Abstract
The aim was to describe the use of and adherence to migraine preventives among insured patients meeting the International Classification of Headache Disorders, 2nd edn (ICHD-II) criteria for migraine headaches. A retrospective, case–control study was conducted using data from a telephone interview linked with health insurance claims data. Subjects were health plan enrollees aged 18–55 years who had incurred at least one encounter between June 2000 and November 2001. Interview responses were used to identify cases meeting the ICHD-II criteria for strict and probable migraine and a random sample of controls. Pharmacy claims data were used to construct measures of use and adherence. Differences in outcomes by adherence status were evaluated using generalized linear models. We identified 2517 cases and 941 controls. Among cases, the prevalence of antidepressant use was 4%, anticonvulsant use was 1.9%, antihypertensive use was 8.9%. Combined use was 13.4% among cases and did not differ significantly from that observed among controls (12.4%). Mean adherence rate between the first and last dispensing during the year was high (88%) and did not differ by migraine status. When the entire 12-month period is considered, adherence was substantially lower (56%). Patients who were adherent between dispensings reported significantly less migraine-related disability and incurred higher prescription drug costs, but did not differ in their total medical care costs. Patients with migraine are unlikely to be users of preventive medications. Among users, few are taking preventive medications continuously. Patients with migraine—especially those without a medical diagnosis for migraine or headaches—are not receiving the benefits available from existing pharmacotherapy options.
Introduction
Pharmacological migraine treatment includes acute treatments taken at the time of attack to relieve pain and restore function, and preventive treatments taken daily to reduce headache frequency. Preventive migraine treatment is recommended for patients with frequent headaches as well as attacks that remain disabling despite optimal acute treatment (1). Although trial evidence demonstrates that migraine management with preventive pharmacotherapy can reduce headache frequency and improve health-related quality of life (2–4), little is known regarding effectiveness in practice.
In the general population, when validated instruments are used to ascertain migraine status, only 12% of migraine sufferers report taking preventive treatments for migraine (5). These data are limited by subject recall of medications and reasons for taking them. Studies have also examined migraine preventive medication use among health plan enrollees (6). These studies arguably provide more accurate information on medication use via the use of pharmacy records, but they are limited by incomplete ascertainment of migraine sufferers.
We link pharmacy and medical claims data with patient self-reports of headache symptoms to describe the use of migraine preventive therapy among insured patients meeting the International Classification of Headache Disorders, 2nd edn (ICHD-II) criteria for strict and probable migraine as well as among controls without migraine. We also describe adherence to migraine preventives and report the association between medication adherence and health status, migraine-related disability and medical care costs.
Methods
Study setting and population
Subjects were identified among enrollees in a non-profit health plan serving residents of south-east Michigan, USA. The plan provides health insurance coverage to > 500 000 enrollees, approximately 20% of the local market. At time of the study, approximately half of care delivered under this plan was provided by a large, salaried, multi-specialty medical group. Both health plan and medical group are affiliated with an integrated health system, facilitating linkage of patient-level data across the two. Eligible study enrollees were the 23 299 men and women aged between 18 and 55 years who were continuously enrolled in the health plan 1 June 1999 to 31 May 2001 who incurred at least one encounter with a medical group provider 1 June 2000 to 31 May 2001.
Enrollees were recruited for study participation as described elsewhere (7–9). Briefly, between June and November 2001, eligible enrollees were mailed letters of study introduction that were followed by recruitment calls. A telephone administered survey, which included a validated diagnostic algorithm—based on reported headache type, characteristics (i.e. age of onset, frequency, duration, disability) and features (i.e. location and quality of pain, effect of headaches on physical activity, and the occurrence of nausea, vomiting, photophobia, phonophobia and visual or sensorimotor aura)—was used to identify migraine cases. Validation studies indicate this diagnostic tool has a sensitivity of 85% and specificity of 97% using a clinical assessment to assign a diagnosis based on the ICHD-II criteria as the gold standard (10,11). A random sample of controls, who did not meet the migraine case definition, was also selected for study participation. Survey respondents were classified as: (i) strict migraine (ICHD II 1.1 and 1.2) if they met all criteria for migraine with or without aura; (ii) probable migraine (ICHD-II 1.6) if they fulfilled all criteria but one for migraine with or without aura; and (iii) control if they did not meet the criteria for either strict or probable migraine. All aspects of the study were approved by the medical group's Institutional Review Board.
Data sources and analytical variables
Interview data
In addition to the headache diagnostic questions, the telephone interview included the Migraine Disability Assessment Scale (MIDAS) (12–14), a standardized quality-of-life assessment (the SF-12 (15)), and socio-demographic questions, thus enabling age, sex, race, education, marital status and health status via self-report. Headache-related disability was assessed using the MIDAS categorized as 0–10 and 11 or greater.
Medical claims data
Interview responses were linked to medical claims records for the 12-month period immediately preceding interview date. Available medical claims data included all services (regardless of diagnosis) delivered by the medical group and those services reimbursed by the affiliated health plan but not delivered by the medical group. Cost estimates were derived using institutional cost-to-charge ratios for those services delivered by the medical group. For prescription drugs (see below) and those services delivered by other organizations, but paid for by the health plan, the amount paid by the health plan was used. Thus, ‘cost’ reflects the perspective of an integrated health system that is at risk for externally delivered services.
We also used claims data to construct a variable reflective of whether or not the individual had a migraine diagnosis [International Classification of Diseases (ICD)-9 = 346.xx], any headache diagnosis (ICD-9 = 784.0, 307.81, 339.0, 346.xx, 672.2), depression diagnosis (ICD-9 = 296.2, 296.3, 298.0, 300.4, 311) or anxiety diagnosis (ICD-9 = 292.89, 293.84, 300.00, 300.01, 300.02, 300.1, 300.10, 300.20, 308.0, 309.21, 309.24, 309.28) at any time in the 12-month period preceding interview.
Pharmaceutical claims data
Prescription drug dispensing information was obtained from pharmacy claims for the 12-month period immediately preceding interview date. Using available National Drug Codes and class codes, medications dispensed were considered in three categories: antidepressants, anticonvulsants, and antihypertensives. Medications considered under each category were those consistent with US Headache Consortium recommendations (16) and known clinical practice. Patients with at least two dispensings in the 12-month period preceding survey administration were defined as users of migraine preventive medications.
Among users, adherence to migraine preventives was calculated in two ways (17,18). The first measure reflected adherence during the entire (or fixed) 12-month observation period (i.e. the cumulative days' supply of medication dispensed per claims data within the 365 days divided by 365 days). The second reflected adherence between the first and last dispensing date observed in the 12-month period (i.e. the cumulative days' supply of medication dispensed per claims data between first and last dispensing divided by the total number of days between first and last dispensing). These two measures effectively reflect a conservative and liberal calculation of a medication possession ratio. By convention, we consider patients with access to medications ≥ 80% of the time as adherent and those with less as non-adherent (19).
Statistical methods
Differences in patient characteristics and medication use by ICHD-II-based migraine status and claims data-determined diagnosis status as well as differences in patient outcomes by adherence status were evaluated using χ2 tests for categorical variables and t-tests for continuous variables. Due to the presence of non-normal distributions, Generalized Linear Model procedures with a gamma distribution and log link were used to assess the association between migraine preventive medication adherence status and medical and pharmaceutical costs. Three null hypotheses were tested: (i) there is no association between medication adherence and patient-reported quality of life; (ii) there is no association between medication adherence and patient-reported headache-related disability; and (iii) there is no association between medication adherence and medical and pharmaceutical care costs. All reported results are unadjusted.
Results
Cohort characteristics
Sample characteristics by International Classification of Headache Disorders, 2nd edn migraine case status

P < 0.1, **P < 0.05, ***P < 0.01 comparison between all migraine cases and controls.
P < 0.05, †††P < 0.01 comparison between strict and probable migraine cases.
Use of migraine preventive medications
Per cent using* preventive medications by International Classification of Headache Disorders, 2nd edn migraine case status and type of preventive migraine medication
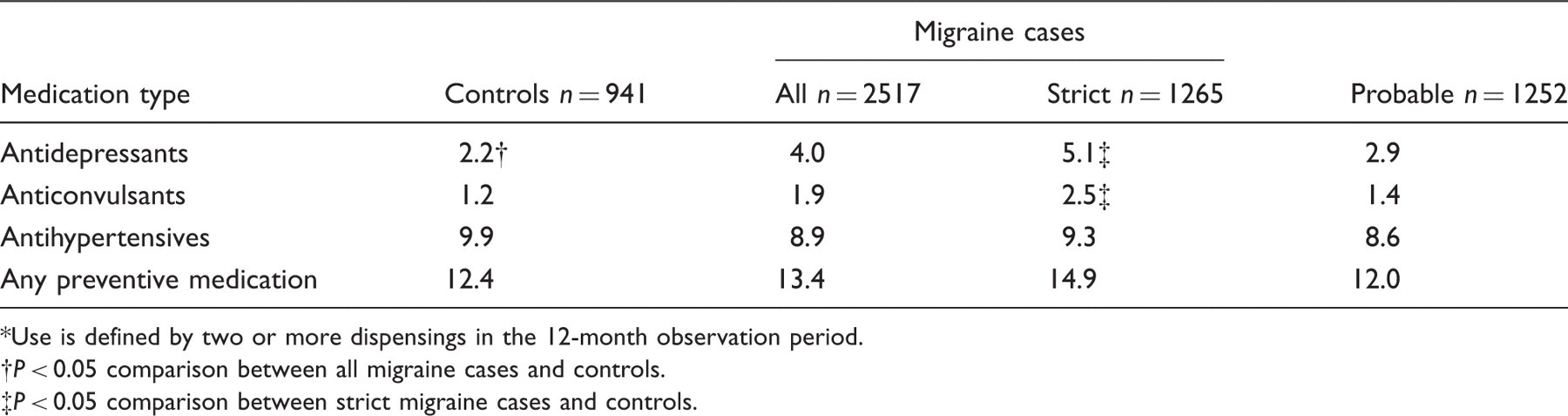
Use is defined by two or more dispensings in the 12-month observation period.
P < 0.05 comparison between all migraine cases and controls.
P < 0.05 comparison between strict migraine cases and controls.
Per cent using preventive migraine medication(s) use* by International Classification of Headache Disorders, 2nd edn migraine case status and medical claims-based diagnosis status

Any antidepressant, anticonvulsant or antihypertensive agent listed in Appendix 1.
P < 0.05, ††P < 0.01 comparison between diagnosed and non-diagnosed for each diagnosis category.
On the other hand, similar to the results observed above, patients not meeting criteria for migraine (i.e. controls) with a claims-based diagnosis of one of these conditions of interest were just as likely (P > 0.01) to have had two or more dispensings for a medication with known migraine preventive properties as their counterparts presenting with migraine symptoms (i.e. either all migraine or strict migraine cases) and a claims-based diagnosis for one of the conditions of interest. (That is, there is no significant difference in the prevalence of migraine preventive medication use by migraine status, controlling for each of the claims-based diagnoses of interest.)
Medication adherence
The mean preventive medication adherence rate between the first and last dispensing in the 12-month period ranged between 88% (for antidepressants) and 100% (for antihypertensives), but did not differ significantly by the type of medication. The mean adherence rate for the fixed 12-month period ranged between 47% (for anticonvulsants) and 58% (for antihypertensives) and likewise did not differ significantly by the type of medication. As there were no significant differences in adherence rates by the type of preventive medication for either measure of adherence (either overall or within cohorts defined by migraine status), we report findings for adherence to preventives as a group.
Mean adherence to migraine preventive medication(s) by International Classification of Headache Disorders, 2nd edn migraine case status and adherence definition

Medication adherence and patient outcomes
Patient outcomes by adherence to migraine preventive medications
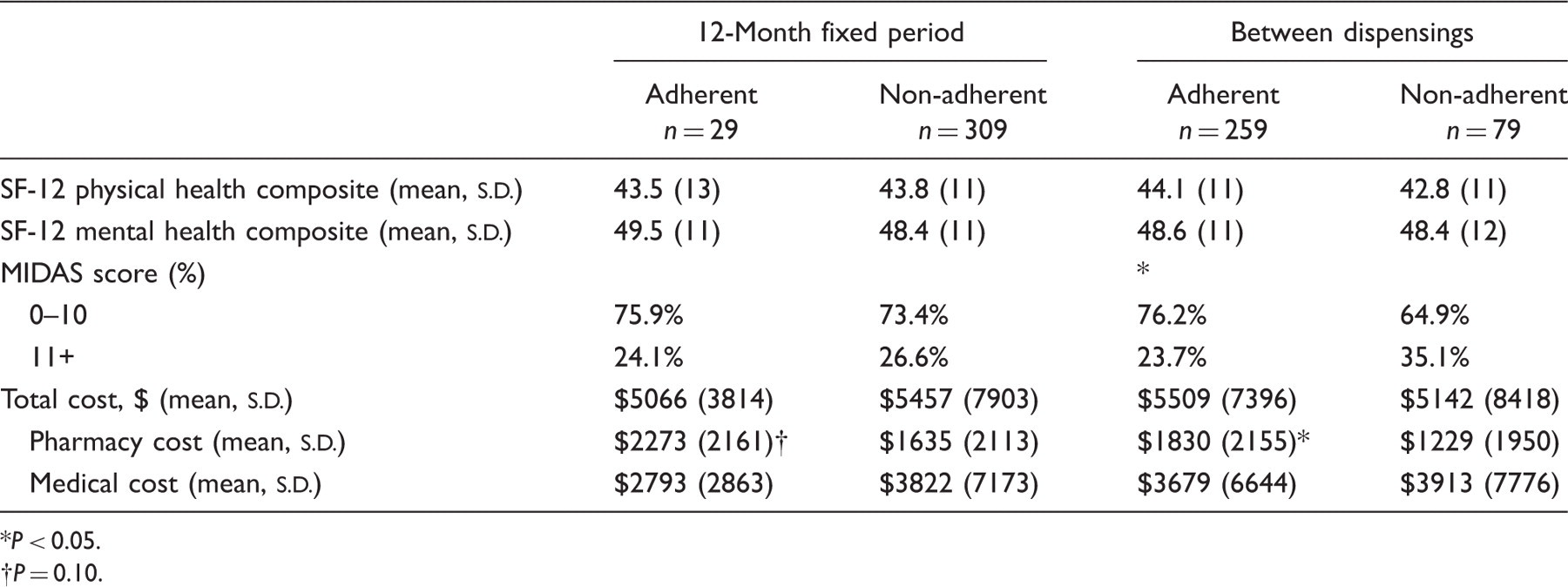
P < 0.05.
P = 0.10.
Discussion
Among an insured population, we found only 13.4% of those meeting the ICHD-II criteria for migraine headaches to have received two or more dispensings for a medication with known migraine preventive properties. Although this rate rose to 14.9% when only those meeting the ICHD-II criteria for migraine with and without aura are considered, we found no significant differences in the use of medications with known migraine preventive properties between patients meeting and not meeting the ICHD-II criteria for migraine headaches. Among preventive migraine medication users, we found adherence between the first and last dispensing during the year to be high (averaging 88%), reflecting consistent use between two anchoring dispensings. However, when the entire 12-month period was considered, adherence dropped substantially, reflecting the fact that users tended not to have these medications available for the full year. Furthermore, adherence between two anchoring dispensings was associated with less reported migraine-related disability and higher pharmaceutical costs, but not significant increases in overall costs.
An extensive body of trial evidence illustrates that preventive pharmacotherapy for migraine can reduce headache frequency and related disability as well as improve health-related quality of life (4,20,21). Based on this evidence, the US Headache Consortium Guidelines recommend preventive pharmacotherapy for migraine sufferers with frequent disabling migraine attacks. Despite the existence of such evidence and guideline recommendations, migraine preventive agents continue to be underused in clinical practice (5,21–24). Our finding that a minority of patients with migraine headache used preventive medications is consistent with other recent population studies (22,25) that report only 12% of migraine suffers used a migraine preventive medication.
Studying medications with known migraine preventive properties using administrative databases is challenging, in part, because these medications can be used for multiple clinical indications (26). Thus, even with reports of 12–13% of migraine sufferers taking such medications, the reason it was prescribed remains uncertain. By comparing the rate of use by migraine status, we are able to shed additional light in this area. We found no significant differences in use between patients meeting and not meeting the ICHD-II criteria for migraine (i.e. between migraine cases and controls) —probably indicating that at least some use among patients with migraine is for other indications. We did, however, find those patients meeting the ICHD-II criteria for strict migraine were significantly more likely to have received two or more dispensings for a medication with known migraine preventive properties compared with those not presenting with migraine symptoms, although this difference (2.5%) was quite small. Our findings also highlight the intermittent use of preventive therapy among those using medications with known migraine preventive properties: although adherence between two anchoring dispensings was high, when a 12-month period is considered, mean adherence was only 56% among migraine cases.
Taken together, these findings illustrate two potential paths to recommended migraine pharmacotherapy. First, patients with migraine are unlikely to be dispensed medications with known preventive properties. Second, among those fortunate enough to use them, many do not use them continually. For patients with migraine, this implies that they are not receiving the likely benefits available within existing pharmacotherapy options. Within the confines of this study, we are not able to determine whether patient factors, provider factors, health system factors, or a combination of these are the barriers to appropriate migraine preventive medication use and adherence, but previous research would support consideration of each of these (19).
Findings also point to the potential existence of benefits from migraine preventive medications in practice. We found that, among migraine cases, those adherent to preventive medications reported less headache-related disability compared with those who were non-adherent to preventive medications. Furthermore, benefits from the use of migraine preventive medications may accrue not only to patients themselves, but also more broadly. Although costs related to prescription drugs were, not surprisingly, higher among patients adherent to migraine preventives, there was a trend towards a reduction in overall medical care costs. Previous studies have shown that migraine patients incur higher medical care costs when compared with the general population—often related to frequent physician and emergency department visits (9,27). However, Silberstein and colleagues have found that medication use (for other conditions), and visits to the emergency department and physicians' offices, were significantly reduced when preventive migraine treatment was introduced in migraine management strategies (28). Others have found savings associated with reduced migraine frequency due to preventive treatment (29). Consistent with these previous findings, our results imply that additional pharmaceutical costs associated with use of migraine preventive medications may be offset by reduced spending related to office visits, emergency departments and hospitalizations.
Our study is not without limitations. First, use of a survey tool to differentiate between those with and without migraines may have led some individuals with migraine headaches to be labelled as controls. Given our previous studies (10,11), we expect this number to be small, as the interview has high sensitivity. Nonetheless, classification errors could cause us to overestimate burden in the control group and make migraine and control groups appear more similar. Similarly, migraine cases may include some individuals who do not have migraine, again potentially attenuating group differences. Second, knowledge of medication use among the study population is limited to that available within pharmaceutical claims data. Thus, although we know which patients were dispensed a medication of interest, we do not know if these medications were consumed by the patient. Similarly, because we used pharmacy claims from one insurer only, we do not know if a patient was given a prescription for a preventive medication and elected not to fill it or elected to fill it under a different policy. Third, data are from a cross-sectional study and therefore do not allow causal inferences. As our sample included small numbers of migraine preventive medication users, small sample size is also a potential issue. Finally, care should be taken when generalizing findings to other populations.
Migraine is a highly prevalent and disabling disorder (30,31). Many migraine sufferers do not consult healthcare professionals explicitly for headache (7,32), and those seeking care often do not receive a diagnosis of or treatment for migraine headache (5,7,32). Not surprisingly, those diagnosed with migraine or headache are more likely to receive a dispensing for a medication with known migraine preventive properties. Thus, one means by which to improve treatment rates is to improve diagnosis rates. Given the availability of existing screening questionnaires with proven sensitivity and specificity (33), community- and practice-based programmes to improve diagnosis rates should be evaluated.
Footnotes
Competing interests
R.B.L. has consulted for or conducted studies funded by Astra Zenica, Ortho McNeil, Glaxo Smith Kline, Pfizer and Merck among other companies.
Acknowledgements
This research was supported by the Blue Cross Blue Shield of Michigan (BCBSM) Foundation based on data collected with the support of AstraZeneca Pharmaceuticals.
Appendix 1 Preventive migraine pharmacotherapy used among the study cohort
| Medication type | |
| Antihypertensives | Anticonvulsants |
| Hypotensives, | Anticonvulsants |
| ACE inhibitors | |
| Prinivil | Carbamazepine |
| Lisinopril | Gabapentin |
| Hypotensives, angiotensin receptors | Topiramate |
| Candesartan | Valproic acid |
| Calcium channel blocking agents | Antidepressants |
| Nifedipine | Selective serotonin reuptake inhibitors |
| Diltiazem | Fluoxetine |
| Verapamil | Tricyclic antidepressants |
| β-Adrenergic blocking agent | Amitriptyline |
| Metoprolol | Serotonin-norepinephrine reuptake |
| Atenolol | Venlafaxine |
| Inderal | |
| Propranolol | |
| Timolol | |

