificity for human cells without cross reaction to rat cells was confirmed by histochemical staining for HLA-ABC and hCOL2 using human chondrocytes (Lonza) (data not shown). The secondary antibodies for each immunostaining are as follows: Alexa Fluor 594-conjugated goat anti-mouse IgG1 (Molecular Probes, Invitrogen Japan K.K., Tokyo, Japan) for HLA-ABC staining and Alexa Fluor 488-conjugated goat anti-mouse IgG2a (Molecular Probes) for hCOL2. DAPI solution (Vector Laboratories, Burlingame, CA, USA) was applied for 5 min for nuclear staining.
Double immunohistochemistry with HLA-ABC and hCOL2 was performed to detect human chondrocytes in articular cartilage. These assessments were performed by three orthopedic surgeons blind to treatment (n = 5 in each group).
Macroscopic Appearance of the Chondral Restoration
The macroscopic appearances of the chondral defect site were taken at week 8 following the transplantation. These procedures were done under anesthesia. Chondral restoration was identified by the smooth surface. Macroscopic assessments of each animal were examined by three observers blinded to each group (n = 10 in each group).
Histological Assessment of the Chondral Healing
Histological evaluation was performed with toluidine blue staining to assess the chondral healing at week 8.
The degree of chondral healing was evaluated at week 8 in each group using Wakitani's score (44). This grading scale was composed of five categories with a score range from 0 to 14 points. Cell morphology was graded from zero for normal tissue (uninjured cartilage) to four points when cartilaginous tissue was absent; matrix staining represented the degree of metachromatic staining compared with adjacent normal cartilage in sections stained with toluidine blue; surface regularity referred to the proportion of the defect surface that appeared smooth compared to the whole defect surface; cartilage thickness was related to the average thickness of the defect cartilage compared to that of the surrounding cartilage; and integration of donor into host adjacent cartilage was graded from zero (no gap between donor and host cartilage) to two (full separation or dissociation). These assessments were performed by three orthopedic surgeons blinded to groups (n = 10 in each group).
Analysis of Transplanted Cell Apoptosis
Fresh frozen sections were obtained, and the terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay was performed at week 4 using an Apop Tag Plus Peroxidase In Situ Apoptosis Detection kit according to the recommendations of the manufacturer (Chemicon, Temecula, CA, USA). Briefly, sections were incubated with 15 μg/ml of proteinase K for 15 min at room temperature and then washed in PBS. Endogenous peroxidase was quenched with 3% H2O2 for 5 min at room temperature. After washing in PBS, sections were immersed in buffer containing terminal deoxynucleotidyl transferase enzyme and incubated for 90 min at 37°C in a humid atmosphere. After washing again in PBS, sections were incubated with antidigoxigenin conjugate for 30 min at room temperature. After washing in PBS and developing color in peroxidase substrate with diaminobenzidine, signals were examined by microscopy (n = 6 in each group).
Statistical Analysis
The results were statistically analyzed using a software package (Statview 5.0, Abacus Concepts, Inc., Berkeley, CA, USA). All values were expressed as mean ± SE. The multiple comparisons among groups were made using an one-way analysis of variance (ANOVA). Post hoc analysis was performed by Fishers' PLSD test. A value of p < 0.05 was considered to denote statistical significance.
Results
Phenotypic Characterization of CD271+ MSCs and PA-MSCs
CD271+ MNCs were isolated from the BM-MNCs by AutoMACS. BM-MNCs contained a CD271+ cell fraction at the rate of about 20% and the freshly isolated CD271+ cell fraction had a purity of >90% as determined by FACS analysis (Fig. 2A). CD271+ MSCs and PA-MSCs, which were obtained after 3 weeks cultivation, were positive for cell surface markers of CD29, CD44, CD105, and CD166 (Fig. 2B) but were negative for cell surface markers of CD34, CD45, and CD133 (data not shown). CD271+ MSCs contained STRO-1 and CD271+ cell fractions at the rate of more than 10–40%, but PA-MSCs at the rate of less than 10% (Fig. 2B).

Phenotypic characterization and expansion potential of CD271+ MSCs and PA-MSCs. (A) The BM-MNCs contained a CD271+ cell fraction at the rate of about 20%. Freshly isolated CD271+ cell fraction had a purity of >90% as determined by fluorescence activated cell sorting (FACS) analysis. (B) Both CD271+ MSCs and PA-MSCs were positive for cell surface markers of CD29, CD44, and CD105. However for the CD271 cell surface marker, CD271+ MSCs contained a CD271+ cell fraction at the rate of 20–40%, while the PA-MSCs rate was less than 10%.
Expansion Ability of Each MSCs
We performed this experiment to compare expansion ability of CD271+ MSCs and PA-MSCs. Analysis of short-term kinetics showed that CD271+ MSCs exhibited significantly higher PD than PA-MSCs until six passages (p < 0.01 for CD271+ MSCs vs. PA-MSCs in passages 2–6). However, after seven passages, there was no significant difference between both populations (Fig. 3). The proliferative ability of both MSCs was retained even at passage 10.

Comparison of in vitro expansion ability. To examine the in vitro cell expansion ability, CD271+ MSCs and PA-MSCs (n = 5) were plated in 60-cm2 dishes, and routine cell expansion was performed every 14 days. Analysis of short-term kinetics showed that CD271+ MSCs exhibited significantly higher population doubling (PD) rates than PA-MSCs until six passages. The proliferative ability of both types of MSCs was retained even at passage 10.
Correlation Between CD271 Expression and Chondrogenesis
To investigate the change of CD271 expression rate during cell culture and the correlation between CD271 expression and chondrogenesis, we examined FACS analysis for CD271 expression and pellet culture using two, five, and eight passages of CD271+ MSCs. In the FACS analysis, passage 2 CD271+ MSCs expressed 38.45% of CD271, and passage 5 cells 22.07%, but the expression of CD271 was decreased to 5.26% by the 8th passage (Fig. 4A). Moreover, the pellet culture using CD271+ MSCs from each passage revealed that the average pellet size was significantly greater in passages 2 and 5 compared with passage 8 (passage 2, 1.39 ± 0.23; passage 5, 1.23 ± 0.12, passage 8, 0.71 ± 0.11, p < 0.05 for passages 2 and 5 vs. passage 8) (Fig. 4B, C).

Comparison of chondrogenesis by pellet culture. (A) In the FACS analysis, passage 2 CD271+ MSCs expressed 38.45% of CD271, and passage 5 cells 22.07%, but the expression of CD271 was decreased to 5.26% in the passage 8. (B, C) The pellet culture using CD271+ MSCs from each passage showed that the average pellet size was significantly greater in the passages 2 and 5 compared with passage 8.
Comparison of Chondrogenesis by Pellet Culture
The chondrogenic ability of each MSC was evaluated by pellet culture. The average pellet size after 3 weeks cultivation of each MSC was significantly greater in the CD271+ group compared to the PA group (CD271+, 1.69 ± 0.25; PA, 1.20 ± 0.16, p < 0.05, respectively.) (Fig. 5A, B). To further verify chondrogenesis, RT-PCR analysis of tissue RNA isolated from the pellets for human-specific chondrogenic markers (hSOX9 and hCOL2) was performed (Fig. 5C). The expression ratio of hSOX9 to hGAPDH was significantly greater in the CD271+ group than the PA group (CD271+, 1.093 ± 0.076; PA, 0.779 ± 0.083, p < 0.05, respectively) The expression ratio of hCOL2 to hGAPDH was significantly greater in the CD271+ group than the PA group (CD271+, 1.289 ± 0.094; PA, 1.082 ± 0.088, p < 0.05, respectively) (Fig. 5D).

Comparison of chondrogenesis by pellet culture. (A, B) Chondrogenic ability of each type of MSC was evaluated by pellet culture. The average pellet size after 3 weeks cultivation of each MSC type was significantly greater for CD271+ MSCs than PA-MSCs. (C, D) By RT-PCR analysis of tissue RNA isolated from each pellet for chondrogenic markers, the expression ratio of human sex-determining region Y box 9 (hSOX9) and human collagen type 2 (hCOL2) to human glyceraldehyde 3-phiosphate dehydrogenase (hGAPDH) was significantly greater in CD271+ MSCs than PA-MSCs.
Comparison of Chondrogenesis at the Chondral Defect Site in Animal Models
To histologically assess the chondrogenesis of each type of MSC, immunohistochemical staining for HLA-ABC for various lineages of human cells and hCOL2 for human cell-derived chondrocytes was performed using the tissue samples obtained 4 weeks after cell transplantation. The transplanted human cells were detected as HLA-ABC-positive cells, and differentiated human chondrocytes derived from the transplanted cells were detected as hCOL2-positive cells in the chondral defect site in the CD271+ and PA groups (Fig. 6A, B), while hCOL2-positive cells were not identified in the PBS group (Fig. 6C). The number of double-stained cells was significantly greater in the animals that received CD271+ MSCs compared to PA-MSCs (CD271+, 851.8 ± 95.5; PA, 348.7 ± 61.6; NC, 0.0 ± 0.0/mm2, respectively. p < 0.01 for CD271+ vs. NC and PA vs. NC, p < 0.05 for CD271+ vs. PA) (Fig. 6D).
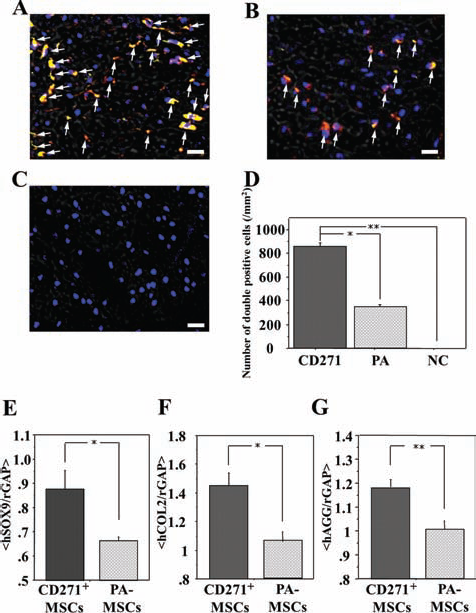
Comparison of chondrogenesis at the chondral defect site. Immunohistochemical staining and real-time RT-PCR for human-specific chondrocyte markers were performed using tissue samples harvested at week 4. Original magnification: 200×. Arrows show double-positive cells. Scale bars: 20 μm. (A–D) Differentiated human chondrocytes were identified as double stained cells with human specific COL2 (green) and human leukocyte antigen (HLA)-ABC (red) at chondral defect site in rats receiving CD271+ MSCs (A) or PA-MSCs (B). No labeled cells are evident in the PBS (no cell or NC) group (C). The number of human cells at chondral defect site was significantly higher in the animals that received CD271+ MSCs than those that received PA-MSCs (D). (E–G) Real-time RT-PCR analysis of tissue RNA isolated from the chondral defect site for human-specific chondrocyte markers [hSOX9, hCOL2, and human aggrecan (hAGG)] was performed. The expression ratio of hSOX9, hCOL2, and hAGG to rGAPDH were significantly greater in the animals that received CD271+ MSCs than those that received PA-MSCs.
We performed real-time RT-PCR to quantify the expression of hSOX9, hCOL2, and hAGG at the chondral defect sites. The expression ratio of hSOX9, hCOL2, and hAGG to rGAPDH at week 4 was greater in the CD271+ group compared to the PA group (hSOX9/rGAPDH: CD271+, 1.131 ± 0.284; PA, 1.085 ± 0.269, p < 0.05. hCOL2/rGAPDH: CD271+, 1.028 ± 0.276; PA, 0.991 ± 0.271, p < 0.05. hAGG/rGAPDH: CD271+, 1.179 ± 0.032; PA, 1.004 ± 0.035, p < 0.01) (Fig. 6E–G).
Macroscopic and Histological Chondral Healing
Macroscopic chondral healing was identified by the smooth surface (Fig. 7). All animals at 8 weeks that received CD271+ MSCs or PA-MSCs demonstrated chondral healing. However chondral defect sites in all animals receiving sponge alone showed chondral defect after 8 weeks, which is consistent with a previous report of the natural course in this animal model (47) (Fig. 7A–C).

Macroscopic and histological chondral healing. (A–C) Gross examination of chondral defects 8 weeks after surgery revealed glossy white and well-integrated repaired tissue in the animals that received CD271+ MSCs (A) or PA-MSCs (B), not in the animals that received collagen sponge alone (C). (D–G) Histological observations at 8 weeks after surgery revealed that chondral defects were filled with repaired tissue that appeared to be well integrated with the surrounding cartilage in the animals that received CD271+ MSCs (D, G) or PA-MSCs (E, H), not in the animals that received collagen sponge alone (F, I). Original magnification: 40× (D–F) and 100× (G–I). Arrow heads show chondral defect sites. Scale bars: 200 mm (D–F) and 80 mm (G–I). The average of histological score (Wakitani's score) in the animals that received CD271+ MSCs was significantly greater than in the other groups (J).
Chondral healing was also histologically evaluated with toluidine blue staining. Histological observations at 8 weeks after surgery revealed that chondral defects were filled with repaired tissue that appeared to be well integrated with the surrounding cartilage in the animals that received CD271+ MSCs (Fig. 7D, G) or PA-MSCs (Fig. 7E, H), but not in the animals that received collagen sponge alone (Fig. 7F, I). The degree of chondral healing at 8 weeks was assessed by Wakitani's score (44) and was significantly better in the CD271+ group compared to the other groups, as well as in the PA group compared to the NC group (CD271+, 3.2 ± 0.13; PA, 5.9 ± 0.46; NC, 11.2 ± 0.98, respectively, p < 0.01 for CD271+ vs. NC and PA vs. NC, p < 0.05 for CD271+ vs. PA) (Fig. 7J).
Apoptosis Analysis at the Cell Transplanted Site
To analyze chondrocyte apoptosis histologically, TUNEL staining was performed using tissue samples obtained 4 weeks after cell transplantation. Chondrocyte apoptosis was less abundant in the cell transplanted site, especially in the CD271+ group compared with the NC group (Fig. 8A–C). Quantification of TUNEL staining showed that the CD271+ group had significantly fewer apoptotic chondrocytes compared with the other groups, as well as the PA group compared with the NC group (CD271+, 55.5 ± 9.4; PA, 84.0 ± 5.6, NC, 203.4 ± 18.5, p < 0.01 for CD271+ vs. NC and PA vs. NC, p < 0.05 for CD271+ vs. PA) (Fig. 8D).

Apoptosis analysis at the cell transplanted site. (A–C) Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining in all groups 4 weeks after transplantation indicated that chondrocyte apoptosis was less abundant in the cell transplanted site, especially in the CD271+ group (A) compared with the NC group (C). Original magnification: 200×. Scale bars: 40 μm. (D) Quantification of TUNEL staining showed that the CD271+ group had significantly fewer apoptotic chondrocytes compared with the other groups, as well as the PA group compared with the NC group.
Discussion
In the present study, we compared CD271+ MSCs with classical PA-MSCs isolated from BM for cartilage regenerative potential and indicated the superiority of CD271+ MSCs over PA-MSCs. CD271 is known as one of the cell surface markers for MSCs (37) and was first reported as the receptor for nerve growth factor (NGF) in human neuronal and nonneuronal cells (43). After the first report, CD271 became well known as a NGF receptor or neurotrophin receptor (p75 NTR), belonging to the tumor necrosis factor receptor super family (10), and CD271 was reported to be much more widely expressed in human cells and tissues (19). Recently, some papers showed that isolated CD271+ BM-MNCs have a one to three log greater expansion ability of MSCs than nonisolated BM-MNCs and a greater capacity to differentiate to adipocytes, osteoblasts, and chondrocytes than classical PA-MSCs in vitro (19,37). Based on these backgrounds, CD271+ MSCs can be expected to have a greater faculty for chondral regeneration than PA-MSCs.
In this study, FACS analysis demonstrated that isolated CD271+ MNC fractions were more than 90% CD271+ cells. However, after the cultivation to obtain MSCs from CD271+ MNCs, the expression of CD271 decreased to 10–40% (Fig. 2B). This phenomenon matched the previous papers that BM-MSCs are losing the CD271 marker in culture and are unable to reacquire it (18,20). From the FACS analysis, both CD271+ MSCs and PA-MSCs were positive for cell surface markers of CD29, CD44, CD105, and CD166 and negative for cell surface markers of CD34, CD45, and CD133, which is a similar finding to the previous reports using BM-MSCs (38,39) (Fig. 2B). CD271+ MSCs contained a CD271+ cell fraction at the rate of 10–40%, whereas PA-MSCs contained less than 10%. This difference may be one of the important factors for explaining the reason why CD271+ MSCs had a greater initial expansion ability than PA-MSCs (Fig. 3). In the case of MNCs, it is reported that CD271+ MNCs can expand at a higher rate than nonisolated BM-MNCs because of less contamination with hematopoietic cells (16). Therefore, it is possible that this superiority of CD271+ MSCs in cell expansion is caused by the same reason as for CD271+ MNCs. Moreover, the results of FACS analysis showed that CD271+ MSCs have more STRO-1-positive cells than PA-MSCs, which also indicated CD271+ MSCs can have greater expansion ability and stemness when compared with PA-MSCs.
To further verify the correlation between CD271 expression and chondrogenesis, the pellet culture using different passages of CD271+ MSCs was performed (Fig. 4), which displayed that the cells from passages 2 and 5 have a greater chondrogenesis when compared with the passage 8 cells. These results support the possibility that the higher expression of CD271 results in the greater ability for chondrogenesis. Further experiments will be needed to highlight the detailed correlation between CD271 expression and chondrogenesis.
According to the previous report, the pellet's size is a convincing indicator of in vitro chondrogenesis of MSCs (39). Our data showed that CD271+ MSCs were significantly greater than PA-MSCs in average pellet size. Moreover, RT-PCR analysis of the pellets demonstrated greater expression of chondrogenic related genes in CD271+ MSCs compared with PA-MSCs (Fig. 5). We have demonstrated, for the first time, CD271+ MSCs had a greater chondrogenic potential than PA-MSCs with a pellet culture method.
In the in vivo study, it has already been demonstrated that chondral repair is obtained by transplantation of PA-MSCs (1,15,27,31,33,44). In the current study, we demonstrated that CD271+ MSCs had a greater faculty of chondrogenesis and potential for repairing the cartilage defects than PA-MSCs in the rat chondral defect models (Figs. 6 and 7). The appearance of histology in our study showed some differences from previous reports, because the defects in the histological figures of our study were a little shallower than the ones in the other papers (15,33, 44). Nevertheless, from the histological figure in the control (NC) group, we were able to confirm that the defects had reached subchondral bone and could not recover naturally. The chondral healing in this study occurred in the same manner as previous reports with PA-MSCs. The thickness and appearance were comparable to normal cartilage and the subchondral plate was similar to that of surrounding cartilage. However, the adjacent normal cartilage did not show any degenerative change as previous papers showed (15,33,44). This difference may be attributed to the method of making a chondral defect or the way of cell transplantation. We used cell pellets to transplant on chondral defect sites with collagen sponge as a cover. The pellet culture system with MSCs or chondrocytes is widely used to produce calcification in the in vitro study (22), and a three-dimensional environment can support spherical cell morphology and prevent chondrocyte dedifferentiation (2,4,9,32). Considering the clinical application, it may be better to use some form of scaffold for transplantation. Further experimental investigation of the transplantation methods will thus be needed to investigate which method we should use for chondral repair.
We also performed apoptosis assays in vivo (Fig. 8). TUNEL assay (apoptosis) results suggested that CD271+ MSCs showed a lower level of chondrocyte apoptosis than PA-MSC in the chondral repair site. It is conceivable as one of the mechanism behind the superiority of CD271+ MSCs to PA-MSCs, as well as the in vitro higher expansion ability of CD271+ MSCs than PA-MSCs.
PA-MSCs have already been applied to chodral defect in the clinical situation and a case report have published (27). Compared with transplantation of isolated CD271+ MSCs, PA-MSC transplantation does not require a magnetic cell sorting process, indicating that time and cost of the cell preparation can be diminished. However, our study revealed that CD271+ MSCs is one of the more effective cell sources compared to the classical PA-MSCs for chondral defect fixation. Thus, additional preclinical/clinical studies would be warranted to compare the feasibility, safety, and efficacy of both strategies for cartilage repair.
In a recently published paper, it was reported that the proliferation rate of the selected CD271+ MSCs was higher than PA-MSCs by in vitro cell culture (36). However, the current report is the first indication that isolated CD271+ MSCs show a greater capacity to differentiate to chondrocytes than classical PA-MSCs in the in vivo situation.
Several limitations should be noted in this study. At first, although we have already clarified the cartilage repair by CD271+ MSC transplantation, we have not evaluated it biomechanically. However, there are some reports that aggrecan can mainly work as a compressive force in articular cartilage (7). Since we showed greater gene expresson of aggrecan in the chondral repair site in the CD271 group compared with the PA group (Fig. 6G), the superior of CD271+ group can be expected in the biomechanical assessment. A second limitation is that the rat models used in the present study were small, and the cartilage healing process occurs at a faster rate in rats than in human. We need further studies to test the effectiveness of this application in a large animal model so as to confirm the clinical feasibility.
In conclusion, our data suggest that CD271+ MSCs have a greater ability than classical PA-MSCs in in vitro and in vivo chondrogenesis. The present findings may provide important clinical insight for regenerative medicine aimed at enhancing cartilage healing in chondral defect injury, and this method using CD271+ MSCs for chondral defect is likely to be a reasonable treatment in the future.
Footnotes
Acknowledgments
The authors are grateful to Miki Horii and Ayumi Yokoyama for excellent technical help. The authors declare no conflict of interest. The authors confirm that there is no financial support in regard to this study.
