Abstract
Mesenchymal stem cells (MSCs) are considered to be effective treatments for various diseases, and a wide variety of clinical studies have been performed worldwide. However, substantial obstacles remain before they can be approved and disseminated as treatments. A major bottleneck is the elucidation of their mechanisms of action, and the molecules that are essential for their efficacy have not been fully characterized. In this paper, I review the studies that attempted to identify the key mediators of MSCs that are involved in their effects on disease using in vivo models. More specifically, studies are discussed in which reductions in the efficacy of MSCs in animal models of disease were induced by the absence of key mediators. The target diseases were lung, joint, cerebral nerve, or cardiac diseases and graft-versus-host disease (GVHD). The following molecules were identified and are discussed herein: TSG-6, VEGF, KGF, HGF, claudin-4, ANXA1, MANF, PYCR1, integrin β1, PDGFRβ, type-II collagen, CD151, TIMP3, TGF-β1, BDNF, COX-2, Botch, IL-1β, CTRP3, CXCR4, miR-34c, FSTL1, IDO, iNOS, IFNγR1, PGES, Chi3l1, and IL-6. These are key mediators of the efficacy of MSCs in vivo.
Graphical abstract was created in BioRender. Nakamura, K. (2025) https://BioRender.com/u98s099
Introduction
Mesenchymal stem cells (MSCs) are said to have been discovered by Friedenstein et al. in 1970, and since then, their effectiveness in the treatment of various diseases has been reported, and many clinical studies have been performed around the world1–8. There are increasing numbers of schemes and regulations concerning the approval of cell-based therapies as pharmaceutical products, and some MSC products have been clinically assessed and/or marketed. Specifically, Temcell has been approved and is now available in Japan as a treatment for graft-versus-host disease (GVHD). In addition, the US Food and Drug Administration (FDA) has approved Remestemcel-L for the treatment of GVHD in 2024, making it the first FDA-approved MSC product. There has been a great deal of research into the treatment of a wide variety of diseases using MSCs because of their efficacy and functionality 9 .
Unlike molecular targeting drugs and biopharmaceuticals, which tend to have their effect through a single mechanism, MSCs may be effective at treating disease through multiple mechanisms, and this explains why they are candidates for the treatment of various diseases. However, this wide variety of mechanisms of action means that they cannot always be fully characterized, which renders their development as therapeutic products difficult and time-consuming10–12. Unlike molecular targeting drugs, which are developed through target screening on the basis of a single mechanism, the development of MSCs often begins with the demonstration of therapeutic efficacy vs disease phenotypes, which makes it extremely difficult to identify the mechanism of action (MoA), and therefore for their progression from basic research to therapeutic products.
The present review focuses on diseases for which MSC treatment has been investigated in clinical studies. In particular, target diseases for which a large number of clinical studies have been conducted are herein collectively summarized. Furthermore, the studies summarized are those that reported the mechanisms of action of MSCs in the treatment of these diseases. This is because the identification of the molecules in MSCs that are involved in their mechanisms of action is important for the quality control of MSCs as a physical entity and product.
Many studies of the efficacy of MSCs for disease treatment have demonstrated the amelioration of signs or symptoms, and therefore a therapeutic effect. However, it is difficult to identify the MoA molecules that are essential for their efficacy by simply confirming the presence or absence of the inhibition or activity of particular biochemical pathways in vitro. Therefore, this review focuses on studies that assessed the pharmacologic effects of MSCs in animal models of disease and identified mediators that are essential for the pharmacologic effects of MSCs in these models. Specifically, the studies searched were those that confirmed the disappearance or attenuation of the therapeutic effects of MSCs when they were administered with particular proteins or micro-RNAs knocked out, knocked down, or blocked in animal models of disease. In this way, the list compiled included proteins or micro-RNAs in MSCs that have been demonstrated to be essential for their efficacy against specific diseases in vivo.
Methods
The clinicaltrials.gov database was searched for clinical studies of MSCs. Thereafter, the diseases under investigation for MSC treatment were listed according to the number of studies performed. PubMed was then searched for articles regarding the diseases treated with MSCs, which were ranked top four in the aforementioned list and GVHD, for which a practical application as a therapeutic product is progressed 13 . The articles were searched for findings that relate the results of in vivo studies of MSCs in which specific proteins or micro-RNAs that are essential for their effects were identified using molecular inhibition techniques such as knockdown, knockout, and the administration of blocking antibodies. Finally, the target diseases and the essential proteins and micro-RNAs were comprehensively evaluated to compile a list of MoA molecules that could be considered to be of significant importance for the pharmacologic effects of MSCs.
Results
Number and classification of clinical studies
A search of clinicaltrials.gov identified 1694 clinical studies of MSCs up to October 2024. The results of their classification by disease are shown in Figure 1 and Table 1. There were 416 cases in phase 1, 477 cases in phase 1/2, 274 cases in phase 2, 35 cases in phase 2/3, 67 cases in phase 3, 16 cases in phase 4, and 409 cases in other. This review does not evaluate the efficacy of each clinical trial.
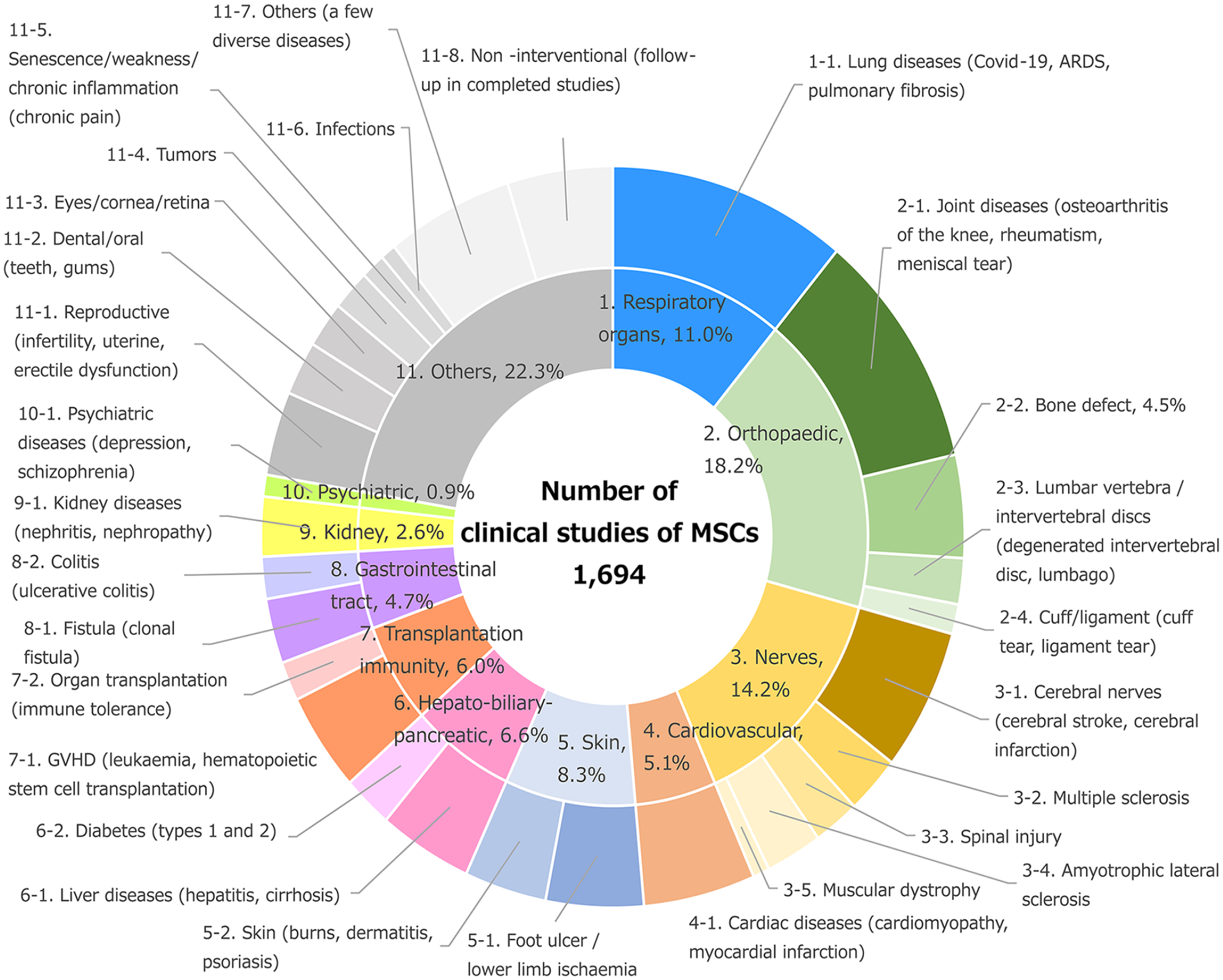
Number of clinical studies of mesenchymal stem cells and classification of target diseases (total n = 1694). ARDS: acute respiratory distress syndrome; COVID-19: coronavirus disease 2019; GVHD: graft-versus-host disease; MSC: mesenchymal stem cell.
Number and percentage of clinical studies of mesenchymal stem cells by disease class.

ARDS: acute respiratory distress syndrome; COVID-19: coronavirus disease 2019; GVHD: graft-versus-host disease.
The following were investigated in many clinical studies of MSCs: lung diseases (e.g., coronavirus disease 2019 [COVID-19], acute respiratory distress syndrome [ARDS], pulmonary fibrosis; n = 186), joint diseases (e.g., osteoarthritis of the knee, rheumatism; n = 177), cerebral nerve diseases (e.g., cerebral stroke, cerebral infarction; n = 105), cardiac diseases (e.g., cardiomyopathy, myocardial infarction [MI]; n = 87), foot ulcers/lower limb ischemia (n = 76), bone defect (n = 76), liver diseases (e.g., hepatitis, cirrhosis; n = 73), and GVHD (n = 73). Of these studies, pulmonary diseases (11.0%), joint diseases (10.4%), cerebral nerve diseases (6.2%), and cardiac diseases (5.1%) were targeted in ≥5% of the clinical studies.
Lung diseases were ranked highest because many clinical studies of MSCs were performed in patients during the recent COVID-19 epidemic 14 . In addition, there have been numerous clinical studies of joint diseases15,16, cerebral nerve diseases 17 , and cardiac diseases 18 , which have conventionally been thought of as candidate indications for MSC treatment. GVHD, for which MSCs are being used therapeutically currently, was ranked eighth in the list of clinical studies, implying that relatively few clinical studies have been conducted regarding this use of MSCs. The 99 clinical studies classified under “Others” in the list targeted a wide variety of diseases, each of which was represented by a few clinical studies. These findings confirm that the use of MSCs for the treatment of a wide range of diseases has been evaluated in humans.
Next the list of studies that attempted to identify the proteins and micro-RNAs essential to the effects of MSCs on the four highest ranked diseases (lung diseases, joint diseases, cerebral nerve diseases, and cardiac diseases) and GVHD was compiled.
Key mediators of the effects of MSCs on lung diseases in vivo
A thorough examination of the 170 articles (PubMed search results up to October 2024) on lung diseases revealed that six articles mentioned key mediators through studies of animal models of disease. These are listed in chronological order in Table 2.
Proteins and micro-RNAs expressed in mesenchymal stem cells that have been demonstrated to be important for their effects in vivo.

GVHD: graft-versus-host disease; MoA: mechanism of action; MSC: mesenchymal stem cell.
Molecules of MSCs that have been shown in vivo to be key to their therapeutic efficacy.
In 2011, Danchuk et al. 19 evaluated the effects of bone marrow–derived MSCs in a mouse model of lipopolysaccharide (LPS)-induced pulmonary inflammation and found that their therapeutic effects were impaired in mice in which TSG-6 was knocked down.
In 2014, Chang et al. 20 found that umbilical cord blood–derived MSCs had significant beneficial effects in a rat model of alveolar dysgenesis, whereas MSCs with small interfering RNA (siRNA)-mediated VEGF knockdown had significantly less marked effects. The number of hyperoxygenation-induced TdT-mediated dUTP Nick End Labeling (TUNEL)-positive cells in rat pulmonary tissue was significantly reduced by untransfected MSCs or scramble siRNA-transfected MSCs, but this effect was significantly abrogated by VEGF siRNA transfection of MSCs.
In 2015, Li and Wu 21 reported that bone marrow–derived MSC administration substantially reduced LPS-induced acute lung injury in rats, whereas the administration of MSCs with KGF knocked down did not have the same effect. The authors found that the mechanism involved recovery of alveolar fluid clearance via alveolar type-II cells, which depend on KGF secreted by the MSCs.
In 2016, Hu et al. 22 administered wild-type (WT) MSCs or MSCs with HGF knocked down to a rat model of ARDS and compared their therapeutic effects. The MSCs were derived from bone marrow. The inflammation and lung damage were mitigated by the WT MSCs, whereas the MSCs with HGF knocked down had a weaker protective/therapeutic effect. In addition, the amelioration of pulmonary vascular permeability, assessed using dye extravasation, was significantly poorer in the rats administered MSCs with HGF knocked down.
In 2016, Zheng et al. 23 reported that bone marrow–derived MSCs protected against pulmonary edema in a rat model of LPS-induced acute lung injury, but this effect was abrogated when claudin-4 was knocked down in the MSCs. The beneficial effect was attributable to inhibition of apoptosis by MSCs, and the inhibitory effect was lost when claudin-4 was knocked down.
In 2023, Zhu et al. 24 evaluated the effects of adipose-derived MSCs in a model of ARDS and reported that the hemorrhage, inflammatory cell infiltration, alveolar collapse, and interstitial expansion in the lungs of the mice were significantly mitigated by the MSCs and the ANXA1-overexpressing MSC group, but the effects were not significant when MSCs with ANXA1 knocked down were administered.
Thus, TSG-6, VEGF, KGF, HGF, claudin-4, and ANXA1 have been identified to be key proteins for the effects of MSCs on lung diseases in vivo. These results, along with those for the four other disease types, are listed in Table 2.
Key mediators in MSCs for the treatment of joint diseases in vivo
A thorough evaluation of 205 articles (PubMed search results up to October 2024) regarding the treatment of joint diseases with MSCs revealed that five articles mentioned key mediators through studies of animal models of disease.
In 2021, Xia et al. 46 reported that bone marrow–derived MSCs had beneficial effects in a rat model of osteoarthritis and that when used in combination with low-intensity pulsed ultrasound, their therapeutic effects were enhanced. They also found that when MSCs were treated with an autophagy inhibitor, 3-MA, both the therapeutic effects of MSCs alone and the combination disappeared. The expression of SDF-1 and CXCR4 was suppressed by 3-MA, and although these may be key mediators of the effects of MSCs, no in vivo studies of direct SDF-1 or CXCR4 inhibition were conducted.
In 2021, Tejedor et al. 25 evaluated the regenerative effects of bone marrow–derived MSCs in a “super healer” strain of mice, Murphy Roths Large (MRL) mice. In MRL mice with osteoarthritis, therapeutic effects of MSCs were identified, but these effects were abrogated by MANF knockdown in the MSCs.
In 2021, Tejedor et al. 26 also studied the repair of injured joint cartilage in MRL mice. They evaluated cartilage tissue regeneration in BL6 mice that had been subjected to an ear punch. Tissue repair, including that of cartilage, was observed in the mice treated with MRL bone marrow–derived MSCs, whereas those that received MRL MSCs with PYCR1 knocked down showed a significantly smaller area of repair, similar to that in untreated mice. They concluded that the expression of PYCR1 is key to the cartilage regeneration induced by MSCs, as confirmed by the results of an in vitro study.
In 2022, Kitahashi et al. 27 evaluated the effects of synovial MSCs in a rat model of meniscal injury and found that they caused meniscal cartilage regeneration, whereas mice that received synovial MSCs treated with integrin β1 or PDGFRβ-neutralizing antibody showed significantly smaller areas of regeneration. They also reported that synovial MSCs in which type-II collagen had been knocked out also caused significantly less regeneration.
In 2024, Xia et al. 28 reported a contribution of CD151 to the effects of umbilical cord–derived MSC treatment using a mouse model of collagen-induced RA. They created a DBA/1J mouse model of rheumatoid arthritis and administered human umbilical cord MSCs (hUC-MSCs) or CD151-knocked-down hUC-MSCs. Signs of inflammation in the joints were ameliorated in the MSC-treated mice, whereas those that received MSCs with siRNA-mediated CD151 knockdown showed less marked amelioration. The authors stated that cell proliferation and migration mediated by CD151 play important roles in the therapeutic effects of MSCs.
Accordingly, MANF, Pycr1, integrin β1, PDGFRβ, type-II collagen, and CD151 were recorded as proteins that are important for the effects of MSCs on joint disease in vivo. These results are listed in Table 2.
Key mediators in MSCs for the treatment of cerebral nerve diseases in vivo
A thorough evaluation of 177 articles (PubMed search results up to October 2024) regarding cerebral nerve diseases revealed that eight articles mentioned key mediators that are important in the effects of MSCs in animal models of disease.
In 2012, Menge et al. 29 showed that TIMP3 is a key mediator of the therapeutic effects of bone marrow–derived MSCs using a mouse model of traumatic brain injury. In a group of mice that were injured and then intravenously administered MSCs, the high level of permeability of the blood–brain barrier (BBB) was reduced, whereas in mice that were administered MSCs with TIMP3 knocked down, this protective effect was significantly impaired, and their BBB permeability was similar to that of mice that did not receive MSCs. The authors concluded that MSCs secrete TIMP3, which promotes vascular endothelial barrier integrity and maintains normal BBB permeability.
In 2013, Yoo et al. 30 reported that the administration of bone marrow–derived MSCs to a rat model of cerebral stroke restored motor function. They also showed that this restorative effect was reduced by knocking down TGF-β1 in MSCs using shRNA, that TGF-β1 knockdown prevented the suppression of CD68+ cell infiltration into the ischemic brain, and that the improvement in motor function was also attenuated by this knockdown.
In 2017, Ahn et al. 31 administered WT human bone marrow-derived MSCs (hUCB-MSCs) or hUCB-MSCs with BDNF knocked down to rats with severe intraventricular hemorrhage and then compared their motor function and other parameters. The MSCs were derived from umbilical cord blood. They found that the therapeutic effects of MSCs, including a reduction in the severity of hydrocephalus and an improvement in motor function, were abrogated by BDNF knockdown. They concluded that BDNF secreted by MSCs has neuroprotective effects.
In 2017, Kota et al. 32 evaluated the effects of human bone marrow-derived MSCs (hBM-MSCs) on neuroinflammation by assessing IBA-1+ microglial accumulation in a rat model of traumatic brain injury. The MSCs were derived from bone marrow. Rats subjected to controlled cortical impacts showed accumulation of microglia and inflammation in the hippocampus and thalamus, which were ameliorated by MSCs. However, the administration of MSCs with COX-2 knocked down was associated with a smaller reduction in microglial accumulation and a less marked anti-inflammatory effect. As a comparison, MSCs with IDO knocked down were also administered, and these had the same effects as the WT MSCs.
In 2019, Liu et al. 33 studied the therapeutic effects of bone marrow–derived MSCs (BMSCs) and a potential mechanism using a rat model of subarachnoid hemorrhage-induced early brain injury. BMSC administration to the rats ameliorated their neurobehavioral disorders and reduced their brain water content (BWC), thus confirming the beneficial effects of the cells. They found that rats administered a Notch1 inhibitor (DAPT) or that had Notch1 knocked down responded in the same way to BMSCs, and that BMSC treatment inhibited the activation of the Notch1 signaling pathway. When Botch was knocked down in BMSCs, the effects of BMSCs to ameliorate neurobehavioral disorders and reduce BWC were abrogated. Botch has been reported to be an inhibitor of Notch1.
In 2019, Sheikh et al. 34 administered a human bone marrow–derived MSC cell line (B10 cells) that has been reported to ameliorate the neurologic defects of a rat model of middle cerebral artery occlusion and evaluated the restoration of brain angiogenesis. Angiogenesis in the lesion (indicated by positivity for endoglin) was increased by B10-MSC administration, whereas rats that received B10-MSCs in which IL-1β expression had been silenced by siRNA treatment showed a smaller area of angiogenesis.
In 2020, Yang et al. 35 reported that the transplantation of intrastriatal BMSCs into rats with brain ischemia/reperfusion (I/R) injury significantly improved their neurological function and that MANF was important for this. The MSCs were derived from bone marrow. The neurologic function of the rats was improved more by MSC treatment than by no treatment or Phosphate Buffered Saline (PBS) treatment. Although MSCs improved the modified neurological severity score and reduced the volume of cerebral infarction, these effects were significantly less marked in rats that received MSCs in which MANF had been knocked down using siRNA. They concluded that MSCs secrete MANF, which promotes the depolarization of M2 microglia.
In 2020, Roura et al. 36 demonstrated that adipose tissue-derived MSC-concentrated conditioned medium (ASC-CCM) derived from adipose-derived MSCs alleviated visual impairment in mice with mild traumatic brain injury. On the other hand, they reported that no similar alleviating effects were observed when using TSG-6 knockdown ASCs. They suggested that TSG-6 takes a central role for MSC therapy.
In 2023, Wang et al. 37 reported that BMSCs improved the neurobehavior of a rat model of intracerebral hemorrhage. In rats with intracerebral hemorrhage, the measures of motor performance were improved by BMSC administration, including the modified Garcia score, forelimb placement testing, and corner turn testing, whereas these effects were significantly less marked in rats that received BMSCs in which CTRP3 had been silenced using siRNA. The BWC, assessed as an index of functional impairment of the BBB, was also ameliorated by BMSCs, but this effect was significantly less marked in the rats administered with CTRP3-silenced BMSCs. The authors concluded that microglial pyroptosis is reduced by CTRP3 secreted by BMSCs.
Accordingly, TIMP3, TGF-β, BDNF, COX-2, Botch, IL-1β, MANF, TSG-6, and CTRP3 appear to be key proteins for the effects of MSCs on cerebral nerve diseases, according to the results of in vivo studies. These results are listed in Table 2.
Key mediators in MSCs for the treatment of cardiac diseases in vivo
A thorough evaluation of 95 articles (PubMed search results up to October 2024) regarding cardiac diseases revealed that nine studies of the effects of MSCs in animal models of disease mentioned key molecules mediating their effects.
In 2010, Huang et al. 38 reported that CXCR4 was a key mediator of the therapeutic effects of bone marrow–derived MSCs in a rat model of MI. Measures of cardiac function, such as left ventricular dimension and left ventricular ejection fraction (LVEF), were significantly improved by the MSCs, but these effects were attenuated by the siRNA-mediated knockdown of CXCR4 in the MSCs. In addition, the effects of the MSCs were enhanced by CXCR4 overexpression. Parallel effects were identified with respect to histopathologic parameters and the amount of angiogenesis, which supported the authors’ findings.
In 2010, Wang et al. 47 reported the therapeutic effects of bone marrow–derived MSCs overexpressing connexin 43 (Cx43) in a rat model of MI. MSCs reduced the infarct size, LVEF, and left ventricular fractional shortening (LVFS), and these effects were enhanced by Cx43 overexpression. Conversely, MSCs in which Cx43 had been knocked down had similar effects to WT MSCs, implying that although the overexpression of Cx43 enhances the therapeutic effects of MSCs, Cx43 is not essential for these effects.
In 2012, Huang et al. 48 reported the therapeutic effects of bone marrow–derived MSCs, CXCR4-overexpressing MSCs, MSCs with CXCR4 knocked down, MMP-9-overexpressing MSCs, and MSCs with MMP-9 knocked down in a mouse model of MI. They found that only the CXCR4-overexpressing MSCs had significantly better therapeutic effects than WT MSCs, with all the other MSC types having similar effects to the WT MSCs. Thus, CXCR4 and MMP-9 appear not to be essential for the therapeutic effects of MSCs on MI.
In 2014, Wang et al. 49 reported the influence of Cx43 on the effects of bone marrow–derived MSCs using a rat model of MI. Cx43-overexpressing MSCs had more marked therapeutic effects, such as reducing infarct size and improving cardiac function, than WT MSCs. However, similar effects were obtained using WT MSCs and MSCs in which Cx43 had been knocked down, implying that Cx43 is not essential for the therapeutic effects of MSCs.
In 2015, Kang et al. 39 reported that miR-34c is essential for the therapeutic effects of BMSCs using a nude mouse model of MI. When BMSCs were administered to mice that had undergone coronary ligation, they reduced the fibrosis and improved cardiac function, assessed using indices such as ejection fraction. However, when BMSCs in which miR-34c had been knocked down were administered, these beneficial effects were not apparent. Thus, angiogenesis induced by miR-34c is an important part of the therapeutic mechanism.
In 2016, Zhang et al. 40 described the relationship between the therapeutic effects of BMSCs and FSTL1 in a rat model of I/R. The area of infarction was reduced by the BMSCs, but not by BMSCs in which FSTL1 had been knocked down: the sizes of the infarcted areas were similar in untreated mice and those receiving the modified BMSCs. The authors concluded that FSTL1, which is a secreted protein, is essential for the therapeutic effects of BMSCs.
In 2017, Wu et al. 50 reported the therapeutic effects of bone marrow–derived MSCs, CXCR4-overexpressing MSCs, and MSCs, in which CXCR4 had been knocked down in a rat model of MI. They found that CXCR4-overexpressing MSCs had more marked therapeutic effects than WT MSCs and/or MSCs with CXCR4 knocked down in terms of the extent of fibrosis, ejection fraction, and other parameters. However, the effects of the MSCs with CXCR4 knocked down did not differ from those of WT MSCs, implying that CXCR4 is not essential for the therapeutic effects of MSCs.
In 2017, Zeng et al. 51 administered MSCs, integrin-linked kinase (ILK)-overexpressing MSCs, and MSCs in which ILK had been knocked down to rats with MI and evaluated their therapeutic effects. The MSCs were derived from bone marrow. Assessments of cardiac function and histology showed that ILK-overexpressing MSCs had more marked therapeutic effects than WT MSCs or those with ILK knocked down. However, the therapeutic effects of the MSCs with ILK knockdown were greater than those of the vehicle and similar to those of the WT MSCs. Hence, although ILK enhances the therapeutic effects of MSCs, it is not essential for their effects.
In 2022, Gong et al. 41 reported the effects of umbilical cord-derived MSCs (UCMSCs) in rats with dilated cardiomyopathy (DCM). They first showed that their therapeutic effects were much more marked after two doses of MSCs than after a single dose in rats with DCM. They then compared the administration of two doses of WT MSCs, IDO-overexpressing MSCs, and MSCs in which IDO had been knocked down. The IDO-overexpressing MSCs had significantly better effects on various aspects of cardiac function, including ejection fraction, than the other MSCs. MSCs with IDO knocked down had significantly less marked therapeutic effects than the WT MSCs. In summary, the therapeutic effects could be ranked in the following order: two doses of IDO-overexpressing MSCs, two doses of WT MSCs, two doses of MSCs with IDO knocked down, and vehicle, and there were significant differences among these groups. Accordingly, it was concluded that IDO is important for the therapeutic effects of MSCs and contributes to the control of inflammation by inducing Treg responses, which leads to a recovery of cardiac function in DCM.
Accordingly, CXCR4, miR-34c, FSTL1, and IDO have been shown to be important for the effects of MSCs on cardiac diseases using in vivo studies. These results are listed in Table 2.
Key mediators in MSCs for the treatment of GVHD in vivo
A thorough evaluation of 118 articles (PubMed search results up to October 2024) regarding GVHD revealed that four articles mentioned key mediators of the effects of MSCs in studies of animal models of disease.
In 2008, Ren et al. 42 reported the results of the administration of MSCs, iNOS−/−MSCs, or IFNγR1−/−MSCs to mice with GVHD. The MSCs were derived from bone marrow. The administration of MSCs improved the percentage survival of the mice with GVHD, but this effect was absent in mice that received iNOS-knockout MSCs or IFNγR1-knockout MSCs, and similar results were obtained when anti-INFγ antibodies or iNOS inhibitors were co-administered with WT MSCs. Thus, these molecules appear to contribute to the therapeutic effects of MSCs in GVHD.
In 2018, Kim et al. 43 reported the therapeutic effects of BMSCs in a mouse model of GVHD. The percentage survival of the mice was significantly improved by BMSC treatment (alongside treatment with scramble siRNA) vs PBS treatment. However, the percentage survival was not improved when BMSCs were administered in concert with the siRNA-mediated knockdown of PGES (there was no difference from PBS-treated mice).
In 2022, Liu et al. 44 reported the identification of molecules necessary for the therapeutic effects of hUC-MSCs using a mouse model of acute graft-versus-host disease (aGvHD). hUC-MSC-treated mice showed beneficial effects, such as superior percentage of survival and clinical scores, and less tissue damage. However, mice that received hUC-MSCs with Chi3l1 knocked down showed less marked therapeutic effects. Furthermore, the IL-17A concentrations in the serum, spleen, and intestine were higher in the Chi3l1-knockdown hUC-MSC–treated mice.
In 2024, Na et al. 45 reported the results of administering human decidua–derived mesenchymal stromal cells (DSCs) to mice with GVHD. DSCs were described as cells with superior heterologous immunity to bone marrow–derived MSCs. The administration of DSCs improved the percentage survival, clinical scores, and histology of mice with GVHD. However, the administration of DSCs in which IL-6 had been knocked down had less marked effects. The authors found that the immunosuppression was attributable to an IL-6-induced increase in the expression of PD-L1 and PD-L2.
Thus, iNOS, IFNγR1, PGES, Chi3l1, and IL-6 have been reported to be key mediators of the effects of MSCs on GVHD on the basis of studies of in vivo models. These results are listed in Table 2.
Conclusion
The present review has discussed selected diseases for which clinical investigations have been performed to determine the effects of MSCs, according to clinicaltrials.gov. The articles that interrogated the roles of specific proteins or micro-RNAs in the effects of MSCs on the diseases in vivo were searched. As shown in Table 2, a broad range of MoA molecules, associated with anti-inflammation, angiogenesis, and the inhibition of apoptosis, were identified in models of lung disease, which was investigated in the largest number of clinical studies, and it was concluded that their paracrine effects are important for the therapeutic effects of the MSCs.
With respect to joint diseases, various groups of proteins have been reported to play roles in the effects of MSCs, including those involved in cell adhesion, migration, and chondrocyte maintenance. This is consistent with the induction of tissue regeneration being important to the therapeutic effects of these cells.
With respect to cerebral nerve diseases, a wide variety of molecular mechanisms have been reported, including BBB stabilization, immune cell coordination, suppression of neuroinflammation, angiogenesis, apoptosis inhibition, and microglial pyroptosis inhibition. A large number of in vivo studies have been conducted to identify key mediators of the effects of MSCs. This may suggest that many mechanisms are involved in the complex functional recovery of the central nervous system, brain.
With respect to cardiac diseases, unlike the other diseases, many studies have investigated the enhancement of the therapeutic effects of MSCs associated with the overexpression of particular proteins, and only a few have attempted to elucidate the mechanisms involved through the inhibition of key mediators. Some of these studies showed that although the effects of the MSCs were enhanced by the overexpression of specific proteins, knockdown of these proteins did not reduce the effects of the cells. This may be because the effects of WT MSCs are insufficient for the treatment of cardiac diseases, and instead, the overexpression of key proteins in MSCs is necessary for a therapeutic effect to be achieved. Research on MoA molecules of MSCs will continue to advance, for example, on the mechanisms of target molecules on monocytes 52 .
In GVHD, for which the use of MSC therapy is a commercial product, only four relevant in vivo articles were available, despite there having been numerous studies of human PBMCs in vitro. Because the present review was focused on results obtained using in vivo models, mediators that have only been evaluated in vitro were excluded; therefore, the latter studies were not included among the articles discussed herein.
As reported by Nakamura et al. 53 in 2024, it is important to elucidate the mechanisms of action of MSCs, to identify the key molecules involved, and to ensure the quality of the cells, in order for MSCs to become viable therapeutic products. Although it is not easy to identify the most important proteins for the treatment of various diseases, the optimal use of the results of published studies of the mechanisms of action of MSCs should aid in their development for the treatment of various diseases. The summary presented in this review should assist with this process. The proteins described herein (TSG-6, VEGF, KGF, HGF, claudin-4, ANXA1, MANF, PYCR1, integrin β1, PDGFRβ, type-II collagen, CD151, TIMP3, TGF-β1, BDNF, COX-2, Botch, IL-1β, CTRP3, CXCR4, miR-34c, FSTL1, IDO, iNOS, IFNγR1, PGES, Chi3l1, and IL-6) can be said to be important for the effects of MSCs in vivo. It is hoped that MSCs will be developed as pharmaceutical products as the key mediators of their therapeutic effects for various diseases are progressively identified.
Footnotes
Acknowledgements
Ethical Considerations
Not applicable.
Author contributions
Conceptualization: K.N.; Writing—original draft preparation: K.N.; Writing—review and editing: K.N.; Investigation: K.N.; Visualization: K.N.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article, and informed consent is not applicable.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
