Abstract
Background
Migraine has been associated with a dysfunctional activation of the trigeminovascular system. Calcitonin gene-related peptide, a neuropeptide released from the trigeminal nerve fibres, has an important role in the pathophysiology of migraine and is a current therapeutic target for migraine treatment.
Methods
We examined the effects of two novel calcitonin gene-related peptide receptor antagonists, ubrogepant and atogepant, on the relaxations induced by α calcitonin gene-related peptide in human isolated middle meningeal, cerebral and coronary arteries. Furthermore, the contractile responses to atogepant and ubrogepant per se were studied and compared to the responses elicited by zolmitriptan in proximal and distal human coronary arteries.
Results
In intracranial arteries, both blockers antagonized the calcitonin gene-related peptide-induced relaxations more potently when compared to the inhibition observed in distal human coronary arteries, with atogepant showing a higher potency. When analysing their antagonistic profile in HCA, ubrogepant showed a competitive antagonist profile, while atogepant showed a non-competitive one. Neither of the gepants had vasoconstrictor effect at any of the concentrations studied in human coronary arteries, whereas zolmitriptan elicited concentration-dependent contractions.
Conclusion
ubrogepant and atogepant differentially inhibit the calcitonin gene-related peptide-dependent vasodilatory responses in intracranial arteries when compared to distal human coronary arteries. Also, both gepants are devoid of vasoconstrictive properties in human coronary arteries.
Keywords
Introduction
The pathophysiology of migraine is becoming increasingly well understood, particularly regarding the role of the trigeminovascular system. A (dysfunctional) activation of the trigeminovascular system has been described (1,2), followed by the activation of the trigeminovascular reflex characterized by the release of calcitonin gene-related peptide (CGRP) from the perivascular trigeminal sensory fibres and vasodilation of cerebral and dural vessels (3,4). Indeed, the vasodilation of cranial arteries in the pathophysiology of migraine is still controversial, as in patients with spontaneous migraine attacks no extracranial arterial dilatation has been observed (5); however, dilatation of dural branches of the middle meningeal artery cannot be categorically excluded, as those small branches have not been analysed due to technical limitations (6). Our group has previously demonstrated the expression and co-localization of the CGRP receptor components in the smooth muscle cells of intracranial arteries (7). Moreover, patients during migraine attacks show elevated release of CGRP in the cranial venous outflow (8), while intravenous infusion of CGRP in migraine patients has been shown to provoke migraine-like attacks (9).
Due to the important role of CGRP in migraine, CGRP receptor antagonists (gepants) were developed for the acute treatment of migraine. Olcegepant and telcagepant were the first gepants and both were shown to be effective in clinical trials (10,11); unfortunately, due to pharmacokinetic and/or hepatotoxic limitations, neither of these gepants reached the market (12) and the synthesis of novel gepants was temporarily halted. In recent years, two novel, orally available, structurally different gepants have been developed for the acute (ubrogepant) and preventive (atogepant) treatment of migraine (Figure 1). Phase II trials with ubrogepant (NCT01613248 and NCT01657370) showed promising results for the acute treatment of migraine (13–16) and phase III trials (NCT02828020, NCT02867709 and NCT02873221) have been completed with positive results. A phase II/III trial (NCT02848326) with atogepant for the preventive treatment of episodic migraine also demonstrated positive findings (14).

As mentioned above, CGRP plays an important role in the pathophysiology of migraine (17); however, this peptide has also been related to several physiological processes (18–20), and expression of CGRP receptors has not only been described in middle meningeal arteries, but also in cerebral and coronary arteries. This raises the possibility that CGRP receptor antagonists could affect vascular tone in different parts of the circulation (21,22). Even though previous studies have shown that telcagepant was devoid of contractile responses in vitro (7,23), and that olcegepant did not affect cerebral and systemic hemodynamics in vivo (24,25), ubrogepant and atogepant are structurally different to the previous gepants; thus, it is important to study the vascular effects of these novel gepants. Therefore, the aim of this study is to assess the functional responses to human αCGRP in isolated human middle meningeal (HMMA), cerebral (HCxA) and coronary arteries (HCA), and to study the effects of ubrogepant and atogepant on the CGRP-mediated vasodilatory responses. Furthermore, we studied the per se effect of both gepants in HCA and compared them to the contractile responses to zolmitriptan.
Materials and methods
Human isolated arteries
Human middle meningeal arteries (HMMA; one male, five females; aged 45 ± 3 years, range 35–53) and cerebral (cortex) arteries (HCxA; three males, four females; aged 60 ± 4 years, range 43–73) were removed peri-operatively from patients undergoing neurosurgical procedures at Lund University Hospital, Sweden. All vessels were placed in buffer solution, for cerebral arteries, composition (in mM): NaCl, 119; KCl, 4.7; CaCl2, 1.5; MgSO4, 1.17; KH2PO4, 1.18; NaHCO3, 25; glucose, 5.5; at a pH = 7.4; for meningeal arteries, composition (in mM): NaCl, 119; KCl, 4.7; CaCl2, 1.25; MgSO4, 1.2; KH2PO4,1.2; NaHCO3, 25 and glucose, 11.1; at a pH = 7.4, and were kept aerated with carbogen and transported to the laboratory immediately. Human coronary arteries were obtained from eight “heart beating” organ donors (four males, four females; aged 41 ± 3 years, range 25–56). The hearts were provided post-mortem by the Heart Valve Bank, Rotterdam (currently ETB-BISLIFE Tissue Bank, Beverwijk), after donor mediation via Bio Implant Services/Eurotransplant Foundation (Leiden, The Netherlands), following removal of the aortic and pulmonary valves for homograft valve transplantation and were stored at 0–4℃ in a sterile organ-protecting solution immediately after circulatory arrest. After arrival at the laboratory, proximal (internal diameter 3–5 mm) and distal (internal diameter 0.5–1 mm) portions of the right HCA were dissected and placed in a cold, oxygenated buffer solution composed as follows (in mM): NaCl, 118; KCl, 4.7; CaCl2, 2.5; MgSO4, 1.2; KH2PO4, 1.2; NaHCO3, 25 and glucose, 8.3, at a pH = 7.4 and were kept aerated with 5% CO2 in O2 (carbogen). It is worth noting that the buffer solution differed among arteries in order to mimic the intracellular and extra cellular osmotic and ionic balance of each artery, as well as our own experimental experience.
The Swedish part of the study was approved by Lund University Ethics Committee (LU99) and had the individual patients’ approval, while the ethics committee dealing with human experimentations at Erasmus Medical Centre, Rotterdam, approved the Dutch part of the study.
Functional experiments
Human arteries were cut into segments of 2 to 4 mm length, excluding distinct, macroscopically visible atherosclerotic lesions. The proximal HCA segments were mounted in 15-ml organ baths, while distal HCA segments and intracranial arteries were mounted on Mulvany myographs (Danish Myo Technology, Aarhus, Denmark) between two small stainless-steel wires (Ø 40 µm) in 6-ml tissue baths. In both cases, the baths were filled with the respective oxygenated buffer solution at 37℃ (see above). After equilibration for at least 30 min and a wash every 15 min, the vessel segments were stretched to a stable tension of about 15 mN for the proximal HCA, the optimal tension as determined in previous studies (26), or stretched to a tension normalized to 90% of l100 for the distal HCA segments and intracranial arteries, the diameter when transmural pressure equals 100 mm Hg (27). Changes in tissue tension were measured using an isometric transducer (Harvard, South Nattick, MA) and recorded on a flatbed recorder (Servogor 124; Goerz, Neudorf, Austria, 15-ml organ baths) or using a LabChart data acquisition system (AD Instruments Ltd, Oxford, UK, 6-ml Mulvany myographs).
After reaching equilibrium, the contractile capacity of HCxA segments was examined by exposure to a potassium-rich (60 mM) buffer solution, which had the same composition as the standard solution, except that the NaCl was exchanged for an equimolar concentration of KCl. Segments of HMMA and distal HCA were exposed to KCl (30 mM) to “prime” the tissue for stable contractions. After washout, the tissue was exposed to KCl (100 mM) to determine the maximal contractile response to KCl.
The relaxant effect of human αCGRP was examined by cumulative application of increasing concentrations of the peptide (1 pM – 1 µM, whole log steps for cranial arteries and half log steps for the coronary artery) in the absence or presence of ubrogepant (0.1 nM, 1 nM, 10 nM, 100 nM or 1 μM) or atogepant (0.01 nM, 0.1 nM, 1 nM, 10 nM, 100 nM or 1 μM). Segments were precontracted with KCl (30 mM) before αCGRP was added. Each segment was exposed to a single cumulative concentration response curve and a matched pair’s protocol was used, where one segment acted as control (no antagonist present) while in another segment from the same artery the agonist response was assessed following equilibration (20–30 min) with the antagonist. When a concentration of antagonist inhibited more than 80% of the CGRP-mediated vasodilatory responses, no further concentrations of antagonist were tested as estimation of pEC50 values was not reliable. Furthermore, in HCA the per se contractile responses to atogepant (1 pM – 100 μM in distal, 100 pM – 30 μM in proximal), ubrogepant (1 pM – 100 μM in distal, 100 pM – 30 μM in proximal) and vehicle were assessed and compared to the contractile responses elicited by zolmitriptan (1 pM – 100 μM in distal, 100 pM – 30 μM in proximal). The functional integrity of the endothelium was verified by observing relaxation to substance P (10 nM), after precontraction with thromboxane A2 analogue U46619 (10 nM).
Compounds
The following materials were used in the in vitro experiments: human αCGRP (PolyPeptide, Strasbourg, France, for the Rotterdam experiments; Sigma-Aldrich, St. Louis, MO, USA, for the Lund experiments), U46619 and zolmitriptan (Sigma-Aldrich, St. Louis, MO, USA) were dissolved in saline. Ubrogepant (((6S)-N-[(3S,5S,6R)-6-Methyl-2-oxo-5-phenyl-1-(2,2,2-trifluoroethyl)-3-piperidinyl]-2'-oxo-1', 2',5,7-tetrahydrospiro [cyclopenta[b]pyridine-6,3'-pyrrolo[2,3-b]pyridine]-3-carboxamide) and atogepant (((6S)-N-[(3S,5S,6R)-6-Methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)-3-piperidinyl]-2'-oxo-1',2',5,7-tetrahydrospiro [cyclopenta[b]pyridine-6,3'-pyrrolo[2,3-b]pyridine]-3-carboxamide) were received from the Medicinal Chemistry Department, Merck Laboratories, USA, in pre-weighed vials that had been marked by Merck and dissolved in dimethylsulphoxide (DMSO). All compounds were stored in aliquots at −20℃. When the compounds were to be used, they were diluted in saline.
Analysis of data
The individual vasodilator responses were expressed relative to the contraction elicited by KCl (30 mM; 100% precontraction). For each segment, the maximum vasodilator effect (apparent Emax, here defined as the response observed at the highest concentration of CGRP applied) was calculated. The concentration response curves for all agonists were analysed by using nonlinear regression analysis, and the potency of agonists was expressed as pEC50 (i.e. the negative logarithm of the molar concentration of agonist inducing half-maximum response) by using Prism 5.0 (GraphPad Software Inc., San Diego, CA). The blocking potency of the antagonists was estimated by calculating EC50 ratios and plotting a Schild plot by using linear regression to get the slope value (28). In case of depression of the maximum relaxation without a change in pEC50, we could only show significant reduction in maximum relaxations, while it should in these cases of course be born in mind that Emax might have been increased at higher concentrations than applied in our experiments. Data are expressed as mean values ± standard error of the mean (SEM), and n refers to the number of subjects from whom the vessels were collected. Statistically significant differences were examined by Mann-Whitney U test.
Results
Functional responses to αCGRP in human isolated arteries
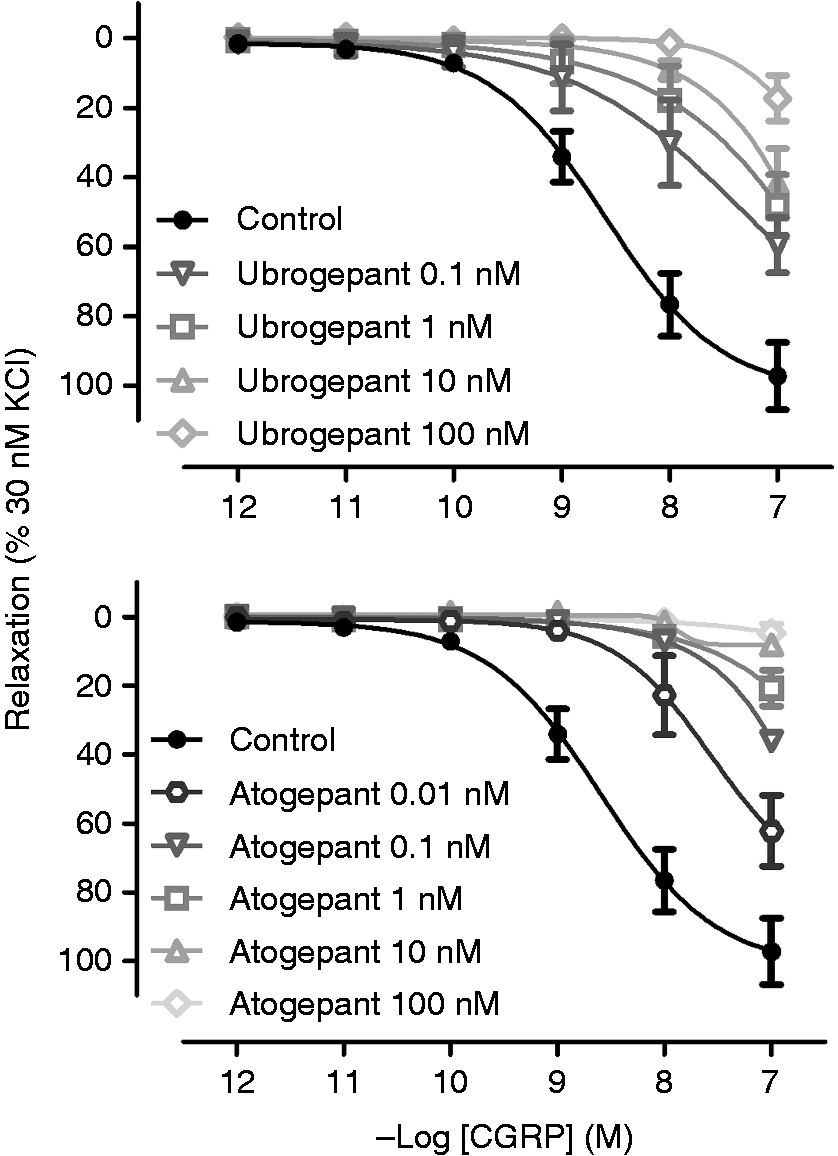
In HMMA (Figure 2), increasing concentrations of αCGRP caused a concentration-dependent vasorelaxation (see Table 1). Similar responses were observed in HCxA (Figure 3). Endothelial function analysis resulted in a mean relaxation to substance P of 57 ± 10% of the precontraction to U46619 in HMMA and 75 ± 5% in HCxA (n = 5 and n = 7, respectively).
Vasodilatory effect of αCGRP on Vasodilatory effect of αCGRP on Vasodilatory responses to αCGRP in human cranial arteries in the absence or presence of atogepanto or ubrogepant. Values given represent mean ± standard error of the mean (n = 3–7 each). Emax: maximal response; ND: not determined; pEC50: negative log of the molar concentration of an agonist needed to reach half of its maximal effect.

In distal HCA segments, concentration response curves to human αCGRP resulted in a concentration-dependent relaxation (Figure 4, Table 2). Conversely, in proximal coronary segments, a small, often absent relaxant response to αCGRP was observed (Emax: 9 ± 8, Figure 5). Quantification of the pEC50 of these concentration-response curves was not feasible in a sensible manner. Endothelial function analysis resulted in a mean relaxation to substance P of 69 ± 3% of the precontraction induced by U466619 in distal HCA and 53 ± 4% in proximal HCA.
Vasodilatory effect of αCGRP on Vasodilatory effect of αCGRP on Vasodilatory responses to αCGRP in human distal coronary arteries in the absence or presence of atogepanto or ubrogepant. Values given represent mean ± standard error of the mean (n = 7–8 each). Emax: maximal response; pEC50: negative log of the molar concentration of an agonist needed to reach half of its maximal effect.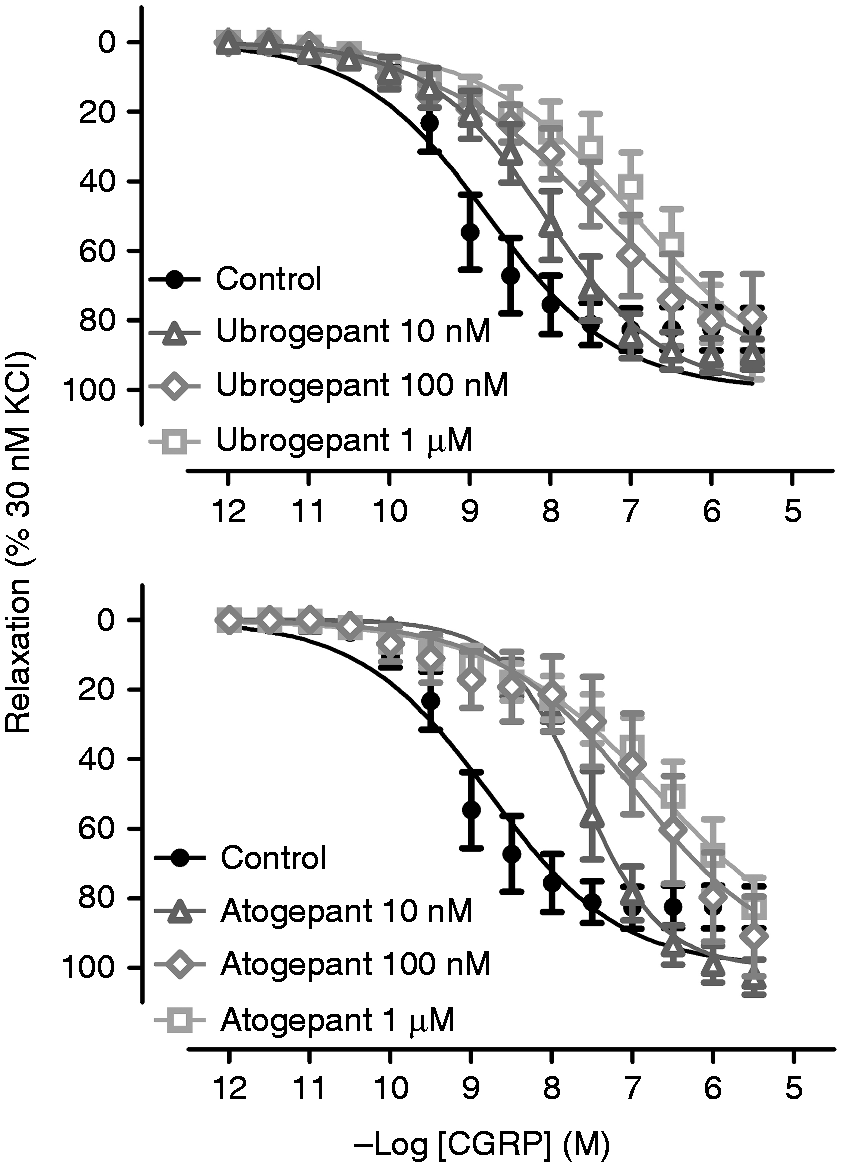


Effects of ubrogepant and atogepant in human isolated arteries
In HMMA (Figure 2), a shift of the concentration-response curve to CGRP was observed at all concentrations tested for ubrogepant and atogepant (Table 1). In HCxA (Figure 3), inhibition of the relaxant responses to CGRP was observed in the presence of ubrogepant and atogepant as seen in Table 1. No estimation of pKb and pA2 values was performed for HMMA and HCxA as none of the concentration-response curves reached a plateau; hence, no accurate values could be calculated.
In distal HCA (Figure 4), a significant parallel rightward shift of the concentration-response curve to CGRP was observed with ubrogepant (10 nM, p = 0.01; 100 nM, p = 0.002; 1 μM pEC50, p = 0.003) and atogepant (10 nM, p = 0.003; 100 nM, p = 0.001; 1 μM, p < 0.001). No significant changes were observed in Emax in the presence of ubrogepant (10 nM, p = 0.22; 100 nM, p = 0.36; 1 μM, p = 0.32) or atogepant (10 nM, p = 0.11; 100 nM, p = 0.35; 1 μM, p = 0.50); therefore, we made an evaluation with the Schild plot (Figure 6). For ubrogepant, the slope of the Schild plot was not significantly different from unity (0.87 ± 0.17), thus we calculated pA2, which amounted to 8.86 ± 0.39 (R2: 0.87). For atogepant, the slope of the Schild plot was significantly lower than unity (0.55 ± 0.01), therefore we did not calculate a pA2 value. Instead, pKb values were calculated per individual concentration of antagonist employed (pKb at 10 nM: 9.42 ± 0.22; pKb at 100 nM: 9.11 ± 0.34; pKb at 1 µM: 8.64 ± 0.21). In the proximal HCA, ubrogepant and atogepant seemed to inhibit the relaxant responses to CGRP (Figure 5). However, as the control responses were rather small, or even absent in some cases, it was not quantifiable in a sensible manner.
Schild plot of the responses observed with atogepant and ubrogepant in distal coronary arteries. A straight line that did not differ from unity was observed for ubrogepant, but not for atogepant.
Vasoconstrictor effect of zolmitriptan, ubrogepant and atogepant in human isolated coronary arteries
Zolmitriptan caused concentration-dependent contractions in distal (pEC50 7.10 ± 0.13; Emax: 72 ± 33%; n = 6; Figure 7) and proximal HCA (pEC50 5.53 ± 0.32; Emax: 38 ± 12%; n = 6; Figure 8). Ubrogepant and atogepant were devoid of significant contractions in concentrations up to 100 µM in both proximal and distal segments of the HCA.
Contractile responses to zolmitriptan (1 pM – 100 μM), ubrogepant (1 pM – 100 μM) and atogepant (1 pM – 100 μM) in Contractile responses to zolmitriptan (100 pM – 30 μM), ubrogepant (100 pM – 30 μM) and atogepant (100 pM – 30 μM) in 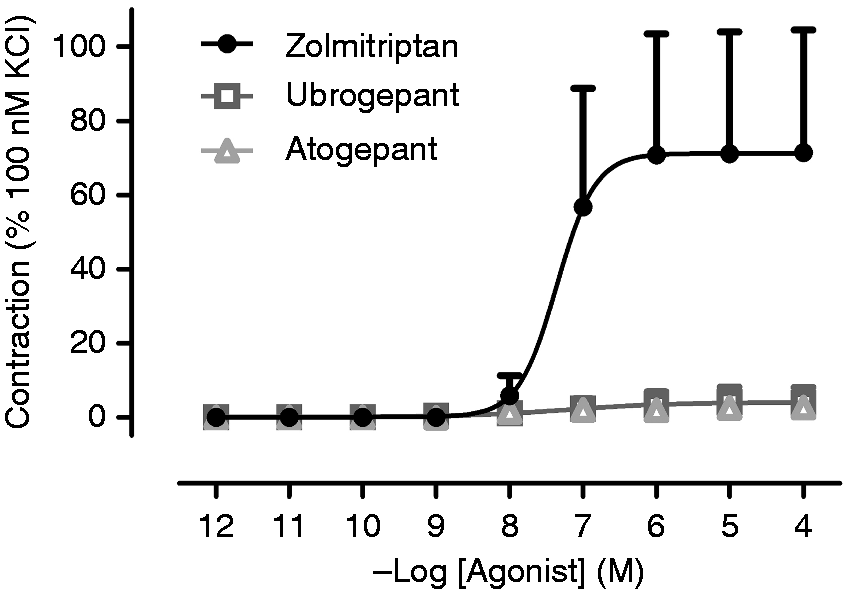

Discussion
In this study, the effect of two novel gepants, ubrogepant and atogepant, on the CGRP-mediated vasodilatory responses in human isolated arteries was investigated.
First, we analysed the inhibition of the vasorelaxant responses to CGRP in HMMA, as this is potentially one of the mechanisms of action of gepants. Our results show that both gepants effectively antagonized the vasodilatory responses to CGRP, with atogepant seeming more potent (Figure 2). In the case of ubrogepant, developed for the acute treatment of migraine, our data suggest that during a migraine attack, reversion of the CGRP-mediated vasodilation may well be one of its mechanisms of action whereas in the case of atogepant, currently being evaluated for the prophylactic treatment of migraine, our results suggest a prevention of the vasodilatory responses. In the latter case, it would be interesting to further assess in vivo, similarly to the CGRP (receptor) antibodies that are currently in development (29), whether the chronic blockade of the CGRP receptor would result in overexpression of CGRP receptors and tolerance over time. Furthermore, if there is indeed an increase in CGRP receptor expression, physicians should be aware that withdrawal of this drug must be gradual, as migraine headaches could worsen if withdrawn abruptly (30).
CGRP has also been related to several physiological processes (18,19), such as regulation of the vascular tone (31,32), raising the concern that CGRP receptor antagonism could affect vascular tone in different parts of the circulation (21). Considering migraine patients have increased cardiovascular risk (33–35), it is important to assess the effect of ubrogepant and atogepant in cerebral (cortex) and coronary arteries. In HCxA, atogepant was more potent than ubrogepant in antagonizing CGRP-mediated vasodilatory responses (Figure 3) Even though the previous gepants were reported to have a limited ability to cross the blood-brain barrier (36), it is important to consider that these novel gepants are not structurally related (see Figure 1), therefore their ability to cross the blood-brain barrier cannot be discarded. In coronary arteries, vasodilatory responses to CGRP in the proximal HCA were rather small, while in the distal portion of the HCA, these were more pronounced. This is in accordance with previous publications from our group (23,37) and shows that CGRP plays a more important role in the vasodilation of the distal portion of the coronary arteries, rather than in the proximal portion. It also reinforces the importance of cardiovascular safety studies when blocking the CGRP receptor (21,38), especially considering that migraine patients have a higher cardiovascular risk, with the grand majority of patients being female and that myocardial ischemic events in women are more common in the distal portion of the coronary artery, where CGRP plays a bigger role. Interestingly, when analysing the effect of both gepants in the vasodilatory responses to CGRP in distal HCA, although atogepant was more potent when compared to ubrogepant, both gepants were less potent in antagonizing the vasodilatory responses in HCA than in HMMA. Though we cannot explain the different potencies of atogepant and ubrogepant in cranial versus coronary arteries, we have previously reported similar results with olcegepant (37,39) and, as will be discussed further, this might be related to CGRP receptor heterogeneity. This property may be of clinical relevance and an advantage for these antagonists, since they would be more potent at blocking CGRP-induced vasodilation in the intracranial arteries than in the coronary arteries, where antagonism of CGRP-induced vasodilation would not be desired. Additionally, in HCA, we analysed the per se vascular effects of both gepants, as current acutely acting antimigraine drugs are contraindicated in patients with coronary artery disease due to their vasocontrictive properties (40). Ubrogepant and atogepant did not contract proximal or distal coronary arteries (Figures 7–8), which represents an advantage for patients to whom triptans are contraindicated.
Finally, we analysed the type of antagonism of both gepants in HCA with a Schild plot (Figure 6). In the case of ubrogepant, the slope was not significantly different from unity, suggesting a competitive type of antagonism. In the case of atogepant, the slope was significantly smaller than unity, thus suggesting a non-competitive type of antagonism, the involvement of multiple receptors or hemi-equilibrium conditions (37,41–43). Certainly, we have previously suggested CGRP receptor heterogeneity in the human coronary artery (37), and that this may be due to the contribution of the amylin type 1 (AMY1) receptor in mediating the relaxations to CGRP in the distal human coronary artery (41), as described in the trigeminal ganglion (42). Additionally, to our knowledge, no studies have assessed the selectivity of ubrogepant and atogepant over the AMY1 (CTR/RAMP1) receptors. However, these results must be taken with caution, as our observations with the Schild plot should be further confirmed with more rigorous experiments, which fall beyond the scope of the current study. Similarly, the mechanism behind the different potency of both antagonists in the cranial arteries when compared to the responses in coronary arteries should be addressed in future studies, as it may shed light on possible targets devoid of the potential for coronary side-effects.
Conclusion
Taken together, these results show that both atogepant and ubrogepant effectively inhibit the CGRP-vasodilatory responses in human meningeal, cerebral, and coronary arteries, and are both devoid of vasoconstrictive properties in HCA.
Key findings
Ubrogepant and atogepant inhibit the vasodilatory responses to CGRP in HCxA, HMMA and HCA. The inhibition of the CGRP-mediated vasodilatory responses is more potent in the cranial arteries than in the coronary arteries. Ubrogepant and atogepant are devoid of vasoconstrictive properties in human coronary arteries.
Footnotes
Acknowledgements
We would like to thank Professor Dr. Ad JJC Bogers, Professor Dr. Clemens Dirven and Dr. Antoon van den Bogaerdt for their invaluable help in obtaining the human tissue.
Availability of data
The dataset supporting the conclusion of this article is available upon reasonable request to the corresponding author.
Authorship contributions
Participated in research design: KYC, AHJD, AMVDB, LE.
Conducted experiments: KYC, LE.
Performed data analysis: ERB, KYC, AMVDB, LE.
Wrote or contributed to the writing of the manuscript: ERB, KYC, AHJD, AMVDB, LE.
Declaration of conflict of interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ERB received travel support from Allergan. AMvdB received research grants and/or consultation fees from Amgen/Novartis, Lilly/CoLucid, Teva and ATI. LE has given talks and received grants for preclinical studies from Amgen, Novartis and Teva. This work was supported by Allergan plc, Dublin, Ireland.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AMVDB was supported by the Netherlands Organization for Scientific Research (Vidi grant 917.113.349). ERB was supported by Consejo Nacional de Ciencia y Tecnología (CONACyT; fellowship No. 409865; Mexico City). This study was financially supported by a research grant from Allergan plc, Dublin, Ireland.
