Abstract
Background
Following the CENTURION phase 3 randomized controlled trial’s four-month double-blind phase, this 12-month open-label extension collected data for up to one year about dose optimization, patterns of use, migraine-related disability, and quality of life during lasmiditan treatment.
Methods
Migraine patients ≥18 years completing the double-blind phase and treating ≥3 migraine attacks could continue into the 12-month open-label extension. The initial oral lasmiditan dose was 100 mg; the dose could subsequently be adjusted to 50 mg or 200 mg at the investigator’s discretion.
Results
477 patients entered and 321 (72.1%) completed the extension; 445 (93.3%) treated ≥1 attack with lasmiditan. Of 11,327 attacks, 8654 (76.4%) were lasmiditan-treated (84.9% of these involved moderate or severe pain). By study end, 17.8%, 58.7%, and 23.4% of patients were taking lasmiditan 50, 100, and 200 mg, respectively. Mean improvements were observed in disability and quality of life. The most common treatment-emergent adverse event was dizziness (35.7% of patients, 9.5% of attacks).
Conclusions
During this 12-month extension, lasmiditan was associated with a high rate of study completion, most attacks were treated with lasmiditan, and patients reported improvements in migraine-related disability and quality of life. No new safety findings were observed with longer exposure.
Introduction
Lasmiditan is a selective serotonin 1F (5-HT1F) receptor agonist approved for the acute treatment of migraine with or without aura in adults (1). In three phase 3 clinical studies, SAMURAI (2), SPARTAN (3), and CENTURION (4), lasmiditan demonstrated significant superiority versus placebo in the percentage of patients who were pain-free at two hours post dose. In CENTURION, consistency of response to lasmiditan was also demonstrated versus placebo, as measured by pain freedom or pain relief at two hours in ≥2 of three migraine attacks (4).
In these prior clinical studies, as well as the GLADIATOR long-term safety study, patients were assigned specific lasmiditan doses without dose adjustment, so optimizing efficacy and tolerability was not an option (2–5). Patients were also required to complete serial assessments during acute migraine attacks. Finally, in previous clinical trials patients waited to treat their headache until it had become moderate-to-severe, whereas many migraine patients may wish to treat their migraine sooner, as is typically recommended (6,7).
We report findings from a 12-month open-label extension (OLE) of the CENTURION study. The purpose of this OLE was to collect data on patients’ use of lasmiditan for up to one year with dose optimization, without electronic diary assessments, and without a requirement to wait until pain became moderate-to-severe. Data are reported on migraine-associated disability as assessed by the Migraine Disability Assessment Score (MIDAS), real-world patterns of use, patient satisfaction, and health-related quality of life.
Methods
Ethics
This study was conducted in accordance with the International Conference on Harmonization Guidelines for Good Clinical Practice and the Declaration of Helsinki and was approved by each institution’s ethical review board. Patients provided written informed consent before enrollment. This study is registered with the European Union Drug Regulating Authorities Clinical Trials Database (EUDRA CT: 2018-001661-17) and ClinicalTrials.gov (NCT03670810).
Study design and patients
This was a 12-month OLE of a four-month, phase 3, randomized, placebo-controlled, double-blind study (Figure 1). The OLE took place at 61 participating sites across 12 countries in Europe and North America. Patients entering the 12-month OLE were initially assigned to 100 mg oral lasmiditan, with flexible dosing (50, 100, or 200 mg) thereafter at the investigator’s discretion to optimize efficacy and tolerability. Patients were asked to treat migraine attacks with lasmiditan when possible and were permitted to take lasmiditan for each new migraine attack with ≥24 hours between doses. If a patient was unable to treat with lasmiditan during a particular migraine attack, they were permitted to use their usual migraine medication for that attack. Patients were not to drive, operate heavy machinery, or engage in other similar activity for eight hours after taking lasmiditan.

Study design.
Selection criteria
During the double-blind study, eligible men and women aged ≥18 years (no upper limit) with migraine with or without aura and with a history of three to eight migraine attacks per month (<15 headache days per month) were to treat four attacks or until four months after randomization. Eligibility criteria are detailed in a previous publication (4). Patients were permitted to continue in the 12-month OLE if they treated ≥3 attacks during the double-blind study with study drug and did not discontinue early from the double-blind study, regardless of randomization.
Endpoints
The primary endpoint was the mean change from baseline of the double-blind main study in the MIDAS total score. Secondary endpoints included: mean change from baseline of the double-blind main study in MIDAS migraine severity, headache days, and score categories; mean changes from OLE baseline in the Migraine-Specific Quality of Life Questionnaire (MSQ) v2.1 total score, and Role Restrictive, Role Preventive, and Emotional Function domain scores; Migraine Treatment Optimization Questionnaire (mTOQ-6) scores at the end of the study (EoS) to evaluate treatment optimization; and time of dosing, severity at time of dosing, rescue and recurrence medications, and reasons for not taking lasmiditan at each postbaseline visit to describe patients’ pattern of lasmiditan use.
Additional secondary assessments were: the treatment satisfaction questionnaire at EoS; the percentage of patients reporting each response on the Patient Global Impression of Change – Migraine Headache Condition (PGIC-MHC) at EoS; the percentage of patients reporting each response on the Patient Global Impression of Severity (PGI-S) at OLE baseline and EoS; the number of migraine attacks per each 30-day interval during the OLE; the mean visitwise change from the baseline of the main study in employment status and the Health Care Resource Utilization (HCRU) questionnaire; and assessment of driving accidents and violations. Detailed descriptions of the assessment tools are available in the online Supplemental Material.
Statistical analysis
Unless otherwise noted, all analyses were as prespecified within the original study protocol addendum. Analyses were conducted on intent-to-treat (ITT) patients who received ≥1 dose of study drug during the OLE. Mean change from main study baseline in MIDAS total score, mean changes from main study baseline in migraine severity and headache days (per MIDAS), and mean changes from OLE baseline in the MSQ v2.1 total score and domain scores were analyzed using mixed model for repeated measures analyses with longitudinal observations.
Summary of frequencies analyses were used to analyze mTOQ-6 scores at EoS. General summaries were used to analyze pattern of lasmiditan use.
Additional assessments analyzed using summaries of frequencies were: the treatment satisfaction questionnaire at EoS; the percentage of patients reporting each response on the PGIC-MHC at EoS; the percentage of patients reporting each response on the PGI-S at OLE baseline and EoS; and the mean visitwise change from the baseline of the main study in employment status. The number of migraine attacks per each 30-day interval during the OLE was analyzed using a summary of counts by month of exposure. The mean visitwise change from the baseline of the main study in the HCRU questionnaire was analyzed using the Wilcoxon signed-rank test.
Results
Patient disposition
In all, 477 patients entered the OLE and 445 treated ≥1 migraine attack with lasmiditan. Of the 445 patients in the ITT population, 167 (37.5%) had been randomized to placebo, 146 (32.8%) had received 100 mg lasmiditan, and 132 (29.7%) had received 200 mg lasmiditan during the double-blind main study before entering the OLE. Patient disposition is detailed in Figure 2.

Patient disposition.
Of the total ITT population (n = 445), 321 patients (72.1%) completed the OLE study. Withdrawal by subject was the most frequent reason for discontinuing from the OLE (n = 38; 8.5%), followed by lack of efficacy (n = 36; 8.1%) and adverse events (n = 22; 4.9%). The reasons for discontinuing from the study are summarized in Figure 2.
Six patients who discontinued study and study treatment (withdrawal by subject or other) during the OLE included coronavirus disease (“COVID”) in the detailed reason: COVID-19 (n = 2); does not wish to take lasmiditan during COVID-19 pandemic (n = 1); due to COVID-19 restrictions (n = 1); COVID mitigation issues (n = 1); patient partner suffering with long COVID-19, therefore, cannot commit to the study (n = 1).
Exposure
During the OLE, 441 patients (99.1%) took ≥1 dose of lasmiditan 100 mg. Per-patient dose modifications made during the OLE are detailed in Table 1. At EoS, the majority of patients (n = 261; 58.7%) were taking lasmiditan 100 mg, 104 (23.4%) were taking lasmiditan 200 mg, and 79 (17.8%) were taking lasmiditan 50 mg.
Summary of baseline demographic characteristics for OLE population.

IQR, interquartile range; MIDAS, Migraine Disability Assessment Score; N, number of patients in the analysis population; n, number of patients in the specified category; OLE, open-label extension; SD, standard deviation.
†Four patients (0.9%) did not take lasmiditan 100 mg at any point during the OLE, but took 50 mg (n = 2), 200 mg (n = 1), or an unknown alternative dose (n = 1).
Exposure for all patients and migraine attacks during the OLE is summarized in Table 2. A median of 15 attacks per patient were treated with lasmiditan.
Summary of exposure for all patients and all migraine attacks during OLE.
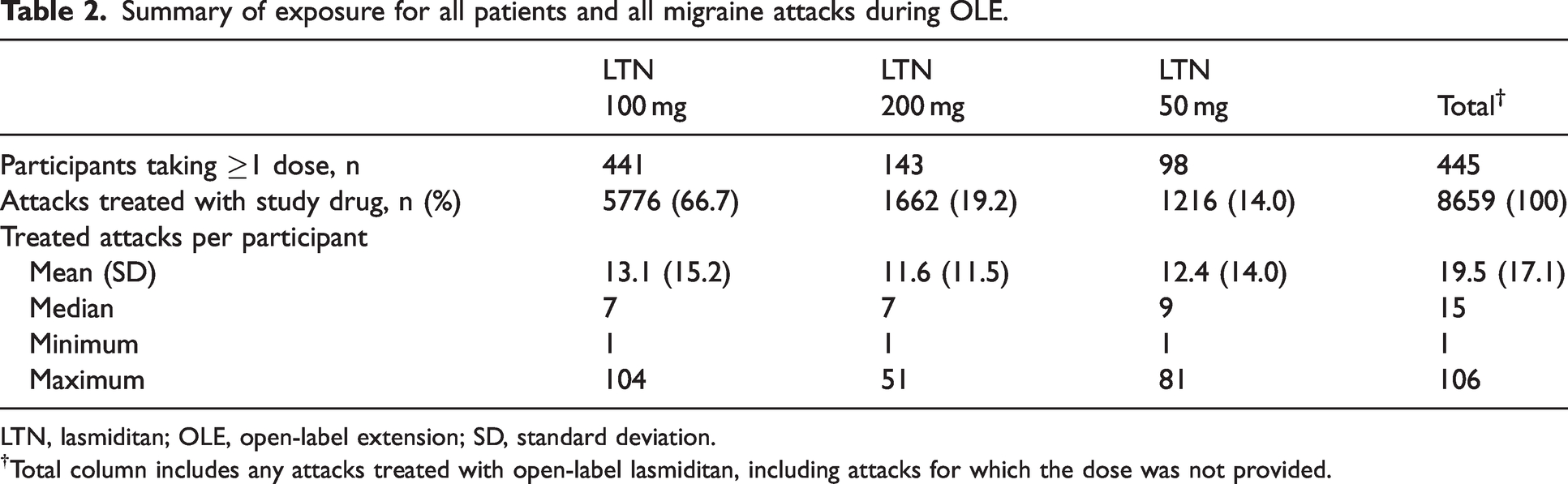
LTN, lasmiditan; OLE, open-label extension; SD, standard deviation.
Patient demographics and baseline characteristics
Demographic characteristics are summarized in Table 1. The OLE patient population was predominantly female (86.1%) and White (83.3%), with a mean age of 42.5 years. The largest number of patients were in the United Kingdom (44.7%).
Effect of lasmiditan on migraine-related disability
Overall, mean improvements in MIDAS total score, MIDAS headache days, and average pain severity were observed in the ITT population during the OLE (Figure 3). Repeated measures analyses revealed significant and sustained mean improvements from double-blind main study baseline in MIDAS total score (p < 0.001 at all OLE timepoints) as well as significant mean improvements in MIDAS headache days and average pain severity (p < 0.001 vs baseline at all OLE timepoints) during the OLE.

Improvements in MIDAS from main study baseline at OLE months 3, 6, 9, and 12: (a) Mean MIDAS total scores, (b) Mean headache days, and (c) Mean average pain severity. Note: Baseline value is measured at Visit 1 in double blind main study.
Shifts in MIDAS total score disability (a 4-point scale on which patients reported little to no disability, mild disability, moderate disability, or severe disability) were compared between baseline and EoS treatment responses. Overall improvement in disability from baseline was observed at EoS (Table 3).
Efficacy outcomes.

EoS, end of study visit (Visit 11/12 months); LTN, lasmiditan; MIDAS, Migraine Disability Assessment Score; mTOQ-6, 6-item Migraine Treatment Optimization Questionnaire; OLE, open-label extension; SD, standard deviation.
Note: Baseline value was measured at Visit 2 in double-blind main study.
All data are reported as n (%) unless otherwise indicated.
Effect of lasmiditan on quality of life
A mean improvement of the MSQ total score from OLE baseline of 11.3 was observed at EoS (Figure 4a). A repeated measures analysis revealed significant mean improvements from baseline in MSQ total score at each OLE visit. In addition, patients showed mean improvements from OLE baseline of 12.7 points in the Role Function-Restrictive domain, 9.6 points in the Role Function-Preventive domain, and 9.8 points in the Emotional Function domain at EoS (Figure 4b–d). Similar to the MSQ total score, repeated measures analyses revealed significant mean improvements from OLE baseline in MSQ Role Function Restrictive, Role Function Preventive, and Emotional Function scores at each OLE visit (p < 0.001 vs OLE baseline at all subsequent OLE timepoints).

Improvements in MSQ v2.1 from OLE baseline at OLE months 3, 6, 9, and 12: (a) Mean MSQ total scores, (b) Mean role function restrictive scores, (c) Mean role function preventive scores, and (d) Mean emotional function scores. Note: Baseline value is measured at baseline visit (Visit 6/Month 0) during the OLE.
Evaluation of treatment optimization
At EoS, an overall improvement for mean change from double-blind main study baseline was observed in the mTOQ-6 total score (Table 3).
Patterns of use
At the beginning of the OLE, all patients were assigned to 100 mg lasmiditan. Dose adjustment was permitted during the OLE to optimize efficacy and/or tolerability. Regarding patterns of lasmiditan use, 47.0% of patients remained at 100 mg lasmiditan throughout the OLE, 21.1% had their dose increased to 200 mg during the OLE and remained at the increased dose for the remainder of the study, 15.5% had their dose decreased to 50 mg during the OLE and remained at the decreased dose for the remainder of the study, and 15.5% had multiple dose adjustments during the OLE (Figure 2).
In this study, patients were allowed to treat migraine attacks without waiting until moderate-to-severe pain. Among 8654 total reported migraine attacks treated with lasmiditan, the median time to dosing during the OLE was 0.5 hours from pain onset. The baseline severity of 7349 (84.9%) of these attacks was characterized as either moderate or severe pain; the remainder had baseline severity characterized as no or mild pain. Among 2673 migraine attacks treated with other medications, for 2031 (76.0%), baseline severity was characterized as moderate or severe pain. Finally, for 1186 attacks (13.7%) treated with lasmiditan, baseline severity was characterized as mild pain; in comparison, baseline severity was characterized as mild for 550 (20.6%) attacks treated with other medications.
Of the 8654 lasmiditan-treated migraine attacks, 749 (8.7%) were treated with other acute treatment within 24 hours before lasmiditan treatment, most commonly paracetamol (2.3%), ibuprofen (1.7%), acetylsalicylic acid/caffeine/paracetamol combination (0.6%), and naproxen (0.6%). An additional 323 lasmiditan-treated migraine attacks (3.7%) were treated with other acute treatment within two hours after lasmiditan treatment, most commonly paracetamol (0.9%), ibuprofen (0.7%), nimesulide (0.3%), and zolmitriptan (0.3%). Finally, 1460 lasmiditan-treated attacks (16.9%) were treated with other acute treatment two to <24 hours after lasmiditan treatment, most commonly paracetamol (3.4%), ibuprofen (2.7%), sumatriptan (2.1%), and acetylsalicylic acid/caffeine/paracetamol combination (0.9%).
Of 8654 lasmiditan-treated migraine attacks, 112 (1.3%) were treated with triptans up to 24 hours before lasmiditan treatment, most commonly sumatriptan (0.5%), rizatriptan (0.3%), zolmitriptan (0.2%), and almotriptan (0.1%). An additional 64 lasmiditan-treated migraine attacks (0.7%) were treated with triptans within two hours after lasmiditan treatment, most commonly zolmitriptan (0.3%), sumatriptan (0.2%), rizatriptan (0.1%), and almotriptan (0.1%). Finally, 365 lasmiditan-treated attacks (4.2%) were treated with triptans two to <24 hours after lasmiditan treatment, most commonly sumatriptan (2.1%), rizatriptan (0.7%), zolmitriptan (0.7%), almotriptan (0.3%), and eletriptan (0.3%).
In all, 2660 attacks (23.5%) were treated solely with other acute treatments concurrent with migraine onset or <24 hours after migraine onset. The four medications most commonly used to treat these attacks were sumatriptan (n = 486 attacks, 18.3%), zolmitriptan (n = 313 attacks, 11.8%), paracetamol (n = 312 attacks, 11.7%), and ibuprofen (n = 290 attacks, 10.9%).
The most common reasons for not treating with lasmiditan were “I planned to drive or operate machinery” (n = 890 attacks, 7.9% of all migraine attacks) or “Thought another medication would work better for this attack” (n = 671, 5.9%), followed by “Thought another medication would have fewer side-effects” (n = 470, 4.1%), “None of the above” (n = 429, 3.8%), and “I did not have study medication available” (n = 213, 1.9%).
Treatment satisfaction at EoS
Patients’ treatment satisfaction with lasmiditan is detailed in Table 3. Of 400 evaluable patients at EoS, 256 patients (64.0%) strongly agreed or agreed with recommending lasmiditan to another patient, 238 patients (59.5%) strongly agreed or agreed they would be willing to take lasmiditan again, and 282 patients (70.5%) selected 1 of 4 “satisfied”-type responses (extremely satisfied, very satisfied, somewhat satisfied, or satisfied) to describe their level of satisfaction with lasmiditan at EoS. Finally, 150 patients (37.5%) preferred lasmiditan in comparison to their previous treatment.
PGIC-MHC
Of the 445 ITT patients, 225 (50.6%) reported “better”-type responses (very much better, much better, a little better) to PGIC-MHC at EoS, and 133 (29.9%) reported their condition was very much better or much better. “Worse”-type responses (a little worse, much worse, very much worse) were reported by 25 patients (5.6%), and 157 (35.3%) reported a neutral response (i.e., no change).
PGI-S
PGI-S responses were available for the ITT population (n = 445). Between OLE baseline and EoS, an overall shift in migraine severity from moderately/markedly/severely/extremely ill to not at all/borderline/mildly ill was observed: 324 patients (72.8%) reported at least moderate illness at baseline, versus 268 (60.2%) at EoS (online Supplementary Table 1).
Migraine frequency
The number of migraine attacks per month, treated or not treated with lasmiditan, is summarized in online Supplementary Table 2. At baseline (Month 1), 1170 migraine attacks were reported by 374 patients, of which 900 attacks (76.9%) were treated with lasmiditan and 270 (23.1%) were not. The percentage of reported migraine attacks treated with lasmiditan was reasonably stable, ranging from 71.8–78.7% throughout the 12 months.
Employment status and HCRU
Mean visitwise change from double-blind main study baseline in employment status
No meaningful changes in employment status were observed versus baseline (data not shown).
Mean visitwise change from double-blind main study baseline in HCRU
In the safety population (n = 445), 56 patients (12.6%) had an outside medical visit due to migraine, seven (1.6%) had a migraine-related hospital emergency room visit, and two (0.5%) had an overnight hospital stay due to migraine.
Shifts in HCRU related to migraine at OLE visits versus baseline were also assessed (Table 4). Baseline values were measured at visit 2 during the double-blind main study. Significantly fewer migraine-related healthcare professional visits (Wilcoxon sign-ranked test, p < 0.001) and migraine-related emergency room visits (Wilcoxon sign-ranked test, p = 0.017) were observed for visits 7–9 versus baseline, as well as for visits 10–11 versus baseline (Wilcoxon sign-ranked test, p < 0.001 for both). In addition, significantly fewer emergency room visits were observed for visits 10–11 versus baseline (Wilcoxon sign-ranked test, p = 0.024).
Shifts in migraine-related HCRU by healthcare type.

ER, emergency room; HCP, healthcare professional; HCRU, Healthcare Resource Utilization; N, number of subjects who have non-missing value at both baseline and final; n, number of subjects in each shift category.
Note: Only patients who completed the HCRU questionnaire at each visit included in the visit grouping and at baseline (Visit 2 from the double-blind main study) were included in the analysis.
Safety
Treatment-emergent adverse events (TEAEs)
TEAEs were defined as any adverse event (AE) with time of onset within 48 hours after a dose of study drug, or any event that worsened in intensity within 48 hours after a dose of study drug. Of the 445 OLE patients, 327 (73.5%) had ≥1 TEAE; these TEAEs ranged in severity from mild (n = 152 [34.2%]) to moderate (n = 139 [31.2%]) and severe (n = 36 [8.1%]). The most common TEAEs, affecting ≥5% of patients, were dizziness (35.7% of patients), paraesthesia (16.2%), fatigue (14.6%), nausea (13.5%), vertigo (11.5%), somnolence (11.0%), and asthenia (5.8%). A summary of TEAEs observed in ≥10% of patients is provided in Table 5.
TEAEs observed in ≥10% of patients or ≥2% of treated migraine attacks.

AE, adverse event; N, number of patients/events in the analysis; TEAE, treatment-emergent adverse event.
All data are presented as n (%).
Note: An AE with the date/time of onset, or that worsens in intensity, at or within 48 hours after the time of dosing is considered treatment-emergent.
MedDRA Version 24.0.
TEAE frequency was summarized by individual migraine attack for the OLE safety population. Of a total of 8659 treated migraine attacks, 2161 (25.0%) were associated with ≥1 TEAE. TEAEs reported in ≥5% of migraine attacks were dizziness (9.5% of attacks) and paraesthesia (5.1%). A summary of TEAEs observed in ≥2% of migraine attacks is provided in Table 5.
Serious adverse events (SAEs) and treatment-emergent SAEs
A total of 19 patients (4.0%) reported ≥1 SAE. Four patients (0.9%) each reported one treatment-emergent SAE: COVID-19 (moderate severity), malignant lung neoplasm (moderate), migraine (severe), and serotonin syndrome (moderate) were reported as SAEs in one patient (0.2%) each. The only SAE deemed related to study drug by the investigator was a reported case of serotonin syndrome, self-reported by the patient and not evaluated by a healthcare professional at the time of the event, lasting 1 hour 40 minutes and not requiring intervention. No cardiovascular SAEs were reported.
Deaths
One patient (0.2%) experienced an SAE from a gardening accident during the OLE. The SAE was not treatment-emergent and was not considered related to the study drug by the investigator. This patient subsequently died due to the SAE.
Discontinuation of lasmiditan due to AEs
In all, 26 patients discontinued lasmiditan during the OLE. One patient discontinued lasmiditan due to “withdrawal by subject, due to ongoing AEs.” The AEs included pleurisy, costochondritis, and right hypochondrium pain, none of which were considered related to study drug by the investigator. Twenty-five patients (5.2%) in the OLE population discontinued lasmiditan due to an AE; of these, 23 patients (4.8%) had taken ≥1 dose of lasmiditan. AEs leading to discontinuation of lasmiditan in ≥2 patients were dizziness (n = 9 [1.9%]), asthenia (n = 2 [0.4%]), and vertigo (n = 2 [0.4%]). Most AEs leading to lasmiditan discontinuation were mild-to-moderate in severity (88.0%). SAEs leading to lasmiditan discontinuation were ophthalmic migraine and serotonin syndrome (n = 1 [0.2%] each).
Driving accidents/moving violations
No patients reported any driving accidents as the driver, or moving violations, in temporal proximity to having treated a migraine attack with lasmiditan. Ten patients were involved in a car accident as the driver; however, none had taken lasmiditan before the accident. There were no violations (including events of speeding, unsafe operation of a motor vehicle, or other moving violations) reported following lasmiditan dosing.
Labs and vital signs
Analyses of laboratory assessments and vital signs did not show clinically meaningful changes in OLE patients.
Pregnancy
A total of two patients became pregnant and had miscarriages during OLE. Neither was considered treatment-emergent or treatment related.
Discussion
During the OLE, lasmiditan achieved the primary endpoint and demonstrated improvements across most secondary endpoints. The improvements in MIDAS total score versus baseline indicate significant improvements in migraine-related disability. Significant improvements were observed at each OLE visit for mean changes from baseline in MIDAS migraine severity and headache days, as well as total score and each subscore of the MSQ v2.1. These findings indicate patients with at least moderate migraine-related disability had less disability while treating migraine attacks with lasmiditan in this study with dose optimization. Patients were generally satisfied with lasmiditan during the OLE, with 64.0% agreeing or strongly agreeing they would recommend lasmiditan to another patient.
Persistent use of lasmiditan was high in the more real-world setting of this OLE. Most patients started on an initial dose of 100 mg lasmiditan and remained on the 100 mg dose throughout the OLE. Patients who changed dose increased to 200 mg more frequently than they decreased to 50 mg lasmiditan. Most attacks were treated with lasmiditan throughout the OLE. When a medication other than lasmiditan was used, the most common reasons were “I planned to drive or operate machinery” (7.9% of attacks) or “Thought another medication would work better for this attack” (5.9% of attacks).
No new safety issues were observed during the OLE. No safety issues were observed when lasmiditan was used in temporal proximity to other drugs, including triptans. This study included patients up to 76 years old, and no safety concerns, including ischemic events, were identified. No driving issues were observed in this study among participants treating a median of 15 migraine attacks over up to a year of observation within the context of the eight-hour driving restriction. Based on these findings, the risk mitigation strategy of not driving for eight hours after dosing with lasmiditan appears to be appropriate.
Limitations
The study was not specifically powered for objectives to evaluate health-related quality of life and disability. As an OLE, the study lacked a placebo control for comparison of migraine-related disability, quality of life, and safety results. Both investigators and study participants were aware of lasmiditan dosages throughout the OLE, and this awareness may have affected patients’ perceptions of their response to treatment and on the reporting of subjective events.
Efficacy data specific to each migraine attack was not collected during the OLE because the efficacy of lasmiditan in the treatment of individual migraine attacks has been evaluated in previous studies (including a previous OLE study: GLADIATOR; NCT02565186) (5,8) and was evaluated during the double-blind portion of CENTURION (4). This study was conducted to assess the impact of lasmiditan on other outcomes without the burden of entering substantial efficacy data into an electronic diary during migraine attacks.
Conclusions
In this 12-month OLE of the CENTURION study, lasmiditan treatment was associated with significant improvements compared to prespecified baseline in migraine-related disability and quality of life, as assessed by MIDAS and MSQ v2.1. In relatively real-world conditions, lasmiditan therapy was associated with a high rate of study completion. Most attacks were treated with lasmiditan and most patients remained on lasmiditan 100 mg throughout the OLE. There were no new safety findings with lasmiditan exposure up to 12 months.
Clinical implications
In relatively real-world conditions, lasmiditan therapy was associated with a high rate of study completion. Most migraine attacks were treated with lasmiditan and most patients remained on lasmiditan 100 mg throughout the OLE. Patients reported improvements in migraine-related disability and quality of life. There were no new safety findings with longer exposure (up to 12 months).
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231161745 - Supplemental material for Long-term treatment with lasmiditan in patients with migraine: Results from the open-label extension of the CENTURION randomized trial
Supplemental material, sj-pdf-1-cep-10.1177_03331024231161745 for Long-term treatment with lasmiditan in patients with migraine: Results from the open-label extension of the CENTURION randomized trial by Messoud Ashina, Caroline Roos, Lily Qian Li, Mika Komori, David Ayer, Dustin Ruff and John Henry Krege in Cephalalgia
Footnotes
Acknowledgements
Medical writing support was provided by Regina E Burris (Syneos Health) and editorial support was provided by Dana Schamberger (Syneos Health).
Data sharing
Eli Lilly and Company provides access to all individual participant data collected during the trial, after anonymization, with the exception of pharmacokinetic or genetic data. Data are available to request six months after the indication studied has been approved in the US and EU and after primary publication acceptance, whichever is later. No expiration date of data requests is currently set once data are made available. Access is provided after a proposal has been approved by an independent review committee identified for this purpose and after receipt of a signed data sharing agreement. Data and documents, including the study protocol, statistical analysis plan, clinical study report, blank or annotated case report forms, will be provided in a secure data sharing environment. For details on submitting a request, see the instructions provided at ![]() .
.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Lily Qian Li, David Ayer, Dustin Ruff, and John Krege are full-time employees of Eli Lilly and Company, Indianapolis, IN, USA and minority holders of company stock. Mika Komori is a full-time employee of Eli Lilly and Company, Japan and a minority owner of company stock.
Messoud Ashina is a consultant, speaker, or scientific advisor for AbbVie, Amgen, Eli Lilly and Company, Lundbeck, Novartis, Pfizer, and Teva and a primary investigator for ongoing AbbVie, Amgen, and Lundbeck trials. Ashina has no ownership interest and does not own stocks of any pharmaceutical company. Ashina serves as an associate editor of Cephalalgia, an associate editor of The Journal of Headache and Pain, and an associate editor of Brain.
Caroline Roos is a consultant for Allergan/AbbVie, Eli Lilly and Company, Lundbeck, Novartis, and TEVA. Roos has received honoraria for promotional speaking from Allergan/AbbVie, Eli Lilly and Company, Homeperf, and TEVA. Roos has received research funding from Allergan/AbbVie, Amgen, Eli Lilly and Company, and Lundbeck.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Eli Lilly and Company in accordance with Good Publication Practice (GPP3) guidelines (![]() ). The authors received no financial support for the research, authorship, and/or publication of this article.
). The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
