Abstract
Background
Direct comparisons of the tolerability and safety of migraine preventive treatments targeting the calcitonin gene-related peptide pathway are lacking. This study aimed to compare the safety and tolerability of anti-calcitonin gene-related peptide monoclonal antibodies and gepants in migraine prevention.
Methods
A network meta-analysis of phase 3 randomized controlled trials assessing the safety and tolerability of anti-calcitonin gene-related peptide monoclonal antibodies (erenumab, eptinezumab, fremanezumab, or galcanezumab) and gepants (atogepant, rimegepant) in migraine prevention was performed. Primary outcomes were treatment-emergent adverse events and serious adverse events. Secondary outcomes included any adverse events, adverse events leading to treatment discontinuation and individual adverse events.
Results
We included 19 randomized controlled trials, comprising 14,584 patients. Atogepant 120 mg (OR 2.22, 95% CI [1.26, 3.91]) and galcanezumab 240 mg (OR 1.63, 95% CI [1.33, 2.00]) showed the largest odds of treatment-emergent adverse events compared to placebo. While eptinezumab 30 mg had greater odds of adverse events leading to treatment discontinuation (OR 2.62, 95% CI [1.03,6.66]). No significant differences in serious adverse events were found between active treatments and placebo. Eptinezumab was associated with the lowest odds of treatment-emergent adverse events and serious adverse events compared to placebo, whereas erenumab was associated with the lowest odds of any adverse events and quarterly fremanezumab with the lowest odds of treatment discontinuation due to adverse events.
Conclusion
Monoclonal antibodies targeting the calcitonin gene-related peptide pathway and gepants are a safe and well tolerated option for migraine prevention.
Introduction
Migraine attacks can be highly disabling, can significantly impair quality of life and lead to loss of productivity (1). Moreover, migraine is first cause of disability in people under 50 (2). Migraine preventive treatments primarily aim to reduce headache frequency, attack severity, patient disability and the risk of medication overuse (3). Effective preventives can also improve patients’ quality of life, and response to acute treatments (4). For preventive treatment decisions, it is important to consider the severity and frequency of migraine attacks, previous treatment history, presence of comorbidities, the use of concomitant medications, as well as treatments effectiveness, safety and side effects. An adequate tolerability is essential to ensure patients’ compliance and therefore treatment effectiveness (5). The most common reasons for poor compliance and adherence to oral treatments commonly used for migraine prevention are poor tolerability and a high number of adverse events (6).
New migraine-specific preventive therapies targeting the calcitonin gene-related peptide (CGRP) pathway have been recently introduced (7). These novel treatments are injectable monoclonal antibodies targeting the CGRP ligand (fremanezumab, galcanezumab, eptinezumab) or its receptor (erenumab), and the orally administrated small molecule CGRP receptor antagonists, the so called gepants (atogepant and rimegepant) (8). Randomized controlled trials (RCTs) demonstrated that these new therapies are safe, well tolerated and effective in migraine prevention, thus leading to their approval for clinical use (9–14).
Direct comparisons of the efficacy, safety and tolerability of anti-CGRP monoclonal antibodies and gepants are lacking. Network meta-analysis is an alternative method that allows to compare distinct treatments indirectly, if both have been previously matched to a common comparator such as placebo (15).
This systematic review with network meta-analysis aims to compare the safety and tolerability of monoclonal antibodies and small molecules targeting the CGRP pathway in migraine prevention. We also aimed to rank these treatments based on their safety and tolerability.
Methods
Search strategy and study registration
As part of a Series Paper, a similar methodology was used in the study evaluating the efficacy of anti-CGRP monoclonal antibodies and gepants. We included phase 3 RCTs assessing the safety and tolerability of anti-CGRP monoclonal antibodies (erenumab, eptinezumab, fremanezumab, or galcanezumab) and gepants (atogepant, rimegepant) in migraine prevention. A systemic literature research was performed using the following online databases: MEDLINE (PubMed), the Cochrane Central Register of Controlled Trials (CENTRAL) and Embase from the inception date up to 11 February 2022. A detailed description of the search strategy for all databases is included in online Supplementary Tables 1–3. The systematic review and network meta-analysis protocol was developed following the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and registered at PROSPERO database (registration number: CRD42022310579, available at: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42022310579 and as online Supplemental material).
Eligibility criteria
Phase 3 RCTs had to enroll adult patients with a diagnosis of migraine according to the diagnostic criteria of the International Classification of Headache Disorders, (ICHD-3 beta and ICHD-3) to be included (16,17). All studies, in which migraine patients received atogepant, erenumab, eptinezumab, fremanezumab, galcanezumab or rimegepant, at any dosage, were included. Each active intervention had to be compared to placebo. Only RCTs that satisfied the population, intervention, comparator, and outcome (PICO) criteria pre-defined in the protocol were included (online Supplemental material). Exclusion criteria were an open-label study design, pediatric or adolescent participants under the age of 18, non-RCTs, use of other languages other than English, report of post-hoc and secondary analyses, studies reporting irrelevant comparisons or outcomes, editorials, conference abstracts and letters.
Outcomes
The primary outcomes of this meta-analysis were treatment-emergent adverse events (TEAEs) and serious adverse events (SAEs). A TEAE was defined as a new event that occurs after treatment administration or an event that already existed but worsen following treatment intake. A SAE was defined as any fatal or life- threatening adverse events (AEs). Secondary outcomes were any AEs, AEs leading to treatment discontinuation and individual AEs most frequently reported in previous RCTs (9–12), including injection site pain, injection site erythema, injection site induration, injection site pruritus, injection site hemorrhage, back pain, neck pain, nasopharyngitis, upper respiratory tract infections, sinusitis, dizziness, fatigue, insomnia, diarrhoea, constipation, nausea and cough. Outcomes selection was based on the guidelines of the International Headache Society for RCTs of preventive treatments in adult patients with episodic and chronic migraine (18,19).
Data collection
After checking for duplicates, four reviewers (RM, FP, FH, ERL) independently screened the title, abstract and full-text of studies obtained from the databases search to determine eligibility according to the protocol. This screening process was performed using Rayyan online software (20). Data were then extracted from included studies and recorded in a predesigned excel spreadsheet by four independent reviewers (RM, FP, FH, ERL). During data screening and extraction, any possible disagreements were resolved by discussion and with assistance of a fifth reviewer or senior author (HCD), when needed. For each study the following information were collected: study title, year of publication, authors’ name, trial registration number, trial acronym, type of active treatment, active treatment dose, study duration, total number of participants, number of patients included in each study arm, patients’ demographic characteristics (age, sex, ethnicity), BMI, migraine type (episodic, chronic), presence of aura, disease duration and history of prior preventive treatments.
Assessment of risk of bias
The quality of the included studies was assessed by two independent reviewers (RM, FH) using version 2 of the Cochrane risk-of-bias tool for randomized trials (RoB 2) (21). The risk of bias assessment was made for five distinct domains: a) bias arising from the randomization process; b) bias due to deviations from intended interventions; c) bias due to missing outcome data; d) bias in measurement of the outcome; e) bias in selection of the reported result. An overall risk-of-bias judgment (low risk of bias, some concerns and high risk of bias) was reported for each study based on the domain-level judgement provided by the RoB2. Any disagreements between the two reviewers were resolved by discussion and with assistance of a third reviewer or senior author (HCD), when needed.
Data analysis
Demographic and clinical characteristics of patients randomized in the included studies were reported as mean and standard deviations or as frequencies. The Mantel-Haenszel method for network meta-analysis was applied to all primary and secondary outcomes to take into account the characteristic of rare events (22). In addition, pair-wise meta-analyses were performed to compare each active treatment to placebo using the Mantel-Haenszel method. Although it is difficult to detect heterogeneity given the small number of studies for the single treatments, it was graphically assessed for pair-wise comparisons. In addition, the I2 index, between-study variance τ2 and p-values for the Cochrane’s Q test were reported. Studies with zero events in all arms were not considered for network or pair-wise analysis. To all other studies with zero events arms, the treatment arm continuity correction was applied, as this method takes into account different sample sizes and shall outperform fixed continuity corrections (23). All treatments were ranked based on primary and secondary outcomes using the P-score, which is a frequentist analogue to the Surface Under the Cumulative Ranking curve (SUCRA) with a similar interpretation (24). Results were reported in terms of odds ratios and 95% confidence intervals. All analyses were performed in R version 4.1.2 (25).
Results
Study selection
A total of 2314 studies were initially identified from electronic databases (PubMed = 256, Cochrane central = 967, Embase = 1091). After removal of 1181 duplicates, 1133 studies were screened for eligibility based on their title, abstract and full-text. Finally, a total of nineteen studies (11,13,14,26–41) were selected and included in the meta-analysis. A flow chart of study selection is represented in Figure 1. The overall risk of bias of the included studies was low. In only one study the randomization process was judged with some concerns (online Supplementary Figure 1) (40). The same studies were included in the network meta-analysis published in the first section of this Series Paper investigating the efficacy of monoclonal antibodies and small molecules targeting the CGRP pathway.

Flow chart of study selection.
Study and participants characteristics
All the included studies were published between November 2017 and November 2021. All studies were multicentre and involved different geographical areas. Two studies (13,34) investigated daily oral atogepant (10 mg, 30 mg, 60 mg, 120 mg), two studies (40,41) examined quarterly intravenous eptinezumab (30 mg, 100 mg, 300 mg), five studies (32,33,35,37,39) investigated monthly subcutaneous erenumab (70 mg, 140 mg), four studies (11,26,36,38) examined monthly subcutaneous galcanezumab (120 mg, 240 mg), one study (14) investigated oral rimegepant (75 mg) administrated every other day and five studies (27–31) examined monthly (225 mg) and quarterly (675 mg) subcutaneous fremanezumab (online Supplementary Table 4). A total of 14,584 patients were randomized in the included studies. Seventy-nine percent of participants were female (11,580 out of 14,584) and the mean age of patients ranged between 38 and 46. A detailed description of the main clinical and demographic characteristics of patients randomized in the included studies is reported in Table 1.
Clinical and demographic characteristics of patients randomized in the studies included in the meta-analysis.
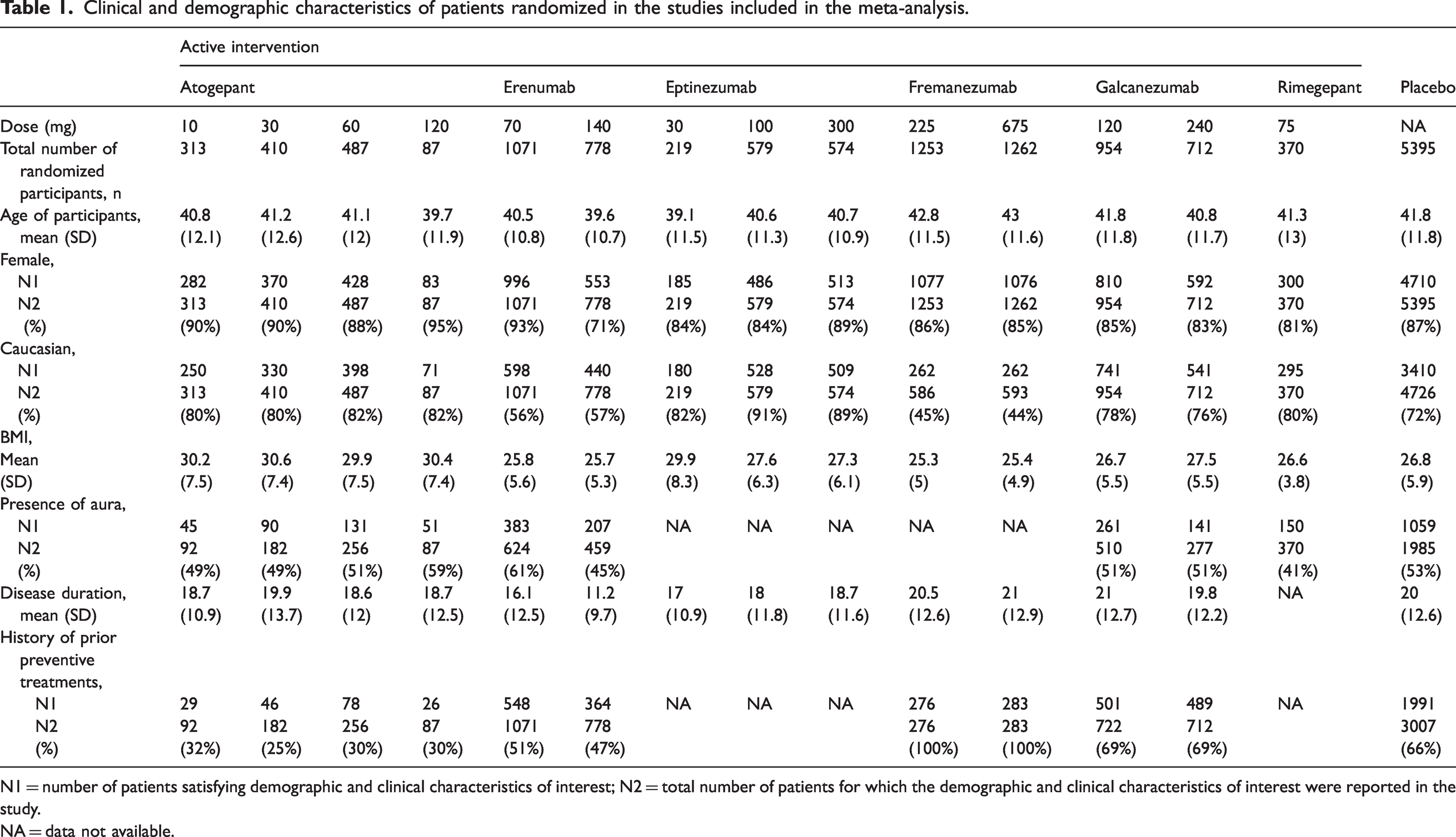
N1 = number of patients satisfying demographic and clinical characteristics of interest; N2 = total number of patients for which the demographic and clinical characteristics of interest were reported in the study.
NA = data not available.
Anti-CGRP monoclonal antibodies versus placebo
Both network meta-analysis and direct comparisons between active drugs and placebo showed significant greater odds of TEAEs during treatment with fremanezumab (225 mg and 675 mg) and galcanezumab (120 mg and 240 mg) compared to placebo ( Figure 2A, Figure 3 and online Supplementary Figure 2). The highest odds of TEAEs was estimated for patients treated with galcanezumab 240 mg followed by galcanezumab 120 mg (Figure 2A). The significant effect associated with fremanezumab can probably be explained by the large number of studies and patients for this treatment. The analysis investigating RCTs in episodic and chronic migraine separately, confirmed higher odds of TEAEs in episodic and chronic patients treated with galcanezumab compared to placebo, with a larger effect in the study including only patients with chronic migraine (Figure 2A).

Forest plots representing results from network meta-analysis comparing active treatments and placebo for primary safety outcomes. Odds ratios higher than 1 indicate that the odds of having a treatment-emergent adverse event (a) or a serious adverse event (b) is higher in the treatment group compared to the placebo. Odds ratios were reported for all RCTs and for studies investigating only episodic or chronic migraine patients. Abbreviations: CI = confidence interval, OR = odds ratio.

League table representing results from network meta-analysis comparing active treatments and placebo against each other for treatment-related adverse events (light blue) and serious adverse events (pink powder). For treatment-related adverse events, column-defining therapies were compared to row-defining treatments. Odds ratios higher than one favor events in the column-defining therapy and odds ratios lower than one favor events in the row-defining treatment. For serious adverse events, row-defining therapies were compared to column-defining treatments. Odds ratios higher than one favor events in the row-defining therapy and odds ratios lower than one favor events in the column-defining treatment. Significant odds ratios (95% CI) are reported in bold. Abbreviations: CI = confidence interval, OR = odds ratio.
No difference in TEAEs can be detected between patients treated with eptinezumab 30 mg or 100 mg and patients treated with placebo with odds ratios almost equal to one, while for all other anti-CGRP monoclonal antibodies at least a slight trend could be seen with TEAEs being more likely in patients receiving the active treatment than placebo (Figure 2A).
In the pair-wise comparisons, some heterogeneity between RCTs can be seen especially for galcanezumab and fremanezumab (online Supplementary Figure 2). The study investigating galcanezumab only in chronic migraine patients (26) showed a much larger effect compared to other studies including both episodic and chronic patients treated with galcanezumab. Regarding fremanezumab, two studies (27,28) showed greater odds of TEAEs compared to placebo, while a higher odds of TEAEs with placebo rather than fremanezumab has been observed in another study (29) and an almost equal odds of having TEAEs with fremanezumab and placebo has been estimated in two other RCTs (30,31).
No significant differences of SAEs were found between active treatments and placebo (Figure 2B, Figure 3 and online Supplementary Figure 3). Both galcanezumab 120 mg and 240 mg showed the largest odds of suffering from SAEs in comparison to placebo with odds ratios of 1.64 (95% CI [0.80, 3.38]) and 1.66 [0.77, 3.56]. All other estimates for the anti CGRP monoclonal antibodies were either smaller than one or close by (Figure 2B).
Data regarding the presence of any adverse events were not reported in RCTs investigating eptinezumab. Results of secondary outcomes showed significant higher odds of reporting any AEs in patients receiving quarterly fremanezumab 675 mg in comparison to patients treated with placebo (Figure 4A and online Supplementary Figure 4). Even if not reaching statistical significance, patients treated with galcanezumab were also more likely to experience any AEs compared to placebo, with estimated odds ratios similar to fremanezumab (Figure 4A). When considering only RCTs in chronic migraine, galcanezumab 120 mg and monthly fremanezumab 225 mg were significantly associated with greater odds of having AEs compared to placebo with similar effects to galcanezumab 240 mg and quarterly fremanezumab 675 mg in chronic migraine patients (Figure 4A). On the other hand, erenumab was the treatment with the lowest odds ratios of having any AEs with estimates smaller than one, even if this result did not reach statistical significance (Figure 4A).
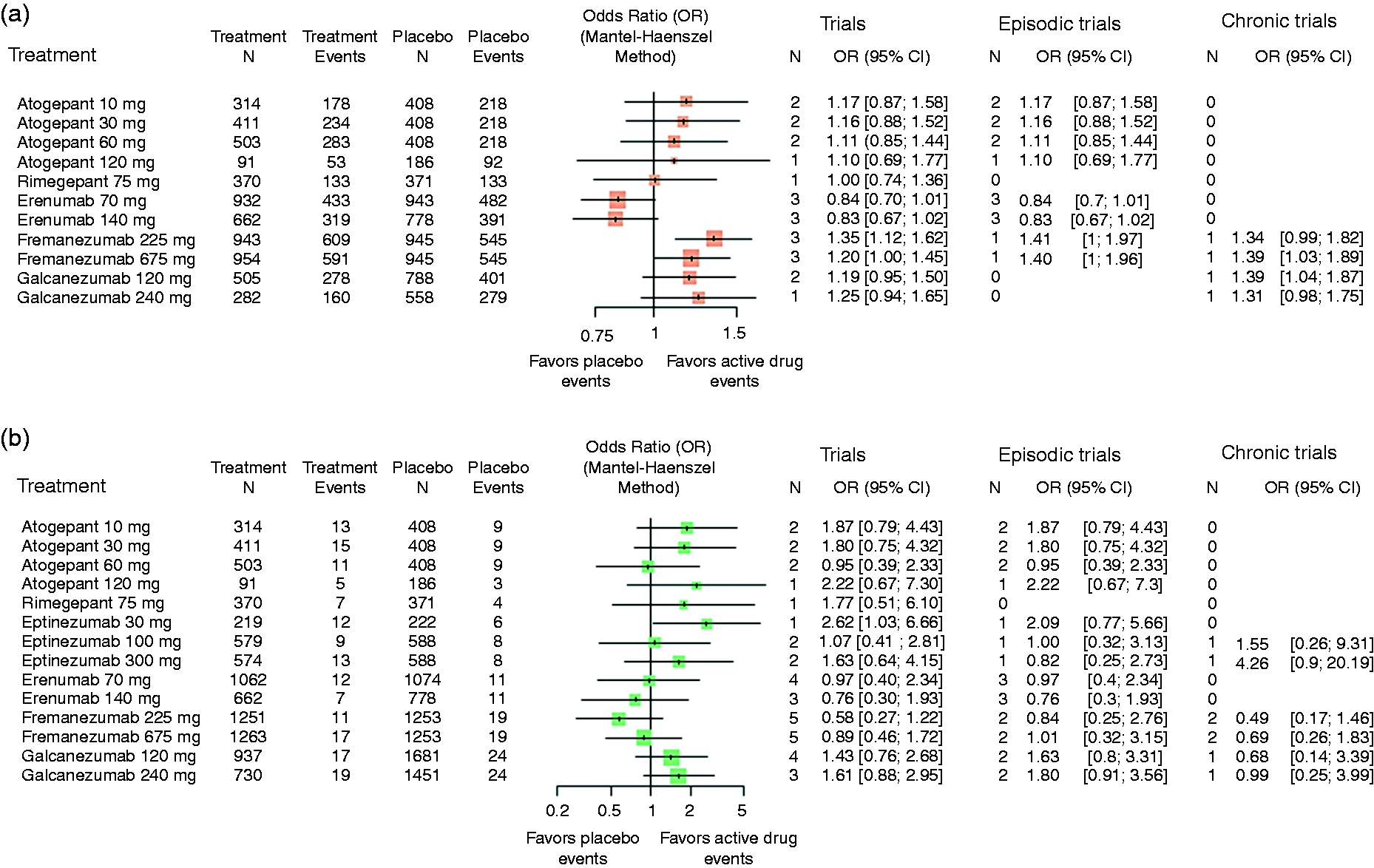
Forest plots representing results from network meta-analysis comparing active treatments and placebo for secondary safety outcomes. Odds ratios higher than one indicate that the odds of having any adverse event (a) or adverse event leading to treatment discontinuation (b) is higher in the treatment group compared to the placebo. Odds ratios were reported for all RCTs and for studies investigating only episodic or chronic migraine patients. Abbreviations: CI = confidence interval, OR = odds ratio.
The network meta-analysis showed that eptinezumab 30 mg was the only treatment significantly associated with higher odds of having AEs leading to withdrawal of treatment compared to placebo (Figure 4B). Although not statistically significant, a similar trend but with smaller effects was observed for eptinezumab 300 mg, as well as for galcanezumab 120 mg and 240 mg (Figure 4B). Whereas, both doses of erenumab and fremanezumab had the smallest odds of AEs leading to treatment discontinuation with estimated odds ratios under one (Figure 4B). The pair-wise meta-analysis comparing eptinezumab 30 mg to placebo did confirm the network meta-analysis’s result, but with a smaller estimated effect (online Supplementary Figure 5).
Compared to placebo, fremanezumab 675 mg and galcanezumab (120 and 240 mg) showed greater odds of injection site erythema, induration and pruritus, while erenumab 70 mg and fremanezumab 225 mg had higher odds of injection site pain. Fremanezumab 225 mg also showed higher odds of injection site erythema and diarrhea, and erenumab 70 mg and 140 mg had greater odds of constipation, compared to placebo. Moreover, the odds of influenza were estimated larger in patients with fremanezumab 675 mg than in placebo patients. Placebo treatment showed higher odds of nasopharyngitis compared to galcanezumab 240 mg and greater odds of sinusitis compared to eptinezumab 100 mg (Table 2). Other effects, e.g., a higher risk of fatigue or a smaller risk of nausea in most of the treatments could be observed in comparison to placebo, but with no significant result in regard to the odds ratio.
Results from network meta-analysis comparing anti-CGRP monoclonal antibodies and placebo for single adverse events. Odds ratios higher than one indicate that the odds of having an adverse event is higher in the treatment group compared to the placebo.

Measures are reported as odds ratio and 95% confidence interval.
NA = data not available.
Gepants versus placebo
Network meta-analysis and direct comparisons showed significant higher odds of reporting TEAEs in patients treated with atogepant (all doses) compared to those receiving placebo (Figure 2A, Figure 3 and online Supplementary Figure 2). Although not statistically significant, it is also estimated that TEAEs are more likely in patients receiving rimegepant than placebo (Figure 2A).
No significant differences in the odds of having SAEs were found between gepants and placebo, with estimated odds ratios under one for all treatments except for atogepant 10 mg. It can be observed that higher doses of atogepant lead to smaller odds of SAEs compared to placebo (Figure 2B and online Supplementary Figure 3). For any AEs it is estimated that patients treated with gepants have slightly larger odds of having an event compared to placebo except for rimegepant, and for AEs causing treatment discontinuation, it is estimated that patients treated with gepants are more likely to have an event compared to placebo with exception of atogepant 60 mg, even if these findings did not reach statistical significance (Figure 4 and online Supplementary Figure 4-5).
In the pair-wise comparisons some heterogeneity between the studies investigating atogepant can be seen especially for TEAEs, any AEs and AEs causing treatment discontinuation (online Supplementary Figures 1, 4 and 5). RCTs investigating the presence of any AEs in patients receiving atogepant 10 mg, 30 mg and 60 mg exhibited an opposite behavior having estimates higher than one in one study and lower than one in the other RCT. A similar trend was observed for atogepant 30 mg in relation to AEs leading to treatment withdrawal.
Compared to placebo, all atogepant doses showed significant greater odds of constipation. The risk of nausea was estimated greater in all gepants compared to placebo, but with significant differences in terms of odds ratios only for high doses of atogepant (60 mg and 120 mg). Fatigue seems to be more important as the atogepant dose increases, starting from odds ratios under one (10 mg and 30 mg) and ending in a significant effect much larger than one for 120 mg (Table 3). Fatigue and constipation were not reported for rimegepant, so no estimations could be done.
Results from network meta-analysis comparing gepants and placebo for single adverse events. Odds ratios higher than one indicate that the odds of having an adverse event is higher in the treatment group compared to the placebo.

Measures are reported as odds ratio and 95% confidence interval.
NA = data not available.
Network meta-analysis comparing active treatments against each other
Patients treated with atogepant (all doses) had higher odds of reporting TEAEs compared to patients receiving any other active drug with the exception of one single comparison, whereof almost 40% of the comparisons were significant. Treatment with galcanezumab (120 mg and 240 mg) showed higher odds of TEAEs compared to all active drugs except atogepant with also almost 40% significant comparisons (Figure 3). Treatment with galcanezumab 120 mg or 240 mg was associated with significant higher odds of reporting SAEs compared to treatment with fremanezumab 225 mg (Figure 3). Although not significant, the risk for SAEs for patients with atogepant 30 mg, 60 mg and 120 mg is estimated smaller in comparison to all of the active drugs except for eptinezumab 100 mg and 300 mg.
Treatment ranking
Treatment ranking measures for primary and secondary outcomes are reported in Table 4. The high-ranking treatments for TEAEs with similar P-scores were placebo, eptinezumab 30 mg and 100 mg followed by eptinezumab 300 mg. Eptinezumab 100 mg and 300 mg showed also the highest ranking for SAEs. Results of secondary outcomes showed that erenumab 70 mg and 140 mg were at the top of the ranking for any AEs, while fremanezumab 225 mg achieved the highest ranking for AEs leading to treatment discontinuation.
Treatment ranking.

Ranking measures are reported as percentage frequentist P-scores.
AEs = adverse events; NA = data not available; TEAEs = treatment-emergent adverse events; SAEs = serious adverse events.
Discussion
This systematic review with network meta-analysis explored the comparative safety and tolerability of novel migraine preventive treatments targeting the CGRP pathway, including anti-CGRP monoclonal antibodies and small molecule CGRP receptor antagonists.
Among monoclonal antibodies, fremanezumab and galcanezumab showed higher odds of a TEAE occurring compared to placebo, with injection site erythema, induration and pruritus being frequently associated to these treatments, whereas these side effects were not studied for most of the treatments.
This result is in accordance with previous studies demonstrating that injection site reactions are the most common AEs reported by patients treated with anti-CGRP monoclonal antibodies (42,43). Our analysis confirmed also that constipation is frequently associated with erenumab treatment (44,45).
The network meta-analysis of AEs leading to treatment discontinuation showed a higher odds of treatment withdrawal in patients receiving eptinezumab 30 mg compared to placebo. However, this finding did not reach statistical significance with a smaller estimate in the pair-wise analysis including the only RCT investigating tolerability of eptinezumab 30, 100 and 300 mg in episodic migraine patients. This discrepancy can be explained by the fact that the network meta-analysis also included another RCT investigating eptinezumab 100 and 300 mg, which has influenced the results obtained for eptinezumab 30 mg, which was studied in one trial only.
Two previous meta-analyses (46,47) explored the efficacy and tolerability of atogepant for migraine prophylaxis compared to placebo, reporting contradictory results in relation to treatment tolerability. In agreement with Lattanzi et al. (46), here we found that orally administrated atogepant was associated with higher odds of TEAEs compared to placebo, and the most common reported events were constipation and nausea. The β-CGRP isoform and its receptor are widely expressed at the level of the enteric system, with a key involvement in regulating intestinal motility. The blockade of the CGRP receptor can affect gastrointestinal motility, thus explaining the higher risk of enteric side effects observed in association with treatments targeting the CGRP receptor, such as gepants and erenumab (48). It is interesting to note that patients treated with the highest dose of atogepant were more likely to experience fatigue. This AE could be explained by the higher blood-brain barrier penetrability of gepants (49).
In regard to SAEs, none of the active drugs showed a significant effect compared to placebo. It is noteworthy that for most of the anti-CGRP monoclonal antibodies and for all gepants, the odds of SAEs were either smaller or comparable to the odds in the placebo group, suggesting that these novel migraine prophylaxes are safe and mostly cause mild AEs. With the present data, only in patients with galcanezumab a trend of an increased risk of SAEs can be suspected.
Although nearly 40% of episodic migraine patients and almost all patients with chronic migraine would benefit from prevention, only 2-13% of migraine patients receive preventive treatments (50,51). This is largely due to the poor tolerability of classical oral migraine treatments. There is evidence showing that persistence to such therapies ranges from 7% to 55% at 12 months (6). It has also been reported that a low persistence to oral treatments is associated to greater use of healthcare resources and costs (52). The poor tolerability and the high degree of adverse events are the most frequent reasons for a poor compliance and adherence to classical oral migraine preventives (53,54). One of the main strengths of novel migraine prophylactic therapies targeting the CGRP pathway is their unequalled favorable safety and tolerability profile compared to classical oral migraine preventives. In addition, compliance with daily intake and forgetting doses is not an issue with anti-CGRP monoclonal antibodies given their monthly administration by injection.
For treatments that were studied in three or more included trials, some heterogeneity was observed between the studies. To better understand this heterogeneity and to explore if it could be present also for the other treatments, more data should be provided.
Notably, RCTs that directly compared anti-CGPR monoclonal antibodies or gepants against each other are lacking. Two previous network meta-analyses compared the efficacy, tolerability and safety of anti-CGRP monoclonal antibodies (43,55). In agreement with these studies, here we found that eptinezumab was associated with the lowest risk of TEAEs and SAEs, while galcanezumab was more likely to cause TEAEs and SAEs compared to the other monoclonal antibodies targeting the CGRP ligand (eptinezumab and fremanezumab) (43,55).
As far as we know, there are no studies comparing the safety and tolerability of anti-CGPR monoclonal antibodies and gepants. Here we observed that, compared to eptinezumab and fremanezumab, treatment with atogepant was associated with a greater risk of AEs.
One of the main strengths of our study is the extensive and sensitive literature search that guarantees the inclusion of all eligible and most up-to-date studies. In addition, two independent reviewers assessed the presence of potential bias and four reviewers were involved in the extraction of data, thus ensuring a high quality of the analysis. As a final point, this is the first network meta-analysis evaluating the comparative safety and tolerability of gepants and anti-CGRP monoclonal antibodies.
Our study also has some limitations. First, the number of RCTs associated to each active treatments were different and, for certain outcomes, some trials reported zero or low number of events. In some cases, data included in the analysis were derived from only one RCT, thus preventing the drawing of any firm conclusions. Second, the studies included in our analysis had a different double-blind period that ranges from three to six months. In addition, we cannot exclude that longer treatment period may lead to a different tolerability profile. Third, the comparison of treatments with different routes of administration might have led to some bias. Fourth, AEs associated to abnormal laboratory findings were not investigated. Finally, possible confounders could be associated to the heterogeneity of patients enrolled in the selected studies, including patients of different race, with and without aura and with a different history of prior preventives failure.
Conclusion
Overall, our network meta-analysis confirmed that monoclonal antibodies targeting the CGRP pathway and gepants are a safe and well tolerated option for migraine prevention, that rarely lead to treatment discontinuation. Albeit similar, we observed some differences regarding the type of TEAEs that were reported during the use of each of the studied drugs. Future direct comparisons and network meta-analyses including also other migraine preventives, like classical oral migraine treatments, are needed to confirm our results.
Article Highlights
Atogepant and galcanezumab showed the largest odds of treatment-emergent adverse events compared to placebo, eptinezumab and fremanezumab. Eptinezumab had greater odds of adverse events leading to treatment discontinuation. No significant differences in serious adverse events were found between active treatments and placebo. Eptinezumab was associated with the lowest odds of treatment-emergent adverse events and serious adverse events compared to placebo, whereas erenumab was associated with the lowest odds of any adverse events and quarterly fremanzumab with the lowest odds of treatment discontinuation due to adverse events. Overall, our network meta-analysis confirmed that monoclonal antibodies targeting the CGRP pathway and gepants are a safe and well tolerated option for migraine prevention, that rarely lead to treatment discontinuation.
Supplemental Material
sj-jpg-1-cep-10.1177_03331024231152169 - Supplemental material for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis
Supplemental material, sj-jpg-1-cep-10.1177_03331024231152169 for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis by Roberta Messina, Eva-Maria Huessler, Francesca Puledda, Faraidoon Haghdoost, Elena R Lebedeva and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-jpg-2-cep-10.1177_03331024231152169 - Supplemental material for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis
Supplemental material, sj-jpg-2-cep-10.1177_03331024231152169 for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis by Roberta Messina, Eva-Maria Huessler, Francesca Puledda, Faraidoon Haghdoost, Elena R Lebedeva and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-jpg-3-cep-10.1177_03331024231152169 - Supplemental material for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis
Supplemental material, sj-jpg-3-cep-10.1177_03331024231152169 for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis by Roberta Messina, Eva-Maria Huessler, Francesca Puledda, Faraidoon Haghdoost, Elena R Lebedeva and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-jpg-4-cep-10.1177_03331024231152169 - Supplemental material for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis
Supplemental material, sj-jpg-4-cep-10.1177_03331024231152169 for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis by Roberta Messina, Eva-Maria Huessler, Francesca Puledda, Faraidoon Haghdoost, Elena R Lebedeva and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-jpg-5-cep-10.1177_03331024231152169 - Supplemental material for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis
Supplemental material, sj-jpg-5-cep-10.1177_03331024231152169 for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis by Roberta Messina, Eva-Maria Huessler, Francesca Puledda, Faraidoon Haghdoost, Elena R Lebedeva and Hans-Christoph Diener in Cephalalgia
Supplemental Material
sj-pdf-6-cep-10.1177_03331024231152169 - Supplemental material for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis
Supplemental material, sj-pdf-6-cep-10.1177_03331024231152169 for Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis by Roberta Messina, Eva-Maria Huessler, Francesca Puledda, Faraidoon Haghdoost, Elena R Lebedeva and Hans-Christoph Diener in Cephalalgia
Footnotes
Author contributions
RM: design of the work, data collection, statistical analysis and drafting/revising the work. E-MH: design of the work, statistical analysis and drafting/revising the work. FP: design of the work, data collection, statistical analysis and drafting/revising the work. FH: design of the work, data collection, statistical analysis and drafting/revising the work. ERL: data collection and drafting/revising the work. HCD: drafting/revising the work.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that they have no conflict of interest related to the publication of this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
