Abstract
Aim
Calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs) are an important new preventative medication class for migraine treatment. While clinical efficacy of CGRP mAbs is well established, less is known about their impact on treatment patterns and work absence. This study examined migraine treatment and work absence patterns before and after CGRP mAbs were reimbursed in 2019 in Sweden.
Methods
We conducted a nationwide non-interventional observational study using Swedish administrative data. Patients with a hospital diagnosis of migraine between 2015 and 2021 were included and split into three cohorts based on the year of their diagnosis: 2015–2016, 2017–2018 and 2019–2021. We assessed time to new treatments after diagnosis using inverse probability weights to adjust for baseline differences. Work absence due to sick leave and disability was assessed via a two-part regression model of the estimated probability of any work absence and the average number of absence days conditional on non-zero absence days in the year following diagnosis.
Results
In total, 40,247 patients were included. The proportion dispensed a new preventative treatment after diagnosis rose from 16.2% in the 2015–2016 cohort to 22.4% in the 2019–2021 cohort. Patients diagnosed in 2019–2021 initiated more new preventative treatments compared to earlier cohorts. CGRP mAbs composed a substantial part of the treatment landscape for those diagnosed in 2019–2021, with 9.2% of patients receiving them as the first new treatment after diagnosis. The regression models showed a lower probability of work absence in the 2019–2021 cohort compared to the earlier cohorts, however the total number of absence days conditional on non-zero days varied across the study cohorts.
Conclusions
After market entry of CGRP mAbs, newly diagnosed patients with migraine initiated and cycled through preventative migraine medications more rapidly than those diagnosed in earlier years. These changes were accompanied by a modest decline in the probability of work absence. Faster cycling through preventative medication after the entry of CGRP mAbs in Sweden may indicate that patients and physicians want to start CGRP mAb treatment as early as possible.
This is a visual representation of the abstract.
Introduction
According to the Global Burden of Disease Study, migraine is a leading cause of disability, particularly among the population under 50. 1 About 15% of the global population suffers from migraine.2,3 Beyond its clinical manifestations, migraine is associated with lost working days and productivity loss. 4
Treatment options exist for both acute migraine (during a migraine episode) and for migraine prevention. Acute treatments include (but are not limited to) non-steroidal anti-inflammatory agents (NSAIDs), paracetamol and triptans, while preventative medications include (but are not limited to) anti-epileptics, tricyclic anti-depressants, beta-blockers and onabotulinumtoxinA. Calcitonin gene-related peptide (CGRP) inhibitors emerged as a new treatment class in recent years and changed the treatment landscape for migraine substantially. 5 They act by blocking the CGRP protein, which is believed to be a factor causing migraine. Two types of CGRP inhibitors exist on the market today, monoclonal antibodies (mAbs) aimed at migraine prevention and receptor antagonists (gepants) aimed to treat acute migraine. The first CGRP mAb, erenumab, was approved by the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) in 2018, and several mAbs have since been approved. Ubrogepant was the first gepant approved by the FDA in 2019.
After new drugs are approved by the EMA, they can be prescribed and used in all European Union countries, including Sweden. However, uptake of new medicines in Swedish clinical practice is highly dependent on the public reimbursement of the drug. The Swedish Dental and Pharmaceutical Benefits Agency (TLV) is the government agency in charge of reimbursement decisions. Erenumab was the first CGRP mAb to receive public reimbursement from 1 January 2019 6 (EMA approval: 26 July 2018), followed by fremanezumab from 1 November 2019 7 (EMA approval: 28 March 2019) and galcanezumab from 28 August 2020 8 (EMA approval: 14 November 2018). In Sweden, eptinezumab, rimegepant and atogepant were approved for reimbursement in 2022, 2023 and 2023, respectively, after the study period in this study. According to the reimbursement criteria for CGRP mAbs set by TLV, only patients with chronic migraine who have tried at least two different classes of preventative medicines without improvements are eligible for CGRP mAb treatment.
There is a growing body of literature studying the use of CGRP mAbs in various settings. These studies typically focus on the characteristics of patients who use CGRP mAbs,9–11 switching patterns, adherence and persistence in CGRP mAb use12–14 and CGRP mAbs’ effectiveness on migraine days.15–18 Historically, despite the availability of multiple preventative treatment options, adherence to and persistence in utilisation of these treatments have been low.19,20 However, the literature on how the entry of CGRP mAbs affected the overall treatment landscape for patients with severe migraine is limited. A US study found the use of non-CGRP preventative medications decreased after CGRP entry in California. 21 Understanding whether the availability of CGRP mAbs affects treatment cycling is important for clinicians making decisions about when to escalate and switch treatments to best support patients. More broadly, our analysis sheds light on the extent to which a breakthrough treatment re-shapes prescribing practices. We also know little about how the entry of CGRP mAbs impacted the ability to work among migraine patients. We aim to address these knowledge gaps.
In this study, we examined treatment patterns and patterns of work absence due to sick leave or disability after a hospital diagnosis of migraine in Sweden, and how these patterns varied over time as CGRP mAbs entered the market. A key analysis was time from diagnosis to initiation of new preventative treatment lines.
Methods
This was a non-interventional observational study using Swedish individual-level administrative and health registers. These registers have complete and nationwide coverage. Through the personal identification number provided to all Swedish citizens, we followed individuals from one data source to another. A detailed description of the data sources used in this study is provided in the online Appendix. The study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Study population
The source population in our analyses included patients who visited a hospital and received a diagnosis of migraine (International Classification of Diseases, 10th Revision (ICD-10) code: G43), recorded in the Swedish National Patient Register (NPR) between 1 January 2005 and 1 March 2022.
From the source population, we created our study population, which consisted of patients who had their first recorded migraine diagnosis in the NPR between 1 January 2015 and 28 February 2021. The chosen inclusion period allowed for potential follow-up of at least 1 year after index (i.e. migraine diagnosis). As we aimed to study patterns of sick leave from work, analyses were restricted to patients of working age between 20 and 60 years of age, as relatively few individuals younger than 20 years are employed.
We created three study cohorts based on whether they were indexed before or after reimbursement of CGRP mAbs. The first two cohorts consisted of those indexed before reimbursement and included a 2017–2018 cohort (those diagnosed closer to CGRP market entry) and a 2015–2016 cohort (those diagnosed further away in time from CGRP market entry). The third cohort was those indexed after CGRP market entry in Sweden, between 2019 and 2021 (until the end of February 2021).
Patient characteristics
We measured age, sex, highest level of education (lower than upper secondary school, upper secondary schools, higher than upper secondary school, tertiary education (bachelor, master, PhD)), disposable income (sum of labour and capital incomes, inflation-adjusted 2024 EUR) and employment status (receiving disability pension, not employed, employed) in the calendar year before index.
Additionally, we considered a patient's treatment history of preventative and acute migraine medications. Drug dispensations were identified using the Anatomical Therapeutic Chemical (ATC) classification system. We considered dispensations of drugs in various preventative treatment classes in the 3 years prior to index (beta-blockers (metoprolol/propranolol, ATC code: C07AB02/C07AA05), angiotensin receptor blockers (candesartan, ATC code: C09CA06), anti-depressants (amitriptyline, ATC code: N06AA09), anti-epileptics (topiramate, ATC code: N03AX11), onabotulinumtoxinA (ATC code: M03AX01) or CGRP mAb (ATC code: N02CD) in the National Prescription Register), number of preventative treatment classes dispensed in the 3 years prior to index and type of acute treatment drugs dispensed in the 3 years prior to index (triptans (ATC code: NO2CC), NSAIDs (ATC code: M01A) or paracetamol (ATC code: N02BE01)).
Propensity score adjustment
To account for differences in observable characteristics in the population diagnosed with migraine across time, we estimated a propensity score model (logistic regression) of the probability of being diagnosed in 2019–2021, the post-CGRP entry era. Age, sex, education, income, employment, acute treatment history and preventative treatment history at the time of index were used in the model. The propensity scores were then converted to inverse probability weights (IPWs) which were used in the time-to-event analyses and regression analysis described below.
Treatment patterns and time-to-event analyses
We analysed time from index to first new treatment (first line), second new treatment (second line) and third new treatment (third line) of preventative migraine medications within the first 365 days following index. New treatments after index were defined as a new dispensation of a preventative medication drug class not previously dispensed in the 3 years prior to index. For instance, for a patient who was dispensed a drug from one of the preventative medication categories before index (e.g. beta-blockers), we assessed how long it took from index to being dispensed a drug from another category (e.g. anti-depressants (first line)), and then potentially to another (e.g. CGRP mAbs (second line)). Patterns related to type of treatment received were presented using Sankey diagrams split by study cohort. The time-to-event analyses were performed using the Kaplan–Meier estimator weighted by IPWs and split by study cohort and the number of preventative treatment classes dispensed in the 3-year pre-index period.
Work absence patterns
Data on government-covered sick leave and early retirement due to disability were extracted from the Swedish Social Insurance Agency. In Sweden, the government provides sick pay for sick leave spells beyond 14 days, and it is these spells that were included in the data. Sick leave spells shorter than 14 days are covered by the employer and not recorded in the register. However, for the spells in the data, we had information on the first true sick day in the spell. Similarly, the government provides disability pension for individuals with permanent disabilities, and this information was also available in the data.
To study work absence patterns, we assessed the mean number of work absence days per patient, per month from 12 months prior to index to 12 months after index. Days were calculated by multiplying the length of sick or disability leave with the extent of leave. For example, a patient with a sick leave spell of 30 days with 80% leave would have 24 sick days during the period (30 × 0.8 = 24). This analysis was also split by study cohort and the number of preventative treatment classes dispensed in the 3-year pre-index period.
Regression analysis
In the regression analysis, we estimated associations between treatment patterns and work absence days in the year following diagnosis and whether these associations varied by study cohort and new treatments received. Many individuals were observed with zero work absence days. Therefore, we estimated a two-part model: first a logistic regression of the probability of having any work absence, and then an ordinary least squares (OLS) regression model of the number of work absence days among those with non-zero work absence days. The predictor variables in this analysis were the study cohorts, the number of new dispensed treatments in the year following index and their interaction. We also used the IPWs as weights in the regression models to account for baseline characteristics.
As we wanted to ease the interpretation of the regression results and compare outcomes across the study cohorts and by the number of new treatments, we used the regression results to obtain estimated marginal means (EMMs). EMM's directly compared the average probability of having work absence and the average number of work absence days for those having non-zero work absence days across the study cohorts and the number of new treatments. We also showed the 95% confidence interval in the regression and EMM analyses.
Ethical approval
The use of Swedish administrative data is subject to approval from ethical review boards and approval for using data in this project was provided by the Swedish Ethical Review Board (2021-02450 and 2023-05437). In accordance with Swedish legislation and the study design, informed consent from individual participants was not required. The authors cannot share the underlying data used in this study for legal reasons; however, the same datasets can be applied for through new data applications to the data holders.
Results
Patient characteristics
In total, 40,247 patients were included in the study population with 13,548, 13,285 and 13,414 patients in the three cohorts of patients diagnosed in 2015–2016, 2017–2018 and 2019–2021, respectively (Table 1). The average age at diagnosis was 39 and about 75% of the patients were female across all three cohorts. More patients had tertiary education in the later years (32% in the 2019–2021 cohort vs. 28% in the 2015–2016 cohort). Disposable income and employment were slightly higher among the later cohort compared to the early cohorts. The number of sick days in the year before diagnosis was the same across the cohorts.
Patient characteristics and treatment history at diagnosis by calendar year cohorts.
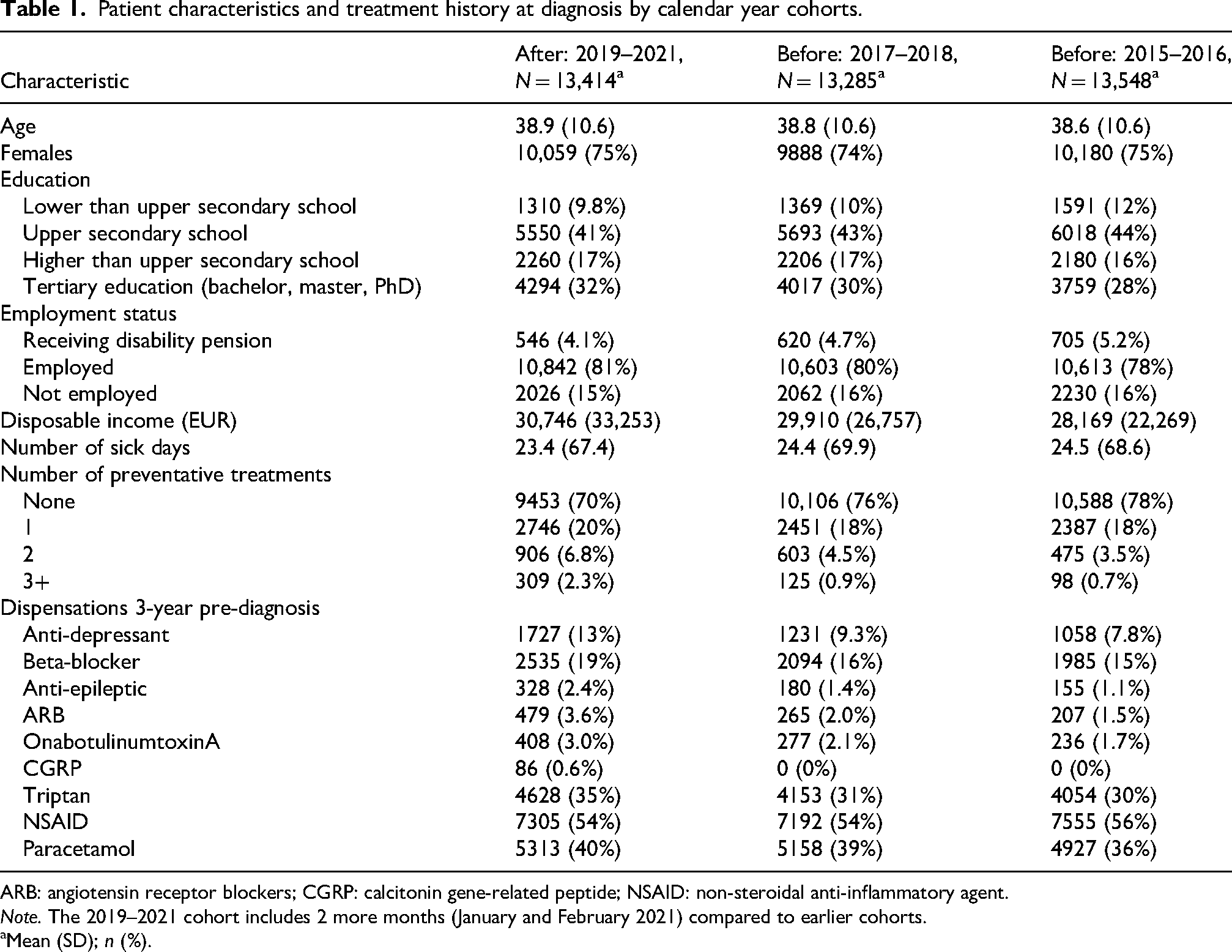
ARB: angiotensin receptor blockers; CGRP: calcitonin gene-related peptide; NSAID: non-steroidal anti-inflammatory agent.
Note. The 2019–2021 cohort includes 2 more months (January and February 2021) compared to earlier cohorts.
Mean (SD); n (%).
For those diagnosed in 2019–2021, 70% of patients were not dispensed any preventative migraine medications in the 3 years preceding diagnosis compared to 78% of patients in the 2015–2016 cohort. Beta-blockers were the most dispensed preventative medication in all three cohorts. More patients in the 2019–2021 cohort had received acute medication in the 3 years before diagnosis than earlier cohorts.
Treatment patterns
In the year after diagnosis, 22.4% (n = 3005) patients in the 2019–2021 cohort (N = 13,414) received any type of preventative treatment medication (Figure 1). The most common first-line treatment after index was anti-depressants (amitriptyline, the only anti-depressant included) (26.4% of the treated) followed by beta-blockers (22.9%). The new CGRP mAbs were dispensed to 9.2% of the treated patients as the first-line treatment. OnabotulinumtoxinA, CGRP mAbs and anti-depressants were the most common second-line treatments. CGRP mAbs were the most common third choice.

Sankey diagram of type of new treatments received by cohort. The treatment categories are mutually exclusive.
For the 2017–2018 cohort, 17.5% (n = 2333) of the patients received at least one preventative medication after index. Beta-blockers (30.9% of the treated) were the most common first-line treatment followed by anti-depressants (25.5%). Anti-depressants were the most common second-line treatment and were dispensed to 29% of patients who received a second-line treatment.
Among patients diagnosed in 2015–2016, 16.2% (n = 2195) received preventative medication after index. Beta-blockers and anti-depressants were dispensed to 38.5% and 28.0% of the treated patients as first-line treatments, respectively. The most common second-line treatments among those receiving treatment were anti-depressants (29.8%) and onabotulinumtoxinA (29.3%).
Treatment patterns stratified by the number of preventative medications dispensed before index are available in the Supplemental materials. For the 2019–2021 cohort, CGRP mAbs were the first-line treatment for 3%, 9.5%, 21.8% and 40.5% of the treated patients with ‘None’ (Supplemental Figure S1), ‘1’ (Figure S2), ‘2’ (Figure S3) and ‘3+’ (Figure S4) dispensed preventative medications before index, respectively.
Time-to-treatment
In Figure 2, inverted Kaplan–Meier estimates (weighted by the IPWs) and the corresponding 95% confidence intervals are shown for time from diagnosis to new treatment lines (i.e. dispensation of a preventive medication class not previously dispensed). Overall, patients diagnosed in 2019–2021 initiated first-line preventative treatments earlier than patients diagnosed in earlier years, regardless of prior treatment history (p-value <0.005). Among patients with no pre-index dispensations of preventative medications, 15.8% in the 2019–2021 cohort received a first-line treatment within 1 year from diagnosis, compared to 13.8% and 13.2% in the 2017–2018 and 2015–2016 cohorts, respectively. For those with one pre-index dispensation, 35.5%, 28.0% and 26.6% received a first-line treatment in the 2019–2021, 2017–2018 and 2015–2016 cohorts, respectively. For those with two pre-index dispensations, 43.5%, 33.4% and 28.6% of patients received a first-line treatment within a year in the 2019–2021, 2017–2018 and 2015–2016 cohorts, respectively.

Time from diagnosis to a new dispensation of a preventative medication not previously dispensed; presented as inverted Kaplan–Meier plots. The columns are stratified by the number of preventative medications dispensed 3 years before index. The rows are stratified by first, second and third new drug after index. The proportions for those with second and third new drug after index are among those patients who already received a first or second new treatment. Shaded areas indicate 95% confidence intervals.
The largest difference in the second-line treatment patterns between the 2019–2021 cohort compared to the other cohorts was for those with one pre-index dispensation. Among those who received one new treatment after index, 28.0% of the 2019–2021 cohort received a second-line treatment within a year compared to 18.9% and 15.6% of the 2017–2018 and 2015–2016 cohorts, respectively. Unweighted Kaplan–Meier estimates are available in the Supplemental materials (Figure S5) and show the same results as the weighted results.
Work absence patterns
Patients with no prior preventative treatments at diagnosis averaged around 2–2.5 work absence days per month in the year preceding diagnosis (Figure 3), with some modest variation across cohorts. After diagnosis, the number of work absence days was on average slightly higher. Overall, the patterns across the calendar year cohorts were relatively similar. For those with one prior treatment, patients diagnosed in 2015–2016 had on average around one additional work absence day per month compared to the other two cohorts. Among those with two prior treatments, the 2015–2016 and 2017–2018 cohorts were relatively similar before diagnosis with around 8 work absence days. After diagnosis, the 2015–2016 cohort had around one additional day compared to the 2017–2018 cohort. The 2019–2021 cohort had consistently fewer work absence days with slightly more than 6 days on average per month. For those with three or more treatments tried before diagnosis, the 2017–2018 cohort had the most work absence days over time, while the 2015–2016 cohort had the least.
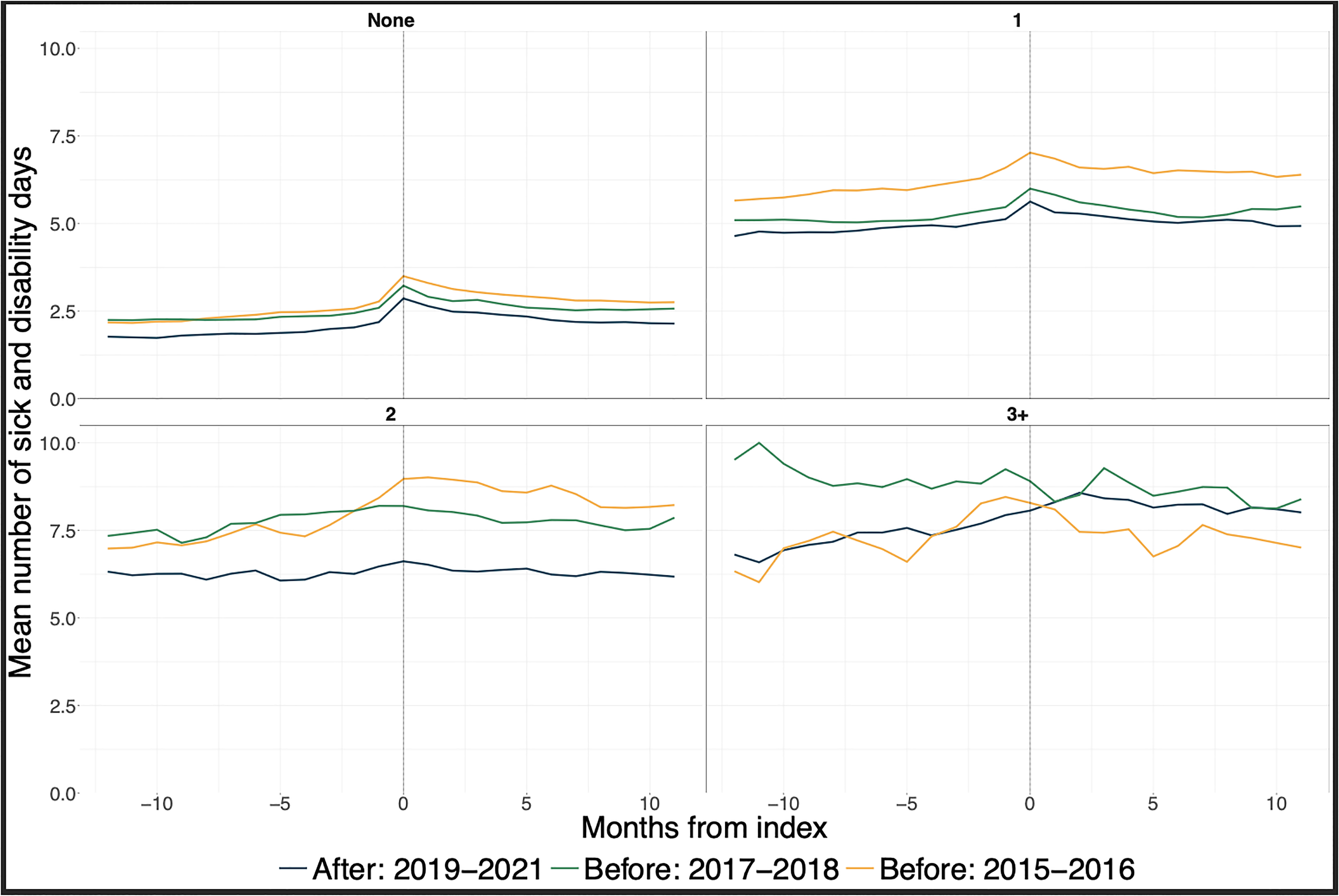
Mean number of combined sick and disability days per month from 12 months before index to 11 months after (including patients with zero sick and disability days). Month 0 is the first month following index. Stratified by the number of preventative medications dispensed 3 years before index.
Regression analysis
In Figure 4, the EMMs of the probability of having at least one work absence day (from logistic regression) and the number of non-zero work absence day (from OLS regression) in the year following diagnosis are presented by the cohorts and the number of treatments received after index. The underlying regression results and the two-way comparisons between cohorts and number of treatments are presented in Supplemental Tables S1 and S2.

Probability of having work absence days and the estimated mean number of work absence days (if having non-zero to begin with) across the cohorts by the number of received treatments in the year following index. Point estimates with 95% confidence intervals.
For patients with no new treatments after diagnosis, the probability of work absence was the highest in the 2015–2016 cohort (28.4%) compared to 27.9% and 26.4% in the 2017–2018 and 2019–2021 cohorts, respectively. Among those with work absence days, the estimated number of days was 134 for the 2019–2021 cohort compared to 147 for the 2015–2016 cohort. For patients who received first-line or second-line treatments after index, the probability of work absence was higher for the 2015–2016 cohort compared to the other two cohorts. However, the number of work absence days was relatively similar. The probability of work absence and the number of absence days among those who received third-line treatment were generally higher than those with fewer treatments, but the confidence intervals were wide. Unweighted EMMs are available in the Supplemental materials (Figure S6) and show the same results as the weighted results.
Discussion
In this study, we examined trends in treatment for patients diagnosed with severe migraine before and after the entry of CGRP mAbs into clinical practice. We found that those diagnosed when CGRP mAbs were on the market were prescribed preventative treatments at a faster rate, as time to the next treatment decreased compared to those diagnosed in earlier years. This was particularly evident for time to first-line treatment after diagnosis for those who had tried other preventative medications prior to their migraine diagnosis in hospital care. These time-to-event analyses were estimated using Kaplan–Meier models and IPWs. Thus, we accounted for differences in observable patient characteristics between the cohorts. The distribution of treatments also changed as CGRP mAbs were dispensed to a substantial share of patients. Based on our regression models, we observed lower probability of work absence post-diagnosis in the later cohorts for those who did not have any prior medication or had one or two previous medications. The EMMs from the regression model between the number of work absence days and faster treatment cycles was generally not statistically significant (overlapping confidence intervals in Figure 4) for those who received preventative medication before index.
One key finding was that treatment patterns differed over time, with shorter observed times to subsequent preventative treatments after CGRP mAb market entry. Our results are consistent with findings from a US study of tertiary headache centres in California, which showed that entry of CGRP mAbs decreased the use of other preventative medications. 21 Despite the differences between the US and Swedish healthcare systems, CGRP mAbs’ market entry was associated with a shift in overall treatment patterns in both settings. We are careful not to make causal statements that faster cycling is due to the market entry of CGRP mAbs, as it may be attributed to other factors. For example, physicians may have increased their knowledge of migraine treatment over time, with or without CGRP mAbs, and made different decisions regarding treatment regimens. We accounted for differences in patient characteristics through the propensity scores and IPWs, and those characteristics are therefore unlikely to drive the faster treatment cycling we observed. Clinicians may also be less willing to continue to prescribe potentially less effective treatments when CGRP mAbs became available. Patients with severe migraine may benefit from earlier escalation to CGRP mAbs rather than staying on traditional treatments with limited efficacy or poor tolerability.
A notable finding was that 78% of the 2015–2016 cohort were not prescribed any preventative medication prior to index compared to 70% of the 2019–2021 cohort, and more patients in the 2019–2021 cohort were dispensed medications after index. A higher use of preventative medication over time has also been found in other countries.22,23 Anti-depressants (e.g. amitriptyline) and beta-blockers were the most common treatments, which aligned with prior literature.22,24,25
The number of sick leave and disability days was also generally lower among the 2019–2021 cohort compared to the other cohorts which may be due to general work absence trends in the population, or better migraine treatment over time. The stabilisation of work absence days among the 2019–2021 cohort with three or more preventative treatments prior to diagnosis may be due to actual use of CGRP mAbs as these drugs were the most common (40.5% of first-line treatments) among this group. Previous observational studies suggest CGRP mAb use decreases healthcare resource use and sick days.17,18,26 However, it is also the case that those who are treated with CGRP mAbs generally experience more severe migraine and higher costs.9,10 Other studies have also found the healthcare burden to be higher among preventative users generally.15,27,28 The finding in our study that those with more dispensed preventative medication at index had higher work absence in general is unsurprising as these patients likely suffer from more severe migraine.
For clinicians, our results suggest that faster cycling of preventative medications and in some cases towards CGRP mAbs is associated with better labour market outcomes. Without proper cycling, patients may stay too long on ineffective treatments and risk both prolonged migraine and work impairment. For policymakers, our findings highlight the importance of timely access to new treatments which may ease the health and labour market burden associated with migraine.
Strengths and limitations
The main strength of this study is the use of nationwide and complete data covering the entire population. Our study population included all patients who visited a hospital and received a diagnosis of migraine in Sweden from 2015 to 2021 and we observed all their dispensed medications and recorded sick and disability days.
A limitation of the sick leave data is that only sick leave spells longer than 14 days are included. In Sweden, the employer provides sick pay for the first 14 days. After 14 days, the state provides sick pay instead, and it is the state-covered sick leave spells that are included in the register. This means we have underestimated the number of sick days as short-term sick leave spells are not included. It is unknown how the distribution of short-term spells is distributed across the study cohorts.
We did not know whether the dispensed medications were prescribed for migraine treatment or other conditions, and many of the preventative treatments are indicated for other conditions. However, as the analyses were centred around those with a diagnosis of migraine, we believe we largely captured prescriptions for migraine.
The index date was set at the first migraine diagnosis in specialty care. It should be noted that this diagnosis, as we observe it, is general. That is, we do not know if the individual suffers from chronic versus episodic or acute migraine. We also do not know the number of migraine days patients had at the time of their diagnosis, nor the number of migraine days during the study period. The validity of ICD-10 diagnoses in the Swedish patient register is considered to be high. 29
Research from 2001 in Sweden suggests around 50% of those with migraine received a diagnosis from a physician. 30 More recent research suggests that over 65% of migraine diagnoses were initially determined by general practitioners (GPs) and 26.3% by neurologists. 31 Over 80% of GPs have reported they treat patients with chronic migraine without referring them to a specialist. 32 Patients treated solely in primary care or by private outpatient specialists are missing from our data. We therefore do not capture the universe of individuals with diagnosed migraine in Sweden, only those with a diagnosis made in a specialist setting. These patients likely suffer from the most severe migraine and are also the patient group for which CGRP mAbs are especially relevant.
Future research
CGRP mAbs are a preventative medication targeting patients treated by (hospital) specialists (e.g. neurologists). It was therefore natural for us to study patients who received a migraine diagnosis in specialty/hospital care. This also means patients who were solely treated in primary care were not captured in our data. That group of patients will increasingly become relevant as CGRP mAbs are being extended to those with episodic migraine. Additionally, CGRP gepants may target a broad migraine population. Future research should assess the impact of the market entry of CGRP gepants aimed at acute migraine.
More research is also needed to fully understand the causal impact of CGRP mAbs on employment outcomes. Given the strong evidence of CGRP efficacy in clinical trials on monthly migraine days, there is potential for these drugs to improve labour market productivity.
Conclusion
The introduction of CGRP mAbs into clinical practice changed the treatment landscape for migraine, and these new drugs have become a common part of severe migraine treatment. Faster cycling through preventative medication after the entry of CGRP mAbs in Sweden may indicate that patients and physicians want to start CGRP mAb treatment as early as possible.
Clinical implications
Patients cycle through different preventative medication classes faster after CGRP mAbs enter the market.
Uptake of CGRP mAbs has been rapid in the most severe migraine population.
Sick leave and disability days vary across users of preventative medications and patients are less likely to be absent from work in recent years.
Supplemental Material
sj-docx-1-rep-10.1177_25158163261421617 - Supplemental material for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study
Supplemental material, sj-docx-1-rep-10.1177_25158163261421617 for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study by Johan A. Liseth Hansen, Meghan Skira and Knut Reidar Wangen in Cephalalgia Reports
Supplemental Material
sj-png-2-rep-10.1177_25158163261421617 - Supplemental material for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study
Supplemental material, sj-png-2-rep-10.1177_25158163261421617 for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study by Johan A. Liseth Hansen, Meghan Skira and Knut Reidar Wangen in Cephalalgia Reports
Supplemental Material
sj-png-3-rep-10.1177_25158163261421617 - Supplemental material for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study
Supplemental material, sj-png-3-rep-10.1177_25158163261421617 for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study by Johan A. Liseth Hansen, Meghan Skira and Knut Reidar Wangen in Cephalalgia Reports
Supplemental Material
sj-png-4-rep-10.1177_25158163261421617 - Supplemental material for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study
Supplemental material, sj-png-4-rep-10.1177_25158163261421617 for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study by Johan A. Liseth Hansen, Meghan Skira and Knut Reidar Wangen in Cephalalgia Reports
Supplemental Material
sj-png-5-rep-10.1177_25158163261421617 - Supplemental material for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study
Supplemental material, sj-png-5-rep-10.1177_25158163261421617 for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study by Johan A. Liseth Hansen, Meghan Skira and Knut Reidar Wangen in Cephalalgia Reports
Supplemental Material
sj-png-6-rep-10.1177_25158163261421617 - Supplemental material for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study
Supplemental material, sj-png-6-rep-10.1177_25158163261421617 for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study by Johan A. Liseth Hansen, Meghan Skira and Knut Reidar Wangen in Cephalalgia Reports
Supplemental Material
sj-png-7-rep-10.1177_25158163261421617 - Supplemental material for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study
Supplemental material, sj-png-7-rep-10.1177_25158163261421617 for Migraine treatment and work absence trends before and after introduction of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs): A nationwide Swedish register study by Johan A. Liseth Hansen, Meghan Skira and Knut Reidar Wangen in Cephalalgia Reports
Footnotes
Consent to participate
The use of Swedish administrative data is subject to approval from ethical review boards and approval for using data in this project was provided by the Swedish Ethical Review Board (2021-02450 and 2023-05437).
Consent for publishing
In accordance with Swedish legislation and the study design, informed consent from individual participants was not required.
Author contributions
JALH: conceptualisation, methodology, formal analysis, writing (original draft preparation) and writing (review and editing).
MS: conceptualisation, methodology, writing (original draft preparation) and writing (review and editing).
KRW: conceptualisation, methodology, writing (original draft preparation) and writing (review and editing).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: JALH is an employee of Quantify Research, part of the Athagoras Group, a consultancy firm providing services to public and private entities, including pharmaceutical companies. MS and KRW declare no conflicting interest.
Data availability statement
For privacy and legal reasons, data used in this study cannot be shared with anyone other than those with explicit approval to access the data. The same data can be applied for with new ethical and data applications to the relevant authorities in Sweden.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
