Abstract
Background:
Small molecule receptor antagonists (gepants), or monoclonal antibodies (mAbs) against calcitonin gene-related peptide (CGRP) have recently become available for migraine prophylaxis and/or acute treatment. Considering their shared mechanisms of action, if the failure to an anti-CGRP(R) mAbs preclude the effectiveness of gepants or vice versa is still unknown. Herein, we report the first case of a patient with refractory migraine responsive to the acute use of rimegepant that previously failed two different anti-CGRP(R) mAbs and with no response to other acute treatments. Finally, we performed a literature review on the use of gepants in patients that failed other anti-CGRP and/or triptans.
Case:
A 56-year-old female with a long history of chronic migraine without aura, fulfilling the EHF definition for a diagnosis of refractory migraine. Overall, the patient treated five not-consecutive migraine attacks. All of them were treated according to the predefined criteria. The mean (±SD) NRS before rimegepant assumption was 7.8 ± 0.9, all attacks cause at least severe impairment at onset, and no rescue medications were used. Pain free at 2 hours was achieved in three out of five attacks (60.0%), with no recurrence of migraine in the following 24 hours. The patient reported also a sustained benefit the day after the drug assumption. The response pain free was achieved after a mean time of 13.3 ± 4.5 minutes considering only attacks successfully treated (three out of five attacks). No adverse events were reported.
Conclusions:
In conclusion, rimegepant for acute treatment may be a viable option in patients with partial or no response to triptans that failed preventive treatments targeting the CGRP pathway, regardless to ligand or receptor. The failure of anti-CGRP(R) mAbs does not necessarily preclude the use of gepants (acute and/or preventive), but further studies are urgently needed to provide evidence on the efficacy of these treatments in managing drug-resistant migraine and to identify novel treatments for patients.
Keywords
Introduction
Migraine is a primary headache with a prevalence of ≅15% worldwide and is a leading cause of global disability for people under 50 years of age. 1 Small molecule antagonists (gepants) of the receptor for calcitonin gene-related peptide (CGRP), the calcitonin-like receptor/receptor activating modifying protein 1 (CLR/RAMP1) (R) or monoclonal antibodies (mAbs) against CGRP (L) or its R (anti-CGRP/R mAbs) have recently become available for migraine prophylaxis and/or acute treatment with proven efficacy and tolerability. Four anti-CGRP mAbs are currently available in Europe. One (erenumab) is directed against the CLR/RAMP1, and three (galcanezumab, eptinezumab and fremanezumab) against the ligand (L). Although anti-CGRP/R mAbs have shown remarkable efficacy, as reported by numerous clinical trials and real-world studies, approximately 25–30% of patients are not responsive to treatment. 2
Gepants (rimegepant, atogepant, ubrogepant, and zavegepant) are all antagonists of CLR/RAMP1. Rimegepant and atogepant are currently available in Europe. 2 Rimegepant is available as an oral tablet (ODT) for the acute treatment of migraine with or without aura and as a preventive treatment of episodic migraine. 3 As all these inhibitors of the CGRP pathway share the same mechanism of action, in principle, a gepant should be ineffective in patients who do not respond to anti-CGRP/R mAbs and vice versa. However, preliminary evidence shows that switching among anti-CGRP/R mAbs could be effective. 4 The European Headache Federation (EHF) guidelines indicate that, while there is insufficient evidence on the potential benefits of switching to date, it may be an option. 5 No data have been reported so far regarding switching gepants and anti-CGRP(R) mAbs. Herein, we report the first case of a patient with refractory migraine who failed two different anti-CGRP/R mAbs, and with no response to other acute treatments, including triptans, but obtained benefit from rimegepant in three out of five non-consecutive attacks.
Patient case
A 56-year-old female patient treated in our headache clinic with a long history of migraine who currently fulfills the International Classification of Headache Disorders (ICHD3) diagnostic criteria 6 for migraine without aura and chronic migraine. She reported having daily headaches (28/28 days per month) that were usually severe in intensity, with a usually bilateral pulsating pain score of 8/10 on the numeric rating scale (NRS), accompanied by phonophobia, photophobia, and/or nausea (these accompanying symptoms became reduced in frequency and intensity over the years).
She has a not better-defined family history of migraine, and a personal history of depressed mood, currently ongoing, treated with venlafaxine 300 mg/day for the last 4 months. She has never fulfilled a diagnosis for major depressive disorder according to Diagnostic and Statistical Manual of Mental Disorders (DMS-5) criteria. Furthermore, the patient had hypercholesterolemia (treated with atorvastatin 10 mg/day), gout (treated with allopurinol 150 mg/day), and controlled arterial hypertension (treated with lisinopril 20 mg/day). These concomitant disorders were stably treated in the last years. General and neurological examinations were normal, and the complete blood workup was within the normal range.
The patient fulfills the EHF definition for a diagnosis of refractory migraine, 7 which includes failure due to ineffectiveness or not tolerability of all classes (more than one molecule per class) with established migraine diagnosis. In particular, the following drugs with evidence in migraine prophylaxis were considered ineffective or not tolerated in the last 10 years: amitriptyline (up to 24 mg/day), topiramate (up to 100 mg/day), fluoxetine (up to 40 mg/day), onabotulinumtoxinA (from 155UI to 195U quarterly according to the PREEMT protocol), atenolol (up to 50 mg/day), metoprolol (up to 100 mg/day), lisinopril (up to 20 mg/day), candesartan (up to 16 mg), and finally erenumab (70 mg first month than 140 mg) then galcanezumab (240 mg loading dose than 120 mg monthly). All drugs were administered for at least the minimum amount of time recommended by international guidelines. 7 Failures were also reported for drugs with very low or no evidence for migraine, including olanzapine (5 mg/day), cinnarizine (75 mg/day), zonisamide (up to 100 mg/day). Furthermore, the patient underwent three greater occipital nerve (GON) block injections (methylprednisolone, 80 mg, and lidocaine 5 ml of 2% lidocaine) without effectiveness.
She received erenumab (70 mg first month than 140 mg) in December 2019 for 3 months with no benefit in monthly migraine days, analgesics use, disability questionnaires, nor according to patient global impression of changes (PGIC). After a washout period, galcanezumab (240 mg loading dose than 120 mg monthly) was started in May 2020. After 3 months of treatment, no benefit was achieved. However, considering the several preventive failures and the preliminary observations that patients with high burden of disease and medication overuse could achieve some amelioration after prolonged treatment, galcanezumab was maintained for 12 months. Nevertheless, excluding a transient, slight reduction in MMDs from month 5 to month 7, the patient discontinued treatment due to ineffectiveness, according to clinicians.
Recently, the patient received a prescription from her general practitioner for carbamazepine (600 mg/day) and lorazepam (5 mg/day), although neither drug is recommended for migraine treatment. The patient continued to experience daily headaches (28/28 days) with severe intensity.
Regarding acute treatment, several drugs alone or in combination were frequently used for the acute treatment of migraine attacks with partial or no beneficial effect, resulting in medication overuse (MO) during treatment with both anti-CGRP mAbs and during the use of rimegepant. Previous ineffective (as self-reported by the patient [not pain relief at 2 hours]) acute treatment included zolmitriptan, eletriptan, frovatriptan, several over-the-counter non-steroidal anti-inflammatory drugs (NSAIDs), an indomethacin-caffeine-prochlorperazine combination, and acetaminophen. The failure of more than two triptans, define the patient as triptan-resistant according to EHF classification. 8 During the last year, the attacks were treated with acetaminophen/codeine (500/30 mg), nimesulide (100 mg), subcutaneous (sc) sumatriptan 6 mg, or the simultaneous assumption of sumatriptan 100 mg, acetaminophen, and codeine. The combination of these oral drugs or sumatriptan sc provides infrequent pain relief after 2 hours, or no benefit. No freedom from pain was achieved in any attack during the last 5 years. No tachyphylaxis was reported with acute medications, but partial or lack of response was always observed from initial drug treatment.
As soon as rimegepant 75 mg became available, the drug was prescribed to the patient as an acute and not as a preventive medication, because the drug is not yet covered by the reimbursement program from the National Health Service nor from private insurance. The patient was instructed to take rimegepant if the criteria in Table 1 were met, and to record data on its effectiveness and tolerability 2 hours after administration for each attack using an eDiary. Briefly, the variables collected included: rating of headache severity, absence or presence of migraine-associated symptoms, use of rescue medications, rating of functional disability (four-point scale: normal, mildly impaired, severely impaired, requires bedrest), and recurrence of headache pain at different time points. The patient had to report all adverse events through the eDiary and/or by contacting the Headache Center. Questions about the use of triptans were collected at baseline and during treatment with rimegepant, as well the patient’s impression of the drug (evaluated on a 0–10 scale). Variables and the timing for taking rimegepant are in line with pivotal trials. 9 The effectiveness was defined as 2 h pain freedom and sustained pain freedom.
Recommended criteria of the migraine attack to allow acute migraine treatment with rimegepant.a

a Criteria modified according to pivotal trials.
Overall, the patient treated five non-consecutive migraine attacks according to the predefined criteria described above, ranging 5–30 minutes from the onset. The features of the attacks are reported in Table 2. The mean (±SD) NRS before rimegepant assumption was 7.8 ± 0.9, as all attacks reached a marked severity after a few minutes from the onset. Pain free at 2 hours was achieved in three out of five attacks (60.0%), with no recurrence of migraine in the following 24 hours (Table 3). Overall, the pain-free response was achieved after 13.3 ± 4.5 minutes considering only attacks successfully treated (five out of five attacks). The pain relief reported by the patient the day of drug assumption was also reported the following day. Notably, in one of the two attacks when no amelioration was achieved, the patient reported a prolonged benefit during the following day. In the other two out of five attacks (40.0%) with no response to treatment, the pain was reported as worsened or not changed at 2 hours after assumption, with a NRS of 8 and 9, respectively (Table 3). Including all attacks, the patient’s impression of the drug effectiveness was rated 7 out of 10, with a perceived benefit clearly superior to triptans. No adverse events were reported.
Migraine attacks features at rimegepant intake.

NRS: numeric rating scale.
a Classified as: normal function, mild impairment, severe impairment, required bedrest.
Effectiveness and tolerability after rimegepant intake.
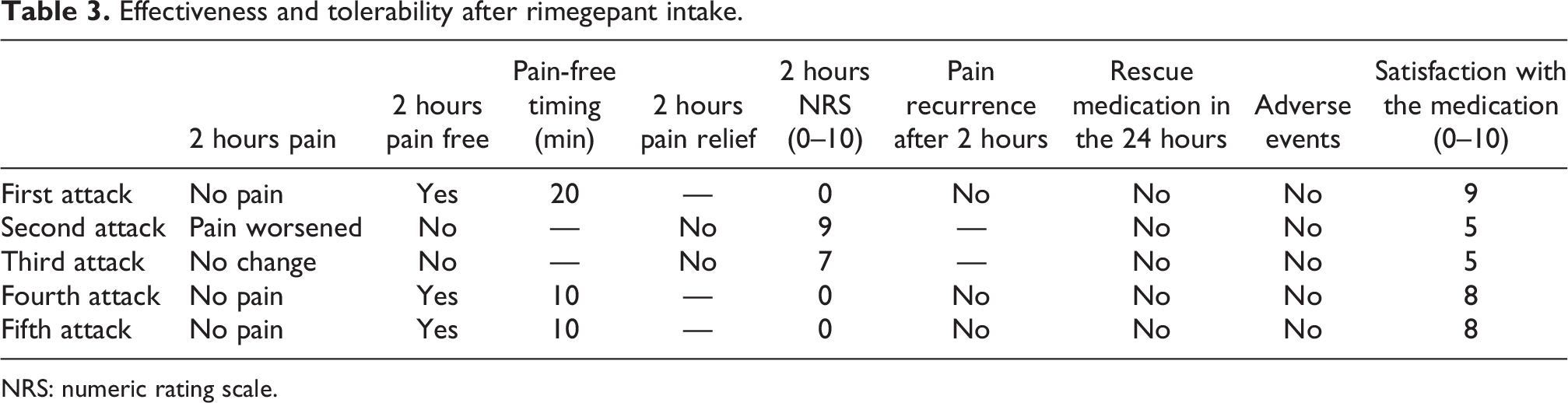
NRS: numeric rating scale.
Discussion
To our knowledge, this is the first case that assesses the use of rimegepant 75 mg as an acute treatment in a patient with refractory migraine (according to EHF definition) 7 who failed two anti-CGRP/R mAbs (erenumab and galcanezumab), one antagonizing the receptor. In this case, several preventive and acute treatment failures, severity of migraine, concomitant treatments, comorbidities, and often-inappropriate self-management of headache attacks, underline the challenge of managing, among different specialists, multifaceted patients in clinical practice.
There is no data from either RCTs or real-world studies on the effectiveness of rimegepant as an acute (or preventive) treatment in patients non-responder to anti-CGRP/R mAbs, regardless of the target. 10 In fact, RCTs that have assessed rimegepant (as well as other gepants) usually excluded all patients that had a previous failure with anti-CGRP drugs, considering the potential risk of inefficacy in these cases. 2,3
Data on the combination of different types of CGRP inhibitors to treat migraine are limited. Three reports (including a phase 1 study) have described use of gepants as acute treatment in patients receiving anti-CGRP(R) mAbs, 11 –13 suggesting that combination therapy of anti-CGRP mAbs with gepants may have the potential to satisfy the unmet need for the subset of patients who reported incomplete amelioration with anti-CGRP/R mAbs. Potential involved mechanisms, including a receptor blockade by small-molecules different from those by the anti-CGRP/R mAb or incomplete neutralization of the circulating free CGRP, are reviewed elsewhere. 14 On the other hand, combining two drug classes, targeting CGRP or its receptor, could potentially increase the risk of adverse events, associated with a complete blockade of CGRP physiological functions. However, adverse events registered with both treatments (mAbs and gepants) are rather infrequent and rarely severe. Furthermore, the three available studies on the combination of anti-CGRP/R mAbs and rimegepant as acute treatment reported few adverse events, of mild or moderate intensity, that spontaneously resolved without therapy. 11 –13 The probability of pharmacokinetic interactions between CGRP-targeted mAbs and gepants is low, considering that the pharmacokinetic profile of ubrogepant has not been changed and no safety issues were described when ubrogepant was co-administered with erenumab or galcanezumab. 10
Furthermore, a preclinical study investigated the effect of atogepant on evoked activity of mechanosensitive unmyelinated C- and thinly myelinated Aδ-meningeal nociceptors. 15 Differences between the actions of atogepant, partially inhibiting both Aδ and C-fibers, fremanezumab, inhibiting Aδ fibers only and onabotulinumtoxinA, inhibiting C-fibers only, were reported. These findings suggest a difference in their mechanisms of action in these preventive treatment acting directly or indirectly on the CGRP pathway. 15
Triptans are agonists of the 5-hydroxytryptamine (5-HT1) receptors of the B, D, and F subtypes with different affinities. At the peripheral level, triptans constrict extracerebral blood vessels (5-HT1B) and reduce trigeminal sensory nerve activation (5-HT1D and 5-HT1F), thus ultimately inhibiting vasoactive peptide release, including substance P and CGRP, 2 in rodents and probably the sole CGRP in humans. 16 Therefore, triptans indirectly share their target with anti-CGRP/R mAbs and gepants. 17 Some observational studies 2 have demonstrated that patients showing a favorable response at any time to at least one triptan had a higher probability of responding to erenumab. However, this finding was not consistently reported though studies, 18 and previous responses to triptans alone should not represent a strict criterion to select patients for erenumab treatment or other anti-CGRP/R mAbs, because a consistent proportion of patients non-responder to triptans responded to anti-CGRP/R mAbs. 17
This issue was also addressed in clinical trials investigating gepants. Post hoc data of a phase 3 trial of ubrogepant showed no differences in response between triptan naive patients and patients who are responders and non-responders to triptans, 8,10 although no prospective evaluation of triptan response has been performed, and responder status was based on retrospective self-reports. Neither general data nor post hoc evaluations have yet been reported for rimegepant. 10 In the present case, rimegepant was effective in a patient with none or very low response to three different triptans, including the sc sumatriptan, a result consistent with the positive response to anti-CGRP/R mAbs in patients non-responsive to triptans. Determining with ad hoc designed studies that non-response to largely used and effective migraine-specific drugs, such as triptans, is not a negative predictor of failure for rimegepant is of critical importance for clinical management. Indeed, a randomized study with rimegepant as acute treatment to evaluate the efficacy and tolerability of rimegepant in patients that are unsuitable for triptans due to a previous intolerance, lack of efficacy, or contraindication, is ongoing (NCT05509400).
It should be noted that the patient was on carbamazepine, a strong inducer of cytochrome 450 3A4 (CYP3A4), involved in rimegepant metabolism. 2,3 CYP3A4 inducers (including carbamazepine) are not recommended, but not contraindicated, in combination with rimegepant, due to potential reduction in drug exposure with a possible loss of efficacy. However, in this case, despite the fact that a possible increased metabolism and the ensuing reduced effect of rimegepant cannot be excluded, given the excellent response to the drug at 2 hours in three attacks, without recurrence, a hypothetical altered metabolism does not seem to have been clinically relevant. Nevertheless, avoiding drugs that induce or inhibit CYP3A4 metabolism is strongly recommended, to reduce the risk of adverse events (with inhibitors) or ineffectiveness (with inducers). The clinical impact of drug-drug interactions needs to be carefully assessed during clinical practice.
Our case report has several strengths. First, the patient has a detailed and extensive history of migraine, which was evaluated by a headache specialist and includes a complete pharmacological anamnesis. Second, we electronically recorded data on the effectiveness of rimegepant, including the timestamp of the migraine onset and rimegepant administration, and the variables reported are consistent with those used in pivotal trials. Third, we treated five non-consecutive migraine attacks according to strict criteria before administering rimegepant. Finally, we carefully monitored the patient for adverse events after each use of rimegepant.
Although they provide early evidence on the use of novel drugs in uncommon pharmacological or disease-related features, case reports are intrinsically limited by design. Further limitations should be considered. Migraine fluctuates over time with spontaneous worsening and improvement; therefore, we cannot exclude that the same or diverse drugs acting on the CGRP could have a different effectiveness and tolerability profile at different time points. However, several studies suggest that poorly treated or untreated migraine, particularly if associated with medication overuse, tends to deteriorate over time. Moreover, the concomitant use of carbamazepine could have reduced the effect of rimegepant. On the other hand, a potential benefit of carbamazepine or lorazepam on migraine is very unlikely. Finally, the contribution of the placebo effect cannot be excluded, 19 considering the reported rapid onset of action of rimegepant during successfully treated attacks. Rimegepant ODT reaches maximum plasma concentrations (Tmax) at 1.5 hours, 3 and although a response can occur before Tmax, in cases of very rapid responses, the influence of the placebo effect should be taken into account. However, it should be noted that patients with long-term chronic migraine who failed numerous preventive/acute treatments are less inclined to a placebo effect and that a positive response was reported for more than one non-consecutive attack, but not in all of them.
In conclusion, the present observation suggests that acute treatment with rimegepant 75 mg may be a viable option for patients with partial or no response to triptans who failed preventive treatments targeting the CGRP pathway, regardless of ligand or receptor. Sustained response to rimegepant in several attacks needs to be addressed. The failure of anti-CGRP/R mAbs does not necessarily preclude the use of gepants (acute and/or preventive), but further studies are urgently needed to provide evidence on the efficacy of these treatments in managing drug-resistant migraine and to identify novel treatments for patients. Prospective, cohort studies, preferably placebo-controlled RCTs, would be particularly valuable.
Clinical implications
Rimegepant may be a viable option for patients with partial or no response to triptans.
Rimegepant may be a viable option for patients who failed preventive treatments targeting the CGRP pathway.
The failure of anti-CGRP mAbs does not necessarily preclude the use of gepants.
Footnotes
Author contributions
All authors critically reviewed the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Informed consent
Informed consent was obtained from the patient to utilize her medical records for the purpose of this report. All information has been appropriately anonymized.
Data availability
All data needed to evaluate the conclusions are present in the paper. Additional data related to this paper may be requested from the corresponding author, upon reasonable request.
Declaration of conflicting interests
The authors declare the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PG: received personal fees from Allergan, Eli Lilly, Novartis, Amgen, TEVA; Grants from Amgen, TEVA, Eli Lilly, Allergan, Chiesi; Scientific Advisory Board, Endosome Therapeutics; Founding scientist of FloNext srl, Spinoff of the University of Florence. FDC received personal fees from TEVA, Eli Lilly, Novartis. LFI received personal fees from Eli Lilly, Lundbeck, Pfizer, Abbvie and TEVA. The other authors have no conflicting interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
