Abstract
Photobiomodulation (PBM) therapy stands as an innovative neurostimulation modality that has demonstrated both efficacy and safety in improving brain function. This therapy exerts multifaceted influences on neurons, blood vessels, and their intricate interplay known as neurovascular coupling (NVC). Growing evidence indicates that NVC may present a promising target for PBM intervention. However, the detailed mechanisms underlying its therapeutic benefits remain to be fully understood. This review aims to elucidate the potential metabolic pathways and signaling cascades involved in the modulatory effects of PBM, while also exploring the extensive repertoire of PBM applications in neurologic and psychiatric conditions. The prospects of PBM within the realm of NVC investigation are intensively considered, providing deeper insights into the powerful capabilities of PBM therapy and its potential to revolutionize neurostimulation treatments.
Keywords
Introduction
Despite constituting a mere 2% of total body weight, the human brain, as the core organ governing essential physiological homeostasis and orchestrating complex cognitive processing, consumes approximately 20% of the body’s energy metabolism. 1 The myriad facets of cerebral activity necessitate substantial metabolic demands, requiring abundant oxygen and glucose consumption. Given the lack of energy reserves in the brain, the supply of nutrients and oxygen relies heavily on cerebral blood flow (CBF). 2 Within the intricate realm of brain energy metabolism, neurovascular coupling (NVC), in which the regional CBF fluctuates continuously in sync with the ever-changing dynamics of brain functional conditions, stands out as a fundamental cornerstone. NVC is distinguished by its swift responsiveness and highly localized CBF change in reaction to neuronal activity, ensuring an excellent spatiotemporal matching between local neural excitability and energy supply. 3 Hence, normal NVC function is vital for preserving cerebral homeostasis and supporting brain functions.
NVC operates via the neurovascular unit (Figure 1(a) and (b)), comprising the extracellular matrix and a diverse array of cells, including cerebral neurons, astrocytes, pericytes, vascular endothelial cells (ECs), and vascular smooth muscle cells (VSMCs). These cells work in harmony through neurovascular signaling pathways and structural connections within the extracellular matrix. 4 Damage to any component of the neurovascular unit can impair NVC function, causing a mismatch between energy supply and neuronal activity. This, in turn, leads to chronic neuronal damage and a decline in cognitive function, which underlies the pathophysiology of various cerebral disorders. 5 However, there is a paucity of specialized pharmacological compounds and therapeutic interventions tailored to address NVC imbalance.
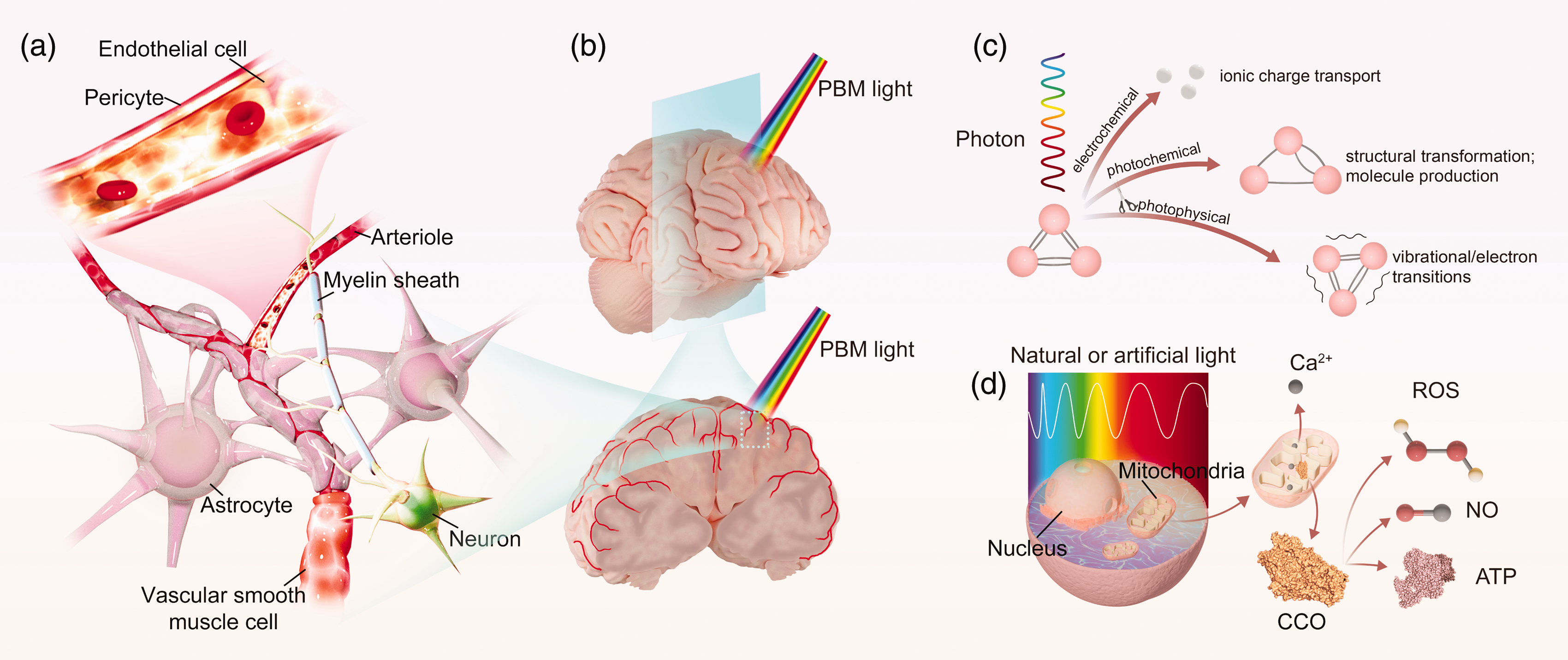
Schematic representation depicting the cellular structure of the neurovascular unit and the biochemical processes triggered by photobiomodulation (PBM). (a) Neurovascular coupling (NVC) operates through the neurovascular unit, which is composed of extracellular matrix and diverse cells, including neurons, astrocytes, pericytes, endothelial cells (ECs), and vascular smooth muscle cells (VSMCs). These cells work in concert through neurovascular signaling pathways that rely on structural connections within the extracellular matrix, ensuring the spatiotemporal alignment between energy requirements of neuronal activities and cerebral blood flow (CBF); (b) Light delivery of PBM on cerebral cortex and peripheral neurovascular unit; (c) PBM stimulation elicits a range of electrochemical, photochemical, and photophysical reactions by transferring light energy to cellular and organelle molecules, resulting in diverse biochemical effects and (d) the fundamental molecular mechanisms of PBM therapy centers around the light-induced activation of mitochondrial functionality, primarily mediated by cytochrome c oxidase (CCO) as the central photo-acceptor and photo-signal transducer. This initial event triggers the modulation of various signaling pathways through the release of diverse cellular factors, including nitric oxide (NO), calcium ions (Ca2+), reactive oxygen species (ROS), and adenosine triphosphate (ATP), subsequently promoting CBF and exerting neuroprotective and neurogenic effects.
Photobiomodulation (PBM) therapy, also termed low-level light therapy, represents a novel treatment modality for brain diseases, employing low-intensity light within the 300 to 1400 nm wavelength range. The initiation of physiological modulation effects in PBM hinges upon the light absorption by endogenous chromophores, which converts light energy into molecular excitations within cells and organelles, subsequently triggering a range of electrochemical, photophysical, and photochemical reactions,
6
as illustrated in Figure 1(c). These reactions encompass:
Electron transitions, excited by light at shorter wavelengths, and electron transfer between molecules.
7
Molecular vibrational dynamics, activated by vibrational absorption of photons at longer wavelengths, induce vibrational energy level transitions, affecting the structure and function reactivity of the molecules.
8
Chemical transformations, leading to structural changes in molecules, transitions in valence states, and corresponding changes in potential energy; and electrochemical transitions, which involve the kinetic migration of mobile ions driven by either an electric field (drift conduction) or a concentration gradient (diffusion current).
9
Subsequently, various biochemical processes are triggered (Figure 1(d)). The therapeutic efficacy of PBM has been demonstrated in proteins, DNA, and chromophores across diverse cell types, notably vascular cells, neurons, and glial cells.10 –12 These effects encompass enhanced tissue oxygenation and ATP synthesis, augmented CBF, attenuated cell destruction caused by oxidative stress, neuroinflammation, and neurotoxicity, as well as facilitated neurogenesis and synaptogenesis.13 –15 Diseases that have been implicated include neurodegenerative diseases, psychiatric disorders, neurodevelopmental disorders, and brain traumatic events.13,14,16
Due to the prerequisite of light absorption by chromophores to induce biological influences, the parameters used in PBM therapy are of great importance for predictive outcomes. One of the most significant parameters is the wavelength, which dictates the responsive molecule. Specifically, the red-to-near-infrared (NIR) spectrum at 600–1100 nm is preferred for neuromodulation due to its superior penetration capabilities. 17 Currently, the preferred wavelengths for application in PBM are within the NIR-I spectrum, specifically around 810 nm, 660 nm, and 980 nm bands. Additionally, NIR-II light is gaining attention for treating brain disorders due to its enhanced penetration capabilities. While a small amount of heat may be produced during PBM, this temperature increase stays within a safe range and does not lead to significant tissue damage. The use of pulsed wave light is equally crucial, which stands out over continuous wave for its capacity to achieve the same peak power density and better penetration depth, but without measurable temperature rise. 18 Furthermore, pulsed light at specific frequencies has been demonstrated to engage with neural oscillations and to activate ion channels, potentially enhancing the therapeutic efficacy.19,20
Among neuromodulation techniques such as vagus nerve stimulation, transcranial magnetic stimulation, and transcranial electrical stimulation, PBM stands out as a non-invasive, radiation-free, well-tolerated, and low-cost therapeutic tool. 21 Nevertheless, the precise mechanisms through which PBM regulates NVC remain incompletely elucidated. This review aims to elucidate the current understanding of how PBM affects NVC at the molecular and cellular levels, with a special focus on the metabolic pathways and signaling cascades involved. We also provide a detailed overview of the application of this technique, to assist clinicians in making better treatment decisions for their patients.
Mechanisms of NVC bio-regulation by PBM
The fundamental mechanisms of PBM revolve around the restoration of mitochondrial dysfunction, which assumes a pivotal role in the pathogenesis of a wide range of brain diseases. The most advocated primary photo-acceptor is the mitochondrial cytochrome c (CCO). 22 After light activation, the viability of various cellular factors is modulated through certain cellular signaling pathways, consequently elevating CBF, exerting neuroprotective and neurogenic effects, and facilitating neurovascular communication. 23 Here, we present the latest insight into the regulatory effects of PBM on NVC with a particular focus on cerebral vessels and neural cells.
Primary bio-regulation by PBM
Modulation of adenosine triphosphate (ATP) levels
ATP serves as the primary currency for brain energy metabolism, primarily driving ion gradient pumping during neuronal signaling. 1 In the brain vascular system, a multitude of factors, including ischemia, hypoxia, and other conditions, may precipitate ATP insufficiency. This deficiency can instigate endothelial dysfunction and trigger a cascade of pathophysiological alterations, involving abnormal vasomotion, an imbalance in reactive oxygen species (ROS) and nitric oxide (NO), inflammatory response, and disruption of the coagulation process of ECs. One notable impact of PBM is the ability to directly increase ATP production, which is proposed to initiate from the light excitation of CCO, 22 also referred to as respiratory chain complex enzyme IV. CCO orchestrates the terminal and purported rate-limiting step within the intricate network of the mitochondrial electron transport chain (ETC). 24 Upon photon absorption, NO, initially bound to the heme iron and copper centers of CCO, is proposed to dissociate, thereby relieving its inhibitory effect on oxygen binding. This further facilitates oxygen consumption and proton translocation across the mitochondrial membrane. 25 Hence, the activation of ETC and an adaptation of the mitochondrial membrane potential (MMP) ensues, eventually culminating in ATP generation 26 (Figure 2(a)).
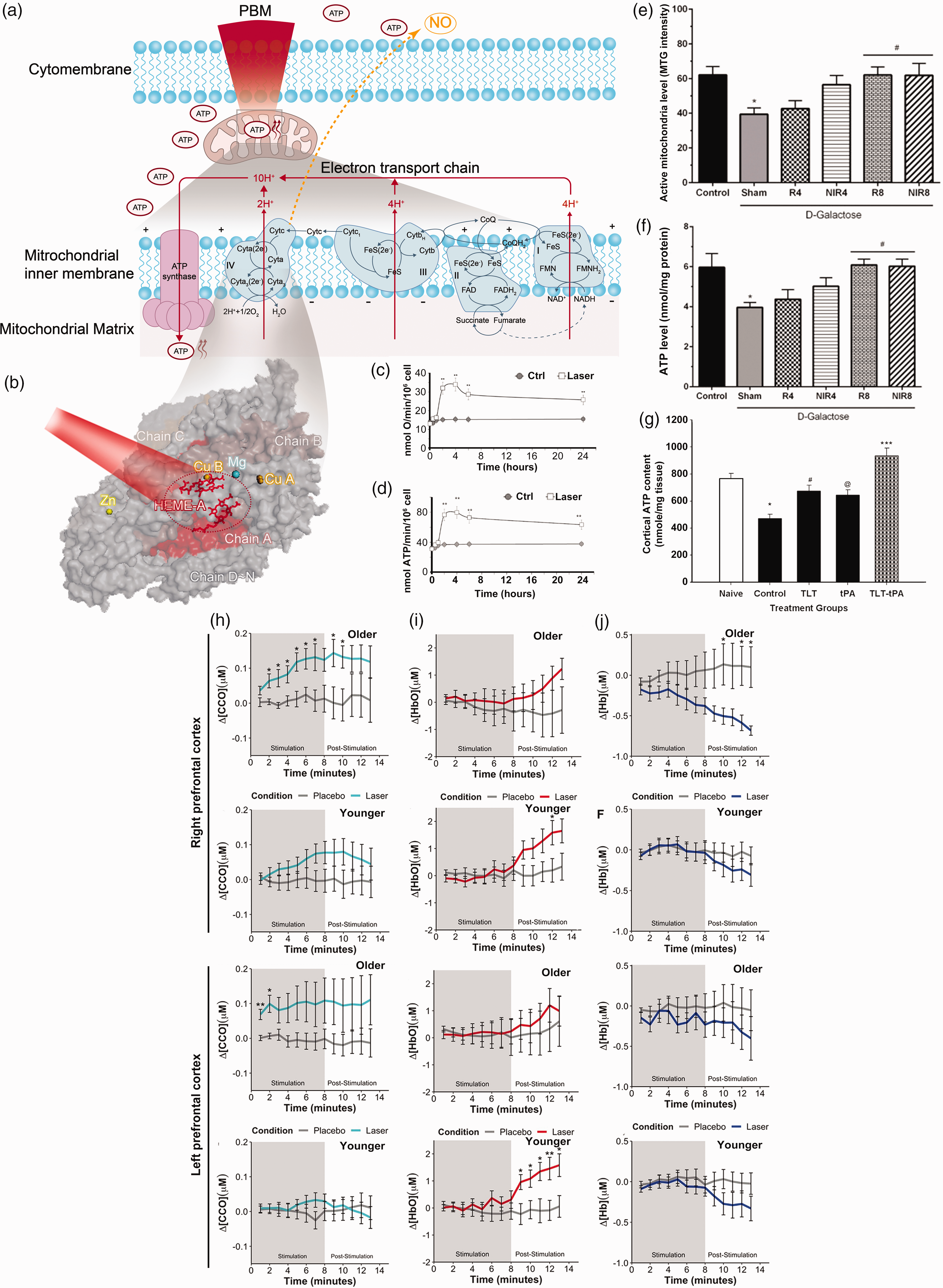
Impact of PBM on mitochondrial function and ATP production. (a, b) Schematic depictions illustrating the impact of PBM on mitochondrial respiratory chain with cytochrome c oxidize (CCO) as the primary photo-absorber. Upon photon absorption, Continued.nitric oxide (NO) is dissociated from the catalytic centers of CCO, subsequently promoting the binding of oxygen to the heme iron and copper centers of CCO. These processes enhance the activity of respiration and ATP production. (c) Comparison of the oxygen consumption and (d) ATP synthesis between PBM-treated and untreated individuals over time. Reprinted from 70 with permission from 2018 Springer Nature. (e) Heightened mitochondrial activity and (f) augmented ATP levels in brain tissue induced by PBM therapy (NIR4, near-infrared light at a fluency of 4 J/cm2; NIR8, near-infrared light at a fluency of 8 J/cm2; R4, red light at a fluency of 4 J/cm2; R8, red light at a fluency of 8 J/cm2). Reprinted from 99 with permission from Elsevier Inc. (g) The decrease in cortical ATP depletion due to embolization, followed by ATP restoration after transcranial laser therapy, was noted. Reprinted from 100 with permission from Elsevier B.V. (h) In older or younger individuals, respectively, PBM-induced changes of oxidized CCO, (i) oxygenated hemoglobin (HbO) and (j) deoxygenated hemoglobin (Hb) within the right and left prefrontal cortex. Reprinted from 101 with permission from Elsevier Inc.
The notion that PBM contributes to ATP synthesis via activating CCO is mainly derived from indirect observations, based on the resemblances between the absorption characteristics of CCO and the action spectrum of PBM effects. The mammalian CCO is composed of 14 subunits (subunits A∼N, Figure 2(b)), with the largest three subunits (I, II, III) forming the central components of the cascade reaction. Among them, the redox-active metal centers: two heme moieties (heme a and heme a3) in subunit I, two redox-active copper sites (CuA in subunit II and CuB in subunit I), along with one zinc and one magnesium site, are potential chromophores for visible light. 27 Specifically, CCO has absorption peaks within the red and NIR spectral range, including heme a at 605 nm, CuA reduced at 620 nm, heme a3/CuB at 655 nm, and CuB oxidized at 680 nm. Extending into the NIR region, additional peaks are observed: CuB is reduced at 760 nm and CuA is oxidized at 825 nm. 28 However, the precise manner in which light affects CCO is not yet known. It was the detection of ATP and NO production from cells during PBM that prompted the conjecture that both reactions transpire within CCO (Figure 2(b)). Recently, an experiment conducted on isolated mitochondria demonstrated a significant increase in mitochondrial biochemical energy density and ATP production following PBM, substantiating the photic stimulation of mitochondrial function by PBM. 29
However, some studies suggest that non-mitochondrial mechanisms, involving various potential chromophores such as transient receptor potential ion channels, transforming growth factor β (TGF-β), superoxide dismutase (SOD1), and catalase, may also play a role. 30 Additionally, there is a controversial hypothesis asserting that the impact of PBM might work separately from CCO. This notion is backed by instances where PBM was observed to augment both ATP and citrate synthase activity levels in cells lacking CCO. 31
Moreover, the elevation in ATP production engenders a cascade of effects and benefits. ATP not only acts as a cellular fuel, facilitating key processes such as axonal transport, cytoskeletal maintenance, microglial movement, gene expression, and protein modification, but also functions as a neurotransmitter, modulating neurogenesis and neuroapoptosis. 32 In the course of ATP biosynthesis of PBM, several additional modulatory molecules are engaged, including NO, ROS, calcium ions (Ca2+), and various signaling molecules. The details of these regulatory factors will be further elucidated in subsequent sections.
Promotion of nitric oxide (NO) release
The liberation of NO also constitutes one of the primary physiological ramifications of PBM. NO, a key orchestrator of endothelium-dependent relaxation, 33 is generated across a diverse array of cellular types, 34 with vascular ECs standing as the primary hub. 35 Once synthesized, NO readily diffuses across the cell membrane and reaches adjacent VSMCs, where it activates soluble guanylate cyclase (sGC), catalyzing an increase in intracellular cyclic guanosine monophosphate (cGMP) (Figure 3(A)). This surge in cGMP levels subsequently modulates protein kinase G, phosphodiesterase, and ion channels, leading to SMC relaxation and vasodilation. 36 In this physiological cascade, NO functions as a pivotal secondary messenger, essential for maintaining homeostasis.
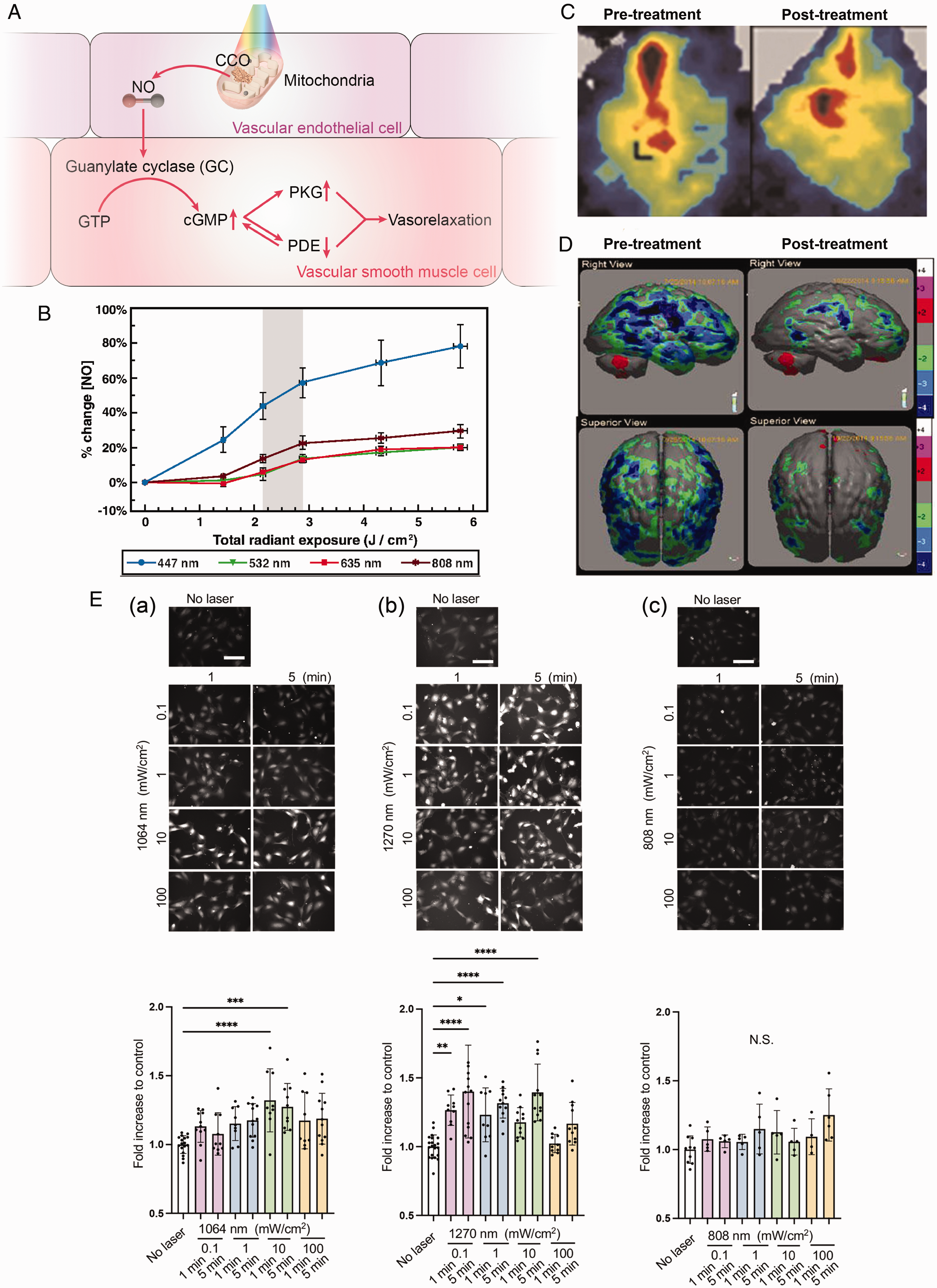
Impact of PBM on mitochondrial NO release and vascular relaxation. (A) A graphical representation of PBM-induced NO release and subsequent NO signaling pathways leading to vasodilation (PKG, protein kinase G; PDE, phosphodiesterase). (B) NO increase induced by light exposure at various wavelengths, in a biphasic dose-response manner. Reprinted from 25 with permission from the Society of Photo-Optical Instrumentation Engineers. (C) Perfusion images before and after 15 min NIR laser irradiation showed an increase in CBF in the irradiated field. Reprinted from 38 with permission from John Wiley and Sons. (D) Quantitative analysis of regional CBF of pre- and post-PBM treatment. Reprinted from 39 with permission from Mary Ann Liebert, Inc and (E) dose- and time-dependent NO generation in ECs induced by laser exposure at 1064 (a), 1270 (b), and 808 nm (c), respectively. Reprinted from 40 with permission from John Wiley and Sons.
Emerging evidence has established a foundation for the phenomenon of light-induced NO dissociation from CCO, leading to increased CBF. Under pathological conditions, NO non-covalently binds to the heme iron and copper centers of CCO. 37 Theoretical computations based on time-dependent density functional theory demonstrate that NIR light illumination can effectively trigger the release of NO. 10 Pope et al. further corroborated this phenomenon, revealing that low-energy laser exposure at various wavelengths significantly enhanced cellular NO levels in retinal pigment epithelium cells immediately following the light intervention, and the process is independent of the function of NO syntheses (Figure 3(B)). 25 The NO generation then promotes CBF increase, as Uozumi et al. found that 808-nm NIR laser irradiation increased local CBF by 30% compared to control mice (Figure 3(C)). 38 In a clinical study, pulsed PBM effectively enhanced the regional CBF in 66.7% of participants with traumatic brain injury (TBI), as determined by single-photon emission computed tomography (SPECT) examination. Quantitative SPECT analysis further unveiled a statistically significant disparity in regional CBF before and after treatment (Figure 3(D)). 39 However, despite these promising findings, direct observation of NO photodissociation from CCO following PBM remains scarce, necessitating advanced structural biology techniques to unravel the underlying molecular mechanisms.
In addition to NIR light in the NIR-I window (630–1000 nm), studies have also looked into the stimulative effects of light in the NIR-II window (1000–1700 nm) on NO release, which boasts less scattering and greater penetration depth. In a recent study, exposure to low-power light at two distinct wavelengths (1064 and 1270 nm) was found to be significantly associated with increased NO in ECs (Figure 3(E)), highlighting the potential of NIR-II window light to improve CBF. 40 Above all, the presence of multiple metal cofactors and subunits within CCO supports its sensitivity to a wide range of wavelengths, potentially elucidating its multifaceted photo-responsive properties. However, this inherent complexity of CCO structure poses great challenges when investigating and characterizing its spectral properties, rendering the spectral analysis of CCO becomes exceptionally demanding in research studies.
Modulation via byproduct reactive oxygen species (ROS)
PBM therapy has a profound impact on the ETC, going beyond the elevation of ATP levels in cells. Within the intricate responses of the ETC, while oxygen acts as the ultimate electron acceptor being converted into water, a proportion of metabolized oxygen generates ROS as natural byproducts, such as superoxide anion (
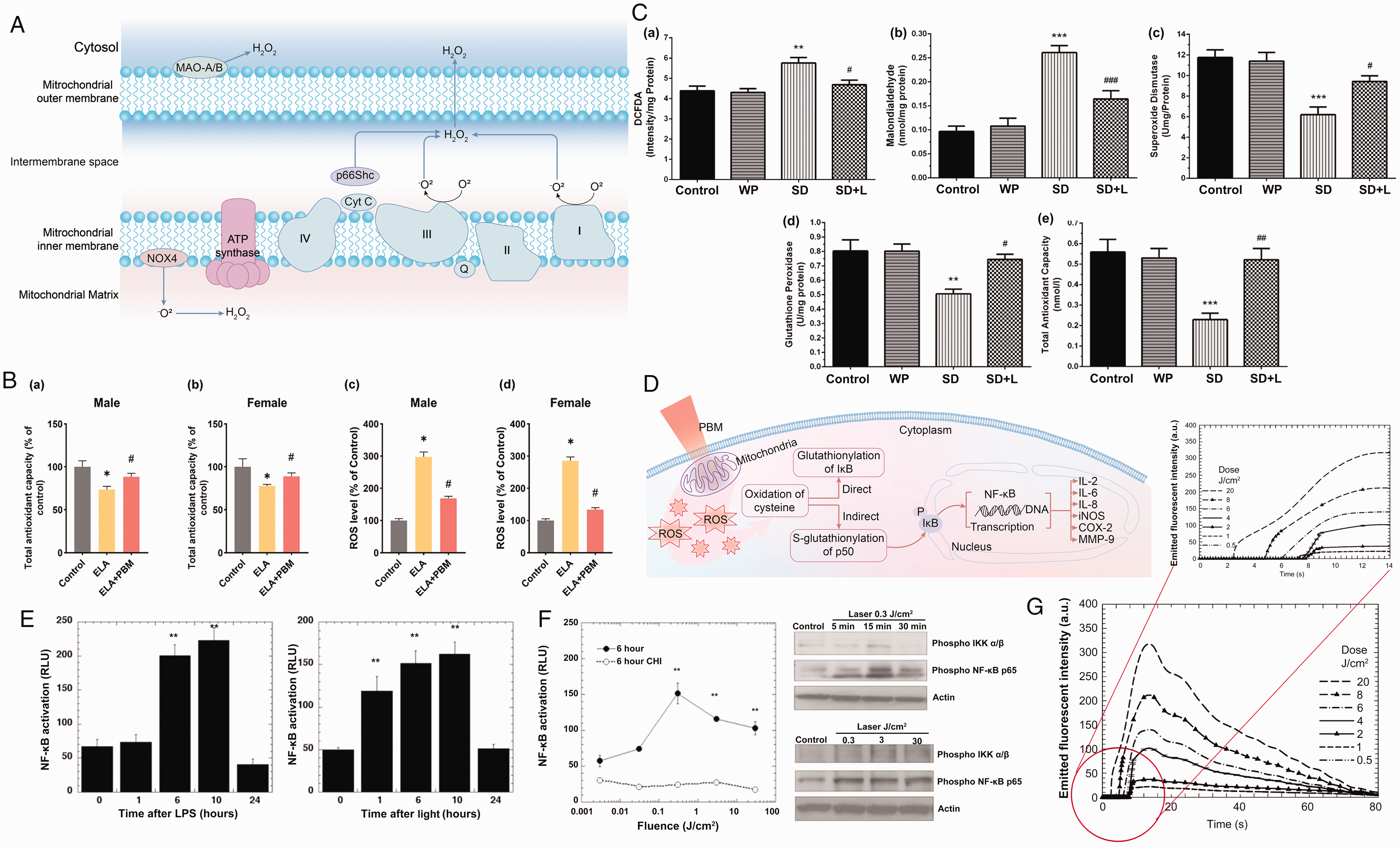
PBM’s modulation via byproduct reactive oxidative species (ROS) of the electron transport chain (ETC). (A) Schematic illustration of the generation of ROS (
Cellular redox homeostasis, based on the delicate balance between ROS production and antioxidant defenses, is crucial for maintaining cellular health. When present at appropriate levels, ROS function as signaling molecules that trigger beneficial antioxidant responses and support cellular function. However, when ROS levels become excessive, they can lead to oxidative stress, damaging cellular structures. PBM therapy has demonstrated its efficacy in enhancing total antioxidant capacity (TAC) (Figure 4(Ba,b)) and reducing ROS levels in various animal models with disrupted redox homeostasis (Figure 4(Bc,d)). 11 In models of sleep deprivation-induced oxidative stress, PBM decreased ROS and malondialdehyde levels while boosting antioxidant enzymes like SOD (Figure 4(C,c)) and glutathione peroxidase (GPx, Figure 4(C,d)), thereby augmenting the TAC in the mouse hippocampus (Figure 4(C,e)). 42 These findings highlight the ability of PBM to mitigate oxidative stress and bolster antioxidant defenses.
The modulation of ROS by PBM also influences several ROS-involved signaling pathways and the corresponding redox-sensitive transcription factors, indicating its cell-protective advantages. ROS possesses the ability to regulate gene expression and crucial processes such as cell proliferation and repair. One illustrative example is nuclear factor kappa B (NF-κB, Figure 4(D)): increased ROS lead to the oxidation of cysteine molecules, thereby activating NF-κB and enabling the transcription of target genes through two distinct mechanisms, including indirect S-glutathionylation of p50 and direct glutathionylation of I-κB. 43 Then, the NF-κB dimers translocate to the nucleus, where they bind to specific DNA sequences and then promote the transcription of target genes, including but not limited to interleukin (IL)-2, IL-6, IL-8, inducible nitric oxide synthase (iNOS), cyclooxygenase 2 (COX-2), and matrix metallopeptidases (MMP-9). 44 Chen et al. found that PBM enhances mitochondrial respiration and activates redox-sensitive NF-κB signaling through ROS generation, potentially explaining the beneficial effects of PBM concerning the expression of anti-apoptosis and pro-survival genes (Figure 4(E) and (F)). 45
ROS have a dual-edged nature, with their effects on cellular physiology depending on concentration, rendering the selection of irradiation parameters critically important. Research has indeed established a link between ROS generation and laser fluence (Figure 4(G)). 46 In a separate study, where the optimal radiant exposure for PBM was found to be 3 J/cm2, the alterations in various redox effect indicators—including ROS, NF-κB, tumor necrosis factor (TNF)-α, IL-1β, intracellular SOD, and TAC—were found to be dose-dependent. 47 Higher doses, such as 120 J/cm2, can lead to excessive ROS generation and activate apoptotic pathways, 48 indicating the biphasic-dose responses of different PBM parameters, which require meticulous management to avoid adverse effects.
Additionally, the effects of PBM on cells seem to be contingent upon their oxidative stress levels. Studies on normal and oxidatively stressed primary cortical neurons in mice have shown that PBM can alter ROS levels in different ways. 49 In cells without oxidative stress, PBM appears to expedite electron transfer within mitochondria, leading to heightened MMP and a transient surge in ROS. 50 Nevertheless, in cells suffering from oxidative stress, PBM can have the opposite effect on ROS. For instance, in a high-glucose model, PBM boosted antioxidant capacity while lowering ROS levels. 47 Given the dual nature of PBM’s effects and the variability in cellular oxidative states, it is important to thoroughly evaluate cellular conditions before adjusting the PBM settings. Further experimental data is needed to provide additional support for these findings.
Activation of light and heat-sensitive ion channels
Emerging research has unveiled the presence of light and heat-gated ion channels to mediate the modulatory effects of PBM, namely transient receptor potential vanilloid (TRPV) channels. TRPV channels have proven to be versatile cellular sensors, orchestrating the cellular response to a range of external stimuli, encompassing light, thermal variations, mechanical forces, gustatory and olfactory signals. 51 PBM was shown to effectively regulate TRPV channels through either light or heat excitation, resulting in a Ca2+ influx. The intracellular Ca2+ elevation triggers a cascade of events, including the activation of factors such as cAMP-response-element-binding protein (CREB), NF-κB, NOS, and the relevant signaling pathways, exerting neuroprotective effects and fostering cellular survival (Figure 5(a)). 52
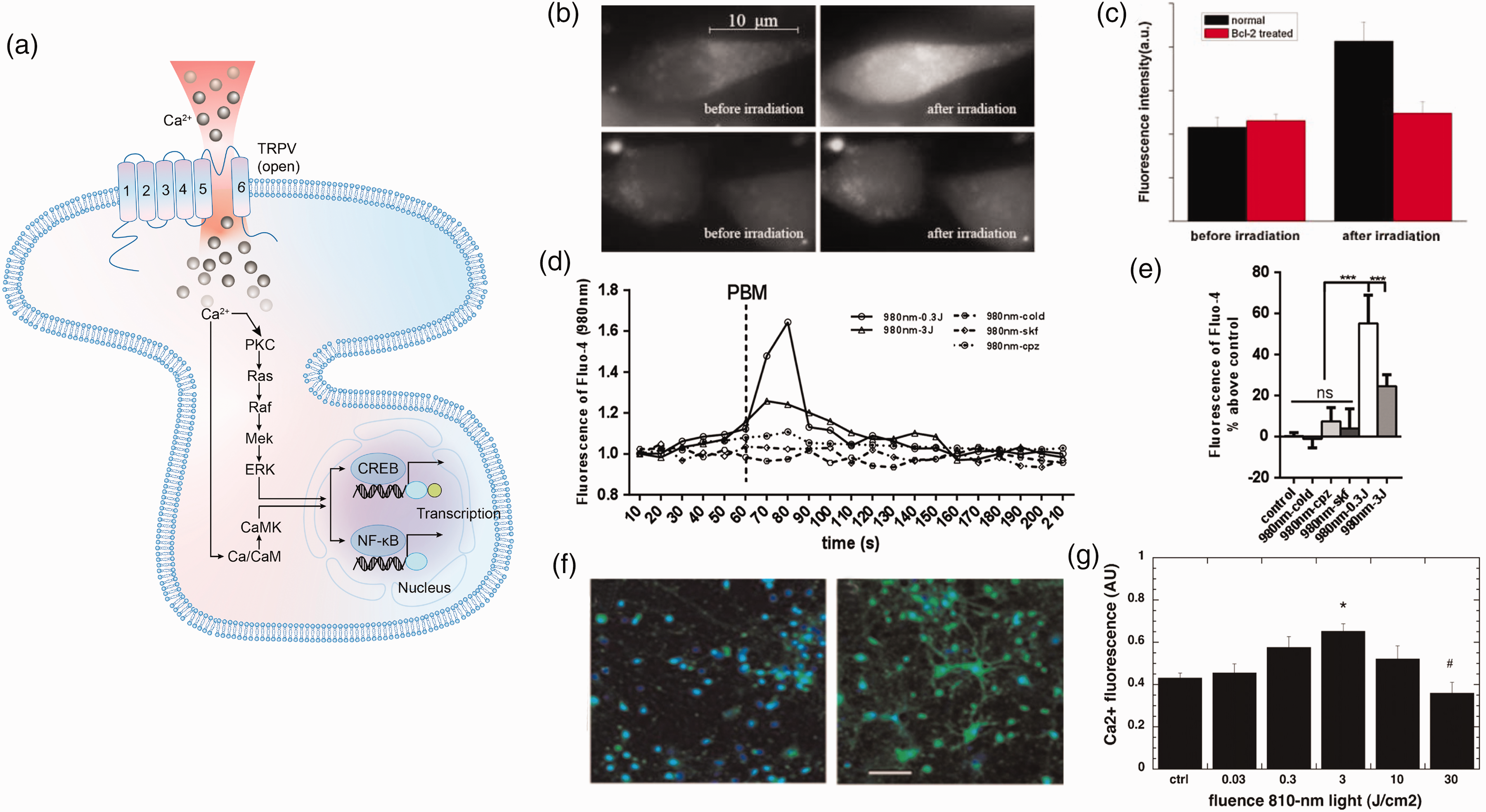
PBM modulation via transient receptor potential vanilloid (TRPV) channels and intracellular Ca2+ elevation. (a) The activation of the TRPV channel initiates the influx of Ca2+, and this intracellular Ca2+ elevation triggers a cascade of events, including the activation of transcription factors such as cAMP-response-element-binding protein (CREB) and NF-κB. (b) The fluorescence images of Ca2+ in an RBL-2H3 cell before (left) and after laser irradiation (right). For the bottom row, cells have been incubated with Bcl-2, a modulator impeding the translocation of mitochondrial signals to the cytoplasm. 57 (c) The increment of intracellular Ca2+ after laser irradiation. Reprinted from 57 with permission from John Wiley and Sons. (d) Time course of cytosolic Ca2+ following 980-nm PBM with different fluence. 58 (e) Rise of cytosolic Ca2+ concentration induced by 980-nm PBM with different fluence. Reprinted from 58 with permission from Elsevier B.V. (f) The fluorescence images of intracellular calcium (green) in control (left) and laser-treated cortical neurons (right) and (g) the biphasic dose response of intracellular Ca2+ to varying fluence levels of 810-nm light irradiation. Reprinted from 59 with permission from John Wiley and Sons.
The most studied subgroup of TRPV channels involved in PBM is TRPV1. As for the light-gated activation, the absorption peak of TRPV1 itself lies in the wavelengths of green light (490–570 nm). 53 However, under red to NIR light, TRPV1 is prone to manifest its temperature-sensitive specificity, with the nanostructured water clusters serving as the initial chromophores. 54 Water clusters, with an absorption peak in the region of 970 nm, undergo vibrational excitation upon photon absorption, elevating their vibrational energy levels. This energy gain creates highly localized temperature gradients, capable of activating the heat-gated ion channels such as TRPV1 families, thus modifying their tertiary structure and functional responsiveness. 8
Furthermore, it is worth noting that the TRPV1 channel plays a significant role in controlling the vascular tone by regulating NO concentration, with the specific pathways differing based on the cell types involved. On the one hand, in endothelial cells, activation of the channel enhances iNOS activity and improves endothelial nitric oxide synthase (eNOS) expression, leading to NO production and in turn, relaxation of vascular smooth muscle. 55 On the other hand, in perivascular nerve fibers, TRPV1 activation facilitates the release of calcitonin gene-related peptide (CGRP), which operates via cAMP-related signaling pathways to enhance TAC, protect vascular ECs and maintain the bioavailability of eNOS, thereby ensuring NO synthesis. This further contributes to neuroprotection and endothelium-dependent vasodilation. 56 Considering the potential of CGRP as a therapeutic target for various conditions, the interaction between CGRP and the TRPV1 channel presents a promising new pathway for PBM that deserves wider investigation.
An indirect activation mechanism of TRPV channels has also been observed during PBM irradiation with light wavelengths below 900 nm, such as 650 nm and 808 nm, mediated by the stimulation of CCO. CCO activation initiates a signaling cascade, transducing the signal from the mitochondria to the cytosol. This leads to cytosolic alkalinization, which triggers the opening of TRPV4 channels, resulting in Ca2+ influx (Figure 5(b) and (c)). 57
It should be emphasized that PBM-induced Ca2+ changes exhibit a similar dose-dependent pattern. According to Wang et al., using 980 nm light, the influx of Ca2+ peaked at a lower fluence (0.3 J/cm2), higher than other fluences tested (Figure 5(d) and (e)). 58 As laser intensity increased, neurons subjected to laser irradiation displayed an initial rise in intracellular Ca2+ levels, followed by a decline (Figure 5(f) and (g)). 59 In a physiological context, the extracellular concentration of Ca2+ markedly surpasses that in the intracellular environment, while excessive Ca2+ influx can trigger cell apoptosis. Currently, there is limited research to determine the precise threshold where calcium entry becomes beneficial or detrimental.
Systemic effects of PBM
Despite the widespread application of transcranial PBM, the therapeutic mechanisms underlying PBM on brain disorders are multifaceted and cannot be adequately elucidated solely through the direct photon absorption by brain tissue. While low-intensity light has shown capabilities of targeting pathologies in cerebral cortex, 60 its penetration into deep-brain structures may be limited due to the attenuation by extracranial tissues. 61 To address this issue, established research has employed remote PBM approaches, which deliver light to other sites of the body rather than directly to the head, and has reported neuroprotective benefits. This pertains to the systemic effects of PBM, providing an alternative explanation for the beneficial outcomes observed when utilizing PBM to treat diseases with injuries localized deep within the brain. 62 In this theory, light is absorbed by blood components, initiating a series of biological responses that can be conveyed to the brain through the circulatory system, thus eliciting neuroprotective and cognitive-enhancing effects. 62 Possible mechanisms of the systemic effects include release of NO, activation of NOS, induction of growth factor expression, modulation of inflammatory cytokines, and the stimulation of CCO in circulating cell-free mitochondria, either in the scalp or throughout the body.22,63,64 A fundamental supporting evidence for this viewpoint is the observation that unilateral irradiation can lead to accelerated wound healing on both sides of the body. 65 In the neurological field, there have been some advantageous outcomes. Ganeshan and colleagues demonstrated that 10-day exposure to 670 nm light targeting dorsum and hindlimbs efficiently mitigates 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridi (MPTP)-induced Parkinson’s disease (PD)-like neuropathology. RNA microarray analysis showed that remote PBM may enhance brain tissue resilience by upregulating cellular signaling, migration processes, oxidative stress response pathways, and modulating the blood-brain barrier (BBB). 66 These findings provide compelling evidence for remote PBM as a potential therapeutic strategy, with neuroprotective capabilities that can overcome penetration limitations. In this sense, the integration of transcranial and remote irradiation emerges as a promising treatment option that may yield superior therapeutic outcomes.
Physiological effects of PBM on cerebral vessels
Regulation of local cerebral blood flow (CBF)
A pioneering investigation employing single-cell spatial transcriptomics within the macaque cortex unveiled a predominant localization of vascular ECs and VSMCs in Layer 1, aligning with the cortical surface according to the six-layer laminar schema. 67 This discovery proclaims the existence of these widely distributed blood vessels before PBM light waves arrive in the cerebral gray matter, potentially prompting the influence of PBM on cerebral vessels. In humans, similarly, a complex vasculature comprising subarachnoid space vessels and cortical perforating arteries pervasively spans the cerebral cortex surface. The cerebral vasculature, composed of ECs sheathed by a basement membrane, arterioles and arteries enveloped by VSMCs, and pericytes surrounding the capillaries, can be influenced by PBM through various mechanisms.
The meticulous regulation of vessel diameter through NVC is acknowledged to be the foundation for neuronal function. However, the question of whether capillaries or arterioles constitute the specific core of vascular response remains a topic of debate.68,69 Theoretically, PBM could enhance ATP production in all cells capable of undergoing aerobic oxidative respiration, demonstrating the capacity to confer beneficial effects on both types of vascular cells.
On the one hand, PBM promotes vasodilation by enhancing ATP production in both ECs and VSMCs. Researchers have demonstrated significant increases in mitochondrial oxygen consumption (Figure 2(c)) and ATP synthesis (Figure 2(d)) in human ECs after exposure to 808-nm pulsed laser. 70 This ATP increase could enhance the release of vasodilatory factors in brain capillary ECs via an endothelium-dependent pathway, which assumes a crucial role in the regulation of NVC. 71 In VSMCs, PBM initiates vasodilation by activating Na+/K+ ATPase and inward rectifier potassium channel (Kir), leading to hyperpolarization of the cell membrane potential. In these processes, ATP is responsible for maintaining the ion gradient across cell membrane and the rapid activation of Kir. Upon hyperpolarization, L-type calcium channels close, thus reducing Ca2+ influx and inhibiting vascular smooth muscle contraction. Moreover, relaxation of VSMCs is further facilitated by the efflux of K+ through calcium-activated potassium channels, which enhances membrane hyperpolarization and induces vascular muscle relaxation. 72 The ATP-generating effects of PBM highlight its potential in regulating local CBF.
On the other hand, PBM induces vasodilation through the generation of bioactive molecules. Among these, photons trigger an increase in intracellular Ca2+ levels via the activation of TRPV channels, 69 which subsequently prompts the release of arachidonic acid metabolites and prostaglandin E2 in both neurons and astrocytes. This cascade of events culminates in vasodilation, facilitated by the activation of EP4 prostanoid receptors on VSMCs and A2a receptors on pericytes. 73 Additionally, NO, as previously mentioned, plays a significant role that interacts with cGMP and Ca2+-mediated NOS activation following PBM irradiation, resulting in vessel vasodilation,74,75 as depicted in Figure 3(a).
Intracerebral hypoperfusion is typically among the initial manifestations of most cerebral diseases. As a promising alternative, PBM therapy has demonstrated its efficacy in augmenting CBF in specific brain regions, alleviating cognitive impairments associated with these areas. In a cohort of individuals with mild cognitive impairments, localized PBM application to the vicinity of the vertebral and internal carotid arteries resulted in a marked CBF enhancement within the prefrontal cortex, anterior cingulate cortex, and occipital lateral cortex, resulting in significant improvements in cognitive performance. 76 Otherwise, A notable 20% augmentation in CBF was observed after 850-nm PBM administered to the forehead of a patient in a persistent vegetative state caused by severe head injury, 77 implying the benefits of PBM on brain circulation.
Stimulation of angiogenesis
Promoting angiogenesis has emerged as a promising therapeutic strategy for brain injuries, particularly those associated with hypoxia and hypoperfusion. 78 The hypoxia-inducible factor-1alpha (HIF-1α) is considered a potential mediator in this context, 79 with pericytes being identified as the fundamental responsive cells. 80
In the adult brain, angiogenesis typically occurs under pathological conditions in the presence of hypoxia and inflammation. 78 Hypoxia-induced cellular responses are primarily mediated by HIF-1α, which degrades in normoxia while attaining stability in hypoxia. 81 Upon activation, HIF-1α moves to the nucleus and regulates the transcription of various downstream genes, encompassing vascular endothelial growth factor (VEGF) and its receptor, erythropoietin receptor, phosphoglycerate kinase genes, and glucose transporter-1. 82 Ultimately, erythropoiesis, angiogenesis, energy metabolism, and cell proliferation are promoted, all of which enhance tolerance to hypoxia in the body. Conversely, a deficiency in HIF-1α can cause brain tissue damage, as demonstrated in rats with chronic cerebral hypoperfusion: knockdown of HIF-1α gene expression for four weeks significantly exacerbated learning and memory deficits, oxidative damage, and decreased angiogenesis in the hippocampus. 83 Therefore, HIF-1α is a potential target for treating neuronal hypoxia and inflammation.
Pericytes exert profound regulatory influence over cerebral capillary diameter, maintaining structural and functional integrity of the neurovascular unit. 84 They wrap around capillary ECs and communicate with them, maintaining the integrity of the BBB and functioning as the primary modulator of capillary diameter in response to neuronal activity. 85 During hypoxia or hypoperfusion, pericytes detach from the capillary wall and secret angiogenic factors, promoting the sprouting of new vessels from ECs. Once the new vessel forms, pericytes reattach to the endothelial wall, facilitating the vessel maturation. 86 On the contrary, pericyte dysfunction will lead to blood flow reduction, capillary destruction, and BBB breakdown, ultimately resulting in neurovascular uncoupling. 87 The absence of pericytes has been demonstrated to associate with neurodegeneration, delayed neuroinflammatory response, and deficiencies in learning and memory. 88
Therefore, regulating the expression of HIF-1α and the activity of pericytes may offer promising therapeutic targets for PBM therapy. 84 When used on pericyte-labeled transgenic mice with back wounds, PBM significantly promoted the mobilization of type-2 pericytes, which are important for blood flow. 89 Cury et al. elucidated the pro-angiogenesis effects of PBM in rat skin flaps at wavelengths of 660 nm and 780 nm, characterized by an increase in HIF-1α and VEGF expression. 79 Furthermore, studies show the impact of PBM on HIF-1α signaling is dose-dependent, exhibiting a biphasic response. 79 However, the interaction between PBM and cerebral pericytes is not well understood, as studies have dominantly concentrated on the peripheral rather than the central neurovascular system. More laboratory investigations are needed to clarify the role of PBM in central neurovascular angiogenesis.
Preservation of red blood cell structure and function
PBM has been shown to positively affect RBCs, offering protection that extends to the surrounding tissues. Given the lack of mitochondria in RBCs, several unique mechanisms have been proposed to mediate the effects of PBM. Porphyrins, which exhibit absorption peaks centered at 900 nm, have been suggested as the principal chromophores to trigger similar downstream pathways within RBCs, where hemoglobin constitutes a significant portion of the intracellular content. 90 When light is absorbed by the heme molecule in hemoglobin, it triggers electron excitation, fortifying the iron ion bond within the porphyrin ring system and thus enhancing the oxygen transport capacity of hemoglobin. 91 A pilot study using an 850-nm wavelength laser diode (LED) on the forearms of healthy individuals demonstrated a significant increase in oxyhemoglobin concentration, as measured by NIR spectroscopy. This finding indicates an acute improvement in local oxygen availability following PBM irradiation, 92 underscoring the role of PBM in promoting hemoglobin function.
Moreover, PBM has been recognized for its role in safeguarding the homeostasis and functionality of RBCs via modulating the key proteins on the RBC membrane, which are essential for the overall integrity and performance of these cells. For example, low-power light irradiation activates aquaporin-1, regulating dehydration and rehydration processes, thereby improving RBC deformability. 93 Additionally, PBM enhances the activity of ion channels, especially the Na+/K+-ATPase, possibly via attenuating detrimental factors such as oxidative stress, and the concurrent strengthening of intrinsic antioxidant defenses. 94 An in vitro study demonstrated that blood exposed to NIR light during hemodialysis maintained lower oxidation levels for 30 minutes compared to untreated blood, where RBC damage increased. 95 This aids in the restoration of normal ionic transmembrane transport and RBC surface charge, which in turn reduces erythrocyte aggregation and preserves RBC membrane stability. 96 Deryugina et al. demonstrated that 890-nm PBM exerts a protective effect on RBCs by reducing adverse morphological changes induced by hyperadrenalinemia. This is manifested by reduced pathological erythrocyte forms, restored normal biconcave shape, increased electrophoretic mobility, enhanced osmotic resistance, and normalization of erythrocyte numbers in peripheral blood. 97 Collectively, these effects improve blood rheology and microcirculation.
Effects of PBM on neural cells
Neuronal bioenergetic dysfunctions
The nervous system is characterized by its high reliance on mitochondria for normal functioning. 98 Beyond the role of energy provision, mitochondria modulate signaling pathways vital for calcium homeostasis, cell proliferation, inflammation, and oxidative balance, all of which are crucial for electrophysiological activities, synaptic plasticity, and neuroprotection.
Dysfunction of mitochondria has been well-documented in various brain disorders. 32 Therefore, employing PBM as a therapeutic strategy for these situations is convincing, as both the disease processes and the interventive mechanisms converge on the shared theme of mitochondrial bioenergetics. PBM therapy beneficially affects all components of NVC, including cerebral vessels and neurons, rescuing their bioenergetic dysfunctions. The beneficial effects are attributed to several mechanisms, encompassing enhanced ATP production, improved brain perfusion and oxygen supply. In this regard, modulation of mitochondrial function by PBM has been documented in both in vitro and in vivo models, as well as in human studies. In cultures of primary cortical neurons, PBM efficiently increased mitochondrial ATP synthesis and suppressed neuronal bioenergetic dysfunction induced by oxidative stress and inflammatory changes. 49 Salehpour et al. noticed better cognitive performance in aging mice given D-galactose, concomitant with elevated CCO expression, heightened mitochondrial activity (Figure 2(e)), and higher ATP levels (Figure 2(f)) after PBM therapy. 99 Similarly, transcranial NIR laser therapy attenuated the cortical ATP decrease caused by embolization in rabbits. When combined with tissue plasminogen activator, the ATP content increased by an additional 22% compared to the control group 100 (Figure 2(g)). After using a continuous wave laser (1064 nm) to stimulate young and aged healthy adults, a discernible surge in oxidized CCO levels was observed (Figure 2(h)), accompanied by a significant elevation in oxygenated hemoglobin (Figure 2(i)) and a decline in deoxygenated hemoglobin (Figure 2(j)), suggesting that transcranial PBM may be used to counteract the age-related reduction in mitochondrial respiration. 101 These findings jointly underscore the therapeutic implications of PBM in modulating neuronal bioenergetics and the associated functional outcomes.
Oxidative stress
A large body of studies have demonstrated increased oxidative stress in brain disorders, especially age-related neurodegeneration. 102 Mitochondrial ETC is the primary source of ROS production. When the MMP deviates from its normative state due to mitochondrial dysfunction or oxidative stress-related disorders, excessive ROS will be generated, because a compromised MMP impairs the efficiency of the ETC, causing electrons to leak and react with oxygen molecules. 103 Upon light exposure, the PBM-induced CCO activity alteration facilitates the restoration of MMP, thereby culminating in the alleviation of oxidative stress. 30
Drawing from the aforementioned evidence, oxidative stress is a compelling target for PBM therapy. In instances of ischemia-reperfusion injury, ischemic exposure enhances ETC complex activity, resulting in excessive ROS generation. After administering a combination of 750 nm and 950 nm light to a rat model of acute stroke, the reperfusion-induced brain injury was significantly reduced, 104 highlighting the potential of PBM in mitigating the detrimental effects of this process. Moreover, PBM has shown increasing promise in improving pathological conditions triggered by oxidative stress. Specifically, in both cell line and organotypic slice cultures of mouse hippocampus, PBM with 660 nm light exerted a profound inhibition of the oxidative stress induced by H2O2, suppressing cell death and promoting the activity of antioxidant enzymes. 105
Elimination of neurotoxicity
Excitotoxicity, a pivotal concept in neuroscience research, is closely associated with a variety of neurological diseases. This phenomenon is primarily triggered by an overload of intracellular Ca2+, leading to neuronal damage. 106 Indeed, Ca2+ acts as a vital secondary messenger in neurons, regulating processes such as neurotransmitter release, synaptic plasticity, and activity-dependent transcription. 107 However, excessive intracellular Ca2+ induces the opening of mitochondrial permeability transition pores, which perturb mitochondrial membrane permeability, leading to mitochondrial dysfunction and ultimately, neuronal death. 108 Ca2+ overload is often ascribed to hyperactivation of N-methyl-D-aspartate receptors (NMDARs), 106 which facilitates the influx of Ca2+ into cells and its release from internal stores such as the endoplasmic reticulum. 109 Emerging evidence suggests that PBM therapy holds promise in mitigating deleterious effects stemming from Ca2+ overload by fostering an equilibrium between intra- and extracellular Ca2+ levels within neurons. The utilization of 810-nm laser light has been shown to lessen Ca2+ overload triggered by activated NMDARs and restore normal MMP, thereby reducing oxidative stress and supporting cell survival. 49 More intriguingly, PBM can counteract the adverse consequences of neurotoxicity by improving ATP synthesis, reducing oxidative stress, and promoting NO generation, collectively exerting its neuroprotective properties.
Inhibition of neuroinflammation
Neuroinflammation represents a key pathophysiological feature observed in brain disorders and cognitive abnormalities. In the CNS, innate immune cells, particularly astrocytes and microglia, assume a central function in the neuroinflammatory response, with related cytotoxic and pro-inflammatory factors including IL-1β, IL-6, iNOS, TNF, and interferon-γ. 110 PBM therapy has demonstrated advantageous effects in mitigating neuroinflammation via the regulation of various cytokines. In spinal cord injury mice, continuous exposure to 808-nm laser was shown to ameliorate motor function impairments and mitigate the neurotoxic impact of proinflammatory macrophages. The successive experiments on neuronal cultures demonstrated that PBM prohibited the secretion of certain inflammatory factors, including IL-1α, IL-6, iNOS, and cyclooxygenase-2, by suppressing the inflammation-related Notch1-HIF-1α/NF-κB signaling pathway. 111 Furthermore, studies in the aging brain have shown that laser treatment at 660 nm can ameliorate inflammatory responses. In the cerebral cortex, PBM increased IL-1α and decreased IL-5 levels, and in the hippocampus, PBM increases IL-1α while reducing levels of IL-5, IL-18, and fractalkine. 112 In a depressive mice model submitted to sub-chronic restraint stress, PBM utilizing an 810-nm laser at a dosage of 33.3 J/cm2 effectively reduced the levels of NF-κB, p38, and c-Jun amino-terminal kinases, and normalized the elevated levels of TNF-α. 113 Additionally, light at wavelengths of 808 and 905 nm was proved to block the NF-κB pathway, significantly attenuating the inflammatory response induced by oxygen and glucose deprivation in the hippocampus. 114
It is noteworthy to acknowledge a paradox wherein certain studies have indicated that PBM may activate NF-κB pathway via ROS. 115 The role of NF-κB in PBM’s anti-inflammatory effects is complex due to the interplay between the wavelength and dosage of the applied light. 30 In a multifaceted manner, a series of modulatory targets participate in the intricate NF-κB signaling pathways.
Neurogenesis and synaptogenesis
Loss of synapses and cell apoptosis constitute pivotal characteristics across a wide spectrum of neurological diseases. PBM has proved to promote neurogenesis and synaptogenesis under various conditions of neuronal pathology. A key mediator in these processes is brain-derived neurotrophic factor (BDNF), 116 a protein that supports various aspects of neuron health, including neurogenesis, synaptogenesis, neuron survival, and synaptic plasticity. Imbalance of BDNF is implicated in numerous neurological disorders. 117 In both amyloid β (Aβ)-treated and APP/PS1 mouse hippocampal neurons, PBM (632.8 nm) rescued the neuronal loss and dendritic atrophy by enhancing BDNF expression through activation of extracellular signal-regulated kinase (ERK)/CREB pathway. This then activates Rho-family GTPase Rac1 and increases postsynaptic density 95 (PSD-95) expression, thereby improving dendritic length, branching, and spine density. 118 Besides, a study of neurotrauma revealed that PBM at 808 nm notably upregulated the mRNA encoding BDNF within the trauma region and boosted BDNF expression in the hippocampus, while downregulating the pro-apoptotic protein caspase-3, 119 thus safeguarding neurons from apoptosis.
Equally important, PBM has been demonstrated to promote the proliferation and differentiation of neural stem cells (NSCs) and neuroprogenitor cells.120,121 As demonstrated by Wu et al., 635-nm PBM stimulates the differentiation of hippocampal APP/PS1 NSCs into neurons through activating TGF-β1/Smad signaling pathways, both in vivo and in vitro. The newborn neurons express key proteins associated with neuronal function, such as microtubule-associated protein-2, α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid receptors (AMPARs), and PSD-95, suggesting their contribution to the improvement of spatial learning and memory in APP/PS1 mice. 122 Otherwise, Yang and colleagues investigated these protective effects in stroke rat model, where PBM stimulated neuroprogenitor cells and modulated the inflammatory microenvironment, thereby enhancing neurogenesis and synaptogenesis, and significantly reducing infarct volume. 123 These findings highlight the role of PBM in neural repair and synapse formation, offering novel possibilities for therapeutic interventions in neural regeneration.
Strengthening neuronal connectivity
There is a hypothesis that PBM stimulation of particular sites has the potential to augment or re-establish neuronal connectivity across diverse intrinsic brain networks, which undertakes critical responsibilities for modulating the higher-order cognitive functions encompassing learning, memory, and emotional process. 124 In this regard, a corpus of human study has been conducted. Urquhart and colleagues reported that PBM using NIR light at a wavelength of 1064 nm delivered to the human head can facilitate the efficiency and reorganization of the brain networks, which is manifested as enhanced information processing speed and functional connectivity within the frontal-parietal network. 125 Graph theory analysis unveiled that PBM considerably improved both global and local network efficiency, shortening the path length of functional connectivity, which ensured more efficient functional integration and communication of small-world networks. 125 Dmochowski et al. utilized functional magnetic resonance imaging (fMRI) to assess the BOLD signal in 20 healthy individuals receiving right frontal pole stimulation with a NIR laser. A significant 15% increase in functional connectivity was revealed during stimulation, with approximately 25% of these augmentations exhibiting statistical significance. This connectivity boost was widespread across the brain, but was more pronounced in the stimulated hemisphere compared to the contralateral side. 126 In a randomized controlled trial conducted on patients with dementia, discernible enhancements in connectivity linking the posterior cingulate cortex and lateral parietal nodes, which are integral constituents of the default-mode network, were ascertained within the cohort receiving PBM. 127
Furthermore, considering the biphasic dose response of NIR light, the augmentation of functional connectivity by different doses of PBM has garnered great attention for investigation. A clinical study employing both transcranial (870 nm) and intranasal (810 nm) PBM systems at two different doses found that only the higher dose (52 J/cm2 transcranially and 18 J/cm2 intranasally) significantly enhanced resting state functional connectivity. This was observed between the left superior frontal gyrus and the left planum temporale, as well as the left inferior frontal gyrus, which are highly associated with working memory, spatial functioning, cognitive control, and executive function. 128 However, functional connection in the lower-dose group (26 J/cm2 transcranially and 9 J/cm2 intranasally) did not witness evident improvement. 129
Overall, to achieve a more precise delineation of the effects of PBM on functional connectivity, the integration of PBM with multimodal functional imaging techniques, such as fMRI and functional NIR spectroscopy, emerges as a promising trajectory for future research endeavors. Given the intricacy of intrinsic brain networks, a rigorous experimental design with diverse analysis methodology is essential to unravel their underlying mechanisms.
Combined PBM effects on neurovascular coupling
Based on previous statements, PBM exerts broad influences on NVC by interacting with CCO and ion channels within cells of the neurovascular unit, initiating a cascade of primary cellular responses, as outlined in Table 1. Subsequently, these initial effects elicit secondary reactions among the neurovascular components, which are detailed in Table 1 and illustrated in Figure 6.
Effects of photobiomodulation (PBM) on cellular molecules and neurovascular unit components.

CCO: cytochrome c oxidase; MMP: matrix metalloproteinase; ATP: adenosine triphosphate; NO: nitric oxide; sGC: soluble guanylate cyclase; cGMP: cyclic guanosine monophosphate; PKG: protein kinase G; PDE: phosphodiesterase; ROS: reactive oxygen species; CREB: cAMP response element binding protein; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; TRPV: transient receptor potential vanilloid; HIF-1α: hypoxia-inducible factor-1 alpha; CNS: Central Nervous System; RBCs: red blood cells; NMDARs: N-methyl-D-aspartate receptors; BDNF: brain-derived neurotrophic factor; ERK: extracellular signal-regulated kinase; NSCs: neural stem cells; TrkB: tropomyosin-related kinase B; VSMCs: vascular smooth muscle cells; BBB: blood-brain barrier; CBF: cerebral blood flow.
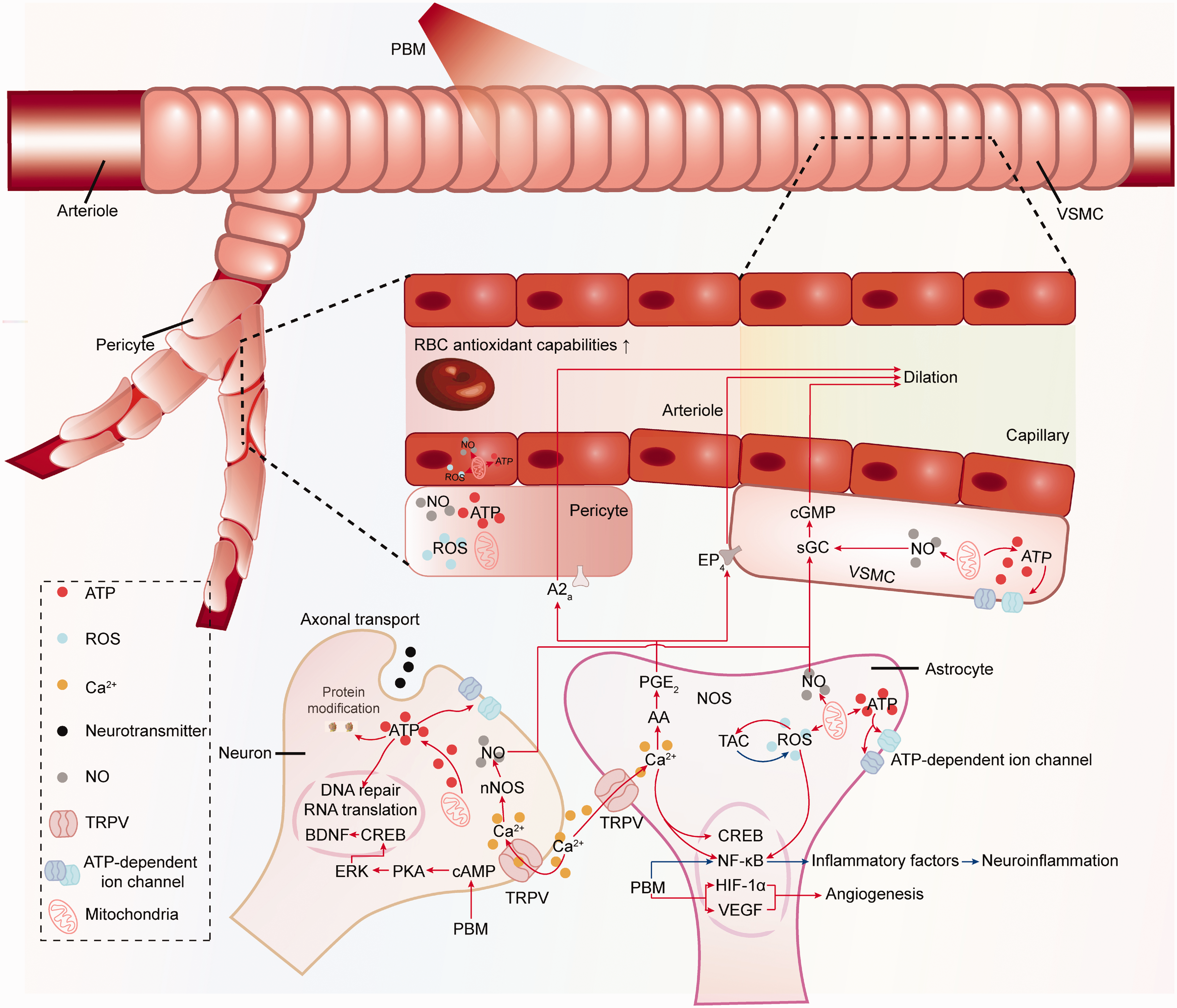
Diverse cellular responses to PBM within the neurovascular unit. At the top of the diagram lies an arterial vessel enveloped by VSMCs. Subsequently, the arteriole bifurcates into capillaries, where pericytes provide coverage. The lower portion of the diagram delineates additional pivotal cell types orchestrating NVC mechanisms. Further elucidation within the diagram expounds upon the impact of PBM on signaling pathways across diverse components of the neurovascular unit, as described in this review. These concerted processes synergistically enhance CBF, bolster neuronal survival, and facilitate neurotransmitter transmission, ultimately ameliorating NVC functionality. RBC, red blood cell; TRPV, transient receptor potential vanilloid; cAMP, cyclic adenosine monophosphate; PKA, protein kinase A; ERK, extracellular signal-regulated kinase; CREB, cAMP response element-binding protein; BDNF, brain-derived neurotrophic factor; AA, arachidonic acid; PGE2, prostaglandin E2; EP4, prostaglandin E2 receptor subtype 4; A2a, adenosine A2a receptor; sGC, soluble guanylate cyclase; cGMP, cyclic guanosine monophosphate.
Application
To date, PBM has shown promising outcomes in a range of brain disorders, including neurodegenerative diseases, neuropsychiatric disorders, neurodevelopmental disorders, and traumatic events, all of which are highly associated with structural and functional disruptions of NVC. Here, we present an overview of the up-to-date application of PBM in these areas.
Neurodegenerative diseases
Alzheimer’s disease (AD)
AD is a neurodegenerative disorder marked by significant memory and cognitive impairments, predominantly stemming from the pathological accumulation of Aβ plaques and hyperphosphorylated tau neurofibrillary tangles. These protein aggregates induce neurotoxicity, leading to mitochondrial dysfunction, oxidative stress, neuroinflammation, neuronal death, and brain atrophy, which collectively exacerbate NVC dysfunction. 130 The multifaceted pathogenic mechanisms of AD underscore the need for a holistic therapeutic strategy. PBM therapy, through its modulatory mechanisms, offers a synergistic approach to addressing AD pathology.
Existing literature documents that PBM has the potential to clear Aβ deposits either in direct or indirect ways. Molecular dynamics simulations have illustrated that NIR laser light at a specific pulse intensity can directly break the hydrogen bonds between Aβ peptides. 131 Furthermore, the enhancement of meningeal lymphatic clearance of Aβ through PBM has garnered significant interest, in which 808 nm and 1267 nm PBM has been shown to decrease Aβ aggregation in diverse brain regions.132,133 Otherwise, 1070-nm PBM at 10 Hz promotes Aβ clearance by activating microglia and facilitating their recruitment, which also promotes angiogenesis. 20
As detailed in Table 2, PBM therapy mitigates the Aβ-induced pathological hallmarks in AD. PBM bolsters the oxidative respiration chain and ATP production in AD, 12 in a dose-dependent manner. 134 PBM also shows effects in treating and preventing AD by promoting neuroplasticity via multiple pathways.118,122 Additionally, PBM exhibits anti-inflammatory and antioxidant properties,12,134 effectively attenuating cognitive and memory impairment induced by Aβ peptides.20,135 Clinical trials have already reported positive outcomes, including reductions in negative emotions and improvements in cognitive function,136,137 particularly evident in memory and attention.138,139
Representative studies on the application of PBM therapy in neurodegenerative diseases.
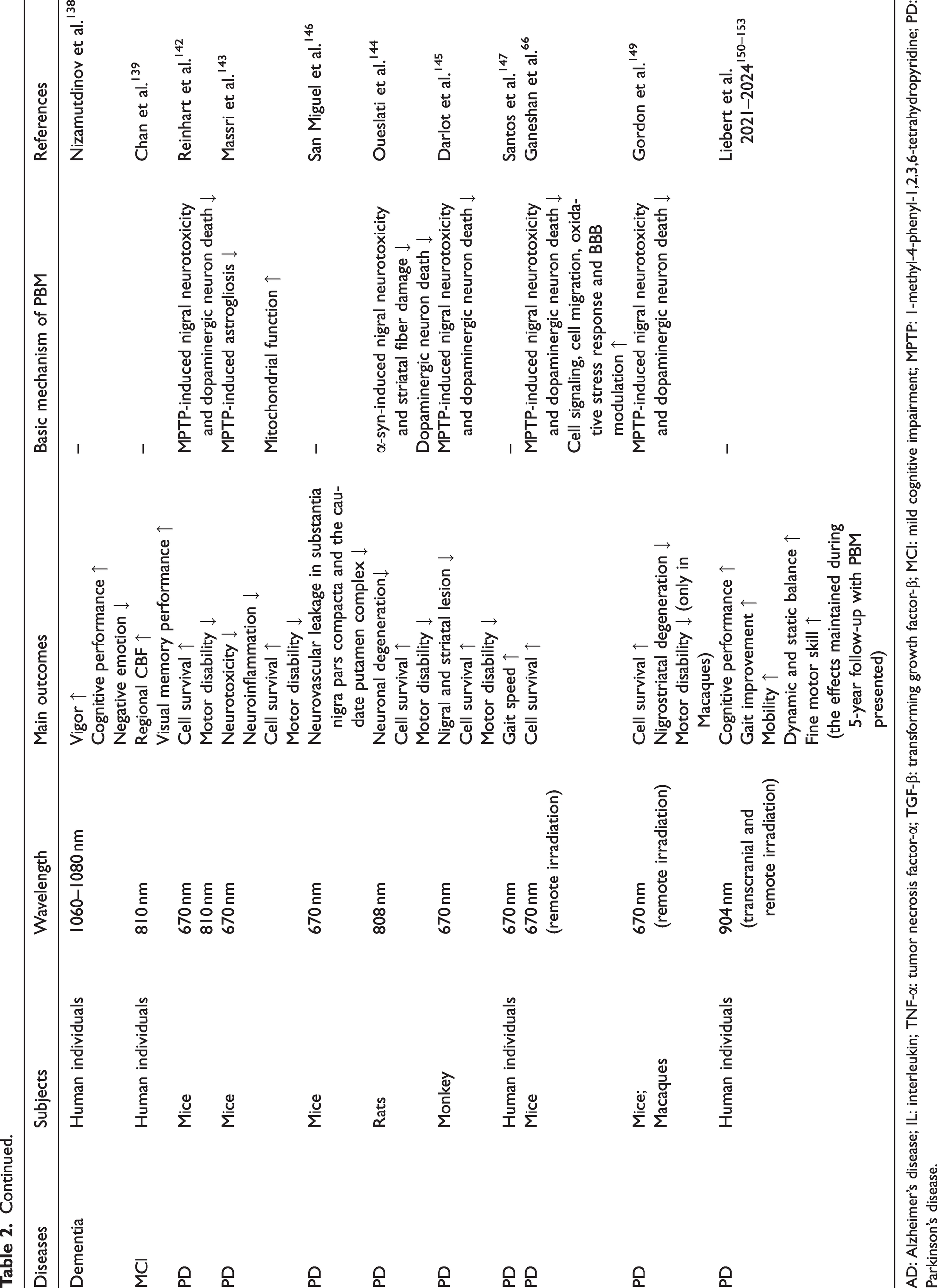
AD: Alzheimer’s disease; IL: interleukin; TNF-α: tumor necrosis factor-α; TGF-β: transforming growth factor-β; MCI: mild cognitive impairment; MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; PD: Parkinson’s disease.
Parkinson’s disease (PD)
PD featuring both motor and non-motor symptoms, represents the second most prevalent degenerative disease. 140 The pathogenesis entails the progressive degeneration of dopaminergic neurons initiating in substantia nigra pars compacta (SNc) of the midbrain and propagating to other cerebral regions, including the cortex. 141 In fact, the precise mechanisms of PBM therapy for PD are not completely clear. The possible mechanisms, as outlined in Table 2, mainly involve rescuing mitochondrial function deficit, protecting dopaminergic neurons from apoptosis, modulating cell signaling, cell migration, oxidative stress response, neurovascular and BBB integrity.142 –146
While limited in number, existing clinical studies have illuminated PBM’s role in PD treatment (Table 2). A double-blinded randomized controlled trial reported gait improvements after 9 weeks of PBM intervention (670 nm) in PD patients. 147 However, one challenge facing the application of PBM in PD is the restricted penetration depth, affecting treatment potency. Recent studies have explored intranasal, transoral, and remote PBM therapy as a promising alternative, 148 demonstrating encouraging efficacy across various irradiating modality.66,149 Notably, Liebert et al. employed transcranial, intranasal, cervical and abdominal PBM interventions in PD subjects, and reported improvements in a range of motor and cognitive outcomes. These gains were sustained over a five-year period, provided that PBM therapy was consistently administered.150 –153 These results highlight the promising application of combining multiple irradiation routes to treat PD.
Psychiatric disorders
Depression
Depression, a highly prevalent psychiatric disorder, is a leading cause of mental health-related disability worldwide. 154 Unlike other neurological conditions, it often arises from non-traumatic factors such as psychological stress and social defeat, which can trigger oxidative stress, inflammation, mitochondrial dysfunction, BBB disruption, and neurovascular function deficits. 155 Despite the documented benefits of antidepressants, many individuals with major depressive disorder (MDD) show poor responses or experience side effects. 156 So far, PBM therapy has emerged as a useful approach for treating varying degrees of depression, with studies in both animals and humans indicating its efficacy and tolerability, even in cases of MDD (Table 3).113,157 –162
Primary studies on the application of PBM therapy in psychiatric disorders and neurodevelopmental disorders.
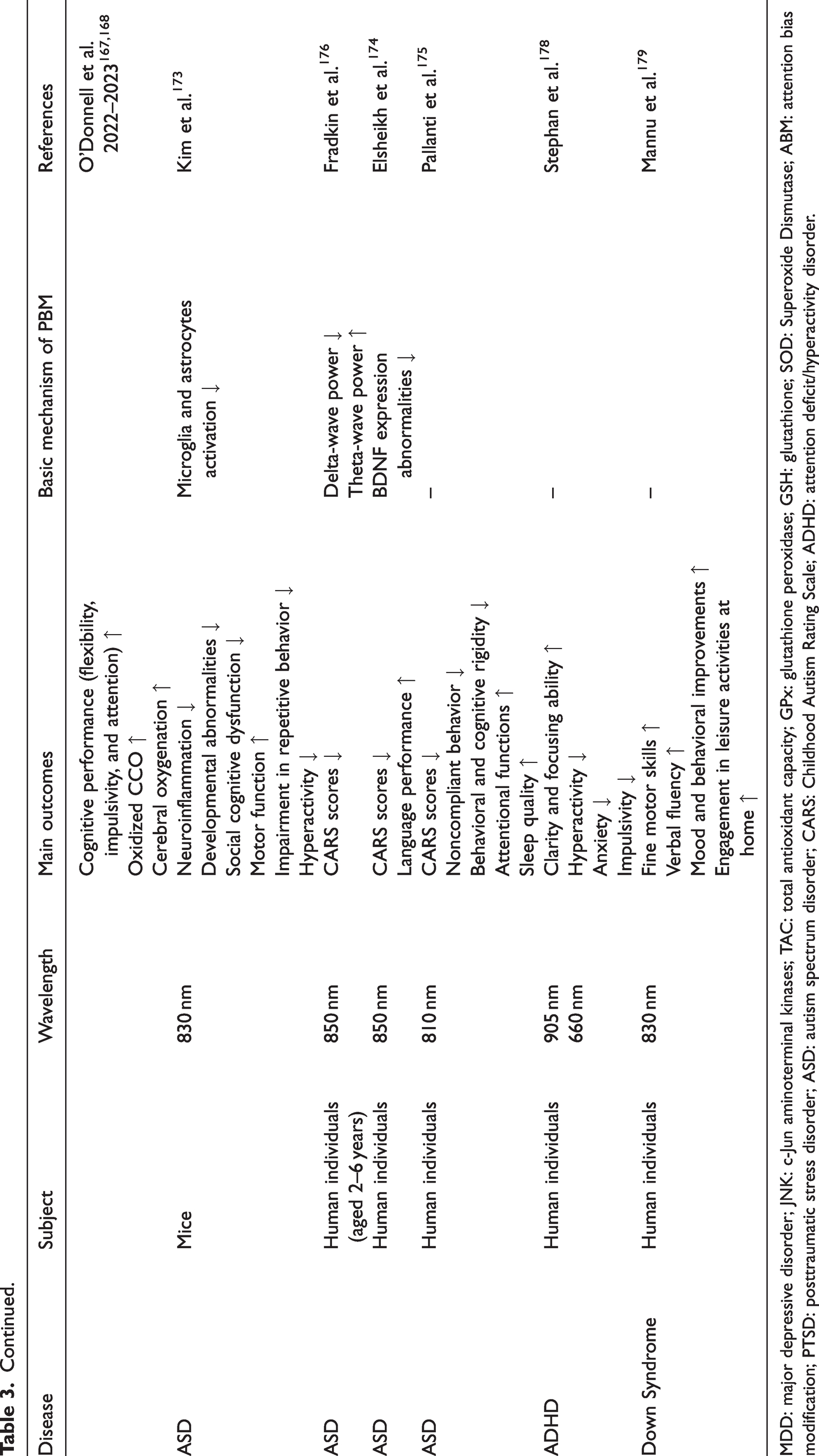
MDD: major depressive disorder; JNK: c-Jun aminoterminal kinases; TAC: total antioxidant capacity; GPx: glutathione peroxidase; GSH: glutathione; SOD: Superoxide Dismutase; ABM: attention bias modification; PTSD: posttraumatic stress disorder; ASD: autism spectrum disorder; CARS: Childhood Autism Rating Scale; ADHD: attention deficit/hyperactivity disorder.
Intriguingly, PBM therapy has captivating preventative properties, reducing the onset of depression in subjects facing significant mental stress. Utilization of 808-nm PBM on adult rats with early life adversity effectively alleviated the stress-induced pathologies like oligodendrocyte dysfunction and oxidative damage, and improved cognitive impairment and depressive behaviors. 11
Despite these encouraging outcomes, the eligible dosage of PBM therapy for depressive disorders remains inconclusive. 163 Future clinical research should endeavor to expand the sample size and prolong follow-up periods to ascertain the optimal therapeutic regimen.
Other psychiatric disorders
The scientific community has embarked upon an exploration of PBM therapy in addressing diverse psychiatric disorders, such as chronic schizophrenia, 164 posttraumatic stress disorder (PTSD)165,166 and bipolar disorder167,168 (Table 3). These compelling findings imply that PBM therapy represents a viable and potentially transformative therapeutic modality for addressing the intricate realm of mental health disorders.
Neurodevelopmental disorders
Autism spectrum disorder (ASD)
ASD is a common neurodevelopmental disorder characterized by sensory anomalies, social communication difficulties, and repetitive behaviors. 169 Typically onset in childhood, ASD is intractable and imposes substantial burdens on families and society. 170 Current treatments, including behavioral therapies and medications, show limited efficacy and may cause side effects. 169 Pathophysiological studies have connected ASD to mitochondrial dysfunction, oxidative stress, neuroinflammation, and aberrant brain connectivity.171,172 PBM has shown promise in addressing these issues (Table 3). Preclinical studies suggest PBM improves behavior and cognitive function by reducing neuroinflammation, 173 while several clinical trials report symptom relief and cognitive benefits with good tolerability.174 –176 However, further large-scale and rigorous research is essential to refine PBM protocols for effective ASD management.
Attention-deficit/hyperactivity disorder (ADHD)
ADHD is a neurodevelopmental disorder with symptoms of inattention, hyperactivity and impulsivity, which has been linked to mitochondrial dysfunction, increased ROS, and abnormal functional connectivity.16,177 While no definitive cure exists, PBM is recognized as a novel non-pharmacological therapy with potential to alleviate ADHD symptoms 178 (Table 3). However, current evidence is limited, highlighting the need for further preclinical and sham-controlled studies to confirm the regulatory mechanisms and feasibility of PBM in ADHD.
Other developmental disorders
PBM has already attracted attention in the treatment of other developmental conditions, such as Down syndrome, 179 demonstrating symptom relief and minimal side effects (Table 3). Future works should also investigate the applicability of PBM to other neurodevelopmental disorders, such as specific learning disorder and Tourette syndrome.
Brain traumatic events
Traumatic brain injury (TBI)
TBI stands as an unignorable global menace, contributing to high mortality rates and lifelong disability. The initial trauma triggers complex molecular and cellular responses, causing BBB dysfunction and neurovascular uncoupling, 180 which consequently result in the development of neuropsychological comorbidities, including dementia, depression, and neurodegeneration.85,181 Given its regulatory potential, PBM exhibits great promise as a cost-effective therapeutic strategy in TBI-related issues.
PBM therapy has been shown to elicit favorable outcomes in both acute and chronic TBI. In acute TBI, it enhances spatial learning and memory, diminishes lesion volume, which was attributed to abated neuroinflammation and promoted neurogenesis.116,182 In chronic TBI, significant improvement in neuropsychological scores and regional CBF have been reported, suggesting promising therapeutic effects of PBM on cognitive function and brain perfusion years after TBI (Figure 3(D)). 39
In clinical practice, imaging techniques have revealed the beneficial effects of PBM in mitigating structural injury. In a pilot randomized clinical trial involving 68 patients with moderate TBI, the administration of three 20-minute sessions of PBM therapy within 72 hours post-injury demonstrated structural repair on MRI, resulting in improved test scores and clinical outcomes. 183 Moreover, PBM has been shown to regulate neuronal interconnectivity and improve cognitive performances in TBI patients, further supporting its ability to positively influence neuronal circuits,184,185 as outlined in Table 4.
Summary of the representative studies on the application of PBM therapy in traumatic brain events.

TBI: traumatic brain injury; MRI: magnetic resonance imaging; NEST: NeuroThera Effectiveness and Safety Trials; eNOS: endothelial nitric oxide synthase; OGD: oxygen-glucose deprivation.
However, the current research on PBM treatment for TBI lacks high-quality design. More well-structured clinical studies are needed to determine the optimal therapeutic parameters and treatment schedules for various TBI conditions.
Stroke
Stroke has emerged as a formidable global challenge, ranging as the second leading cause of mortality and permanent disability. 181 The pathological changes of stroke provide diverse targets for PBM therapy.
As summarized in Table 4, PBM has exhibited neuroprotective and neuroreparative capabilities for ischemic stroke, significantly diminishing infarct size, neuronal cell death, and behavioral deficits, with the mechanisms potentially involving attenuation of neuroinflammation, promotion of energy production, as well as stimulation of neurogenesis and synaptogenesis, among others.15,123 Importantly, recent evidence has confirmed PBM’s efficacy in modulating neurovascular functions, with both NIR-I and NIR-II window light demonstrated to activate eNOS and enhance NO bioavailability, thus improving CBF and reducing neuronal damage.186,187 These findings suggest that PBM may offer a promising therapeutic option for the cerebrovascular diseases, especially ischemic stroke.
So far, three major clinical studies, known as the “NeuroThera Effectiveness and Safety Trials” (NEST), have been executed to assess the effectiveness of 810-nm laser therapy for stroke. While the first two trials reported superior outcomes in laser-treated groups compared to controls, 188 the third trial was prematurely terminated during interim analysis and showed no significant differences, likely due to limitations in light penetration depth and variability in stroke locations. 189 Importantly, none of these studies raised safety concerns regarding the use of this technique on stroke. More recently, a case series involving chronic stroke patients further underscored the therapeutic potential of PBM in stroke, highlighting significant improvements in naming ability and functional connectivity within intrinsic neural networks following red/NIR LED therapy. 190 To fully harness the potential of this innovative technique, future research should prioritize optimizing the delivery of PBM to specific stroke-affected brain areas.
Conclusion
In conclusion, PBM holds promise in modulating NVC through a multifaceted interplay of pathways, offering potential therapeutic avenues for a wide range of neurological and psychiatric disorders. Various PBM therapy approaches can serve as effective noninvasive modalities for neuronal disorders. Considering its portability, low cost, and absence of side effects, the salutary and economic influence of PBM in the treatment of brain disorders could be substantial. However, given the intricate interactions between various cell types and signaling pathways involved in NVC, significant knowledge gaps persist in our understanding regarding the precise mechanistic underpinnings through which PBM exerts its modulatory effects on distinct brain disorders. Additionally, it is imperative to take the biphasic pattern of PBM actions in mind. Future investigations are warranted to ascertain the optimal selection of target sites, dosage protocols, and potential synergistic therapies combined with novel techniques.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation for Young Scholars of China (grant no. 82302199), Key Research and Development Project of Sichuan Science and Technology Plan Projects (grant no. 2023YFG0322), the Young Fund of Sichuan Natural Science Foundation (grant no. 24NSFSC8075), Postdoctor Research Fund of West China Hospital, Sichuan University (grant no. 2024HXBH181) and the National Science Foundation of China (grant no. 82371977, 82071940, 81671702).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
