Abstract
Background:
Rimegepant, a small molecule oral calcitonin gene-related peptide (CGRP) receptor antagonist, is approved for the acute and preventive treatment of migraine. We hypothesized that intermittent CGRP receptor blockade with rimegepant 75 mg acute treatment as needed (PRN) might result in reductions in monthly migraine days (MMD) over time, and was evaluated as the study objective.
Methods:
This was a post-hoc analysis of adults with ≥6 MMD at baseline who self-administered rimegepant 75 mg orally PRN for acute treatment of migraine up to 52-weeks in an open-label safety study (BHV3000-201; NCT03266588). Outcome measures (defined as median time to) and response rates (defined as proportion of patients reporting) were captured for ≥30% and ≥50% reduction of baseline MMD.
Results:
1044 participants with ≥6 MMD at baseline were analyzed. Median time to ≥30% reduction in MMD was 12 weeks (IQR; 4–40 weeks); median time to ≥50% reduction was 32 weeks (IQR; 12-NR weeks). Reduction in MMD was observed over time regardless of baseline migraine frequency, however higher baseline MMD were associated with a longer time to achieving ≥30% or ≥50% MMD reduction.
Conclusion:
In participants presenting with ≥6 MMD, PRN acute treatment of migraine attacks over 52-weeks with oral rimegepant 75 mg was observed to confer reductions in migraine frequency.
Trial registration:
NCT03266588
Introduction
In recent years, calcitonin gene-related peptide (CGRP) targeting drugs have rapidly expanded the treatment landscape in migraine, providing important differentiation from standard of care therapies. CGRP is a potent neuropeptide involved in nociceptive and neuroinflammatory signaling associated with migraine. 1 Experimental sensitivity to the neuropeptide has been demonstrated by migraine-like attacks being induced by exogenous CGRP in patients with a range of migraine frequencies. 2,3 A hallmark feature of people with migraine is indeed a profile of hypersensitivity, which drives the avoidance of common triggers such as light, barometric changes, and smell. 4,5 CGRP is thought to be a key mediator of sensitization as well as an initiator of neurogenic inflammation, and might play a role in 2.5% or more of episodic migraine patients progressing to chronic migraine. 6
Rimegepant (BHV3000) is an oral, small molecule (gepant) that blocks CGRP signaling by binding to the CGRP receptor, with efficacy established in both the acute and preventive treatment of migraine in phase 3 trials. 7 –10 Historically, there have been two overarching modes of migraine pharmacotherapy, acute and prevention, however the FDA approval of rimegepant for both indications in the United States challenges this paradigm. 11 –13 With the approval of the single drug rimegepant in the United States for both acute and preventive therapy at the same dose, patients may take one medication for immediate migraine relief and also use the same agent for prevention. For the acute treatment of migraine, patients use rimegepant as needed (PRN) to relieve the disabling pain and associated symptoms of migraine, and to restore normal function. 7,8,11,12 With preventative treatment, rimegepant is taken every-other-day (EOD) to reduce attack frequency and severity, to decrease the need for acute medications, to prevent migraine progression, and to reduce overall migraine related disability. 10,14,15
Rimegepant’s blockade of the key neuromodulator (CGRP) in the pathogenesis of migraine, and its established efficacy as both an acute (PRN) treatment and as a preventive (EOD) treatment, raises the possibility that PRN use may also have some preventive benefits (by reducing a participant’s sensitivity and reactivity to typical migraine triggers). These potential benefits cannot be studied in a typical acute treatment trial because, by design, relatively few attacks are treated, nor can this hypothesis be tested in a prevention trial, as in that setting treatment is taken in the absence of an attack. However, long-term safety studies where participants use the drug as needed for the acute treatment of migraine, over extended periods of time, offer an opportunity to explore this hypothesis.
To this aim, a post-hoc analysis of adults with ≥6 MMD at baseline who self-administered rimegepant 75 mg orally PRN for acute treatment of migraine up to 52-weeks in an open-label safety study (BHV3000-201; NCT03266588) was conducted. Time to ≥30% and ≥50% reduction from baseline in MMD were selected as clinically meaningful endpoints to explore the preventative effect of PRN rimegepant dosing over time. The proportion of patients achieving 30% and 50% reduction in MMD from baseline are endpoints currently recommended for use in preventive trials. 16,17 While most preventive studies report the proportion of participants in each treatment arm achieving this endpoint over a pre-specified time interval (e.g. at 12-weeks), there is no precedent for assessing the benefits of repeated PRN acute treatment on prevention. In prior work on this same cohort, rimegepant 75 mg has already been shown to be associated with reduced MMD and improvement in MSQv2 domains over time, leading to improvement in EQ-5D-3L utilities. 18 Accordingly, we elected to evaluate the time to at least a 30% and 50% reduction in MMD using Kaplan-Meyer (KM) curves over the course of 52-weeks. We also assessed the influence of baseline monthly migraine frequency on time to ≥30% or ≥50% MMD reduction.
Methods
Study BHV3000-201 was a phase 2/3 multi-center, open-label, long-term safety study of rimegepant 75 mg for the acute treatment of migraine (Figure A1). A detailed description of Study BHV3000-201 is provided in Appendix 1. Key inclusion criteria were adult patients with at least a 1-year history of migraine of moderate or severe pain intensity Table A1. Key exclusion criteria included a history of hemiplegic or basilar migraine Table A1. BHV3000-201 was conducted in accordance with Good Clinical Practice guidelines as defined by the International Conference on Harmonisation and in accordance with all applicable local regulations. The protocol was approved by centralized and local Institutional Review Boards, and participants provided written informed consent before they were screened for the study.
This post-hoc analysis included patients enrolled in BHV3000-201 who exhibited ≥6 MMD during pre-treatment baseline assessment, and who self-treated as needed (PRN) during the study period. In clinical practice, these patients would be considered for preventive treatment. For the purpose of BHV3000-201, PRN dosing was specified as participants taking rimegepant 75 mg up to once daily for a migraine of any intensity (i.e. mild, moderate, or severe) for up to 52-weeks. Participants in the enrollment group from BHV3000-201 who were treated with rimegepant EOD+PRN were not included in this analysis
The primary outcome measures for the current post-hoc study were median time to ≥30% and median time to ≥50% reduction in MMD over the BHV3000-201 study period. These outcomes were based on the accepted criteria for definition of a preventive benefit (≥50% reduction in MMD from baseline), 14,16,19 and/or a threshold deemed clinically significant in certain migraine populations (≥30% reduction in MMD). 16,17 These endpoints were assessed at 4-week intervals from baseline up to 52-weeks. The MMD analyses included participants who self-administered at least one dose in their eDiary within any 4-week time period. A secondary outcome measure involved examining the ratio of rimegepant doses taken per MMD throughout the study duration.
The analysis focused on time to ≥30% and ≥50% reduction in MMD from baseline, overall and within three groups defined via cluster analysis as distinct groups based on MMD over the study period. MMD trajectories were expected to be different for patients with varying MMD severity at baseline. These cluster analyses permitted an assessment of response according to varying levels of MMD trajectory that might indicate different results. This method involved the creation of clusters of participants with similar characteristics within a specific cluster, but with differing characteristics among clusters. Cluster classification was performed using the expectation maximization algorithm, which, given a set number of clusters, aims to compute a cluster membership probability for each participant. 20 The aim of the algorithm is then to maximize the summed membership probabilities across all participants, given the final clusters. 20 Differing numbers of clusters were explored, and three were settled upon in order to preserve larger sample sizes. In the expectation maximization algorithm, cluster models evaluated MMD as the response variable and time was used as a predictor, with participant ID included as a random-effect to account for repeated measurements.
Cox proportional hazard models were fit on the three cluster groupings evaluating the time to each patient achieving either a minimum of 50% or 30% reduction from their baseline MMD. Age and sex were included as additional covariates. Summary statistics and graphs were formulated from the data to gain an understanding of changes in treatment patterns. The proportional hazards assumption was confirmed via the method described in Kalbfleisch and Prentice as well as a global test evaluating if the slope of the time-dependent coefficients was zero. 21 All analyses were performed in R (v3.6.1) using the flexmix (v2.3-15) package for cluster analyses and the survival (v2.44-1.1) package for Cox models.
Results
1044 participants from BHV3000-201, with ≥6 MMD from the PRN enrolment groups were included in this analysis. The mean age was 43.2 years and 91.1% of the participants were female. The mean number of historical monthly migraine days was 10.9 and 40.6% of participants had a history of migraine with aura. 163 (15.6%) of participants used a concomitant prophylactic migraine medication of which the most commonly used medications among all patients were: topiramate (3.1%), botulinum toxin type A (1.4%), and amitriptyline (1.0%). All these participants were on stable prophylactic migraine medication prior to study entry except for a small subset who were prescribed preventive agents during the course of BHV3000-201. 22,23 Detailed baseline demographics for this cohort are described in Table 2. Of the 1044 participants with 6 or more MMD at baseline, 635 (61%) completed the study while 409 (39%) discontinued. Participants who discontinued before achieving an outcome were censored at the time of discontinuation. There were negligible missing MMD data from participants who did not discontinue, therefore no adjustments were made in this regard.
Cox models for PRN ≥6 MMD analysesa—≥30% and ≥50% reduction in MMD from baseline.

CI = confidence interval; MMD = monthly migraine days; PRN = as needed; vs. = versus; three clusters: C1 (mean = 14.8 MMD); C2 (mean = 11.5 MMD); C3 (mean = 8.7 MMD).
a Participants had ≥6 MMD at baseline—consisting of participants from the combined PRN 2–8 and PRN 9–14 groups, and took rimegepant as needed.
Note: Neither Cox regression was in violation of the proportional hazards assumption based on the global chi-square test (both p > 0.05).
Study BHV3000-201—baseline demographics for patients with ≥6 MMD at baseline treated with PRN rimegepant.
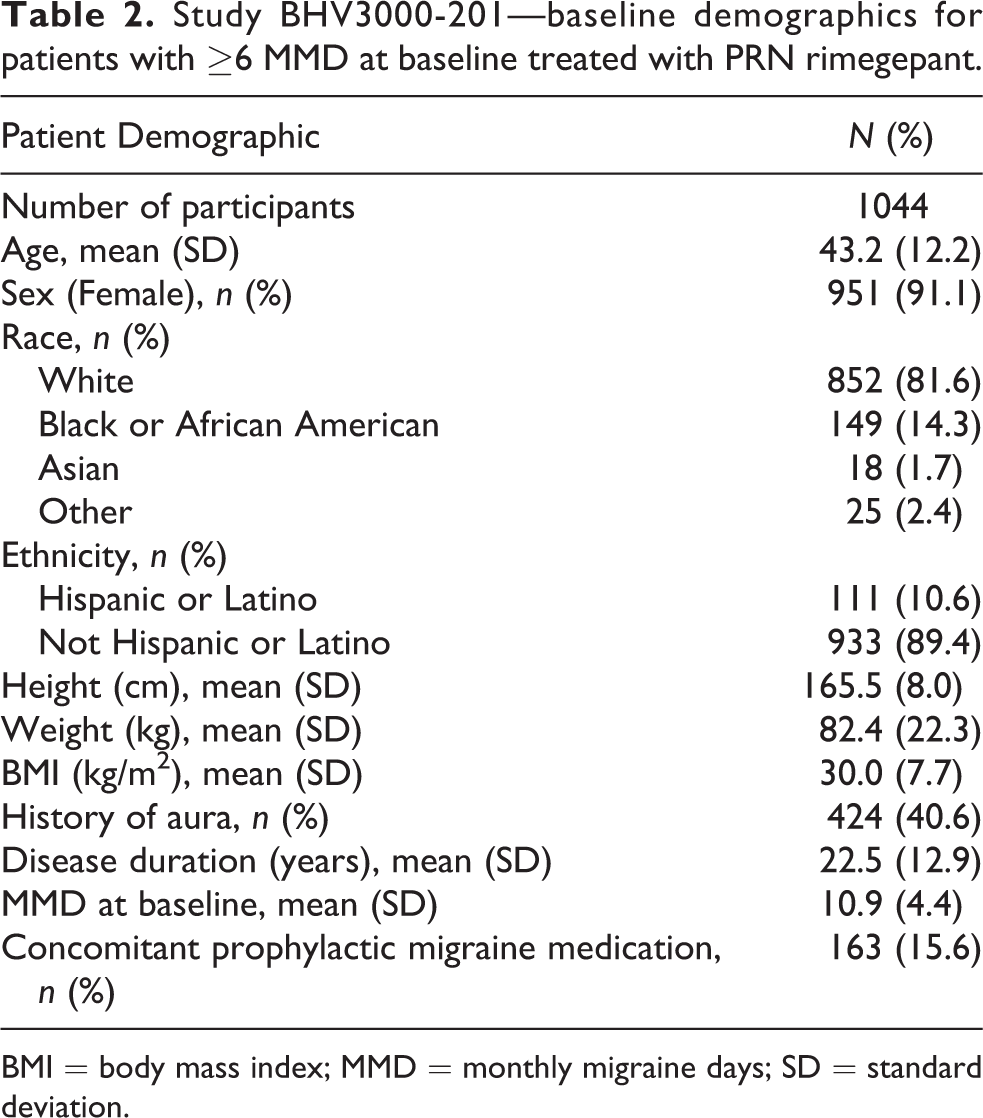
BMI = body mass index; MMD = monthly migraine days; SD = standard deviation.
Cox models for the probability of achieving at least a 30% or 50% MMD reduction from baseline are presented in Table 1. Considering all participants (all migraine frequency clusters combined), the median time to ≥30% reduction of baseline MMD was 12 weeks (IQR; 4-40 weeks), while median time to ≥50% MMD reduction was 32 weeks (IQR; 12-NR weeks; Figure 1). At 52-weeks, 78.6% of participants achieved a ≥30% reduction of baseline MMD and 63.3% of participants achieved a ≥50% MMD reduction.

Kaplan–Meier plots of time to ≥30% or ≥50% reduction in MMD from baseline in the ≥6 MMD group (N = 1,044)* treated as needed (PRN) with rimegepant 75mg. MMD = monthly migraine days; PRN = as needed *The percentage of participants with at least a 30% or 50% decrease in MMD from baseline. Participants had ≥6 MMD at baseline—consisting of participants from the combined PRN 2–8 and PRN 9–14 groups, and took rimegepant as needed; no clustering Note: Patients can be removed from the number at risk by either achieving the outcome (having an event) or discontinuing from the study (being censored).
MMD reduction trends were non-linear across the migraine frequency clusters over 52-weeks. In general, looking at the trends across the three clusters over time, there was an initial more rapid decline in MMD, followed by a smaller more incremental reduction of the MMD rate for the remainder of the follow-up period (Figure 2).

Cluster plots of MMD throughout the study duration by cluster for PRN ≥ 6 MMD analyses.
Figure 3(a) and (b) display KM curves for time to ≥30% and ≥50% MMD reductions respectively, stratified by MMD baseline frequency clusters defined via the cluster analysis. Cluster 1 (C1) was defined as the cluster of patients with the highest MMD over time (mean baseline MMD 14.8), followed by cluster 2 (C2) with more moderate MMD over time (mean baseline MMD 11.5), and cluster 3 (C3) with the lowest number of MMD over time (mean baseline MMD 8.7). Higher baseline monthly migraine frequency was found to be associated with a reduced rate of achieving ≥30% or ≥50% MMD reduction. In Cox proportional hazards analysis, adjusting for age and sex, the hazard ratios and 95% CIs (reflecting increased rate of achieving reduction) for C3 versus C1 were 5.47 (4.27, 7.00) for a ≥30% reduction and 5.74 (4.22, 7.81) for a ≥50% reduction. For C2 versus C1 the hazard ratios and 95% CIs were 2.13 (1.64, 2.76) and 1.91 (1.37, 2.65), respectively, reflecting a less pronounced difference from the C1 group.
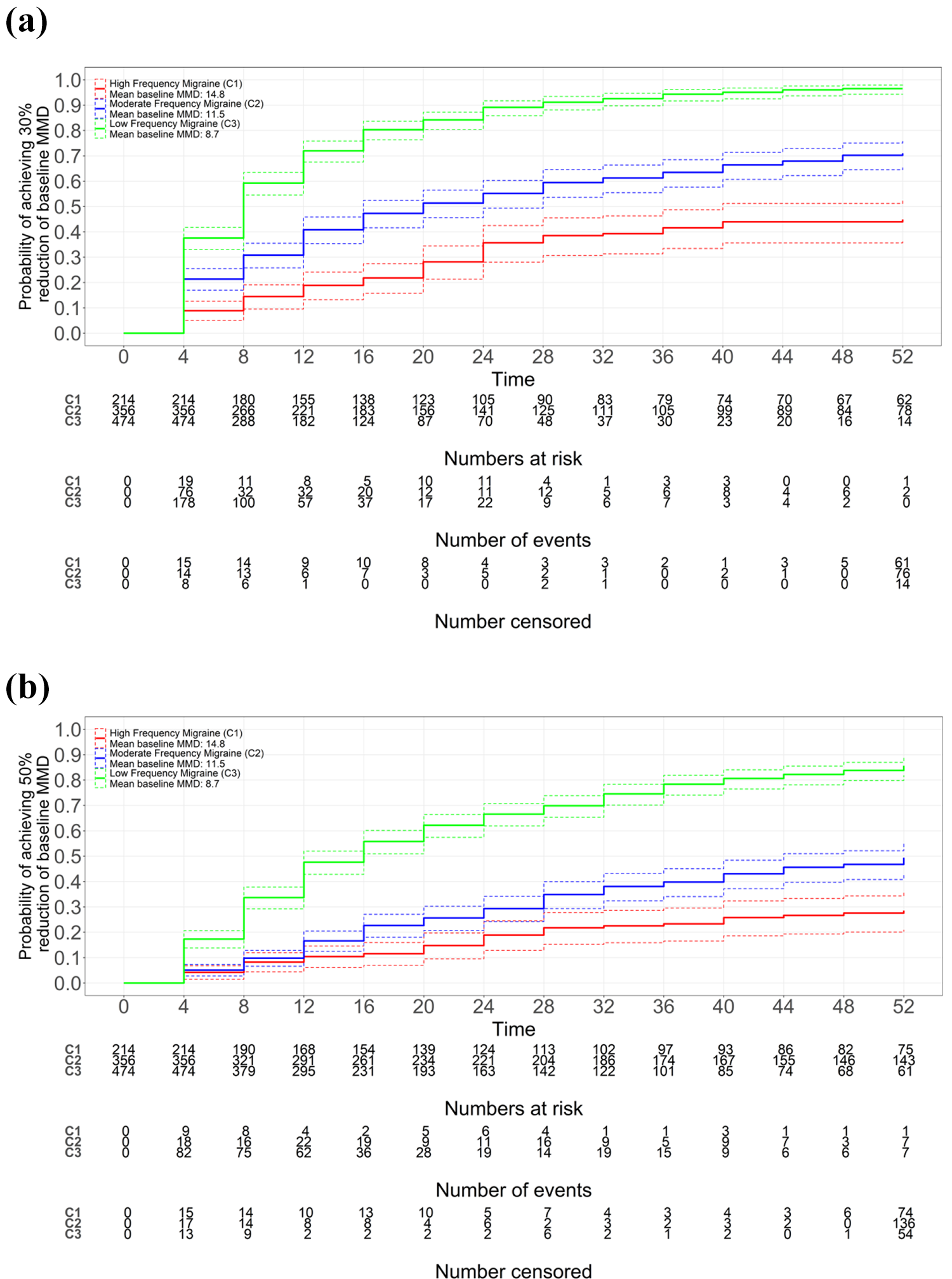
KM curves for PRN ≥6 MMD analyses, (a) ≥30% reduction and (b) ≥50% reduction in MMD from baseline by mean baseline MMD group. C = cluster; MMD = monthly migraine days; PRN = as needed. Note: Patients can be removed from the number at risk by either achieving the outcome (having an event) or discontinuing from the study (being censored).
Lower baseline monthly migraine frequency (C3) achieved ≥30% reduction of baseline MMD more quickly than the moderate (C2) and high (C1) frequency clusters. Median time to ≥30% MMD reduction (Figure 3(a)) was not reached for Cluster 1 (baseline MMD = 14.8 (SD; 5.5)), 20 weeks (IQR; 8-NR weeks) for Cluster 2 (baseline MMD = 11.5 (SD; 3.7)), and 8 weeks (IQR; 4–16 weeks) for cluster 3 (baseline MMD = 8.7 (SD; 2.5)). Likewise the low-frequency cluster (C3) reached ≥50% reduction of baseline MMD more rapidly than the moderate (C2) or high (C1) frequency clusters. Median time to 50% MMD reduction (Figure 3(b)) was not achieved by 52-weeks for cluster 1 (baseline MMD = 14.8 (SD; 5.5)) nor for cluster 2 (baseline MMD = 11.5 (SD; 3.7)), and 16 weeks (IQR; 6-36 weeks) for Cluster 3 (baseline MMD = 8.7 (SD; 2.5)).
Sensitivity analyses of the Cox proportional hazards models were conducted for both time to ≥30% and ≥50% MMD reductions where concomitant prophylactic medication was included as an additional covariate. For both outcomes, this additional covariate was deemed insignificant and not included in the final model configuration.
For all Cox proportional hazards models, the proportional hazards assumption was confirmed via the presence of parallel curves using the method described in Kalbfleisch and Prentice. 21 All p-values were also >0.05 when conducting the global test evaluating if slope =0 for the time-dependent coefficients.
The ratio of rimegepant doses to MMD is illustrated in Figure 4 (green lines). After an initial period of 4–8 weeks, pill utilization stabilized across the three MMD clusters. Overall, these ratios were consistently less than 1 implying that patients chose not to treat some migraine attacks with study medication, although they increased to approximately 0.8–0.85 over follow-up across clusters. The mean number of tablets per 4-weeks showed a reducing trend across 52-weeks for the low (C3) frequency cluster from around 6 tablets per 4-weeks to about 4 tablets per 4-weeks (Figure 4 blue dotted line). The mean number of tablets per 4-weeks was fairly constant for the moderate (C2) and high (C1) frequency clusters running at about 8–9 tablets per 4-weeks for the moderate cluster and increasing from 11 to 15 for the high-frequency cluster (Figure 4 blue dashed and solid lines).

Ratio of rimegepant doses per MMD and tablet frequency by cluster group.
Discussion
The current study used Kaplan–Meier and Cox Proportional Hazards methodology to describe longitudinal changes in MMD associated with the PRN acute treatment of migraine for oral rimegepant 75 mg up to 52-weeks. In participants with ≥6 MMD at baseline, PRN acute treatment of migraine with rimegepant demonstrated a progressive reduction in migraine burden.Median time to ≥30% and ≥50% reduction of baseline MMD was achieved at 12- and 32-weeks, respectively. This study is the first to demonstrate a preventive treatment signal in migraine when an approved oral CGRP receptor antagonist is dosed as needed for the acute treatment of migraine. The use of cluster analysis further provides an objective method to define natural subgroups of people with migraine based on migraine frequency.
It is estimated that 2.5–7.6% of people with episodic migraine will develop chronic migraine every year. 24 –26 Use of several acute treatment classes are associated with an increased risk of medication overuse headache or of chronic migraine onset. 27,28 In this study, PRN acute treatment with the oral CGRP receptor antagonist rimegepant 75 mg at a dosing frequency of up to 15 tablets per 4-weeks was associated with long-term reductions in MMD, differentiating it from other acute treatments for migraine.
These analyses suggest that a substantial percentage of patients (between 63% and 79% overall) presenting with ≥6 MMD will achieve clinically significant reductions in MMD with treatment of rimegepant 75 mg over a time period ranging from 12- to 32-weeks. Changes were nonlinear with greater reductions in the first weeks of treatment, followed by a stable rate over the remainder of the follow-up period. These patterns of change were consistent across the three MMD cluster groups.
As shown in Figure 4 (left axis), the ratio of rimegepant doses to MMD is consistently less than one in each MMD cluster and over the entire follow-up period. This dosing profile in a PRN study that allowed up to daily dosing for 52-weeks may relate to the previous demonstration of durable 48 h of benefit observed with a single dose of rimegepant 75 mg in the acute treatment of migraine, including sustained 2–48 h: pain freedom, pain relief, most bothersome symptom freedom and return to normal function. 8 Of note, there is little separation between the three migraine frequency clusters, indicating that the need for less than one tablet per migraine is a property that may convey to all people with migraine regardless of baseline migraine frequency, including here from low-frequency (baseline MMD 8.7) to high-frequency (baseline MMD 14.8) clusters. Alternatively, the need for less than one dose per MMD may be reflective of either a reduction in MMD over time with rimegepant treatment or less severe attacks over time, providing patients with greater confidence to self-manage their migraine based on ready access to a drug that they know works and is well tolerated, or an awareness that their triggers are less likely to cause a migraine, or a reflection that the burden of disease is reducing over time. The design of BHV3000-201 among participants in the PRN enrollment group is more reflective of an observational, real-world study. Thus, these ratios provide some insight as to rimegepant use in the context of a longitudinal reduction in MMD out to 1 year.
Hypersensitivity to common environmental triggers and to the effects of CGRP in participants pre-disposed to migraine attacks makes managing migraine a challenge for patients and for providers. 4 As a critical neuropeptide in somatosensory neurons, CGRP is thought to play a role in changing the sensitivity threshold in migraine patients. Being abundant in the peripheral trigeminal ganglion and the trigeminal nerve, dysregulation and excessive CGRP signaling is thought to lead to hyper-responsive neuronal signaling in the afferent neurons by lowering their activation threshold and increasing excitability. 5,29 Further potentiation and augmentation of this signal can result in long-term hypersensitivity to common triggers to migraine. In the current study, the reductions in MMDs in patients with established migraine burden following intermittent acute treatment with a CGRP receptor antagonist indicates that these sensitizing events may be reversible, at least in some number of individuals. These findings further support that migraine is a CGRP-mediated disorder, and therapeutic regimens such as rimegepant which disrupt this dysfunctional CGRP signaling, may reduce the overall migraine burden and lower the risk of progression to CM over time (avoiding chronification). Patients in this study who had fewer migraine attacks per month at baseline had a more rapid reduction in their MMD than patients with higher baseline number of attacks per month, suggesting that in those with a more severe underlying hypersensitivity state, and hence more attacks, it may take longer for intermittent CGRP receptor antagonist use to work.
A strength of this study was its duration (52-weeks) coupled with the real-world dosing regimen permitting use of rimegepant on an as needed basis. This provides some assurance that the observed MMD changes are indeed reflective of real-world practice. A limitation pertains to the lack of a reference group, which is not mandated by regulators in a long-term safety study. In an uncontrolled study, a placebo effect is often a potential source of bias. For example, in a recent study, headache and migraine frequency was observed to improve over time among regular users of an electronic headache diary. 30 Future studies could include participants from special populations, with active treatment comparators, and measure other outcomes, particularly biomarkers of the interictal neuro-immune state over time, and patient-reported outcomes to assess additional clinically meaningful benefits of treatment.
Conclusion
In participants with ≥6 MMD at baseline, repeated abortive use of oral rimegepant 75 mg taken as for the acute treatment of migraine resulted in achieving a ≥30% and ≥50% reductions in MMD out to 1 year of follow-up, without definitive evidence of medication related increases in headache frequency. This observation suggests that intermittent rimegepant use for acute treatment may confer an additional preventative benefit. Long-term intermittent acute treatment of migraine with rimegepant 75 mg resulted in reduced migraine frequency which persisted over up to a year of dosing in participants with baseline MMD of ≥6.
Article highlights
This paper describes outcomes from patients treated with as needed (PRN) rimegepant to treat migraine attacks, expressed as median time to ≥30% and ≥50% reduction in MMD. The study population was defined as patients with migraine who had ≥6 MMD at pre-treatment baseline.
This study is, to our knowledge, the first to utilize Kaplan–Meier and Cox Proportional Hazards methodology to describe longitudinal changes in migraine frequency.
This study showed that long-term PRN acute treatment of migraine attacks with rimegepant may lower their monthly disease burden by reducing the number of migraine days.
Reduction in MMD frequency was observed over time regardless of baseline migraine frequency, including low-frequency (baseline MMD 8.7), moderate frequency (baseline MMD 11.5) and high-frequency (baseline MMD 14.8) cluster groups.
Footnotes
Appendix 1
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EP, KJ, and LP are employees of Broadstreet HEOR, which received funds from Biohaven for this work. GL, DM, CC, LH, RC, and VC are employees of and own stock/stock options in Biohaven. NK was a prior employee of Biohaven.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Biohaven Pharmaceuticals Inc.
