Abstract
The differentiation of adipose-derived stem cells (ADSCs) into tendon cells is a key process in tissue engineering and regenerative medicine. The Wnt signaling pathway plays a key role in regulating cell fate and tissue-regeneration decisions, making it a promising target for improving tendon differentiation. Photobiomodulation (PBM) is a non-invasive therapeutic approach that has been shown to modulate cellular processes, including stem cell differentiation. The aim of this review is to provide an understanding of the effects of PBM and Wnt signaling on ADSC differentiation. The complexities of interactions between PBM and dynamic Wnt pathway exist in different ways during the differentiation of ADSCs into tendon cells. The results highlight the potential therapeutic application of PBM in promoting tendon healing and regeneration. This review explores the clinical importance of PBM-mediated Wnt signaling regulation in tendon injuries. The results of this review will provide valuable information for the rational design of therapeutic strategies to enhance tendon differentiation and improve clinical outcomes and will also contribute to increasing knowledge of the synergistic relationship between PBMs, Wnt signaling pathways, and stem cell differentiation.

Introduction
Tendon injuries frequently result in persistent discomfort, reduced mobility, and diminished quality of life owing to the tissue’s poor regeneration ability. Tendons, mostly consisting of tenocytes and collagen fibers, are vital for joint stability and mobility, rendering successful tendon healing crucial in both sports and regenerative medicine 1 .
Adipose-derived stem cells (ADSCs), a kind of mesenchymal stem cells, are viable candidates for tendon regeneration owing to their accessibility, multipotency, and capacity to develop into tenocytes, the principal cellular element of tendon tissue. Augmenting the tenogenic differentiation of ADSCs is a principal research objective in tissue engineering to expedite tendon repair and enhance functional results 2 . Two primary techniques have emerged to facilitate this differentiation: photobiomodulation (PBM) and modulation of the Wnt signaling pathway. PBM is a non-invasive, light-based therapy that enhances cellular proliferation, migration, and differentiation by targeting mitochondrial chromophores, augmenting adenosine triphosphate (ATP) production, diminishing oxidative stress, and activating regenerative signaling pathways 3 . The Wnt/β-catenin pathway is a recognized regulator of cell destiny, tissue remodeling, and stem cell differentiation, including tenogenesis 4 . Recent evidence suggests that PBM can affect this system, augmenting the expression of Wnt target genes implicated in tendon growth5 –7. This indicates a synergistic interaction between PBM and Wnt signaling that may enhance ADSC-mediated tendon healing.
This review examines the interplay between PBM and Wnt signaling in guiding ADSC differentiation into tenocytes, emphasizing the molecular mechanisms involved and the therapeutic implications for tendon regeneration.
PBM overview
PBM is a non-invasive treatment that uses light-emitting diode (LED) light to stimulate cellular functions associated with tissue regeneration and repair 8 . Compared to high-intensity lasers that can generate heat and damage tissue, PBM uses soft lighting, which produces a heat-free and non-destructive effect. It primarily works by targeting cellular chromophores, such as cytochrome c oxidase in mitochondria, which absorb light energy and initiate a cascade of cellular responses that improve cellular function and viability 9 .
PBM mechanism of action
PBM’s effects are initiated when light energy penetrates the tissue and is absorbed by chromophores in mitochondria. This energy absorption enhances mitochondrial function, leading to increased production of ATP which is the cellular energy currency. The resulting boost in ATP production supports various cellular activities essential for tissue repair, including cell proliferation, migration, and differentiation 10 . Additionally, PBM reduces oxidative stress whereby it modulates reactive oxygen species (ROS) levels and promotes anti-inflammatory responses, which contribute to an environment that is favorable for healing and regeneration indicated in Fig. 1.

Photobiomodulation mechanism of action created with BioRender.com under an academic license.
PBM works by increasing the production of ROS, ATP, and nitric oxide (NO) in mitochondria, thereby inducing various cellular responses such as antioxidant responses, anti-inflammatory effects, and improved cellular function. PBM has been shown to improve mitochondrial function, including increased biogenesis, membrane potential, and respiration, thereby increasing ATP production and improving cellular function. The Wnt signaling pathway is tightly regulated by cellular energy levels (ATP), ROS, β-catenin stability, and NO. When Wnt ligands bind to Frizzled and LRP5/6 receptors, ATP-dependent activation of Disheveled (DVL) inhibits the degradation complex composed of Axin, APC, GSK-3β, and CK1. Normally, in the absence of Wnt signaling, β-catenin is phosphorylated by GSK-3β and degraded by the proteasome. However, Wnt activation inhibits GSK-3β, preventing β-catenin degradation and allowing it to accumulate in the cytoplasm before translocating to the nucleus to drive gene transcription. ROS levels can influence this process, as oxidative stress can enhance or inhibit Wnt signaling by modulating the activities of kinases and phosphatases involved in β-catenin stability. Similarly, NO plays a dual role: Low levels of NO promote β-catenin degradation via S-nitrosation, while high levels of NO stabilize β-catenin by inhibiting GSK-3β. The overall outcome of Wnt signaling depends on the balance between the availability of ATP, ROS, and NO, which together regulate β-catenin stability and downstream gene expression, thereby affecting cell fate, proliferation, and survival. PBM enhances ATP synthesis by activating mitochondria and regulates ROS, which contributes to downstream Wnt/β-catenin pathway activation9,10.
PBM devices and applications for tendon repair
PBM devices vary in wavelength, output power, and application, allowing for targeted treatment at different tissue depths. Table 1 below summarizes the different types of PBM devices and their applications related to tendon repair. These devices, particularly those operating in the near-infrared (NIR) band (600–1200 nm), are highly effective for tendon repair due to their ability to reach deeper tissue layers and promote cellular repair processes essential for tendon healing 12 .
Overview of photobiomodulation (PBM) device types and specifications.

This table provides an overview of the different types of PBM devices, detailing their typical wavelength ranges, output powers, and therapeutic applications. These devices range from low-energy lasers, LEDs, and infrared lasers to more complex systems such as dual-laser units, multi-mode platforms, and clinical-grade lasers. The wide range of uses for these technologies from topical home pain relief to professional clinical interventions for deep tissue healing and regenerative therapies highlights the adaptability and broad clinical relevance of the PBM approach.
Comparative overview of tenogenic strategies including PBM
PBM, through its activation of mitochondrial pathways and downstream Wnt/β-catenin signaling, holds considerable promise in inducing tenogenic differentiation of ADSCs. However, it must be considered alongside other well-established strategies in tendon tissue engineering. One such approach involves growth factor-based induction using agents like transforming growth factor-beta (TGF-β) and bone morphogenetic protein-12 (BMP-12) seen at Table 2. These molecules activate SMAD signaling pathways to directly stimulate tendon-related markers such as Scleraxis (Scx), Tenomodulin (Tnmd), and Type I Collagen (Col1a1), with BMP-12 preferred for its reduced osteogenic side effects 22 . Mechanical loading, another cornerstone strategy, uses cyclic tensile strain to simulate the tendon’s natural biomechanical environment, leading to the upregulation of similar markers via integrin-mediated activation of MAPK, PI3K/Akt, and Wnt/β-catenin pathways 23 . Although highly effective, mechanical loading is often complex to implement and lacks scalability. Biomimetic scaffolds, including aligned nanofibers and hydrogel matrices, provide structural cues that guide cell alignment, elongation, and extracellular matrix (ECM) production. These scaffolds may also be combined with PBM or growth factors to synergistically enhance tenogenesis. PBM offers a uniquely non-invasive alternative that modulates intracellular differentiation mechanisms, and when integrated with scaffolds, it may offer a powerful combinatorial approach to tendon regeneration.
Comparative summary of tenogenic induction strategies.
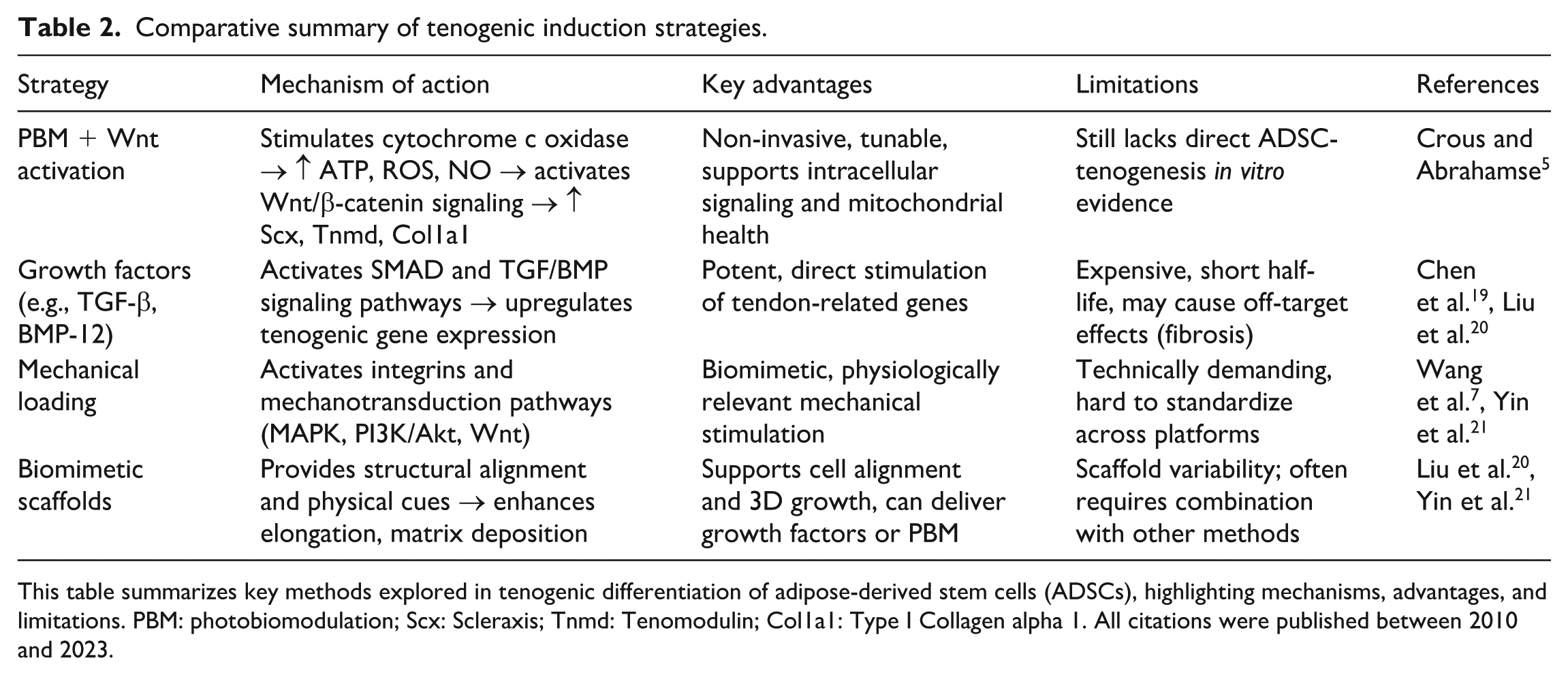
This table summarizes key methods explored in tenogenic differentiation of adipose-derived stem cells (ADSCs), highlighting mechanisms, advantages, and limitations. PBM: photobiomodulation; Scx: Scleraxis; Tnmd: Tenomodulin; Col1a1: Type I Collagen alpha 1. All citations were published between 2010 and 2023.
PBM’s influence on Wnt signaling in tenogenic differentiation
PBM, through the use of red or NIR light, has emerged as a promising, non-invasive tool to modulate stem cell behavior and guide to differentiation. One of its key mechanisms involves the activation of the canonical Wnt/β-catenin signaling pathway, which plays a central role in the tenogenic differentiation of ADSCs. This pathway regulates the expression of key tenogenic markers, including Scx, Tnmd, and Col1a1, which are essential for tendon formation and regeneration 24 .
PBM enhances Wnt signaling primarily through mitochondrial stimulation. Upon light absorption by cytochrome c oxidase, PBM leads to elevated levels of ATP, ROS, and NO, which all play crucial roles in intracellular signaling. These changes support the stabilization and nuclear translocation of β-catenin, which is essential for the transcriptional activation of Wnt target genes5,9. ATP serves as an energy source for signaling enzymes like DVL, while ROS and NO modulate GSK-3β activity, an inhibitor of β-catenin degradation 25 . This synergy enhances β-catenin activity in the nucleus, thereby promoting tenogenic gene expression.
Although limited, emerging experimental studies support this mechanistic link. For instance 26 , Fallahi et al. demonstrated that dual-wavelength PBM (660 and 810 nm) significantly enhanced the expression of Scx and Tnmd in ADSCs, along with increased β-catenin levels, confirming the activation of the canonical Wnt pathway. Similarly, Zhang et al. 24 reported that Wnt3a treatment of ADSCs led to increased nuclear β-catenin and upregulation of tenogenic markers, and this would suggest that PBM-induced Wnt signaling could achieve similar outcomes.
PBM was shown to upregulate Wnt-related genes and modulate mitochondrial pathways in mesenchymal stem cells undergoing lineage differentiation 5 . Even though these studies focused more on osteogenesis, the shared pathways suggest a broader application of PBM-Wnt crosstalk in lineage specification, including tenocytes. This is particularly relevant because mitochondrial regulation through PBM not only fuels cellular energy needs but also affects redox balance and transcriptional programs necessary for tendon development.
Wnt pathway’s dual role in regulating stem cell differentiation
The Wnt pathway has a dual regulatory function in stem cell differentiation interestingly, depending on the cellular context and the level of pathway activity. For ADSCs, moderate activation of Wnt signaling promotes differentiation toward specific lineages, such as tenocytes. However, excessive Wnt signaling promotes stem cell proliferation at the expense of lineage commitment, thereby inhibiting differentiation. This balance underscores the importance of precise regulation, which PBMs can help achieve by modulating Wnt pathway activity to provide just the right level of signaling for optimal tenocyte differentiation 27 . The synergistic effect of PBM and Wnt signaling pathways holds great therapeutic promise, providing a non-invasive approach to enhance ADSC differentiation into tenocytes. This combined strategy not only facilitates the development of ADSCs into tendon-like cells but also opens new avenues to improve tendon repair and regenerative therapies, thereby significantly shortening recovery time and improving the prognosis of patients with tendon injuries 28 .
PBM’s modulation of Wnt pathway components
The interaction between PBM and Wnt signaling represents an emerging area of stem cell research, particularly in its application to promote ADSC differentiation into tenocytes. By modulating cellular states and signaling pathways, PBMs have shown the potential to upregulate Wnt signaling components, thereby promoting lineage-specific differentiation and enhancing ADSC differentiation toward the tenocyte lineage, which is essential for tendon regeneration 29 .
PBM has multiple effects on the Wnt/β-catenin signaling pathway. It enhances ATP synthesis by activating mitochondria and regulates ROS, both of which are important components of cell signaling. This bioenergetic boost promotes the activation of Wnt signaling pathway components seen in Fig. 2, particularly in stem cells, leading to healthier cellular responses. Studies have shown that Wnt ligands that bind to receptors such as Frizzled and LRP5/6 initiate a cascade that stabilizes β-catenin, preventing its degradation, thereby allowing it to accumulate and translocate into the nucleus where it activates transcription of target genes involved in proliferation, differentiation, and tissue regeneration 25 .

Wnt pathway components created with BioRender.com under an academic license.
WNT proteins bind to the Frizzled receptor and its co-receptor LRP, triggering the activation of the WNT pathway. This interaction recruits DVL, which inhibits the destruction complex composed of GSK, Axin, CK1-alpha, and APC as this complex phosphorylates β-catenin, marking it for degradation. However, when inhibited, β-catenin stabilizes and accumulates in the cytoplasm before translocating to the nucleus. There, it binds to TCF/LEF transcription factors, activating the WNT target genes that regulate cell proliferation, differentiation, and survival.
The Wnt pathway in ADSC differentiation toward tenocytes
Tissue balance, cell proliferation, and differentiation are all regulated by the Wnt signaling pathway, a signaling cascade that has been preserved. When Wnt ligands bind to Frizzled receptors on the cell membrane, this pathway then gets activated. The activation of the Wnt pathway sets off a sequence of intracellular processes (see Table 3), leading to the stabilization and movement of β-catenin from the cytoplasm into the nucleus. Inside the nucleus, β-catenin engages with TCF/LEF transcription factors, triggering the expression of genes that dictate cellular fate and differentiation 37 .
Factors involved in the activation of the WNT pathway and function during stem cell differentiation into lineage specific cells.

The table provides an overview of the major molecular components of the Wnt signaling pathway, highlighting their functions and associated cell types. Central to this pathway are Wnt ligands, which initiate signaling by binding to Frizzled and LRP5/6 receptors, thereby activating the downstream Disheveled (Dvl) protein and inhibiting GSK3β protein expression. This prevents the degradation of β-catenin, allowing it to translocate to the nucleus and regulate gene expression via TCF/LEF transcription factors. Other regulators such as CK1α/β kinase and NF-κB also influence pathway activity. These components function in a variety of cell types, from immune-related cells, cancer cells, stem cells, and epithelial cells, reflecting the broad biological importance of this pathway. The inclusion of scaffold proteins such as AXIN involved in β-catenin degradation is essential for a comprehensive understanding of pathway regulation.
Role of Wnt signaling in ADSC differentiation toward tenocytes
ADSCs can differentiate into tenocytes through biochemical, mechanical, and PBM stimuli. ADSCs have shown promising differentiation capabilities toward tenocytes, which are tendon cells. Several studies have demonstrated the potential of ADSCs to differentiate into tenocytes in vitro and in vivo 38 . Several growth factors, such as TGF-β and insulin-like growth factor-1 (IGF-1), mechanical stimulation, and particular culture conditions that resemble the tendon microenvironment, can cause ADSCs to differentiate into tenocytes. ADSC-derived tenocytes have been shown to exhibit tendon-specific markers and characteristics, making them a valuable cell source for tendon regeneration and repair 19 . PBM (red/NIR light) enhances ATP production, ROS signaling, and NO modulation, which activate the Wnt/β-catenin pathway at key stages of differentiation 5 . Activation of the Wnt/β-catenin pathway promotes the expression of tendon-specific markers, such as sclerin (Scx), Tnmd, and Col1a1, which are essential for tendon structural integrity and function. Studies show that stimulation of the Wnt pathway in ADSCs enhances their differentiation into the tenocyte lineage, accelerating the formation of cells with tendon-like characteristics 24 .
A key molecular mechanism guiding this tenogenic commitment is the Wnt signaling pathway, which is divided into two major branches: canonical (β-catenin-dependent) and non-canonical (β-catenin-independent) pathways. The canonical Wnt pathway is activated when Wnt ligands such as Wnt3a, Wnt1, and Wnt10b bind to Frizzled (Fzd) receptors and co-receptors LRP5/6. This interaction stabilizes β-catenin, allowing it to accumulate and translocate into the nucleus, where it binds to TCF/LEF transcription factors to regulate downstream target genes 39 . In ADSCs, activation of this pathway enhances tenogenic differentiation, promoting increased expression of Scx, Tnmd, and extracellular matrix components essential for tendon integrity 40 . The inhibition of this pathway significantly reduces tenogenic marker expression, confirming its importance 41 .
The non-canonical Wnt pathway, which includes ligands such as Wnt5a, Wnt5b, and Wnt11, does not stabilize β-catenin. Instead, it operates through other signaling cascades like Wnt/Ca2+ and planar cell polarity (PCP) pathways, modulating cytoskeletal organization, intracellular calcium levels, and cell polarity 42 . Non-canonical signaling contributes to tenogenesis by promoting tenocyte alignment, mechanical responsiveness, and morphogenesis, which is the key process in tendon development and repair 42 .
Treatment of stem cells with Wnt ligands, such as Wnt3a, or chemical activators, such as lithium chloride, has been shown to upregulate tenogenic markers and promote the formation of a tendinogenic phenotype in these cells. Conversely, inhibition of Wnt signaling using inhibitors or RNA interference (RNAi) significantly suppressed tenogenic differentiation, suggesting that Wnt activity is essential for ADSC differentiation into tenocytes 41 .
Interaction between PBM and Wnt signaling in tenogenic differentiation
PBM has been shown to affect the Wnt pathway, thereby enhancing ADSC differentiation into tenocytes 43 . PBM indirectly affects Wnt signaling by stimulating mitochondrial activity and modulating oxidative stress, thereby creating favorable conditions for cellular responses 5 . Studies have shown that PBM activates Wnt/β-catenin signaling, particularly in stem cell populations, by upregulating the expression of Wnt ligands or modulating Wnt-related transcription factors 6 .
One study examined the effects of PBM on ADSCs and found that light of a specific wavelength and intensity could enhance the activation of the Wnt/β-catenin pathway, thereby promoting the expression of tenogenic markers (Table 4). This activation is thought to be due to PBM’s ability to reduce oxidative stress and promote ATP production, thereby creating an environment that supports Wnt pathway activity and accelerates ADSC differentiation into tenocytes. By complementing Wnt pathway signaling, PBM enhances the cell signaling required for ADSCs to develop a tenocyte-like phenotype, thereby improving the efficiency and speed of tendon regeneration 26 .
Regulation of WNT pathway by photobiomodulation.

This table summarizes recent findings on how PBM influences Wnt/β-catenin signaling across diverse cell types. The effects of PBM appear highly context-dependent: while it suppresses Wnt signaling in osteoblasts, human mesenchymal stem cells (hMSCs), and breast cancer cells—leading to outcomes like reduced proliferation or migration, it enhances Wnt activity in ADSCs and periodontal ligament (PDL) cells, promoting differentiation. The diverse effects are influenced by variations in wavelength (ranging from 254 nm to 980 nm), energy density, and exposure duration, highlighting the importance of tailored PBM parameters to achieve desired biological responses. ADSC: adipose-derived stem cells; ROS: reactive oxygen species.
Therapeutic implications of PBM-Wnt interactions
PBM regulation of Wnt signaling has important therapeutic implications. By using PBM to influence the Wnt pathway, clinicians may be able to more effectively direct ADSC differentiation to the tendon, providing an advanced non-invasive approach to tendon repair. As research progresses, optimizing PBM parameters such as wavelength, intensity, and duration will be critical for maximizing the benefits of Wnt pathway modulation in ADSCs and enhancing tenogenic differentiation outcomes in clinical settings.
The interaction between PBM and the Wnt pathway may become a powerful tool to promote ADSC differentiation into tenocytes. By selectively modulating Wnt components, PBM enhances cellular responses and supports tendon regeneration, laying the foundation for improved targeted therapies in regenerative medicine.
Conclusion and future directions
Although PBM has been demonstrated to affect the Wnt/β-catenin signaling pathway in different situations, there is currently limited direct evidence that PBM specifically regulates Wnt signaling to promote the differentiation of ADSCs into tenocytes. Studies have demonstrated PBM’s effects on Wnt signaling in other cell types and differentiation processes 46 . For instance, research indicates that PBM can modulate Wnt/β-catenin signaling during neurogenesis, promoting neuronal differentiation of stem cells 47 . PBM has been observed to affect the Wnt signaling pathway in hair regeneration, suggesting a broader role in stem cell differentiation 48 . This review highlights the potential of PBM to address gaps in current regenerative medicine strategies, including combining stem cell–based therapies with light-induced modulation of cellular pathways. In the context of tenogenic differentiation, the Wnt signaling pathway is known to play a complex role. Studies have shown that Wnt signaling can both promote and inhibit tenogenic gene expression, depending on the cellular context and specific Wnt ligands involved 49 . However, direct studies investigating the impact of PBM on Wnt-mediated tenogenic differentiation of ADSCs are currently lacking.
The existing body of research exhibits numerous methodological deficiencies that constrain the translational applicability of PBM-based therapy for tendon repair. Absence of standardization in PBM protocols: Research varies significantly in wavelength (e.g., 660 nm vs 810 nm), energy density, exposure durations, and dosage intervals. The inconsistencies impede inter-study comparisons and obstruct reproducibility5,26. Lack of in vitro ADSC tenogenic models utilizing PBM with direct evaluation of the Wnt pathway: The majority of data is indirect or derived from unrelated lineages (e.g., osteogenic, neural), and there are few investigations utilizing lineage-tracking markers or β-catenin-responsive reporter assays in tenogenic systems. Inadequate molecular profiling and investigations on pathway-specific inhibition: Future research should utilize methodologies like as RNAi, Wnt inhibitors, and CRISPR/Cas9 to confirm the role of Wnt in PBM-induced tenogenesis. Restricted integration with alternative regulatory pathways: Although PBM may engage with Wnt, it likely also interacts with TGF-β, BMP, Notch, and MAPK/ERK signaling, all of which play roles in tenogenesis. These interactions are inadequately examined50,51. Insufficient advanced preclinical models: There is a lack of rigorously controlled in vivo research utilizing tendon damage models to assess the efficacy of PBM-Wnt-ADSC. Furthermore, the immunomodulatory effects and dose-response kinetics are inadequately comprehended in big animal models or human explants 28 .
Despite encouraging findings, several gaps remain in our understanding of PBM’s precise molecular mechanisms in tenogenic differentiation. Future research should focus on explaining the interactions between PBM, Wnt signaling, and other regulatory pathways, such as TGF-β and BMP signaling, to optimize treatment protocols. Converting PBM-regulated ADSC therapy into practical clinical uses will also require developments in gene editing, preclinical models, and clinical trials 51 . Investigating PBM’s immunomodulatory effects and computational modeling of its impact on cell signaling could further refine its therapeutic potential 52 . In conclusion, the interplay between PBM, Wnt signaling, and tenogenic differentiation may represent a transformative approach in tendon tissue engineering. This technology has the potential to become a crucial part of regenerative medicine by bettering our understanding of the molecular mechanisms underlying PBM and standardizing treatment protocols. It will also give clinicians a useful tool for improving tendon repair and patient outcomes.
Footnotes
Acknowledgements
The authors thank the University of Johannesburg (UJ) and Laser Research Center (LRC) for use of their facilities and resources.
Ethical Considerations
This article is a review of previously published studies and does not contain any new data involving human participants or animals. Therefore, ethical approval and informed consent were not required.
Statement of human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors. All data discussed are from previously published studies, which are duly cited.
Statement of informed consent
This article does not contain any studies with human participants performed by any of the authors. Therefore, informed consent was not required.
Author contributions
MM and AC: Conceptualization, Writing—Original Draft, and Writing—Review & Editing; MM: Visualization; AC and HA: Supervision and Funding acquisition. All authors read and approved of the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Research Foundation of South Africa Thuthuka Instrument, grant number TTK2205035996; the South African Medical Research Council (SAMRC) through its Division of Research Capacity Development under the Research Capacity Development Initiative from funding received from the South African National Treasury. The content and findings reported/illustrated are the sole deduction, view, and responsibility of the researcher and do not reflect the official position and sentiments of the SAMRC, grant number 57050; the Department of Science and Innovation (DSI) funded African Laser Center (ALC), grant number HLHA23X task ALC-R007; the University Research Council, grant number 2024URC00813; the Department of Science and Technology’s South African Research Chairs Initiative (DST-NRF/SARChI), grant number 98337; the University of Johannesburg Global Excellence and Statue (GES) 4.0 master’s scholarship award. The funding bodies played no role in the design of the study, collection, analysis, and interpretation of the data or writing the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
