Abstract
Background
In this study, we aimed to evaluate the differing global access to acute and preventive medications for migraine and tension-type headache.
Methods
A custom-built questionnaire created by members of the International Headache Society Juniors Group was sent to International Headache Society members worldwide, including a list of acute and preventive treatments for migraine and tension-type headache. This list was based on evidence-based medicine guidelines. For each treatment, participants were asked about availability, type of reimbursement and variability of access within their country.
Results
Eighty-four members completed the questionnaire providing data for 84 countries. The majority were neurologists (88%) and worked at an academic/university hospital (62%). Of participants, 36% were located in high-income economy countries and 13% were located in low-income economies. Common preventive treatments such as propranolol and topiramate were available in most countries (respectively in 99% and 92% of responding countries). Sumatriptan was available in most countries (95%), whereas other triptan availability was lower. Novel migraine treatments such as rimegepant and erenumab were only available in 14% and 46% of the assessed countries, respectively.
Conclusions
Availability of headache medications, ranging from simple analgesics to novel therapies migraine-specific drugs, varied greatly across the world. Actions are needed to improve effective drug availability in many countries to ensure an adequate management of people living with headache.
This is a visual representation of the abstract.
Introduction
Primary headache disorders are chronic lifelong illnesses that have a direct impact on billions of affected individuals, as well as on their families, employers, and society at large (1). Although headache causes disability and economic losses across all world regions, cultures, and socio-economic statuses, insufficient resources in low- and middle-income countries determine increased challenges, including reduced access to care, lack or limited access to specialized headache services or even to doctors and nurses, inadequate education and awareness of headache disorders among healthcare professionals, and, ultimately, impaired diagnosis and treatment (2,3).
The management of primary headaches is experiencing an unprecedented shift in treatment paradigms thanks to the introduction of novel medications that target disease-specific pathways, such as monoclonal antibodies (mAbs) against calcitonin gene-related peptide (CGRP), gepants (CGRP antagonists) and lasmiditan (serotonin receptor agonist) (4–6). These treatments are mostly available in some high-income countries, and their elevated costs might in fact contribute to increasing the health disparities of headache medicine. Variability of access to medication across different regions of the world hinders the possibility of creating specific international treatment guidelines for primary headaches (7).
In this questionnaire-based study, led by the members of the Junior Group of the International Headache Society (IHS), we aimed to assess the availability of the main acute and preventive treatments for the most common forms of primary headache, across the majority of countries worldwide. Our overarching goal was to obtain a geographic map of availability of medications for migraine and tension-type headache (TTH) in a systematic way, paving the way for future advocacy activities and policy changes to reduce the availability gap.
Methods
We developed an online questionnaire listing the most commonly used medications for migraine and TTH, divided by drug class, for both acute (i.e., analgesics, antiemetics, triptans, neuromodulation, gepants and lasmiditan) and preventive (i.e., anti-hypertensives, antiepileptics, antidepressants, onabotulinumtoxinA, CGRP-targeting mAbs and CGRP antagonists) treatments (Table 1). The questionnaire was based on, but not limited to, treatments recommended in current national guidelines, and encompassed widely used medications in clinical practice.
Written version of the online questionnaire on acute and preventive treatments for migraine and tension-type headache.

Please comment below if you believe the IHS could aid your country or region with specific unmet needs:
The questionnaire captured demographics of participants (age, sex, role, place of employment and country of employment), and, for each drug: i) within-country availability (yes/no/unknown/expected within 5 years); ii) type of prescription (prescription-only/pharmacy or over the counter/general sales list/private prescription); iii) type of reimbursement (full/partial/none); iv) within-country variability of access. The questionnaire was developed by the junior authors (FP, IdB, RM, DGA, MNPS, MAK, OBdD) and approved by the senior authors (CT, AM) as well as by the IHS board of trustees. Before the final launch, the questionnaire was revised by a total of ten headache specialists from different countries and regions of the world, to ensure adequate representation of treatments. The survey was then developed into an electronic online version and circulated among headache specialists who were members of the IHS. In order to be eligible for the survey, participants had to be qualified medical doctors with a minimum of three-year experience in treating headache disorders. We only included in the analysis the questionnaires that provided responses to at least 30% of the questions.
The primary endpoint was the availability of different headache treatments in different regions of the world. For analyses purposes, countries were divided by regions and income based on the World Bank definition (8). Statistical analyses were run in R, version 4.2.1. If multiple participants from the same country answered the questionnaire, two investigators (FP and IdB) checked the questionnaires separately and then selected the one that was most complete.
Results
We received a total of 147 questionnaires. Of these, 18 were excluded due to insufficient data. Of the remaining 129, 84 belonged to individual countries and were included in the evaluation; the remaining 45 were less complete questionnaires from countries for which data was already available, and were thus excluded.
The large majority of respondents were fully trained neurologists (n = 74; 88%). The other respondents where neurology residents (n = 5; 6%), fellows (n = 3; 4%) and general practitioners (n = 2, 2%). The F:M ratio was 45:39. Over 60% worked in an academic hospital setting (n = 52), 24% in a tertiary center, 8% in a regional hospital, 5% in a general practice. Forty percent (n = 34) also conducted private practice. Thirty-three percent (n = 28) of responders were based in Africa, 30% (n = 25) in Europe, 17% (n = 14) in the American continent, 18% (n = 15) in Asia and 2% (n = 2) in Oceania. Of these, 41% were located in high-income countries, 28% in upper-middle income countries, 30% in lower-middle income countries and 15% in low-income economies.
With regard to acute medications, paracetamol/acetaminophen and ibuprofen are available in over 90% of the countries (Figure 1(a) and 2(a)). Drugs such as indomethacin and sumatriptan are reported as mostly available, except for certain countries in Africa, Latin America and Asia (Figure 1(a)), however full reimbursement is rare (see Figure 3 and Online Supplementary Table 1). Availability for other triptans is significantly lower, with rizatriptan and frovatriptan being available in only 53% and 38% of countries, respectively. Lasmiditan and ubrogepant are available in less than 25% of the countries, while rimegepant is available in some countries in Europe and unavailable in most remaining world regions.
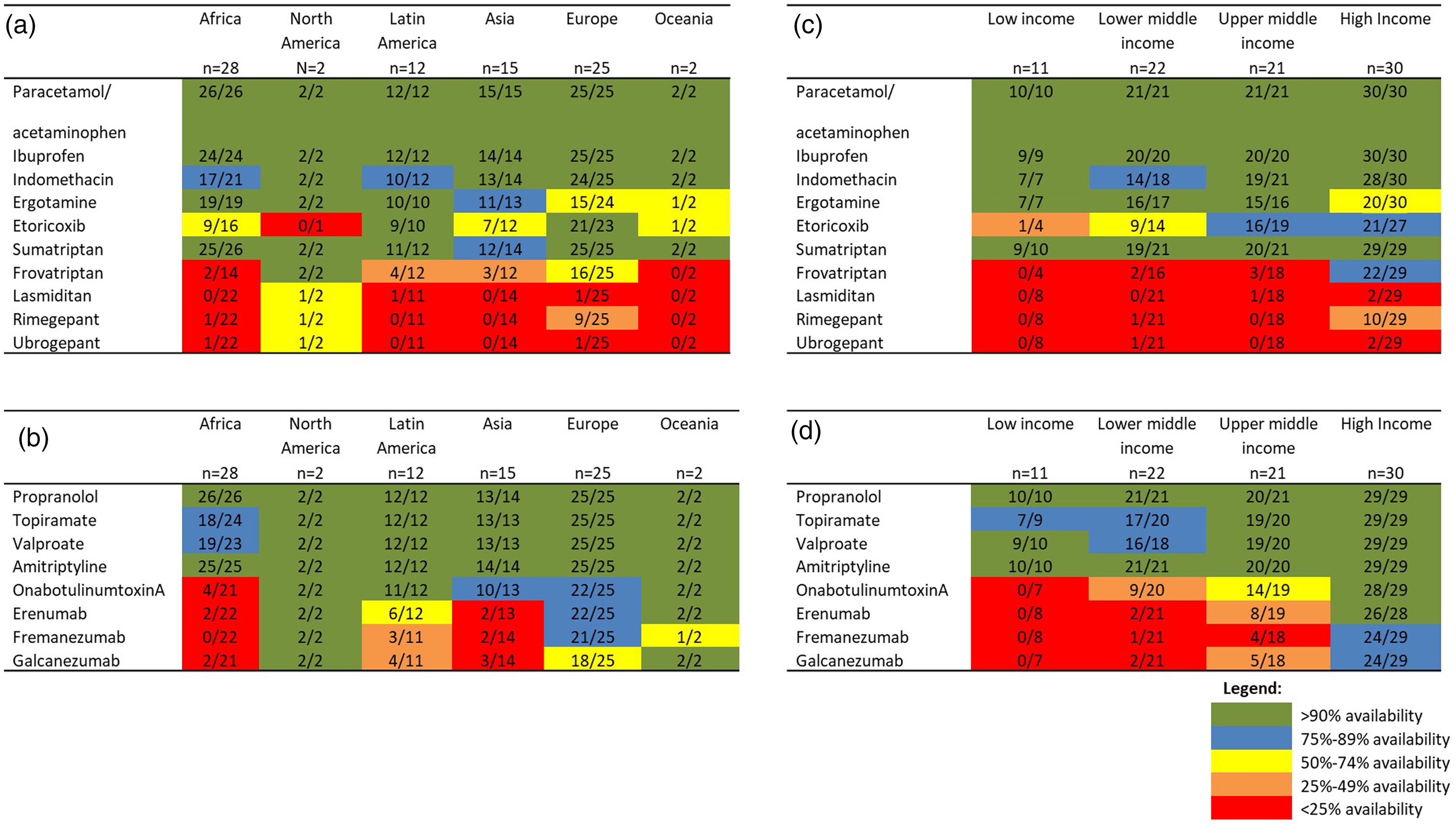
Availability of some traditional and new acute (a) and preventive (b) drugs for the treatment of migraine and tension-type headache across the five world regions and economies (c and d) as of December 2023. For a complete overview of drug availability and reimbursement type per country, see Online Supplementary Table 1.
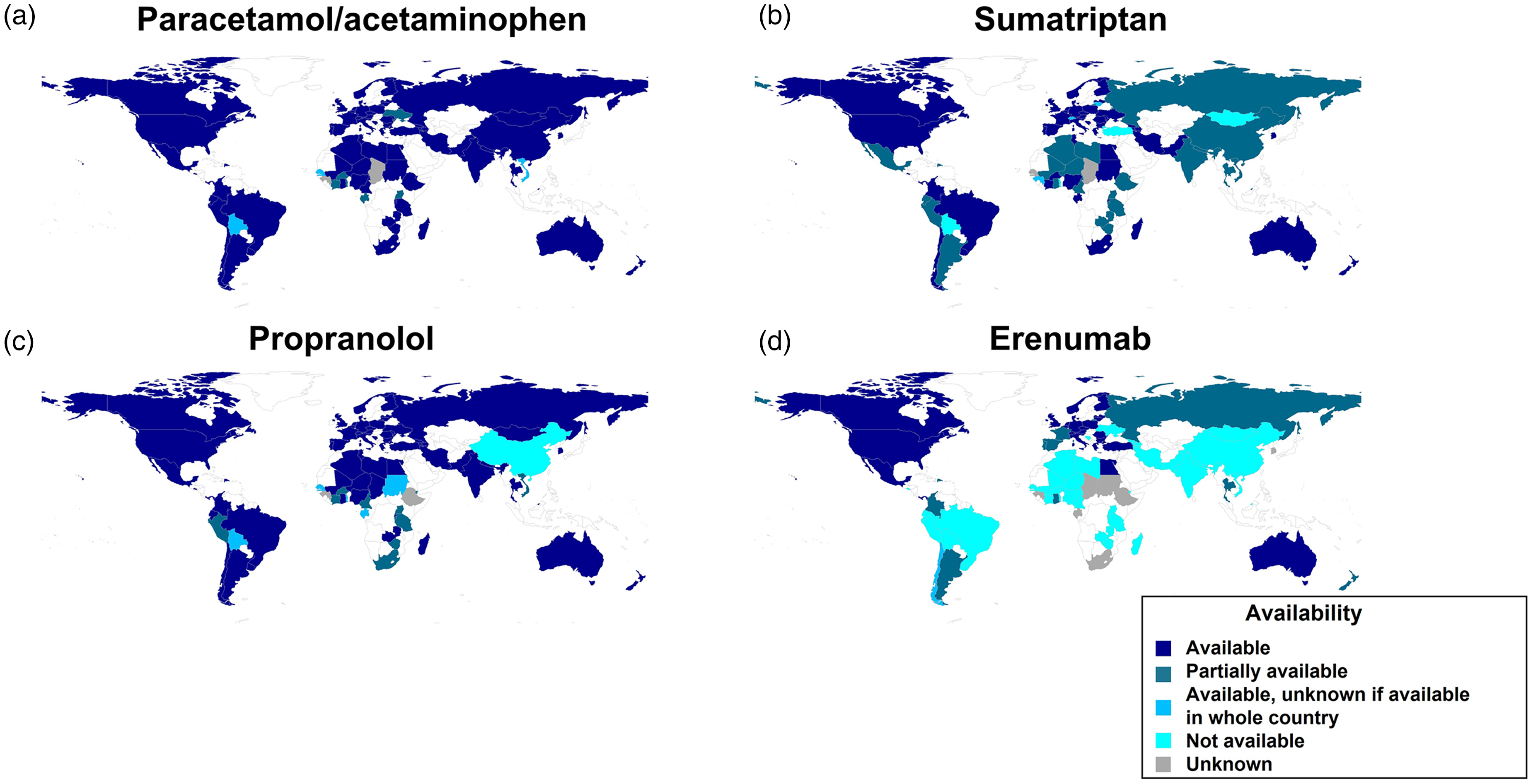
Worldwide distribution and within-country availability for paracetamol/Acetaminophen (a), sumatriptan (b), propranolol (c), and erenumab (d) as of December 2023. These drugs have been selected as they represent widely used acute (a and b) and preventive (c and d) options for migraine, that are both non-specific (a and c) and targeted to migraine-specific disease mechanisms (b and d).
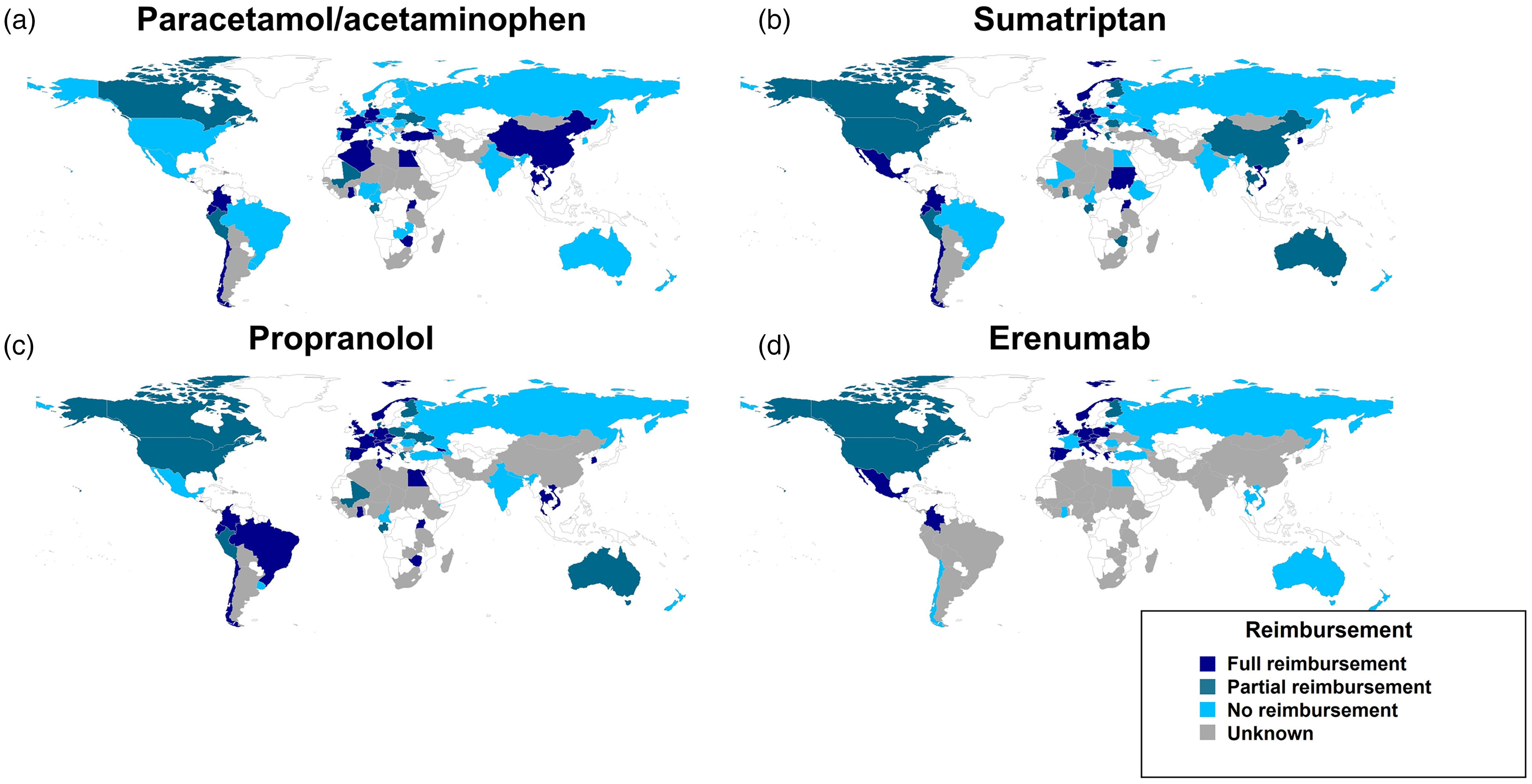
Per country reimbursement for paracetamol/Acetaminophen (a), sumatriptan (b), propranolol (c), and erenumab (d) as of December 2023. These drugs have been selected as they represent widely used acute (a and b) and preventive (c and d) options for migraine, that are both non-specific (a and c) and targeted to migraine-specific disease mechanisms (b and d).
For preventives, common drugs such as propranolol and amitriptyline are available in almost all responding countries (Figure 1(b) and 2(c)), whereas topiramate and valproate, although present in most countries, have a lower availability in Africa (75–89%). OnabotulinumtoxinA is available in almost all countries in Latin America, Europe and Oceania, while availability is <90% in Asia and it is only available in four of the 21 countries for which data was collected in Africa. Among the novel anti-CGRP mAbs, erenumab is largely available in North America, Western Europe and Oceania, but mostly unavailable in Latin America, Asia and Africa (Figure 1(b) and Figure 2(d)). Neuromodulation, including transcranial magnetic stimulation and non-invasive vagus nerve stimulation, is largely unavailable in most interviewed countries (see Online Supplementary Table 1).
Figure 3 shows reimbursement policies in different countries for paracetamol, sumatriptan, propranolol and erenumab. Figure 4 is an infographic representing the absolute number of people in the world who have access to either sumatriptan, propranolol, onabotulinumtoxinA or erenumab.
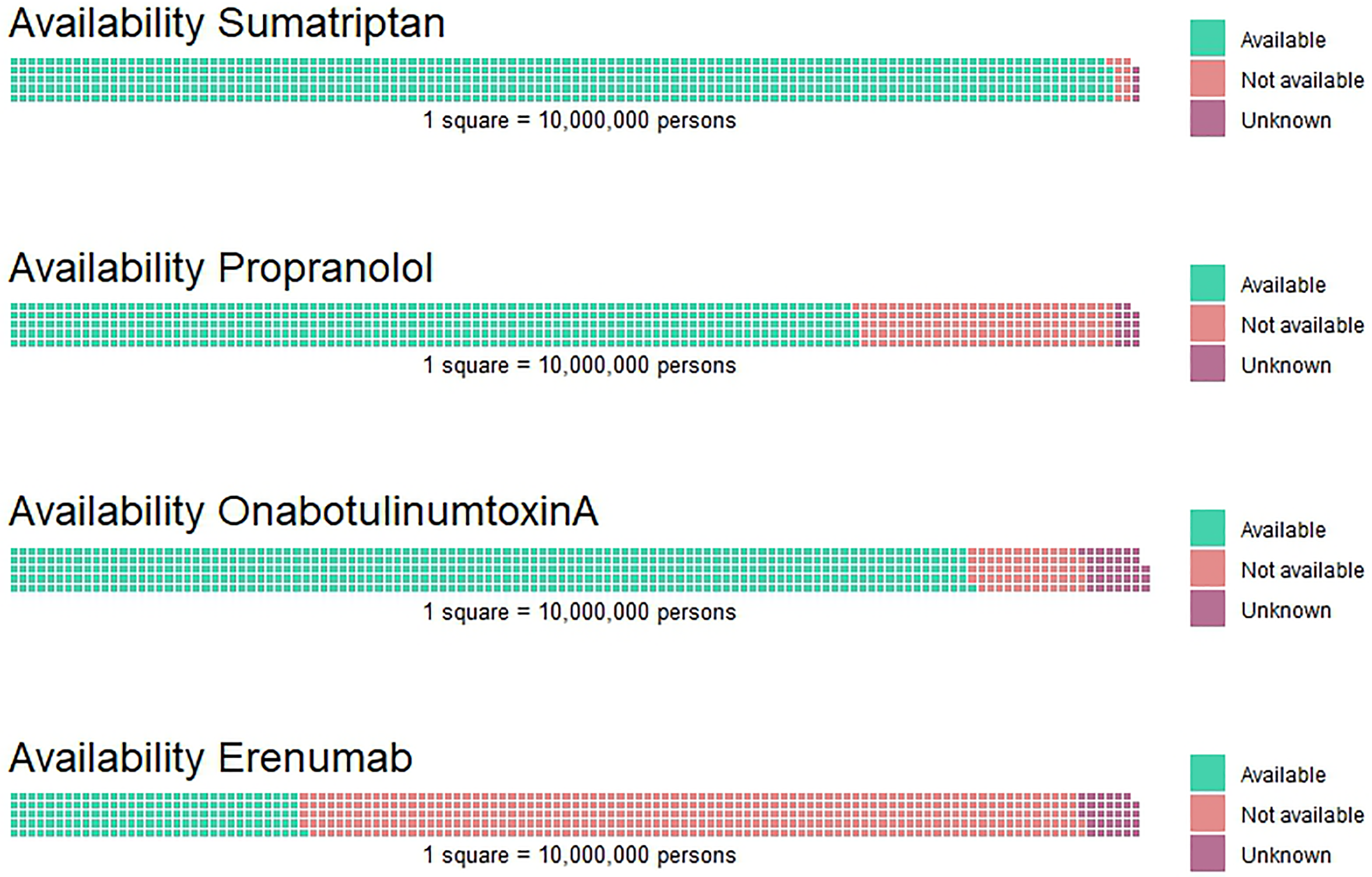
Global availability of sumatriptan, propranolol, onabotulinumtoxinA, and erenumab, represented by the number of people who have access to each medication. Each square in the image represents 10 million people.
A complete list of medication availability and type of reimbursement per country is presented in Online Supplementary Table 1.
Discussion and conclusion
Our survey shows that a large variability exists across the globe when it comes to availability and reimbursement of migraine and TTH medication. The disparities in availability and reimbursement of headache medications are disproportionate to migraine prevalence and related disability across the world (9). This survey demonstrated that the greatest areas in need of access to headache medications are Africa, Asia and Latin America.
The World Health Organization (WHO) Model List of Essential Medicines (10) is a list of medications updated every two years by the WHO, indicating drugs that are considered to be the most essential to meet the needs of a healthcare system, and is often used by countries to develop national lists of essential medicines. Notwithstanding, many low-income countries lack most of the drugs on the Essential List (11), therefore it is not surprising that the more expensive and novel treatments are not available at all (Figure 1(c) and 1(d)). The migraine medications listed on the WHO List of Essential Medicines are acetylsalicylic acid, ibuprofen, paracetamol (acetaminophen), sumatriptan, and propranolol (10). Our survey unfortunately shows that their availability is far from optimal in Africa, Asia and Latin America (Figure 2(a)–2(c)). Other commonly used preventive drugs like valproic acid and amitriptyline can be found in the list under indications different from migraine, e.g., epilepsy and depression, and might thus be available, albeit not reimbursed for treating headache, in some countries. Even when medications are available, their costs may be prohibitive and there may be disparities regarding their availability within a given country. When paracetamol, acetylsalicylic acid, ibuprofen, sumatriptan, and propranolol were available in low-income countries, their access still differed across sub-areas. For example, in Ethiopia, a prescription is required from a specialist to get sumatriptan, but the medication is not reimbursed, while in Ghana the drug is partially reimbursed.
Reimbursement is also a major issue for novel headache treatments such as ditans and drugs targeting the CGRP pathway. While these drugs are currently unavailable in the majority of regions of the world, they are also unlikely to be reimbursed in the next future in most healthcare systems that are not prepared for sustaining their high costs.
The main limitation of this study is the selection bias from the survey being promoted through IHS membership, as we expect the countries of the greatest needs in terms of headache care to be those with no doctor nor health provider affiliated with the IHS. It must further be noted that for novel drugs that have only recently been marketed, availability may rapidly change. However, we feel that the fact that we managed to collect data from a very large number of countries across the globe is a very important step toward the future, more capillary initiatives, and these findings are helpful for identifyingtarget areas that are most in need of supportive efforts by IHS and other relevant organizations. Additionally, this survey relied completely on the knowledge the participants had of the healthcare system in their country. We tried to take this into account by requiring that each participant had three years experience with headache care. Finally, this study unfortunately contained missing data, which led to medication information not being available for all countries.
In summary, these findings highlight a large unmet need for equitable access to headache medications across the world. This survey also represents the first effort at studying the differences in headache medication access around the world. We trust that the current findings and regular updates, which will aim to reflect the quick changes of drug availability in the coming years, will facilitate the creation of up-to-date and truly international treatment guidelines for primary headaches and will foster initiatives for a more equitable access of headache drugs.
Now is the time for headache medicine to actively participate in the Intersectoral Global Action Plan (IGAP) (12). International collaborations between headache specialists, community and health workers, global professional organizations (such as IHS), global patient advocacy coalitions, local professional and patient organizations pharmaceutical companies, political leaders, regulatory authorities, health ministries, and stakeholders represent an important way forward to ensure advocacy and policy efforts that work towards reducing this important gap. Higher-income economies and organizations such as the IHS also hold a responsibility towards lower-income countries in working towards increased availability and affordability of novel headache medications.
Article highlights
Availability of headache medications, ranging from simple analgesics to novel therapies migraine-specific drugs, varied greatly across the world.
Common preventive treatments such as propranolol and topiramate were available in most countries that completed the questionnaire.
Sumatriptan was available in most countries, whereas other triptans showed low availability.
Novel migraine treatments such as rimegepant and erenumab were largely unavailable throughout Latin America, Asia and Africa
Supplemental Material
sj-docx-1-cep-10.1177_03331024241297688 - Supplemental material for Worldwide availability of medications for migraine and tension-type headache: A survey of the International Headache Society
Supplemental material, sj-docx-1-cep-10.1177_03331024241297688 for Worldwide availability of medications for migraine and tension-type headache: A survey of the International Headache Society by Francesca Puledda, Irene de Boer, Roberta Messina, David Garcia-Azorin, Marcio Nattan Portes Souza, Mohammad Al-Mahdi Al-Karagholi, Olivia Begasse de Dhaem, Cristina Tassorelli and Arne May in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
