Abstract
Preventive therapy is aimed at reducing migraine frequency, but should also improve the much deteriorated quality of life of the migraneur. We aimed to evaluate the impact of preventive therapy with two widely employed drugs (topiramate and nadolol) on the quality of life of migraine patients. A population of consecutive migraineurs aged ≥16 years, with frequent migraines, was selected prospectively for evaluation at baseline and after 16 weeks of therapy with nadolol or topiramate (40 mg and 100 mg daily, respectively) by generic and specific quality of life questionnaires (SF-36 and MSQOL) and by an anxiety and depression scale (HADS). Preventive therapy resulted in a statistically significant improvement in physical domains of the SF-36, whereas mental domains remained almost unchanged. Despite this improvement, all domains remained below the population norms. The HADS revealed a moderate depressive state at baseline that did not change with therapy. The MSQOL global score also revealed statistically significant improvement. Both drugs were similarly effective, although topiramate was superior on the role physical domain compared with nadolol. Preventive therapy with nadolol and topiramate significantly improves the quality of life of migraineurs, although additional efforts are needed to place them in a nearer-to-normal situation compared with the general population.
Keywords
Introduction
Migraine is a common, incapacitating disorder that interferes severely with the activities of daily life and with productivity (1–4). As an episodic disorder, migraine causes marked fluctuations in the quality of life of the sufferer. Patients with more frequent attacks suffer a greater burden, in that patients with more than two to three episodes per month require preventive therapy (5).
Preventive medication should not only reduce the frequency of attacks, but should also result in an improvement in the patient's quality of life. Assessing quality of life is an important complement to those clinical end-points traditionally employed in determining the effectiveness of antimigraine drugs, be they abortive or preventive.
Quality of life has been defined as the ability of an individual to play a role in society and to enjoy fully their role as a citizen, with an independent social status (3). The assessment of quality of life relies fundamentally on answers to questionnaires that are generic or specific to a particular illness. Generic questionnaires such as the Short Form 36 Health Survey (SF-36), which was developed from the Medical Outcome Study General Health Survey Instrument, have been used for migraine (6). The SF-36 has the advantage of having been widely employed in different conditions (including migraine), in general populations, and has been validated in many countries and languages. However, the SF-36 was not designed to measure limitations or restrictions specifically associated with migraine. In this regard, disease-specific instruments, such as the migraine-specific quality of life questionnaire (MSQOL), may be more sensitive in evaluating variations in the quality of life of the patient with migraine (7). It is of particular interest to measure the long-term effects of migraine and migraine treatment on quality of life. As with the SF-36, standardized versions of the MSQOL exist in Spanish.
Although quality of life in migraine has been assessed following treatment for the acute attack (8), the long-term effects of migraine prevention on quality of life have received little attention.
In this study, we sought to investigate whether preventive therapy for migraine with nadolol or topiramate, two widely employed preventive drugs, results in improved quality of life for the migraineur. To this end, we employed a generic questionnaire, the SF-36, and a specific questionnaire, the MSQOL. Also, the Hospital Anxiety and Depression Scale (HADS) questionnaire, a measure of anxiety and depression, was administered, since it is known that depression is comorbid with migraine, contributing to the deterioration in the quality of life of the patient (2, 9), and accounts for low scores on many SF-36 scale scores (10), thus confounding the comparisons of SF-36 scales among patients with different headache diagnoses.
Patients and methods
Patient selection and procedures
This was a prospective study in which patients were required to be ≥16 years old and to have had a diagnosis of migraine with or without aura according to the International Headache Society (IHS) criteria for at least 6 months before screening, with more than three attacks per month, but no more than 15 headache days per month. They should be naive in terms of migraine prevention or should not have had medication in the 6 months prior to study onset. Patients with analgesic overuse, as defined by the IHS criteria, were excluded, as were those patients who were not able to properly fill out the headache calendar, and those intolerant to triptans or non-steroidal anti-inflammatory drugs (NSAIDs). Diagnosis was established by one of the neurologists in the study (N.F. or J.C.G-M.), who evaluated all patients. Patients were recruited consecutively in the Department of Neurology of our hospital, a publicly funded community hospital serving a population of 300 000. Patients were referred by their primary physicians for headache evaluation and were unselected except for their migraine frequency.
During the initial visit, appropriate abortive therapy was indicated, which consisted of NSAIDs or triptans. Patients were directed to take one of them and leave the other as rescue medication, according to usual clinical practice. If patients were already on acute attack medication, pertinent modifications were introduced if needed. Comorbid conditions were recorded. A headache calendar was given for a period of 6 weeks on which patients recorded the frequency (attacks per month) and characteristics of migraine attacks. The study was explained to the patient and informed consent was obtained. Any medication with possible migraine-preventive effect was forbidden. Fertile women were required to practise an acceptable method of birth control before study entry. Prior diagnosis of depression requiring specific therapy was also an exclusion criterion.
During the second visit, scheduled 6 weeks later, the need for preventive therapy was confirmed with the calendar. At this point, patients filled out the questionnaires of quality of life and of anxiety and depression (HADS).
Therapy with nadolol 40 mg/day or topiramate 100 mg/day, both slowly titrated over 2 weeks, was prescribed as clinically indicated. The dose of topiramate was chosen on the basis of previous studies, which have shown that 100 mg/day provides the best risk–benefit ratio. The optimal dose of nadolol is not well established, with a wide range in published studies. Our own experience with >700 patients treated with this drug (unpublished results) indicates that a dose of 40 mg/day provides a good risk–benefit profile. Preventive therapy was chosen in a sequential manner, i.e. patient 1 would receive nadolol, patient 2 topiramate and so on, unless some kind of contraindication was seen.
Patients were kept on medication for 16 additional weeks, during which they kept a calendar and recorded the frequency (attacks per month) and characteristics of headaches, as well as side-effects. At the end of the period, patients came back to the clinic for the final visit and again filled out the quality of life questionnaires and the HADS.
The study was approved by our local Ethics Committee.
Instruments
We employed two questionnaires of quality of life, one generic (SF-36) and one specific for migraine (MSQOL), as well as a scale to evaluate the degree of anxiety and depression (HADS).
SF-36
The SF-36 is a structured, self-report questionnaire which the patient can generally complete with little or no intervention from an interviewer. It includes 36 items measuring health across eight domains of health status. The scoring system for the SF-36 is relatively complex and generates subscale scores for physical functioning (PF), role limitations due to physical problems (RP), bodily pain (BP), general health perceptions (GH), vitality (VT), social functioning (SF), role limitations due to emotional problems (RE) and mental health (MH). Two summary scores can also be derived from the SF-36: the physical component and the mental component summary. Four scales (PF, RP, BP and GH) contribute most to the scoring of the physical component summary (PCS) measure (Ware et al. 1995). The VT, SF, RE, and MH scales contribute most to the scoring of the mental component summary (MCS) measure. Summary scores allow a much simpler interpretation of results and are very useful when interpreting differences across scales in the SF-36 profile and for monitoring disease groups over time. The SF-36 scores range from 0 to 100, with a higher score indicating better health status. In addition, normalized values can be estimated so that it can provide a reference value from the general population. To do so, each SF-36 score was first standardized using the mean and standard deviations (SD) obtained from the Spanish population, and then transformed to norm-based (mean = 50, SD = 10) scoring, as suggested by the authors of the questionnaire (11). The SF-36 has been translated into Spanish and validated in Spanish populations and the measurement properties have been published previously (12).
MSQOL
The MSQOL questionnaire is a specific instrument that has been shown to be a valid and reliable measure in migraineurs (7, 13). Self-administered, it consists of 20 items, each of which is rated using a response scale with four categories (1 = very much to 4 = not at all). It has a multidimensional scale comprising 20 items grouped into three dimensions: avoidance (10 items), social relationships (six items) and feelings (four items). The scores were determined by adding the items for each domain. An overall score was also determined by adding the 20 items. The data were standardized to a range of values from 0 to 100, where 0 represents the worst health status and 100 the best health status. The original questionnaire has been translated and validated into Spanish (13, 14).
HADS
This is a self-reporting screening scale that measures depression and anxiety simultaneously whilst giving a separate score for each (15). The HADS was designed to provide clinicians with a reliable, valid and practical screening tool for identifying and quantifying anxiety or depression in the medical out-patient clinic for non-psychiatric patients such as, for example, migraineurs. The HADS consists of seven items for anxiety (HADS-A) and seven for depression (HADS-D). The items are scored on a four-point scale, from 0 to 3. The item scores are added, giving subscale scores on the HADS-A and the HADS-D from 0 to 21, with higher scores indicating more degree of anxiety or depression. The two subscales, anxiety and depression, have been found to be independent measures. In its current form the HADS is now divided into four ranges: normal (0–7), mild (8–10), moderate (11–15) and severe (16–21). HADS is a reliable and valid instrument in and outside hospital and community settings (16). The HADS has been translated into Spanish and validated in Spanish populations (17) and the measurement properties have been published previously (18).
Statistical analysis
Sample size was calculated based on our own previous pilot studies considering an α value of 0.05, a β value of 0.8 and an average detectable change of four points in the therapeutic gain of the SF-36 or MSQOL domains. We employed the sampsi procedure of the Stata version 8. By using a paired t-test, this resulted in a sample size of 50 individuals, which was increased to >60 to compensate for drop-outs.
Descriptive statistics of sociodemographic and clinical variables included frequencies, percentages and mean, SD and median. Patients' characteristics at baseline were compared between those who finished the study and those who did not. Descriptive statistics according to the drug employed were also included. χ2 and Fisher's exact tests were used to test for statistical significance among proportions. For continuous variables, Student's t-test and non-parametric Wilcoxon rank sum tests were performed. For the comparison of migraine frequency at baseline and after therapy a paired t-test was used. All data were analysed blindly by a statistician, coauthor of the manuscript (A.B.).
Since the primary objective was to assess the overall impact of migraine-preventive medication on patients' quality of life, we analysed the effects of the combination of the two drug groups on the different questionnaires. The therapeutic gain, defined as the change between baseline values and those obtained after therapy was concluded, was obtained for every quality of life item measured. Since the scale measures for the SF-36 and those for the MSQOL and HADS follow an opposite direction (i.e. higher values in the SF-36 represent better status, contrary to the MSQOL and HADS), the gain for SF-36 represented post-therapy values minus baseline values, whereas the opposite was true for the MSQOL and the HADS. Thus, positive values in gain consistently represented an improvement in quality of life.
Mean scores at the first and second visits and mean changes were calculated for the domains of SF-36, MSQOL and HADS. A paired t-test was used to determine if the change detected from the first to the second visit was significant. The analyses were also performed separately for each drug. For comparison of the therapeutic gain of topiramate vs. nadolol, Student's t-test and non-parametric Wilcoxon rank sum tests were performed.
The SF-36 normalized values at the first visit and after therapy were compared with the norm-based results for the Spanish general population.
Finally, although the study was not designed to this end, we evaluated whether there was a relationship between quality of life in the pooled topiramate and nadolol patient population and their migraine attack frequency. We analysed therapeutic gain for every domain, among those patients in whom frequency reduced by ≥50% and among those with a <50% reduction, by means of paired t-test.
Patients' information was entered into a Microsoft Access database. In all analysis, P < 0.05 was considered to be significant. All statistical analyses were performed using SAS for Windows statistical software, version 8.0 (SAS Inc., Cary, NC, USA).
Results
Study population and demographics
Seventy-six patients met the criteria and entered the study; 61 completed the study and 15 dropped out, seven in the topiramate group and eight in the nadolol group. Reasons for drop-out were as follows: (i) topiramate group: decision to become pregnant (n = 2), dizziness (n = 1), decision not to take preventive therapy on second thoughts (n = 1), fear of side-effects (n = 1), depression (n = 1) and insomnia (n = 1); (ii) nadolol group: decision not to take preventive therapy (n = 3), spontaneous improvement before preventive therapy started (n = 1), fear of side-effects (n = 2), depression (n = 1) and anxiety (n = 1).
No differences were present between those patients who finished the study and those who did not in terms of age, diagnosis, comorbidities or frequency of migraine attacks (data not shown).
Baseline characteristics of patients in the two drug groups were similar in terms of age, diagnoses and frequency of migraine attacks (Table 1). Comorbidities showed some statistically non-significant differences. There were two untreated asthma patients in the nadolol group, and four in the topiramate group who received long-term inhaled therapy with a combination of steroids (budesonide or fluticasone) and β2-agonist (salmeterol or formoterol), as well as occasional doses of inhaled terbutaline. Epilepsy was present in two patients in the nadolol group and in none in the topiramate. Although topiramate could have served the double purpose of preventing migraine and epilepsy, its use was approved only as an add-on antiepileptic therapy in our country at the time of this study, and these patients were already on a first-line drug (phenytoin). Depression, according to the patient's own assessment, was present in almost one-third of the overall sample (26.1–31.3%) and was slightly more frequent in the nadolol group. However, it has to be stressed that none of them had a prior, formal diagnosis of depression, nor were they being treated to this end. Hypertension (mild and untreated) was present in three patients and thyroid abnormalities in one patient of each arm, and both were on hormonal replacement therapy. Peptic ulcer or gastritis was more frequently reported among patients in the nadolol group.
Demographics and characteristics of the patients who entered the study

Data are given as n (%) unless otherwise indicated.
P-value for reduction in migraine frequency <0.0001.
P-value for reduction in migraine frequency = 0.0007.
P-value for reduction in migraine frequency = 0.0176.
The number of patients who felt they had depression (self-diagnosis). None of them had a prior formal diagnosis of depression.
Migraine frequency reduction effectiveness
There was a statistically significant reduction in migraine frequency when global therapy was considered, as well as when each drug was considered separately, although topiramate achieved a greater statistical significance in frequency reduction (Table 1).
Overall impact of preventive medication on quality of life
Preventive treatment resulted in a statistically significant improvement in those aspects of the SF-36 related to physical health, such as physical functioning and role limitations due to physical problems, as well as in general health perception (Table 2). As a result, the standardized physical component score was also significantly better than at baseline. The gain in the other aspect related to physical health, bodily pain, did not reach statistical significance, nor did mental components (vitality, social functioning, role limitations due to emotional problems, and mental health). Gain in role emotional and mental health was negative, indicating a moderate, statistically non-significant, worsening respect to baseline, just like the standardized mental component score. The lack of improvement in the role emotional factors could be associated with the depressive state of patients (see below, HADS results), since this component reflects problems with work or other daily activities as a result of emotional problems.
Results of the quality of life and Hospital Anxiety and Depression Scale questionnaires
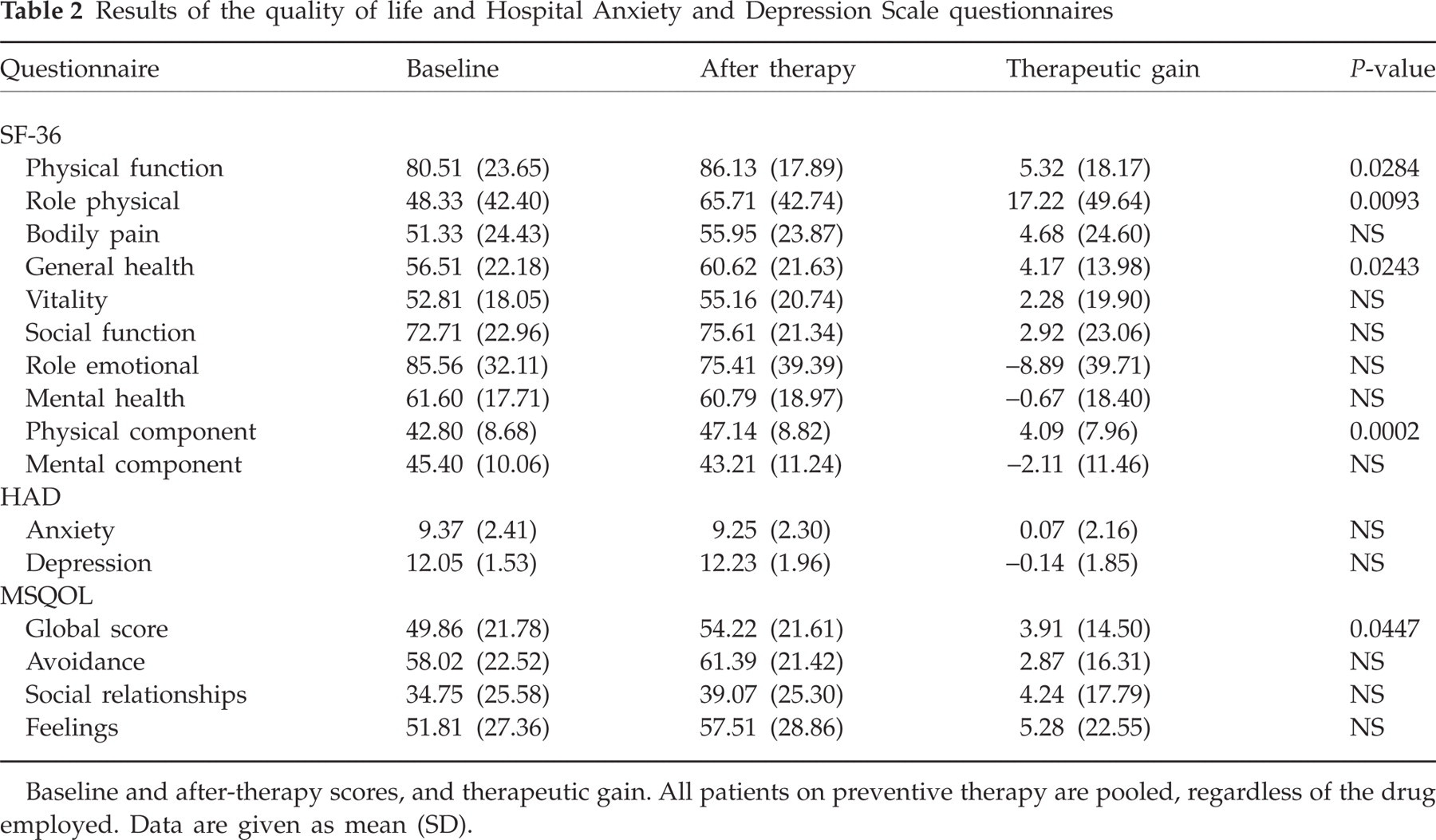
Baseline and after-therapy scores, and therapeutic gain. All patients on preventive therapy are pooled, regardless of the drug employed. Data are given as mean (SD).
When general population norms were applied, it was evident that the role emotional was the least affected of the SF-36 domains at baseline (Fig. 1). Despite showing a significant improvement on several domains after therapy, norm-based SF-36 scoring disclosed that patients' health status remained below the average of the general population (i.e. a score of 50) on every domain (Fig. 1).
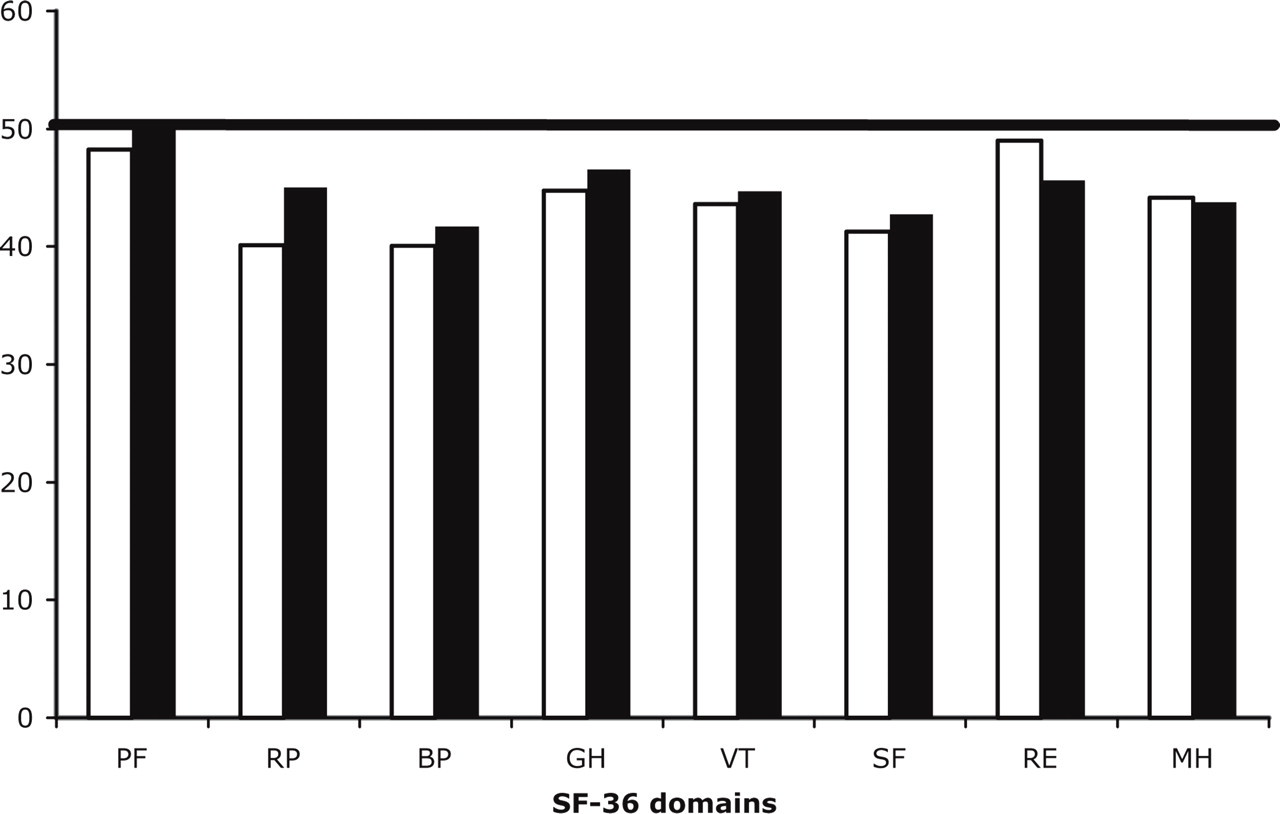
Norm-based SF-36 scores at baseline and after preventive therapy (topiramate and nadolol pooled). General population score ≥50 on each item. □, Baseline; ▪, after therapy. PF, Physical functioning; RP, physical role; BP, bodily pain; GH, general health perceptions; VT, vitality; SF, social functioning; RE, emotional role; MH, mental health.
The HADS indicated a mild baseline anxiety state, with a mean score of 9.37 that did not change significantly after therapy. Consistent with this, patients as a whole had a moderate depressive state at baseline, with a mean score of 12.05 that remained unchanged at the end of the study (Table 2).
All the domains in the specific questionnaire for migraine, the MSQOL, improved, although only the global score showed a statistically significant improvement. The other three domains did not reach statistical significance (Table 2).
Comparison of topiramate and nadolol gains
After the overall evaluation of the impact of preventive medication on patients' quality of life, the impact of each drug separately was assessed by analysing the therapeutic gain of each drug (Table 3). There were no differences between them in any domain of the questionnaires employed, except for the superior effect of topiramate on the role physical domain of the SF-36, whose gain was statistically significant compared with nadolol.
Quality of life therapeutic gain according to the drug employed for migraine prevention
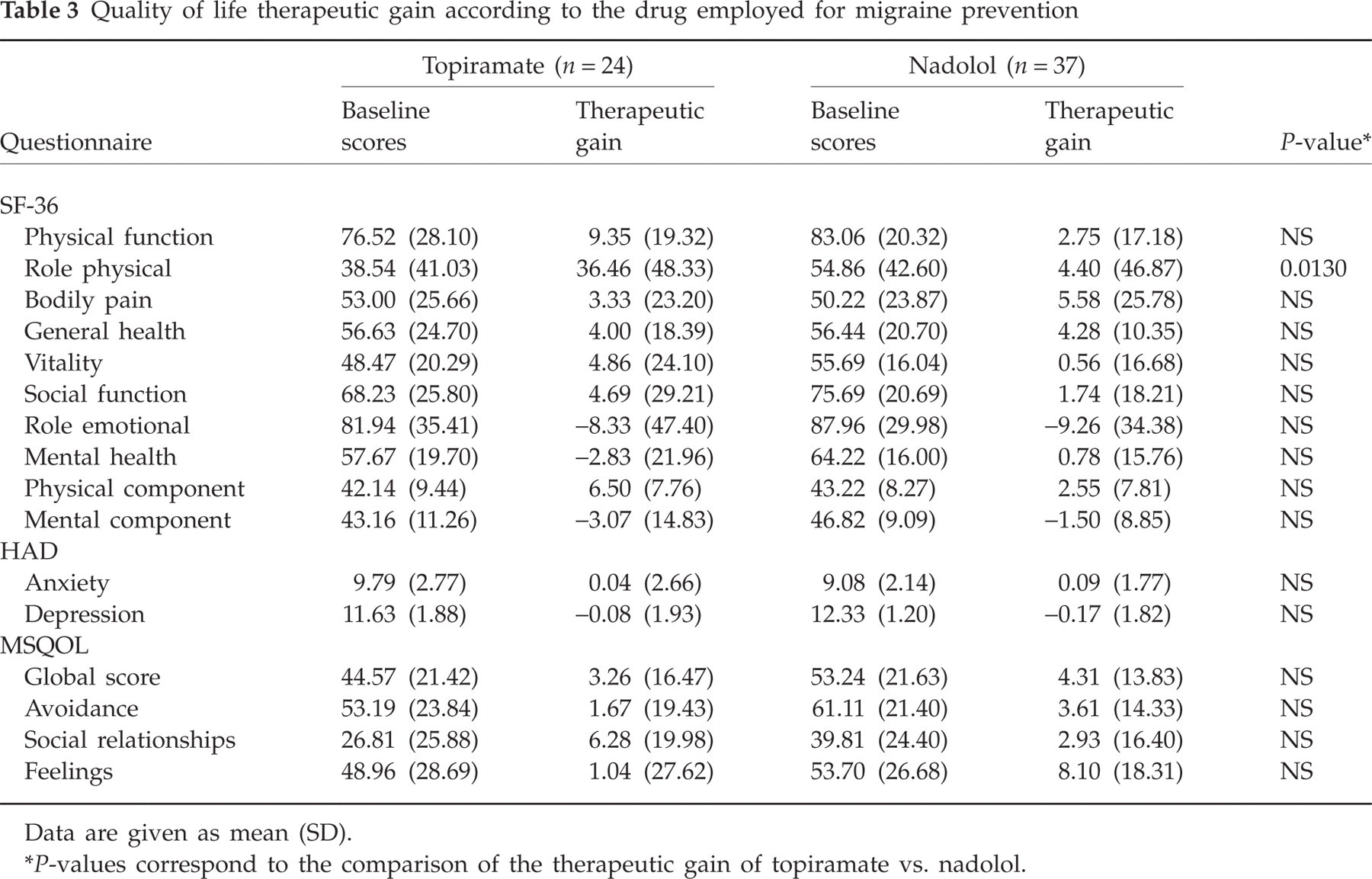
Data are given as mean (SD).
P-values correspond to the comparison of the therapeutic gain of topiramate vs. nadolol.
Quality of life and migraine frequency
Although the study was not designed for this purpose, we evaluated whether there was an association between quality of life in the pooled topiramate and nadolol patient population and their frequency of migraine attacks. Among those patients in whom frequency was reduced by ≥50%, there was a statistically significant improvement in the therapeutic gains of role physical, general health, and physical component summary of the SF-36, whereas only the physical component summary was significantly improved among those with a <50% reduction (data not shown).
Discussion
Measuring the subject's perspective of migraine adds an important dimension to the assessment of treatment response that the classical physical measures of clinical efficacy do not take into account. In this sense, generic and specific instruments offer complementary views, and an instrument assessing anxiety and depression is crucial to interpret the quality of life questionnaires.
This study indicates that preventive medication with 40 mg/day nadolol or 100 mg/day topiramate results in a positive impact of the patient's quality of life as measured by the SF-36 and the MSQOL. Interestingly, it was the physical domain of the SF-36 that showed a significant improvement, while the mental domain did not change with therapy. This is consistent with the fact that the HADS, a measure of depression and anxiety, did not change after therapy, and has two important implications. First, neither depression nor anxiety was responsible for low SF-36 scores at baseline. Second, depression and anxiety did not account for the improvement in the SF-36 results, which rules out a potential antidepressant effect of preventive therapy as the reason for improvement.
Our results resemble those from clinical studies comparing scores for patients before and after treatment that have shown that three of the scales (PF, RP, GH and BP) with the most physical factor content are most responsive to the benefits of knee or hip replacement (19) and heart valve surgery (20). Along these lines, studies that used the SF-36 with chronic headache patients in primary care treated with acupuncture have confirmed that the treated group only reached significant differences in the physical domains as well (21, 22). Another study in patients with non-malignant pain has confirmed that these patients have poor physical functioning and worse bodily pain according to the SF-36 (23). In contrast, the four scales with the most mental factor content (VT, MH, RE and SF) have been shown to be most responsive in comparisons of patients before and after recovery from depression (24). In this context, our patients' depression score did not change after migraine prevention treatment. A recent study found a positive impact of migraine-preventive therapy on all SF-36 domains (25). The fact that the authors used a variety of drugs (six different pharmacological groups) and did not include evaluation of anxiety and depression makes it difficult to compare their results with the present study.
Despite reductions in migraine frequency after preventive therapy, and a significant positive impact on several domains of the SF-36, the patients remained below the scores of the general population. This fact stresses again the deteriorated quality of life of migraineurs. Furthermore, it stresses the need to go a step further than simply reducing migraine frequency, and to try to place the patient in a closer-to-normal situation in terms of quality of life.
The global score of the MSQOL showed a significant gain after preventive therapy, and there were no significant differences between the two drugs employed. However, the separate domains of the MSQOL did not reflect a significant change with therapy. These findings are consistent with the scant modification of the mental components of the SF-36 and of the anxiety and depression scores of the HADS, since mental factors are important when assessing avoidance, social relationships and feelings in the MSQOL. Also, the global score could be more sensitive to changes than the individual fields in this setting, with the latter requiring larger samples. This aspect requires clarification, since the items within the questionnaire possess a high degree of internal consistency (13).
Several methodological aspects deserve mention. First, we employed a fixed-dose schedule. It could be argued that some patients would have obtained greater benefit with a higher dose. A variable dosage schedule, however, would have required a considerably larger number of patients in each group and could have resulted in results less comparable with other studies. Furthermore, both drugs reduced significantly the frequency of migraine attacks at the doses employed. Second, patients who consult for migraine frequently use an inadequate regimen to treat acute attacks, and a modification in the abortive medication could result in an improvement in perceived quality of life erroneously attributed to preventive therapy. We avoided this by adjusting the acute therapy medication 4 weeks before the quality of life was first measured, which ensured that all patients treated an attack in a similar manner. Our patients were recruited after verification that they actually had the frequency of attacks required in the study. Finally, the test–retest period for the MSQL is 24 weeks and we employed a shorter evaluation period (but more conventional in terms of preventive therapy), which could have been suboptimal.
Despite topiramate and nadolol being two drugs widely employed in migraine prevention due to their effectiveness in decreasing the frequency of migraine attacks, little is known about their impact on the quality of life of the patient. The effect of topiramate 100 mg/day on various aspects of health was evaluated by a post hoc analysis of pooled data from controlled trials using a specific instrument developed by Glaxo, the Migraine-Specific Questionnaire (MSQ) data. Therapy with topiramate improved the three domains of the MSQ (role restriction, role prevention, and emotional function) compared with placebo, and the effect sizes were considered moderate and likely to be of clinical significance (26). Unlike ours, this study did not have as the main objective quality of life evaluation, and did not include generic questionnaires or anxiety and depression evaluation, which is an important consideration, since it is well known that depression is highly comorbid with migraine and contributes to deterioration of the quality of life of the patient (2) and accounts for low scores on many SF-36 scale scores acting as a confounder (10).
Nadolol has been proven effective in migraine prevention in terms of reducing migraine frequency, but no evaluation has been made in terms of quality of life. At a dose of 40 mg/day, it significantly reduced the frequency of migraines and resulted in a positive impact on the quality of life, similar to topiramate, except for a lesser effect on physical role limitations.
Our analysis suggests an association between quality of life improvement and reduction in migraine frequency, suggesting that improvement of quality of life in migraine is mediated, at least in part, by a reduction in the frequency of attacks. Other aspects are important, as reflected by the norm-based scores obtained after therapy. This important issue requires studies specifically aimed at this objective.
In summary, preventive migraine treatment with topiramate or nadolol positively impacts on the quality of life of the migraineur, as manifested by their effects on the generic questionnaire SF-36 and on the specific MSQOL. Physical factors significantly improve while the emotional components remained unchanged, in keeping with the unmodified anxiety and depression scores. Despite this improvement, patients still score below the general population average on the norm-based scoring of the SF-36, and this is an indication for further efforts to return these patients as close to normality as possible.
