Abstract
Background
Therapeutic patient education (TPE) has been understudied for the management of chronic migraine (CM).
Objective
To study the effects of TPE with standard of care (SOC) versus SOC alone on patient-related outcome measures (PROM) in CM.
Methods
CM patients who were not on any preventive drugs for the past 3 months, aged 18–65 years, were randomized to receive TPE with SOC versus SOC alone and followed up for 12 weeks. TPE was imparted by a medical student following a rigorous 4-week training program by two senior headache specialists. TPE was imparted in the form of interactive group sessions lasting at least 20 minutes every 2 weeks. Patients in the control group received only SOC and instructions on dosing. The primary endpoint was the change in the Migraine-Specific Quality of Life (MSQOL) scores at Week 12. The key secondary outcomes were changes in the Headache Impact Test-6 (HIT-6) and Migraine Disability Assessment (MIDAS) scores at Week 12. Other secondary endpoints were also assessed. Efficacy analyses were performed in a modified intention-to-treat population who had at least one follow-up visit at Week 4. The trial was registered with the Clinical Trial Registry of India (CTRI/2022/06/04361).
Results
One hundred twenty-six patients were randomized. One hundred and seventeen patients (57 in the treatment and 60 in the control group) were available for the final efficacy analysis. The treatment group, compared with the control, showed a significant improvement from baseline to Week 12 in primary and key secondary endpoints. The mean change in MSQOL score was −11 (95% CI: −15.8 to −6.1); p-value < 0.001; HIT-6 scores, −6.4 (95% CI: −9.4 to −3.3); p-value < 0.001, and MIDAS scores −8.9 (95% CI: −17.2 to −0.6); p-value = 0.04, favoring the treatment group. Other secondary endpoints also showed superior outcomes for the treatment group.
Conclusion
TPE as an adjunct to SOC improved migraine-specific quality of life and headache-related disability in patients with CM.
This is a visual representation of the abstract.
Keywords
Introduction
The International Classification of Headache Disorders (ICHD)-3 defines chronic migraine (CM) as headaches occurring for ≥15 days/ month for more than 3 months, with migraine headaches occurring for ≥8 days/month. 1 With an estimated worldwide prevalence of 1.4–2.2%, 2 CM has been associated with severe disability, reduced quality of life, increased psychiatric toll, and higher healthcare costs. 3 CM has also been shown to have a negative impact on key life decisions, including those related to career, finances, and relationships. 4
Parallel to the increased burden of disease, there is a dearth of optimum pharmacological interventions for treating and preventing chronic migraine. 5 Current FDA-approved drugs, such as Onabotulinum toxin-A and calcitonin gene-related peptide antagonists, are costly and particularly inaccessible in low-resource countries. Standard oral preventives either lack evidence of efficacy or have significant side effects, resulting in poor adherence and suboptimal treatment outcomes. 5 The high prevalence of medication overuse headaches co-existing with CM makes its management even more challenging. 5 There is thus a pressing need to shift our focus toward non-pharmacological interventions. Therapeutic patient education (TPE) is one such modality, which entails a “structured person-centered learning process” so that individuals with chronic conditions can “self manage their health by drawing on their own resources,” as per the World Health Organization. 6 Despite promising evidence for TPE in various chronic pain conditions, its utility in managing CM is understudied. A recent scoping review has noted that data on TPE has mostly come from high-income countries, and there is a need to study its efficacy in middle and low-income countries as well. 7
Given this premise, we conducted a 12-week randomized controlled trial to study the efficacy of TPE as an adjunct to standard-of-care (SOC) treatment versus SOC alone for the management of CM at a tertiary public healthcare institute in India. We hypothesized that TPE as an adjunct to SOC treatment would improve the patient-reported outcome measures (PROMs) compared with SOC alone in patients with CM.
Methods
Trial oversight
Approval of the study protocol, informed patient consent forms, validated questionnaires, and accompanying study materials was obtained from the Institutional Ethics Committee of Maulana Azad Medical College and the associated Govind Ballabh Pant Institute of Postgraduate Medical Education and Research (GIPMER), New Delhi, India. Informed patient consent was taken from all participants prior to screening. The study was conducted in accordance with the International Council for Harmonisation Good Clinical Practice Regulations and Guidelines and followed the principles of the Declaration of Helsinki. The authors take full responsibility for the conduct of the trial, adherence to the protocol, the accuracy and completeness of the data and analyses, and the reporting of adverse events. The study is registered with the Clinical Trial Registry of India (CTRI/2022/06/04361).
Trial participants
CM patients (with or without aura) fulfilling the ICHD-3 criteria who were not on any preventive drugs for the past 3 months but were only on acute treatments, attending a tertiary care headache clinic at an academic institute (GB Pant Institute of Post Graduate Medical Education and Research, New Delhi), aged between 18–65 years were screened for the study from 7 July 2022 till 19 January 2023. Patients with a clinical phenotype of CM but, on further investigation, found to have a secondary cause for their headache were excluded.
Trial design
This randomized controlled study employed TPE as an intervention in combination with SOC for treating CM and compared it with SOC alone. The CONSORT guideline for randomized controlled trial was followed. TPE was imparted by MG, a medical student, following a rigorous 4-week training program that included weekly hour-long lectures on CM education with a structured course material (Supplementary Table 1) provided by DC and AD (senior consultant neurologists and headache specialists), as well as weekly observation at the Headache Clinic. This was followed by the screening phase (Weeks -4 to 0), during which baseline parameters of consecutive patients (including demographic details, headache characteristics, and baseline variables) were recorded using a structured proforma. Patients were instructed to complete a paper headache diary for 4 weeks, in which they recorded headache characteristics, including migraine and headache days, headache intensity using a visual analog scale (VAS) ranging from 0 to 10, and acute migraine treatment (AMT) days, which included the intake of analgesics and triptans.
Patients fulfilling the inclusion criteria were randomly assigned using a computer-based block randomization chart (blocks of 4 and 6) and allocated to treatment or control groups (1:1). Sequentially numbered opaque sealed envelopes and 126 labels were prepared for each group labeled A or B. They were put into envelopes numbered from 1 to 126 according to the randomization chart by investigator 3 (AD). Envelopes were attached to the patient's proforma. Patients were recruited sequentially and were given enrollment numbers starting from 1, which was mentioned on their outpatient cards by investigator 3 (AD). Proforma carrying the same number as the patient enrolment number was then opened, and the patients were assigned to receive intervention by investigator 2 (MG). Patients in the treatment group received TPE alongside SOC medication in the form of interactive group sessions lasting at least 20 minutes every 2 weeks. Patients in the control group received only standard-of-care medications and were given instructions on dosing. Both groups were assessed at the end of Weeks 4, 8, and 12 by investigators 1(DC) and 3 (AD) (Figure 1). Patients who had at least one follow-up visit (at Week 4) following intervention (TPE or standard-of-care alone) were included in the final efficacy analysis.

Trial design.
Therapeutic patient education
TPE was imparted by investigator 2 (MG), a medical student, as a pre-formed script to ensure treatment fidelity across different sessions. The session included elements of key patient competencies of knowledge, skill, and confidence as described by WHO 6 (Supplementary Table 2). The contents of the material clearly stated the diagnosis. Briefly, the pathogenesis was first explained (as a primary headache disease with both genetic and environmental factors that cause increased sensitivity to external stimuli and a decreased pain threshold). Patients were then explained the features of a migraine headache versus a non-migraine headache and the non-headache symptoms associated with migraine (such as nausea, photophobia, phonophobia, cranial autonomic symptoms, etc.) so that patients were better able to identify their headaches. Designed as an interactive session, patients were allowed to share their experiences at any point during the discussion to facilitate better understanding and recognition of features amongst all patients. Patients were reassured that their headache was not life-threatening and that, contrary to the apprehensions of many patients, there were no underlying structural abnormalities, brain tumors, or infections. Patients were then briefed about possible triggers and how to identify them. They were introduced to the concepts of acute and preventive medications and given a detailed explanation of the proper dosing of each, as well as the importance of compliance and avoiding medication overuse. Lastly, patients were explained the chronic nature of the disease and the need for lifestyle modifications that could significantly improve quality of life, including regularizing sleep and eating habits, identifying and avoiding triggers to the best possible extent, incorporating regular exercise, and avoiding excessive weight gain, as well as maintaining a regular headache diary. The session concluded with an open discussion about patients’ perceptions of the information and clarification of any doubts. Six TPE sessions each lasting between 20 and 30 minutes were offered to CM patients in groups no more than 10 patients each.
Trial efficacy endpoints
The primary endpoint of this study was the Migraine-Specific Quality of Life (MSQOL) version 2.1 questionnaire 8 scores at Week 12 adjusted to baseline. The key secondary outcomes were the Headache Impact Test-6 (HIT-6) 9 and Migraine Disability Assessment (MIDAS) 10 scores at Week 12 adjusted to baseline. Other secondary endpoints included changes in mean migraine days per month, mean headache days per month, mean acute migraine treatment (AMT) days per month, and mean visual analog scale (VAS) score (0–10) from baseline, measured as repeated measures every 4 weeks for 12 weeks. A migraine day and a headache day were defined as a calendar day on which the patient recorded ≥4 continuous hours of migraine phenotype headache (as per ICHD-3) or any form of headache, respectively, in the headache diary. Additional secondary endpoints included the patients in each group with at least a 50% reduction in headache days from baseline till Week 12 (50% responder rate), changes in mean headache management self-efficacy (HMSE), 11 and pain catastrophizing scale (PCS) 12 scores from baseline to Week 12 and documentation of adverse effects (AE) and serious AE.
Sample size calculation
The expected treatment effect difference between the treatment and control groups was estimated from a previous trial comparing TPE with mindfulness. 13 In this study, the mean MSQOL score at 3 months was 38.6 (95% CI: 35.6–41.6) versus 43.5 (95% CI: 40.0–47.1) at baseline for the group receiving TPE. With a two-sided alpha of 0.05 and a beta of 80%, the estimated total sample size was 114 patients. To account for a 10% loss to follow-up, a minimum of 126 (63 in each group) subjects were enrolled in the study.
Statistical analysis
Categorical data were summarized as frequencies and percentages, while continuous data were summarized as means (±standard deviation). Student t-test was used to compare the means. The X2 test with Yates correction and Fisher's exact test was used when appropriate, along with the Odds ratio (OR) and 95% confidence interval (CI), to compare proportions between the groups. The primary and key secondary endpoints at Week 12, which were measured at baseline and the end of Week 12, such as changes in mean MSQOL scores, MIDAS scores, and HIT-6 scores, were analyzed using a generalized linear model with treatment group and medication overuse [presence vs absence] as factors and baseline MIDAS (−12 to 0 weeks), MSQOL and HIT-6 scores during observation period (−4 to 0 weeks) and years since migraine onset as covariates without imputing the missing data. For the other secondary efficacy endpoints that were measured every 4 weeks following randomization, the least-squares mean at each time point was calculated with a linear mixed effects model that included participant as a random effect and number of headache days during the 4-week observation period (baseline: −4–0 weeks) and years since migraine onset as covariates. The model included fixed effects for the treatment group, medication overuse [presence vs absence], study month in the treatment phase (month 1 was 4 weeks, month 2 was 8 weeks, and month 3 was 12 weeks), and the month-by-treatment group interaction without imputation of missing data. We reported the least-squares mean change from baseline for each treatment group, treatment difference compared with placebo, 95% CI, and p-values for pairwise comparison. The 50% responder rate was estimated using a stratified Cochran-Mantel-Haenszel test. Missing patients were considered as non-responders. The level of significance was set at p<0.05. A prespecified hierarchical gatekeeping strategy was used to maintain the overall type I error rate at a two-sided alpha level of 0.05 in which the significance of the key and other secondary endpoints would be evaluated only if a significant treatment effect in the primary efficacy endpoint was achieved. Statistical analysis was done with SPSS version 25 and R statistical software (v 4.1.2; R core Team 2021).
Results
One hundred twenty-six patients were randomized, with 63 patients in each of the treatment and control groups. Baseline data for one patient from the treatment group were lost during processing and hence were not included in the analysis. Demographic data and baseline headache characteristics of the remaining 125 patients, comprising 62 in the group that received TPE and SOC and 63 in the group that received SOC alone, are presented in Table 1 and are comparable. The mean ages of the patients in the two groups were 36 (±10.8) years and 34.6 (±10.5) years, with most patients being female: 52/62 (83.9%) and 57/63 (90.5%), respectively. The baseline MSQOL scores of the patients receiving TPE and SOC were 58.3 (±1.7), compared with 58.1 (±1.5) for those receiving SOC alone. Other baseline measures of trial efficacy endpoints were also comparable between the two groups and are tabulated in Table 2. The flow of the patients through the study as per CONSORT guidelines is shown in Figure 2.

Flow of the study.
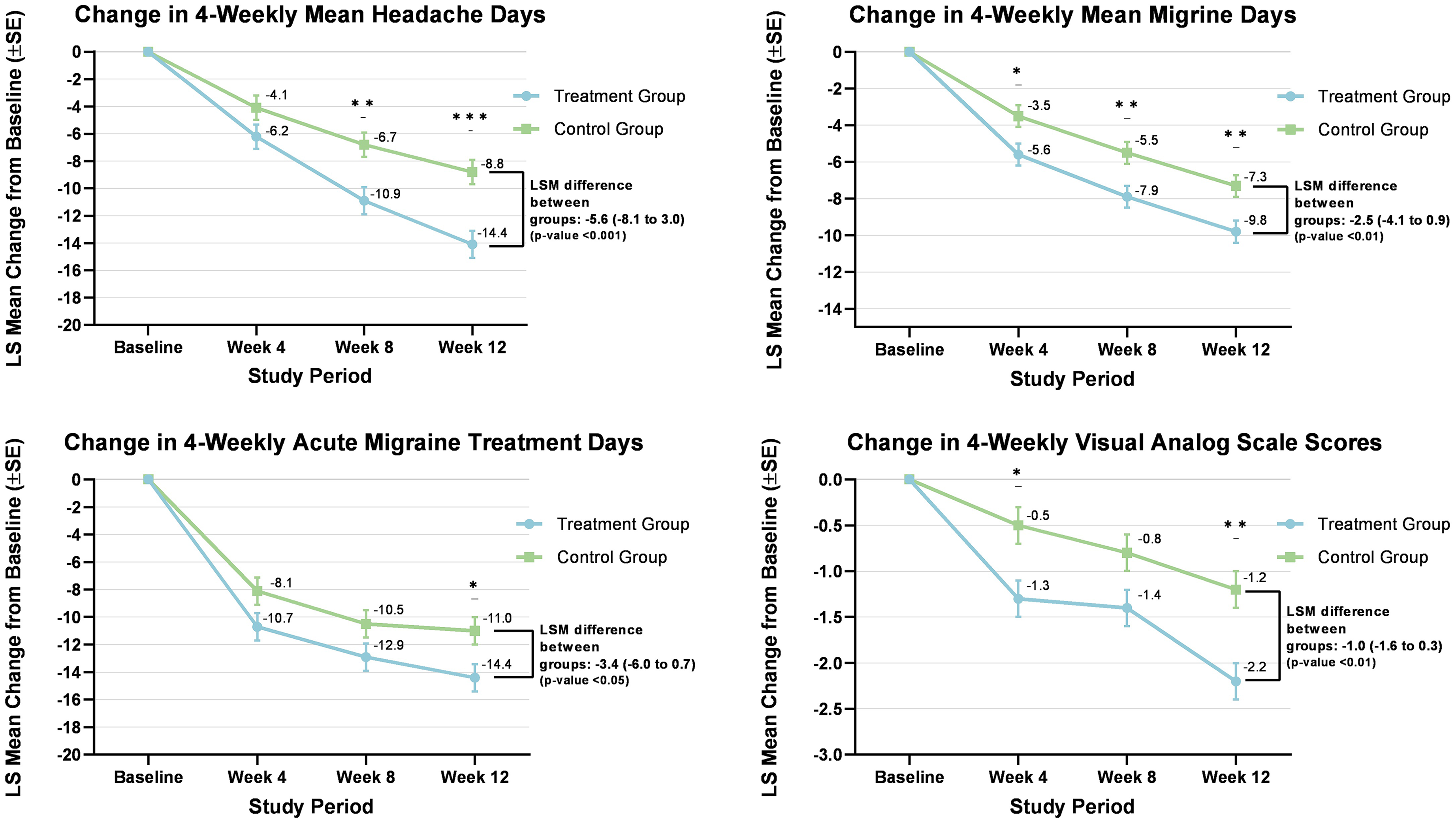
Change in other secondary endpoints.
Baseline characteristics of study participants according to trial groups*

Plus-minus values are means ± SD. The intention-to-treat population included all patients who underwent randomization for analysis of baseline characteristics. Patients in the treatment group received a combination of therapeutic patient education and standard-of-care medications, while patients in the control group received standard-of-care medications alone.
Baseline measures of trial efficacy endpoints
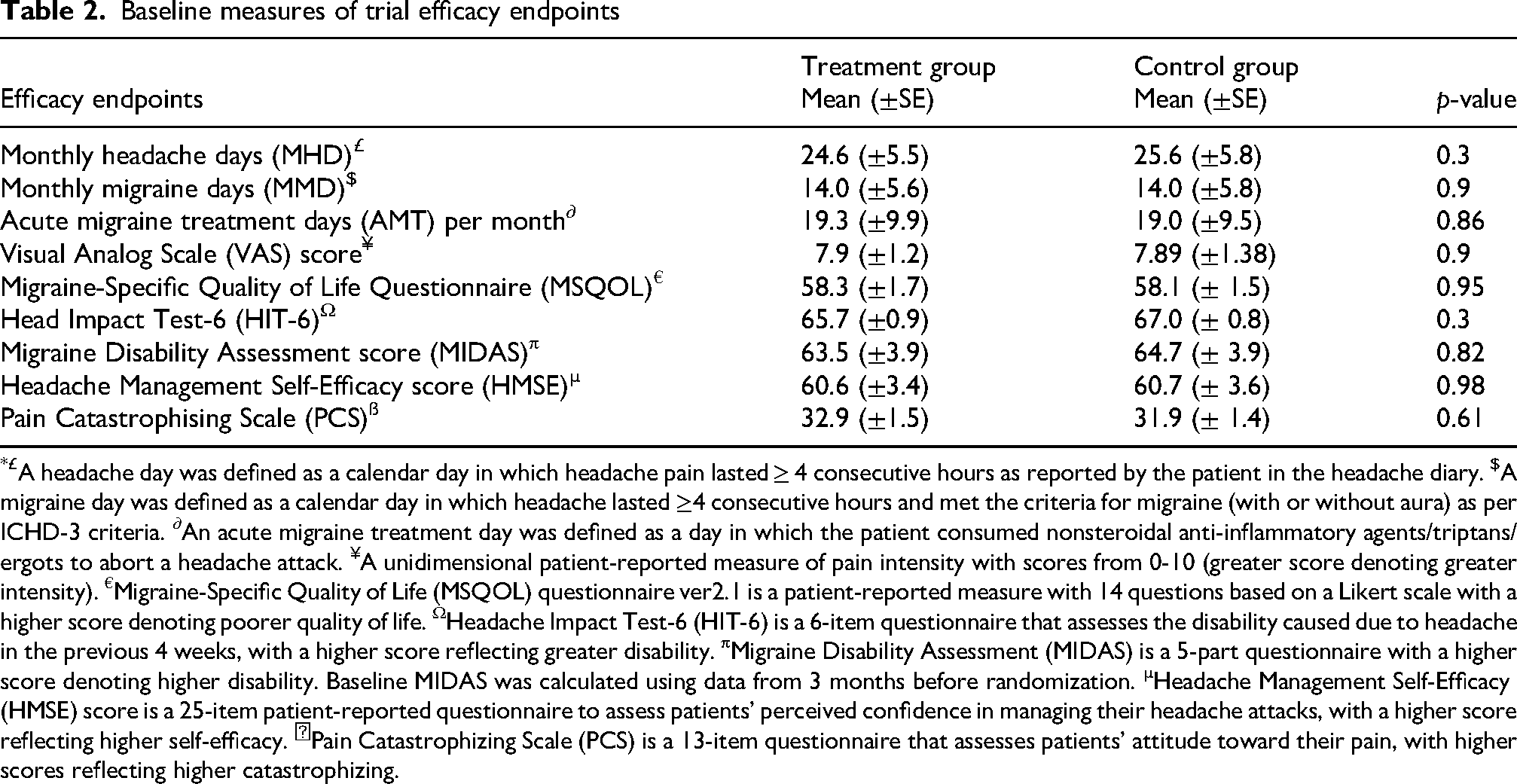
£A headache day was defined as a calendar day in which headache pain lasted ≥ 4 consecutive hours as reported by the patient in the headache diary. $A migraine day was defined as a calendar day in which headache lasted ≥4 consecutive hours and met the criteria for migraine (with or without aura) as per ICHD-3 criteria. ∂An acute migraine treatment day was defined as a day in which the patient consumed nonsteroidal anti-inflammatory agents/triptans/ergots to abort a headache attack. ¥A unidimensional patient-reported measure of pain intensity with scores from 0-10 (greater score denoting greater intensity). €Migraine-Specific Quality of Life (MSQOL) questionnaire ver2.1 is a patient-reported measure with 14 questions based on a Likert scale with a higher score denoting poorer quality of life. ΩHeadache Impact Test-6 (HIT-6) is a 6-item questionnaire that assesses the disability caused due to headache in the previous 4 weeks, with a higher score reflecting greater disability. πMigraine Disability Assessment (MIDAS) is a 5-part questionnaire with a higher score denoting higher disability. Baseline MIDAS was calculated using data from 3 months before randomization. µHeadache Management Self-Efficacy (HMSE) score is a 25-item patient-reported questionnaire to assess patients’ perceived confidence in managing their headache attacks, with a higher score reflecting higher self-efficacy. ẞPain Catastrophizing Scale (PCS) is a 13-item questionnaire that assesses patients’ attitude toward their pain, with higher scores reflecting higher catastrophizing.
One hundred and seventeen patients were included in the final efficacy analysis. Of the 62 patients who received TPE, 49 (79%) attended 4 or more TPE sessions out of possible 6. The distribution of the TPE session is shown in Supplementary Table 3. The SOC consisted of prescriptions of paracetamol, NSAIDS, naproxen and domperidone, rizatriptan, sumatriptan with naproxen, and zolmitriptan nasal spray as acute medications. For prophylaxis, both the TPE and control groups received propranolol, topiramate, amitriptyline, botulinum toxin-A, and greater occipital nerve blocks either alone or in combination (details of the treatment are shown in Supplementary Table 4).
Primary efficacy endpoint
The mean MSQOL score at Week 12 showed a significant improvement in the treatment group compared with the control group, with an LSM difference of −11 (95% CI: −15.8 to −6.1) (Table 3).
Primary and key secondary efficacy endpoints at week 12

Plus-minus values are means ± SE. Efficacy analyses were done using a modified intention-to-treat population, which included all patients who received at least one dose of intervention (TPE + SOC or SOC alone) and completed at least one follow-up visit (at week-4); The endpoints were analyzed using a generalized linear model with treatment group and medication overuse [presence vs absence] as factors and baseline MIDAS (−12 to 0 weeks), MSQOL and HIT-6 scores during observation period (−4 to 0 weeks) and years since migraine onset as covariates without imputing the missing data. MSQOL: Migraine-Specific Quality of Life; HIT-6: Headache Impact Test-6; MIDAS: Migraine Disability Assessment; Plus-minus values are means ± SE
Key secondary efficacy endpoints
The patients who received TPE showed significant mean reductions at Week 12 in HIT-6 and MIDAS scores compared with the control group (51.2 (±1.2) vs 57.6 (±1.1); LSM difference of −6.4 (95% CI: −9.4 to −3.3); p-value < 0.001; 25.5 (±3.2) vs 34.4 (±3.1); LSM difference of −8.9 (95% CI: −17.2 to −0.6); p-value = 0.04, respectively). (Table 3)
Other secondary endpoints
Among the patients receiving TPE, 73.7% recorded a reduction of 50% or more in headache days compared to baseline by Week 12, compared with 46.7% who received SOC alone (OR 3.2; 95% CI:1.5 to 7.0; p-value 0.003). The HMSE scores for headache self-efficacy showed significant improvement [LSM difference 26.4 (95% CI: 14.4 to 38.4); p-value < 0.001] and PCS scores for pain catastrophizing showed a significant reduction [LSM difference −7.6 (95% CI −11.8 to −3.3); p-value < 0.001] in patients who received TPE. MMD, MHD, AMT, and VAS scores showed significantly greater improvements at Week 12 in the group receiving TPE (Figure 3). Between the groups who received TPE with SOC versus those who received SOC alone, 4-weekly MMD changed to −5.6 vs. −3.5, −7.9 vs. −5.5, and −9.8 vs. −7.3 days at Weeks 4, 8, and 12, respectively. The LSM difference between the two groups at Week 12 was −2.5 days (CI: −4.1 to 0.9; p < 0.01). AE attributable to the pharmacotherapy was reported equally between the two groups, and no AE was related to TPE (Supplementary Table 5). No serious adverse effects were reported.
Discussion
This study demonstrated that TPE, in the form of group sessions led by a trained medical student, combined with SOC, is superior to SOC alone in enhancing the migraine-related quality of life in CM patients. Furthermore, the group that received TPE with SOC showed greater reductions in migraine-related disability, headache frequency, and severity, the number of days of AMT taken, pain catastrophizing, and an increase in headache self-efficacy, suggesting that all these parameters may have contributed to achieving a better quality of life in these patients. Although the pathophysiology of CM remains uncertain, it is well understood that it involves complex mechanisms, and headaches are just one component of the clinical spectrum. The non-headache symptoms can be equally debilitating for the patients. 14 Therefore, merely relieving the pain is not adequate for its optimal management. As this study focused on the effects of combining TPE with SOC on PROMs, three PROMs, namely MSQOL, HIT-6, and MIDAS, were chosen as primary and key secondary outcome measures. MSQOL has been extensively studied as a reliable tool for evaluating patients’ emotional and functional responses to their disease, which in turn translate to their quality of life. 15 Additional tools, such as the HIT-6 9 and MIDAS, 10 which have been extensively used in migraine research, quantify the disability caused by the disease and, together, help paint a picture of the impact of migraine on patients’ lives beyond just symptoms. The findings of this study of greater improvements in these PROM for the patients receiving TPE compared with those receiving only SOC is, therefore, reassuring. Furthermore, the absolute difference in these measures was also clinically meaningful. For example, we achieved an improvement in MSQOL of 11 and HIT-6 score of 6.4 points for the group receiving TPE compared with those receiving SOC alone. Studies have shown that the between-group minimal clinically important difference for the HIT-6 is a decrease of at least 2.3 points for CM patients. 16 For MSQ domains of the MSQ-RR, MSQ-PR, and MSQ-EF, the corresponding minimal clinically important differences are 3.2, 4.6, and 7.5 points, respectively. 8 For MIDAS, the improvement was 8.9 points for the treatment group, compared to a suggested cut-off of 4.5 points, the minimal clinically important change after non-pharmacological treatment. 17 Thus, this study showed that the primary and key secondary endpoints were not only statistically significant, but changes were also clinically meaningful for the treatment group. To the best of our knowledge, this is the first study that specifically assessed the efficacy of TPE in CM. Many previous studies have employed multiple bio-behavioral interventions simultaneously, such as education, relaxation techniques, and biofeedback, thus limiting the evidence of the efficacy of any one intervention.18–20 This fact was also commented upon in a recent systematic review. 21 This study employed only TPE and did not directly control patient behavior outside the outpatient department (OPD) setting, thus providing stronger evidence for its efficacy. Previous studies employed various modes of TPE delivery, including CD-DVD, 22 brochures, 23 and web-based interventions. 24 Compared with non-human forms of education, human-provided education has been shown to provide greater patient satisfaction. 25
It is also pertinent to note that the educational intervention in the present study was provided by a medical student who had received prior training in headache medicine from headache specialists. Previous studies employed nurses, 22 pharmacists, 26 and even trained lay migraineurs 27 to impart the TPE, with promising results. This highlights the fact that TPE need not be imparted only by headache specialists, and this can greatly reduce the burden of care on specialists. Further, this study employed TPE in the form of group teaching. A systematic review 21 suggests that group teaching might be superior to individual teaching, as it increases patients’ confidence and allows for the creation of a community of fellow migraineurs who can motivate each other to take behavioral action more effectively than a doctor's prescription alone. Despite the utility of TPE in various other chronic pain conditions, TPE approaches for CM have not been validated due to variable content, methods and frequency of administration, and a variety of instructors delivering the program. The superior efficacy of TPE, as shown in this study, therefore, should be interpreted with this caveat in mind. Despite this limitation, it has been argued that a biopsychological model for treating migraine patients holistically would be useful, and patient education forms an important tool in this model to tackle problems arising out of social environments, stressors, and patient behavior. 28
The present study recruited CM patients from a tertiary-level academic public healthcare institution in New Delhi, India. It may have biased the selection toward more severe cases of CM. However, given that a public hospital offers free healthcare, patients from low-income backgrounds with lower health literacy comprised a large section of the participants. Inadequate health literacy is known to be strongly associated with a low socioeconomic position, poor health status, inactivity, and overweight. 29 Appropriate education thus becomes even more significant in this population. It has also been shown that patients with a high chance locus of control (believing their disease is controlled by fate or outside factors) benefited more from the psychological intervention compared with patients with a low chance locus of control, possibly because they have the most to learn from such interventions. 20 TPE and support could be very important components for management of CM, especially in poor-resourced countries where access to the latest and costly migraine medications may be limited.
Two other secondary endpoints merit our attention. The first is pain catastrophizing, a well-studied phenomenon where a patient has an exaggerated negative attitude toward their pain, with easier recollection of past painful memories and feelings of doom associated with the present condition. This study found a significantly greater improvement in PCS scores in the TPE group, which we believe stemmed directly from the educational intervention, wherein we taught the patients the pathophysiology of CM in lay terms, reassured the patients about the non-fatal nature of the disease, and empowered them with the tools for lifestyle modifications that can help reduce their attack frequency. The other secondary endpoint was well-studied concepts of self-efficacy and locus of control, 20 which relate to patients’ perceived control over their health. Higher self-efficacy also helps combat the potential development of overdependence on medications as the sole means of headache relief (external locus of control), which is often seen in patients with chronic diseases. This study's findings of improved HMSE scores in patients who received TPE suggest that education about concepts of self-efficacy and locus of control can help them achieve a better quality of life, as reported in an earlier study. 20
There are some limitations of the present study. First, this is a single-center study. Second, this study did not employ a double-blind protocol, as devising an ideal “behavioral placebo” for patient education was difficult. Instead, the control group was given only SOC. Third, the intensity of treatments varied between the two groups as those receiving TPE plus SOC visited every 2 weeks, compared to 4 weekly visits by those receiving SOC alone. Hence, the possible Hawthorne effect, wherein subjects alter their behavior when they are aware that they are being studied more intensely, cannot be ruled out. Fourth, this study followed patients for only 12 weeks, and the long-term effects of TPE remain uncertain. At least 6 to 12 months follow up would have provided more conclusive data. Hence the results of this study should be considered as preliminary evidence for the efficacy of TPE in CM patients. Long-term randomized controlled trials (RCTs) are necessary to strengthen the evidence for TPE further. Finally, most of the patients in this study were females and hence the results might not be generalizable for males. Despite these limitations, this study provided evidence of efficacy for TPE as an adjunct to SOC for CM patients in improving MSQOL, migraine-related disability, headache self-efficacy, and several clinical headache characteristics, including headache frequency, severity, and AMT days, over a 12-week period.
Clinical implications
Therapeutic patient education (TPE) as an adjunct to standard of care improved migraine-specific quality of life and migraine-related disability.
Improvement in MSQOL, HIT-6, and MIDAS scores in the treatment group was not only statistically significant for those receiving TPE but was also clinically meaningful.
TPE, when administered every 2 weeks over a 12-week period with standard of care, also improved clinical headache characteristics such as headache frequency, severity, and acute migraine treatment days over a 12-week period.
The patients receiving TPE had significantly greater improvements in pain catastrophising and self-efficacy scores, signifying that education plays a vital role in patient self-empowerment.
Supplemental Material
sj-docx-1-rep-10.1177_25158163251391185 - Supplemental material for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures
Supplemental material, sj-docx-1-rep-10.1177_25158163251391185 for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures by Debashish Chowdhury, Mridula Garg and Ashish Duggal in Cephalalgia Reports
Supplemental Material
sj-docx-2-rep-10.1177_25158163251391185 - Supplemental material for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures
Supplemental material, sj-docx-2-rep-10.1177_25158163251391185 for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures by Debashish Chowdhury, Mridula Garg and Ashish Duggal in Cephalalgia Reports
Supplemental Material
sj-docx-3-rep-10.1177_25158163251391185 - Supplemental material for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures
Supplemental material, sj-docx-3-rep-10.1177_25158163251391185 for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures by Debashish Chowdhury, Mridula Garg and Ashish Duggal in Cephalalgia Reports
Supplemental Material
sj-docx-4-rep-10.1177_25158163251391185 - Supplemental material for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures
Supplemental material, sj-docx-4-rep-10.1177_25158163251391185 for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures by Debashish Chowdhury, Mridula Garg and Ashish Duggal in Cephalalgia Reports
Supplemental Material
sj-docx-5-rep-10.1177_25158163251391185 - Supplemental material for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures
Supplemental material, sj-docx-5-rep-10.1177_25158163251391185 for A randomized controlled trial on combining therapeutic patient education with standard care versus standard care alone for chronic migraine: Effects on patient-reported outcome measures by Debashish Chowdhury, Mridula Garg and Ashish Duggal in Cephalalgia Reports
Footnotes
Acknowledgments
We thank Debanjan Chowdhury, PhD Scholar, Heidelberg University, Germany, for his invaluable suggestions and inputs. Some of the results of this trial were presented during the International Headache Congress at Seoul in 2023.
Ethical considerations
The study was conducted in accordance with the International Council for Harmonisation Good Clinical Practice Regulations and Guidelines and followed the principles of the Declaration of Helsinki.
Consent to participate
Written informed patient consent was taken from all participants prior to screening.
Consent for publishing
Yes, the authors agree to publish with Cephalalgia Reports.
Author contributions
DC conceptualized the design of the trial. DC, MG, and AD organized the study. MG acquired the data. DC analyzed the data and performed the statistical analysis. DC and MG wrote the first draft of the manuscript and interpreted the findings. AD and MG critically reviewed the manuscript. All authors provided their input to the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was an academic trial, and the hospital provided the drugs free of cost.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The trial data will be made available on request, from the time of publication of the article, after approval of a formal written request. The corresponding author needs to be contacted for access to trial data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
