Abstract
Aim
We aimed to evaluate the efficacy and safety of eptinezumab for the preventive treatment of chronic migraine in a predominantly Asian population.
Methods
The multi-regional, randomized, double-blind, placebo-controlled, phase 3 SUNRISE trial randomly assigned adults with chronic migraine to receive eptinezumab 100 mg, 300 mg, or placebo. The primary endpoint was change from baseline in monthly migraine days (MMDs) during Weeks 1–12. Key secondary efficacy endpoints were 50% reduction in MMDs (Weeks 1–12) and 75% reduction in MMDs (Weeks 1–4, Weeks 1–12), and the percentage of participants experiencing migraine on Day 1.
Results
Overall, 978 participants received treatment, including 621 (63.5%) from Asia. Both eptinezumab doses met the primary and all key secondary efficacy endpoints. The mean change from baseline in MMDs (Weeks 1–12) was −7.2 for eptinezumab 100 mg, −7.5 for eptinezumab 300 mg, and −4.8 for placebo. Between-group differences were −2.4 for eptinezumab 100 mg versus placebo (p < 0.0001) and −2.7 for eptinezumab 300 mg versus placebo (p < 0.0001). Both eptinezumab doses also demonstrated an odds ratio of >2 versus placebo for all migraine responder rates (p < 0.0001), and a lower percentage of participants experiencing migraine on Day 1 versus placebo (p ≤ 0.01). Safety outcomes were similar across treatment groups.
Conclusions
Eptinezumab demonstrated statistically significant greater reductions in MMDs compared with placebo, beginning on Day 1 and sustained through Week 12, with a well-tolerated safety profile consistent with prior clinical trials.
Trial registration
ClinicalTrials.gov (Identifier: NCT04921384); EudraCT (Identifier: 2020-001657-42).
Keywords
Introduction
Migraine is a disabling disorder characterized by headache and is often accompanied by symptoms including nausea, vomiting, photophobia, and phonophobia (1). Worldwide, migraine is the second leading cause of disability for all ages, and the leading cause for persons aged 15–49 years (2,3). Among neurological disorders in Asia, migraine is the second most burdensome condition, accounting for 24.6 million disability-adjusted life years in 2019 (4). Across the World Health Organization–defined area of the Western Pacific (which includes Asian countries such as China, Japan, and South Korea), it was estimated that >260 million people were affected by migraine in 2019, with an age-standardized prevalence of 12,187 per 100,000 (5). Furthermore, while the age-standardized prevalence of migraine has remained largely stable among Central and South Asian countries over the past 30 years, it has shown a marked increase in East Asia (estimated annual percentage increase of 0.28%) (6).
Despite the substantial burden and impact of migraine across Asia, there remain multiple challenges to effective management, including a need for improved medical education in the field of headache disorders and more accurate diagnosis of migraine, a historical lack of standardized treatment guidelines, and variability in the availability of pharmacological treatments (7,8). The recently published practical recommendations from the International Headache Society (IHS) for acute and preventive pharmacological treatment of migraine are an important step toward improving migraine management globally (9,10). However, there remains a high unmet need for greater utilization of preventive treatment options for migraine within Asian countries (11–13).
Clinical trials of new preventive migraine treatments in Asian populations are critical, both to gain regulatory approval and to increase confidence in their efficacy and safety among clinicians and patients. Eptinezumab is a humanized monoclonal antibody that inhibits the action of calcitonin gene-related peptide (CGRP) (14), a protein that plays an important role in migraine by facilitating the transmission of pain within the central nervous system (15,16). Results from two placebo-controlled, large-scale phase 3 trials showed that eptinezumab led to statistically significant reductions in the number of monthly migraine days (MMDs) in participants with episodic and chronic migraine (CM), with migraine reduction from as early as Day 1 after the first infusion, sustained efficacy over 6–12 months, and an acceptable tolerability profile with low incidences of treatment withdrawal due to adverse events (17,18). Data from the smaller phase 3 SUNLIGHT trial (n = 193) in a mainly Asian population with a dual diagnosis of CM and medication-overuse headache (MOH) reported that all efficacy endpoints numerically (but not statistically) favored eptinezumab 100-mg treatment compared with placebo, and no new safety signals were identified (19).
We report the primary results from the large-scale phase 3 SUNRISE clinical trial, which evaluated the efficacy and safety of eptinezumab 100 mg and 300 mg for the preventive treatment of migraine in a predominantly Asian population with CM. The primary estimand was the mean difference in change from baseline in MMDs (Weeks 1–12) in patients with chronic migraine treated with eptinezumab (100 mg and 300 mg) and placebo, with or without use of other preventive migraine treatment (except without use of other anti-CGRP medications), and regardless of infusion interruption or termination before full dose is received.
Methods
Trial design
SUNRISE was an interventional, multi-regional, multi-site, randomized, double-blind, placebo-controlled phase 3 clinical trial to confirm the efficacy and safety of eptinezumab in predominantly Asian participants with CM who were eligible for preventive treatment. The final protocol and statistical analysis plan are presented in Online Supplement 1 and Online Supplement 2, respectively. The total trial duration was approximately 36 weeks and included a screening period (28–30 days), double-blind placebo-controlled period (12 weeks), dose-blinded extension period (eptinezumab only; 12 weeks), and a safety follow-up period (8 weeks) (Figure S1 of Online Supplement 3). The trial was initiated in May 2021; the completion date for the primary analysis of the placebo-controlled period was October 2024, and the final completion date for the extension and safety follow-up periods was February 2025.
The trial was designed in accordance with the Declaration of Helsinki and was conducted in compliance with the protocol, Good Clinical Practice guidelines, and all applicable regulatory requirements. The trial was approved by the Ethics Committee or Institutional Review Board at each site. Participant safety was regularly monitored by an independent Safety Data Monitoring Committee. SUNRISE is registered on ClinicalTrials.gov (NCT04921384) and EudraCT (2020-001657-42).
Participants
Participants were recruited from specialist headache/migraine clinical settings. Full details of the inclusion and exclusion criteria are provided in Online Supplement 1 (pages 44–47). In brief, eligible participants were required to have a diagnosis of CM as outlined in the IHS International Classification of Headache Disorders, 3rd edition, guidelines (1) at the screening visit, with a history of migraine onset ≥12 months previously, and ≥8 migraine days and ≥15 headache days per month within the three months prior to screening. Documented confirmation of migraine was required during the screening period as confirmed by electronic diary (eDiary) entries indicating ≥15 to ≤26 headache days, of which ≥8 days were assessed as migraine days. All participants were adults aged ≥18 years (≥20 in Taiwan) and ≤75 years, with migraine onset on or before 50 years of age. A concurrent diagnosis of MOH was allowed. Key exclusion criteria were prior use of medications targeting the CGRP pathway as a preventive migraine treatment, and the presence of confounding and clinically significant pain syndromes, active temporomandibular disorder, other types of headache syndromes (beyond MOH), unusual migraine subtypes, or any other conditions considered clinically relevant in the context of the trial. All participants provided written informed consent prior to trial participation.
Randomization, blinding, and interventions
Participants were assigned an eDiary at the screening visit (either on a trial device or uploaded to their personal electronic device), and were asked to complete the eDiary daily from the time of screening until either the primary outcome visit (Week 12) or withdrawal. Participants with adequate eDiary compliance (defined as data entry for ≥24 of 28 days) and who continued to meet eligibility criteria were randomly allocated to receive eptinezumab 100 mg, eptinezumab 300 mg, or placebo (1:1:1 ratio), stratified by location and by MMDs at baseline (<17 or ≥17 days). Participants assigned to placebo in the placebo-controlled period were randomly allocated to enter one of the two treatment groups: eptinezumab 100 mg or 300 mg (1:1 ratio) at Week 12, while those assigned to eptinezumab continued on their originally assigned dose. The sponsor's biostatistician was responsible for the creation of the randomization scheme and draft randomization codes, an unblinded randomization administrator generated the randomization codes, and allocation was implemented using interactive response technology. Eptinezumab and placebo were administered via intravenous infusion over 30 min. Participants, investigators, and the sponsor's trial personnel were blinded to treatment allocation (with dose-blinding maintained in the extension), while the pharmacist or designee responsible for receiving, storing, preparing, and dispensing eptinezumab and placebo infusions was unblinded and was not responsible for other aspects of the trial where blinding was necessary. Medication for the acute treatment of migraine was allowed for the entire duration of the trial.
At the end of the 12-week placebo-controlled period, a subset of the SUNRISE population—specifically 160 of the first participants in Japan—were given the opportunity to be enrolled in the SUNSET trial. SUNSET evaluated the long-term (60-week) open-label tolerability and effectiveness of eptinezumab (ClinicalTrials.gov: NCT05064371) and aimed to provide data from ≥1 year of exposure to proposed therapeutic doses of eptinezumab in ≥100 participants, in accordance with ICH E1. Results from SUNSET are reported separately. After SUNSET had enrolled 160 participants, the SUNRISE protocol was amended so that Japanese participants would continue in SUNRISE and receive active treatment during the 12-week safety extension period; thus, outcomes over Weeks 13–24 in SUNRISE include a subset of the Japanese population from Weeks 1–12.
Trial endpoints
The primary objective of the SUNRISE trial was to evaluate the efficacy of eptinezumab for the prevention of migraine in participants with chronic migraine. To achieve this, the primary endpoint was the change from baseline in number of MMDs during Weeks 1–12. Key secondary efficacy endpoints were ≥50% and ≥75% MMD responder rates (i.e., ≥50% and ≥75% reduction from baseline in MMDs during Weeks 1–12, and ≥75% reduction from baseline in MMDs during Weeks 1–4) and the percentage of participants experiencing migraine on the day after dosing (Day 1). Other secondary efficacy endpoints were: monthly headache day (MHD) responder rates (≥50% reduction from baseline during Weeks 1–12 and ≥75% reduction during Weeks 1–4); change from baseline in the number of MHDs (Weeks 1–12); percentage of participants experiencing migraines with severe pain intensity (Weeks 1–12); percentage of participants experiencing headaches with severe pain intensity (Weeks 1–12); number of MMDs with use of acute medication (Weeks 1–12); Patient Global Impression of Change (PGIC) score at Week 12; and patient-identified most bothersome symptom (PI-MBS) change score at Week 12. The PGIC is a single patient-reported item reflecting the participant's impression of change in their disease status since the start of the trial, rated on a 7-point scale from ‘very much improved’ to ‘very much worse’ (20). Participants were also asked to rate the improvement in PI-MBS (nausea, vomiting, sensitivity to light, sensitivity to sound, mental cloudiness, fatigue, pain with activity, mood changes, or other) on a 7-point scale identical to the PGIC scale (21,22). For completeness, ≥50% MMD responder rate (Weeks 1–4), ≥50% MHD responder rate (Weeks 1–4), and ≥75% MHD responder rate (Weeks 1–12) are also presented. There were no assessments of efficacy endpoints during the 12-week extension period.
Assessments of safety included the occurrence of treatment-emergent adverse events (TEAEs), classified according to the Medical Dictionary for Regulatory Activities version 27.0; clinical safety laboratory test values; vital signs, weight, and ECG parameter values; and Columbia–Suicide Severity Rating Scale (C-SSRS) scores (23). TEAEs were initially analyzed for all participants based on the 12-week placebo-controlled period. For participants entering the extension period, safety outcomes were captured for a further 20 weeks (12-week extension period plus 8-week safety follow-up period) and tabulated. Participants who did not enter the extension period had a total of 20 weeks of safety follow-up data; TEAEs reported after withdrawal in the placebo-controlled period but before Week 12 were allocated to the placebo-controlled period, and TEAEs reported during the subsequent eight weeks were allocated to the safety follow-up period.
Statistical analyses
Based on recent results of previous randomized, placebo-controlled, phase 3 anti-CGRP studies conducted in Asian patients with CM (19,24), the design was adapted while the trial was ongoing; following the adaptation a treatment difference versus placebo in change from baseline in MMDs (Weeks 1–12) of −1.6 days for eptinezumab 100 mg and −2.2 days for 300 mg, with a common standard deviation (SD) of 6.9 was assumed. The sample size was increased from 175 to 315 participants per treatment group, providing 97% power to show an effect in the 300-mg group, 81% power for an effect in the 100-mg group, and combined 80% power for both doses.
Demographic and placebo-controlled safety data were assessed using the all-participants-treated set (i.e., all randomized participants who received an infusion of double-blind trial medication). Efficacy data were assessed using the full-analysis set (i.e., all participants in the all-participants-treated set who had a valid assessment of baseline MMDs and ≥1 valid post-baseline 4-week assessment of MMDs in Weeks 1–12). Safety data over the extension period was assessed using the all-participants-treated-extension set (i.e., all randomized participants who received an infusion of dose-blinded trial medication during the extension period).
For the primary efficacy endpoint, the change from baseline in the number of MMDs per 4-week interval (Weeks 1–4, 5–8, and 9–12) were modeled using a mixed model for repeated measures (MMRM) with month, treatment, and location as fixed factors, and baseline MMDs as a continuous covariate. Interaction terms were included for month–treatment, and month–baseline score. Within-participant variance was modeled using an unstructured variance matrix. The model used Kenward-Rogers approximation of degrees of freedom. The change from baseline in MMDs for Weeks 1–12 was estimated as the average across the three time intervals, and the treatment effect of each of the two doses of eptinezumab compared with placebo was calculated. The trial applied a 5% significance level, and a combination of sequential testing and splitting of the significance level by dose was applied for testing the primary and key secondary efficacy results. The primary endpoint was first tested for 300-mg eptinezumab versus placebo using a 5% significance level and, if this showed effect, testing for the primary endpoint continued for 100-mg eptinezumab versus placebo using a 5% significance level. Following this, the significance level was divided between the two doses, with 2.5% for each; for each subsequent step, testing continued only if all prior effects in the hierarchy were found to have p-values below 2.5% (Figure S2 of Online Supplement 3).
For responder rate endpoints, response was defined as a ≥50% or ≥75% reduction in the average MMDs (or MHDs) for Weeks 1–4, or the average across the 4-week intervals for Weeks 1–12. Each eptinezumab treatment group was compared with placebo using a logistic regression model, with treatment as a factor and baseline MMDs as a continuous covariate. The percentage of participants experiencing migraine on Day 1 was analyzed using an extended Cochran–Mantel–Haenszel test, adjusting for MMDs at baseline (<17 vs ≥17 days).
Other continuous endpoints related to MMDs and MHDs were analyzed using an MMRM similar to the one described for the primary endpoint, using their corresponding baseline days as a covariate. For endpoints such as PGIC and PI-MBS, analysis used an MMRM including treatment group, visit, site location, and the stratification factor (<17 vs ≥17 MMDs) as fixed factors, their corresponding baseline value as a covariate, and treatment group by visit, baseline value by visit, and stratification factor by visit interactions. The estimated least squares mean differences between each dose group and placebo were calculated, together with 95% confidence intervals.
Outcomes during the placebo-controlled period were assessed by treatment group (i.e., eptinezumab 100 mg, 300 mg, or placebo) and during the extension period were assessed by treatment sequence (i.e., eptinezumab 100 mg–100 mg, eptinezumab 300 mg–300 mg, placebo–eptinezumab 100 mg, and placebo–eptinezumab 300 mg). All statistical analyses were conducted using SAS software version 9.4 or later (SAS Institute, Inc., Cary, NC).
Results
Trial participants
Participant disposition is shown in Figure 1. Of 983 participants randomized to treatment, 978 (99.5%) were treated and 939 (95.5%) completed the placebo-controlled period. The full-analysis set comprised 323 participants in the eptinezumab 100-mg group, 324 in the 300-mg group, and 325 in the placebo group. Of the 972 participants who completed the placebo-controlled period, 777 (82.7%) were treated in the extension period (160 Japanese participants enrolled in SUNSET, and two other participants were not treated). Across both treatment periods, “other” reasons and withdrawal of consent were the most common reasons for study withdrawal (Figure 1).
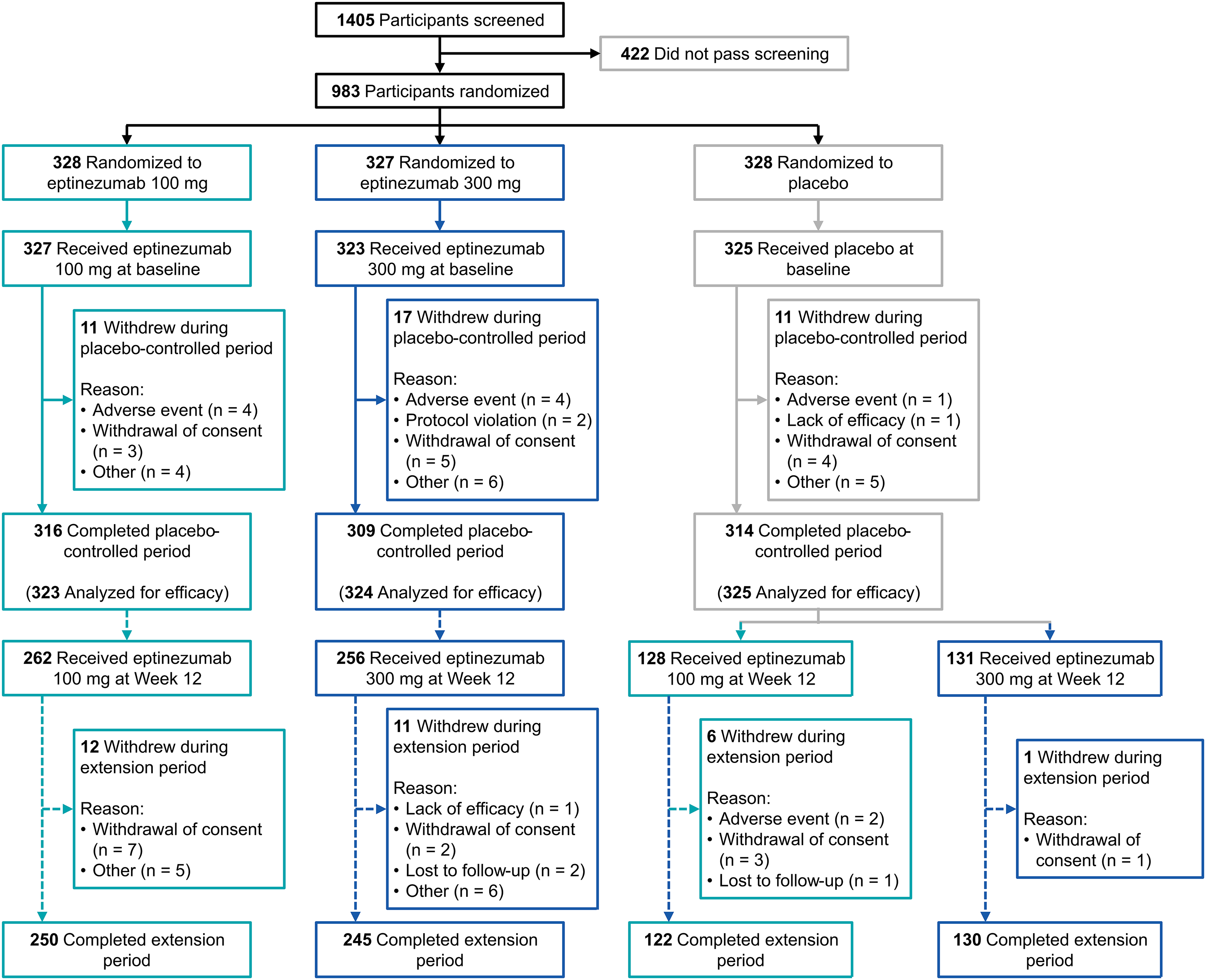
Participant disposition through the placebo-controlled period of the SUNRISE trial. At the end of the 12-week placebo-controlled period, 160 of the first participants in Japan were given the opportunity to be enrolled in the SUNSET trial (NCT05064371). After SUNSET had enrolled, the SUNRISE protocol was amended so that Japanese participants continued in the SUNRISE safety extension period.
Baseline demographic characteristics are reported in Table 1. Approximately two-thirds of participants were from Asia (n = 621; 63.5%), with the remainder from Europe (n = 357; 36.5%). Most participants were female (n = 840; 85.9%), and there was a mean (SD) age of 42.0 (11.0) years and a mean of 17.4 (4.2) MMDs at baseline. Overall, 410/978 (41.9%) participants had a concurrent MOH diagnosis.
Baseline participant demographics.

Data are from the all-participants-treated set (i.e., all randomized participants who received an infusion of double-blind trial medication), except where noted.
Data are from the full-analysis set (i.e., all participants in the all-participants-treated set who had a valid assessment of baseline MMDs and ≥1 valid post-baseline 4-week assessment of MMDs in Weeks 1–12).
One participant in the placebo arm was missing a response, thus percentages are based on 324 participants in the placebo arm and 977 in the total population.
MHDs, monthly headache days; MMDs, monthly migraine days; MOH, medication-overuse headache; SD, standard deviation.
Efficacy outcomes
Both eptinezumab doses showed statistically significant advantages compared with placebo for the primary (Figure 2) and all key secondary efficacy endpoints (Figure S2 and Table S1 of Online Supplement 3). The results for the primary endpoint showed that the mean change from baseline in number of MMDs (Weeks 1–12) was −7.2 for the eptinezumab 100-mg group, −7.5 for the eptinezumab 300-mg group, and −4.8 for the placebo group (Figure 2). Between-group differences were −2.4 for eptinezumab 100 mg versus placebo (p < 0.0001) and −2.7 for eptinezumab 300 mg versus placebo (p < 0.0001).

Change from baseline in MMDs over (a) Weeks 1–12 and (b) 4-week intervals. aP-values are from the analysis of the primary endpoint. Analyzed in the full-analysis set (i.e., all participants treated in the placebo-controlled period who had a valid assessment of baseline MMDs and ≥1 valid post-baseline 4-week assessment of MMDs in Weeks 1–12). The estimated means, mean differences from placebo, and 95% CIs were calculated by a mixed models for repeated measures analysis with month (Weeks 1–4, Weeks 5–8, Weeks 9–12), site location, and treatment as factors; baseline score as a continuous covariate; and interaction terms for treatment-by-month and baseline score-by-month. Estimates and tests over Weeks 1–12 used equal weights for each 4-week interval. ****p < 0.0001 vs placebo. Δ, least-squares mean difference from placebo (95% CI); CIs, confidence intervals; MMDs, monthly migraine days; SE, standard error.
Regarding the key secondary endpoints, both doses of eptinezumab demonstrated an odds ratio versus placebo of >2 for ≥50% MMD response (Weeks 1–12, p < 0.0001) and ≥75% MMD response (Weeks 1–4, p < 0.0001; Weeks 1–12, p < 0.0001) (Figure 3). Both doses of eptinezumab also demonstrated a lower estimated percentage of participants experiencing migraine on Day 1 following dosing compared with placebo (Figure S3 of Online Supplement 3); the estimated percentage of participants with migraine on Day 1 was 41.0% with eptinezumab 100 mg, 38.8% with eptinezumab 300 mg, and 50.9% with placebo (difference vs placebo: 100 mg, −9.9%, p = 0.01; 300 mg, −12.1%, p = 0.002).

MMD responder rates: (a) ≥50% reduction from baseline and (b) ≥75% reduction from baseline. aP-values are from analyses of key secondary endpoints, controlled for multiple comparison. Analyzed in the full-analysis set (i.e., all participants treated in the placebo-controlled period who had a valid assessment of baseline MMDs and ≥1 valid post-baseline 4-week assessment of MMDs in Weeks 1–12). The ≥50% and ≥75% MMD responder rates were defined as the proportion of participants with ≥50% and ≥75% reduction from baseline in MMDs, respectively. Responder rates across the three 4-week intervals (i.e., Weeks 1–12) were calculated as the mean percentage change in MMDs (based on the available monthly values of MMDs). The comparisons were based on logistic regression models including baseline MMDs as a continuous covariate and treatment as a factor. Odds ratios (95% CIs) vs placebo are provided. If the MMD value was missing for a given month, the responder status was derived based on the available values. CI, confidence interval; MMDs, monthly migraine days.
Subgroup analyses of the primary and key secondary efficacy endpoints by site location are shown in Table S2 of Online Supplement 3. Similar results were noted across locations and efficacy results were similar to those of the total trial population.
Table S1 of Online Supplement 3 summarizes results of other secondary endpoints. Both doses of eptinezumab showed better outcomes than placebo for the percentage of participants experiencing migraines with severe pain intensity and the number of MMDs with use of acute medication (Figure 4). Data for change in number of MHDs, MHD responder rates, and proportion of headache episodes with severe pain intensity followed similar trends as the MMD-based endpoints (Figure S4 of Online Supplement 3). Additionally, improvements across patient-reported outcomes (PGIC and PI-MBS) were greater with both doses of eptinezumab compared with placebo at each timepoint (p < 0.0001) (Figure 5).
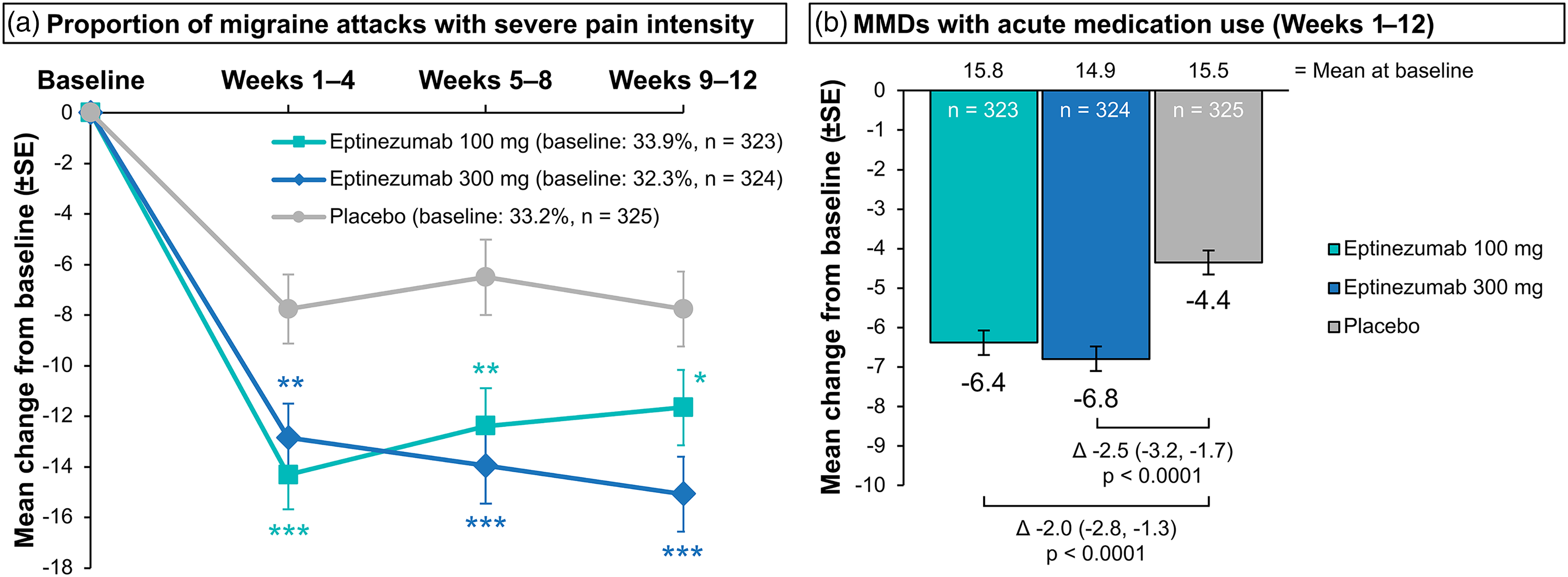
Change from baseline in (a) proportion of migraine attacks with severe pain intensity over 4-week intervals and (b) change from baseline in MMDs with acute medication use over Weeks 1–12. Analyzed in the full-analysis set (i.e., all participants treated in the placebo-controlled period who had a valid assessment of baseline MMDs and ≥1 valid post-baseline 4-week assessment of MMDs in Weeks 1–12). The estimated means and mean differences from placebo were from an MMRM analysis. For panel (a), the MMRM included month (Weeks 1–4, Weeks 5–8, Weeks 9–12), site location, stratification factor (MMDs at baseline [<17 or ≥17]), and treatment as factors; baseline score as a continuous covariate; and interaction terms for treatment-by-month, stratum-by-month, and baseline score-by-month. Estimates and tests over Weeks 1–12 used equal weights for each 4-week interval. For panel (b), the MMRM included month, site location, and treatment as factors; baseline score as a continuous covariate; and interaction terms for treatment-by-month and baseline score-by-month. Estimates and tests used equal weights for each 4-week interval. *p < 0.05 vs placebo; **p < 0.01 vs placebo; ***p < 0.001 vs placebo; ****p < 0.0001 vs placebo. MMDs, monthly migraine days; MMRM, mixed model for repeated measures.

Mean (a) PGIC score and (b) PI-MBS score. Analyzed in the full-analysis set (i.e., all participants treated in the placebo-controlled period who had a valid assessment of baseline MMDs and ≥1 valid post-baseline 4-week assessment of MMDs in Weeks 1–12). The PGIC scale ranges from 1 (very much improved) to 7 (very much worse) with 4 = no change. The PI-MBS scale ranges from 1 (very much improved) to 7 (very much worse) with 4 = no change. The estimated means were calculated using mixed models for repeated measures with visit (Week 4, Week 8, Week 12), location, stratification (baseline MMDs [<17 or ≥17]), and treatment as factors and interaction terms for stratum-by-visit and treatment-by-visit. ****p < 0.0001 vs placebo. MMDs, monthly migraine days; PGIC, Patient Global Impression of Change; PI-MBS, patient-identified most bothersome symptom; SE, standard error.
Safety outcomes
TEAEs during the initial 12-week double-blind treatment period are summarized in Table 2. The proportion of participants with TEAEs was similar between treatment groups: 37.6% (eptinezumab 100 mg), 32.2% (eptinezumab 300 mg), and 33.5% (placebo). There were few serious TEAEs (<2% across groups), TEAEs leading to withdrawal (<2% across groups), or TEAEs leading to infusion interruption (<1% across groups). There were no marked differences between treatment groups in the rates of TEAEs with an incidence of ≥2% during the placebo-controlled period (Table 2).
Summary of treatment-emergent adverse events during the 12-week double-blind placebo-controlled period.

Data are from the all-participants-treated set (i.e., all randomized participants who received an infusion of double-blind trial medication).
COVID-19, coronavirus disease 2019; SAE, serious adverse event; TEAE, treatment-emergent adverse event.
TEAEs during the 12-week dose-blinded extension period are summarized in Table 3. As observed during the initial 12 weeks, the percentage of participants with TEAEs was similar across groups (eptinezumab 100 mg–100 mg, 42.4%; eptinezumab 300 mg–300 mg, 39.1%; placebo–eptinezumab 100 mg, 35.9%; placebo–eptinezumab 300 mg, 42.7%), with low rates of serious TEAEs (<4% across groups), TEAEs leading to withdrawal (two participants total), and TEAEs leading to infusion interruption (three participants total). Similar rates of nasopharyngitis, upper respiratory tract infection, and urinary tract infection occurred during both treatment periods (Table 3).
Summary of treatment-emergent adverse events during the during the 12-week dose-blinded extension period.

Data are from the all-participants-treated-extension set (i.e., all randomized participants who received an infusion of dose-blinded trial medication during the extension period); groups refer to the randomly allocated treatment sequence assigned at baseline (i.e., eptinezumab throughout, or placebo followed by eptinezumab).
COVID-19, coronavirus disease 2019; SAE, serious adverse event; TEAE, treatment-emergent adverse event.
There were no clinically relevant safety findings in terms of vital signs, weight, laboratory values, ECGs, or C-SSRS during either the placebo-controlled or extension periods. No new safety signals were identified relative to prior 12- and 24-week migraine trials and the known safety profile of eptinezumab (18,19,25–27).
Discussion
The results of the SUNRISE clinical trial, conducted in a predominantly Asian population, demonstrated robust efficacy for both eptinezumab 100 mg and 300 mg based on clinically relevant measures which were established for regulatory approval of preventive migraine treatments (28). Eptinezumab produced statistically significant reductions in MMDs versus placebo, beginning the day after dosing and sustained through Week 12, with a well-tolerated safety profile consistent with prior trials.
The robust efficacy and safety data generated from this predominantly Asian population provide high-quality regional evidence that can facilitate approvals of new treatments in Asian countries as well as inform future updates to local and regional treatment guidelines for migraine prevention. Both eptinezumab doses evaluated in this trial showed statistically significant advantages compared with placebo for the primary and all key secondary efficacy endpoints, with eptinezumab reducing migraine frequency, pain severity, and the percentage of participants experiencing migraine on Day 1 to a greater extent than placebo. The data for the primary endpoint, change from baseline in MMDs (Weeks 1–12) for eptinezumab 100 mg and 300 mg compared with placebo, indicated robust and statistically significant (p < 0.0001) efficacy. The sequential hierarchy testing by dose that was applied to the efficacy results in the current trial also demonstrated statistically significant advantages for both doses over placebo for all key secondary endpoints. We observed a rapid onset of effect after the first infusion, with reductions in migraine observed as early as Day 1, with the effect continuing through each 4-week period and sustained over Weeks 1–12. In addition, greater improvements in PGIC and PI-MBS scores were also observed with eptinezumab compared with placebo (p < 0.0001), which were of particular interest since these measures reflect the global impact of treatment and take into consideration different components of improvement. Together, these data offer a clear, evidence-based rationale for incorporating eptinezumab as a recommended preventive therapy, building upon the recent recommendations from the IHS (9,10).
These data indicate comparable efficacy to that observed in the previous pivotal phase 3 PROMISE-2 trial (18) in participants with CM (which included a similar proportion [40.2%] of participants with concurrent MOH compared with the current trial [41.9%]), confirming the benefits of eptinezumab in a predominantly Asian population. In PROMISE-2, the mean number of baseline MMDs was 16.1 days, and the difference in change from baseline in MMDs (Weeks 1–12) was −2.0 days for eptinezumab 100 mg versus placebo and −2.6 days for eptinezumab 300 mg versus placebo. In SUNRISE, the mean number of baseline MMDs was 17.4, and the respective reductions from baseline versus placebo were −2.4 and −2.7. Moreover, the respective odds ratios for eptinezumab 100 mg and 300 mg versus placebo for MMD responder rates during Weeks 1–12 in PROMISE-2 were 2.1 and 2.4 (≥50% MMD response), and 2.0 and 2.8 (≥75% MMD response) (18). In SUNRISE, the corresponding odds ratios were 2.2 and 2.7 (≥50% MMD response), and 2.9 and 3.0 (≥75% MMD response). The consistent numerical superiority of eptinezumab over placebo observed in the preceding small SUNLIGHT trial (n = 193), conducted in a mainly Asian population of participants with CM and MOH (19), was confirmed in the current SUNRISE trial. Importantly, the subgroup analyses of SUNRISE in Asian, Japanese, and Chinese populations also provided efficacy results similar to those of the total trial population (Online Supplementary Table S2). This adds weight to the recent observational study that reported real-world effectiveness of eptinezumab among 30 patients with CM in Singapore, during which mean MMDs decreased by 4.3 days at Month 3 compared with baseline, with 20% of patients reaching ≥50% response (29).
Although differences in trial methodology preclude direct comparisons between studies, SUNRISE demonstrated numerically similar or better efficacy, as measured by treatment differences in MMDs and responder rates, than the phase 3 DRAGON trial of erenumab conducted in participants with CM from nine Asian countries or regions. In DRAGON, the mean change from baseline to Weeks 9–12 in MMDs was −8.2 days for erenumab 70 mg and −6.6 days for placebo (difference −1.57; p = 0.015), and the odds ratio for ≥50% MMD responder rate (erenumab vs placebo for Weeks 9–12) was 1.54 (p = 0.014) (24).
In the SUNRISE trial, eptinezumab was well tolerated and demonstrated a similar safety profile to that seen in prior CM trials (18,19,25,26,30). During the placebo-controlled period, the numbers of serious TEAEs and TEAEs leading to withdrawal were low and were similar across treatment groups. Common TEAEs of nasopharyngitis and upper respiratory tract infections were expected, based on the known safety profile (27,31), while the TEAE of COVID-19 was also unsurprising given the trial was initiated during the pandemic. Data from the extension period confirmed the tolerability of eptinezumab, and no new safety signals were identified.
Results from the SUNRISE trial may help address the underutilization of preventive treatments in Asian countries (11–13) by bolstering clinician confidence and providing crucial regional evidence underscoring the efficacy of migraine-specific anti-CGRP treatment. The high retention rate, clear and consistent benefits across multiple efficacy endpoints—including the rapid reduction in migraine incidence observed as early as the day after dosing (Day 1)—and well-tolerated safety profile may provide reassurance to both clinicians and patients of the clinical value of eptinezumab in Asia.
The main limitation of the SUNRISE trial was the exclusion of participants who had previously used a preventive migraine medication targeting the CGRP pathway, and those with other headache and pain syndromes, meaning that the findings may not be generalizable to the wider population of real-world patients who seek treatment for migraine.
In conclusion, the SUNRISE trial, conducted in adult participants predominantly from Asia, demonstrated the efficacy of eptinezumab 100 mg and 300 mg over 12 weeks after the initial dose. Eptinezumab was also well-tolerated during both the placebo-controlled and extension periods. These results were comparable with those observed in the global phase 3 CM trials (such as PROMISE-2) and confirm the benefits of preventive eptinezumab treatment in a wider patient population.
Clinical implications
The phase 3, randomized, double-blind, placebo-controlled SUNRISE trial evaluated eptinezumab 100 mg, 300 mg, or placebo for prevention of chronic migraine among a predominantly Asian population.
Eptinezumab produced statistically significant greater migraine-preventive effects than placebo over Weeks 1–12, starting from Day 1 after administration.
Eptinezumab treatment was well tolerated, with a safety profile consistent with prior clinical trials.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024251386095 - Supplemental material for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial
Supplemental material, sj-pdf-1-cep-10.1177_03331024251386095 for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial by Shengyuan Yu, Yasuhiko Matsumori, Byung-Kun Kim, Anna Gryglas-Dworak, Gvantsa Giorgadze and Patricia Pozo-Rosich, Mette Krog Josiassen, Kristina Ranc, Anders Ettrup, Aurélia Mittoux, Bjørn Sperling, Takao Takeshima in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024251386095 - Supplemental material for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial
Supplemental material, sj-pdf-2-cep-10.1177_03331024251386095 for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial by Shengyuan Yu, Yasuhiko Matsumori, Byung-Kun Kim, Anna Gryglas-Dworak, Gvantsa Giorgadze and Patricia Pozo-Rosich, Mette Krog Josiassen, Kristina Ranc, Anders Ettrup, Aurélia Mittoux, Bjørn Sperling, Takao Takeshima in Cephalalgia
Supplemental Material
sj-docx-3-cep-10.1177_03331024251386095 - Supplemental material for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial
Supplemental material, sj-docx-3-cep-10.1177_03331024251386095 for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial by Shengyuan Yu, Yasuhiko Matsumori, Byung-Kun Kim, Anna Gryglas-Dworak, Gvantsa Giorgadze and Patricia Pozo-Rosich, Mette Krog Josiassen, Kristina Ranc, Anders Ettrup, Aurélia Mittoux, Bjørn Sperling, Takao Takeshima in Cephalalgia
Supplemental Material
sj-docx-4-cep-10.1177_03331024251386095 - Supplemental material for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial
Supplemental material, sj-docx-4-cep-10.1177_03331024251386095 for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial by Shengyuan Yu, Yasuhiko Matsumori, Byung-Kun Kim, Anna Gryglas-Dworak, Gvantsa Giorgadze and Patricia Pozo-Rosich, Mette Krog Josiassen, Kristina Ranc, Anders Ettrup, Aurélia Mittoux, Bjørn Sperling, Takao Takeshima in Cephalalgia
Supplemental Material
sj-pdf-5-cep-10.1177_03331024251386095 - Supplemental material for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial
Supplemental material, sj-pdf-5-cep-10.1177_03331024251386095 for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial by Shengyuan Yu, Yasuhiko Matsumori, Byung-Kun Kim, Anna Gryglas-Dworak, Gvantsa Giorgadze and Patricia Pozo-Rosich, Mette Krog Josiassen, Kristina Ranc, Anders Ettrup, Aurélia Mittoux, Bjørn Sperling, Takao Takeshima in Cephalalgia
Supplemental Material
sj-pdf-6-cep-10.1177_03331024251386095 - Supplemental material for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial
Supplemental material, sj-pdf-6-cep-10.1177_03331024251386095 for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial by Shengyuan Yu, Yasuhiko Matsumori, Byung-Kun Kim, Anna Gryglas-Dworak, Gvantsa Giorgadze and Patricia Pozo-Rosich, Mette Krog Josiassen, Kristina Ranc, Anders Ettrup, Aurélia Mittoux, Bjørn Sperling, Takao Takeshima in Cephalalgia
Supplemental Material
sj-pdf-7-cep-10.1177_03331024251386095 - Supplemental material for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial
Supplemental material, sj-pdf-7-cep-10.1177_03331024251386095 for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial by Shengyuan Yu, Yasuhiko Matsumori, Byung-Kun Kim, Anna Gryglas-Dworak, Gvantsa Giorgadze and Patricia Pozo-Rosich, Mette Krog Josiassen, Kristina Ranc, Anders Ettrup, Aurélia Mittoux, Bjørn Sperling, Takao Takeshima in Cephalalgia
Supplemental Material
sj-pdf-8-cep-10.1177_03331024251386095 - Supplemental material for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial
Supplemental material, sj-pdf-8-cep-10.1177_03331024251386095 for Efficacy and safety of eptinezumab in a predominantly Asian population with chronic migraine: Results of the randomized, double-blind, placebo-controlled SUNRISE trial by Shengyuan Yu, Yasuhiko Matsumori, Byung-Kun Kim, Anna Gryglas-Dworak, Gvantsa Giorgadze and Patricia Pozo-Rosich, Mette Krog Josiassen, Kristina Ranc, Anders Ettrup, Aurélia Mittoux, Bjørn Sperling, Takao Takeshima in Cephalalgia
Footnotes
Acknowledgments
The authors thank the participants, their families/caregivers, and the sites that participated in this trial. Medical writing and editorial support were funded by H. Lundbeck A/S (Copenhagen, Denmark) and provided by Sally-Anne Mitchell, PhD, and Nicole Coolbaugh, CMPP, of The Medicine Group, LLC (New Hope, PA, USA) in accordance with Good Publication Practice guidelines.
Author contributions
Conceptualization: AM, BS, MKJ. Methodology: BS, KR, MKJ. Software: KR. Validation: BS. Formal analysis: KR, MKJ. Investigation: SY, YM, B-KK, AG-D, BB, PP-R, TT. Writing – Original Draft: All authors. Writing – Review & Editing: All authors. Visualization: All authors. Supervision: AM, BS, MKJ. Project administration: AM.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SY declares no conflicts of interest. YM reports personal consultancy fees from Amgen Astellas BioPharma K.K., Daiichi Sankyo Company, Limited, Eli Lilly Japan K.K., and Otsuka Pharmaceutical Co., Ltd. B-KK has received honoraria as a consultant and speaker from AbbVie, Teva Korea, Lundbeck Koreas, Organon Korea, Pfizer Korea, and SK Pharmaceuticals. His research group has received research grants from Pfizer Korea and funding for clinical trials from AbbVie, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals. He is on editorial boards of Headache and Pain Research and Journal of Clinical Neurology. AG-D has received fees from AbbVie, Amgen, Lundbeck, Pfizer, and Teva while serving as a principal investigator in clinical trials, as well as for speaking and training. GG received fees from Lundbeck while serving as a principal investigator in clinical trials. PP-R has received honoraria, in the last 36 months, as a consultant and speaker from AbbVie, Almirall, Dr Reddy's, Eli Lilly, Lundbeck, Medscape, Novartis, Organon Pfizer, and Teva Pharmaceuticals. Her research group has received research grants from AbbVie, AGAUR, EraNet Neuron, FEDER RIS3CAT, Instituto Investigación Carlos III, MICINN, Novartis, and Teva Pharmaceuticals, and has received funding for clinical trials from AbbVie, Amgen, Biohaven, Eli Lilly, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals. She is the Honorary Secretary of the International Headache Society, is on the editorial board of Revista de Neurologia, and is an associate editor for Cephalalgia and Neurologia. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She is the founder of ![]() , a platform to give information and tools to physicians and people who suffer from migraine and other headaches. MKJ and BS are full-time employees of H. Lundbeck A/S and own stock or stock options in H. Lundbeck A/S. KR, AE, and AM are all full-time employees of H. Lundbeck A/S. TT is an advisor to Hedgehog MedTech, Inc. and Sawai Pharmaceutical Co., Ltd.
, a platform to give information and tools to physicians and people who suffer from migraine and other headaches. MKJ and BS are full-time employees of H. Lundbeck A/S and own stock or stock options in H. Lundbeck A/S. KR, AE, and AM are all full-time employees of H. Lundbeck A/S. TT is an advisor to Hedgehog MedTech, Inc. and Sawai Pharmaceutical Co., Ltd.
Ethical considerations
The trial was designed in accordance with the Declaration of Helsinki and was conducted in compliance with the protocol, Good Clinical Practice guidelines, and all applicable regulatory requirements. The trial was approved by the ethics committee or institutional review board at each site. All participants provided written informed consent prior to trial participation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was sponsored and funded by H. Lundbeck A/S, including medical writing support for the development of the manuscript.
Data sharing
In accordance with EFPIA's and PhRMA's “Principles for Responsible Clinical Trial Data Sharing” guidelines, Lundbeck is committed to responsible sharing of clinical trial data in a manner that is consistent with safeguarding the privacy of patients, respecting the integrity of national regulatory systems, and protecting the intellectual property of the sponsor. The protection of intellectual property ensures continued research and innovation in the pharmaceutical industry. Deidentified data are available to those whose request has been reviewed and approved through an application submitted to ![]() .
.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
