Abstract
Objective
To assess the efficacy of erenumab across the spectrum of response thresholds (≥50%, ≥75%, 100%) based on monthly migraine days (MMD) reduction in patients with chronic migraine from a 12-week, randomized study (NCT02066415).
Methods
Patients (n = 667) received (3:2:2) placebo or erenumab 70/140 mg once-monthly. The proportion of patients achieving a given response threshold was assessed. A post-hoc analysis was conducted to contextualize the actual treatment benefit in subgroups of patients achieving (or not) specified response thresholds. Outcome measures included MMD, acute migraine-specific medication treatment days (MSMD) and disability.
Results
The proportion of patients responding to erenumab exceeded that of placebo at the ≥50% and ≥75% response thresholds. At month 3, 39.9% and 41.2% of patients on erenumab 70 and 140 mg, respectively, achieved ≥50% response versus placebo (23.5%). Similarly, at month 3, 17.0% and 20.9% of patients on erenumab 70 and 140 mg, respectively, achieved ≥75% response versus placebo (7.8%). Compared with the overall erenumab-treated population (change in MMD: −6.6 [both 70 and 140 mg]), ≥50% responders showed MMD reductions of −12.2/−12.5 for 70 mg/140 mg versus −2.6/−2.2 for those not achieving ≥50% response. ≥75% responders showed MMD reductions of −13.9/−14.8 for 70 mg/140 mg versus −5.0/−4.3 for those not achieving ≥75% response. Relative improvements in MSMD and disability were observed in responders versus overall erenumab-treated population.
Conclusion
For erenumab-treated patients achieving ≥50% response, the actual reduction in MMD was almost twice that of the overall population. These findings provide context for setting realistic expectations regarding actual treatment benefit experienced by patients responding to treatment.
Introduction
Chronic migraine (CM) affects approximately 1–2% of the global population (1). According to the International Classification of Headache Disorders (ICHD-III) criteria, CM is defined as headache occurring on 15 or more days per month for more than 3 months, which has the features of migraine headache on at least 8 days per month (2). The high frequency of migraine and non-migraine headache days has a substantial impact on the socioeconomic status of patients, their role functioning and on their quality of life (QoL) (3). Patients with CM have more healthcare provider and emergency visits than those with episodic migraine (4). Due to the high frequency of headaches and severe impact on QoL, patients with CM are generally prescribed preventive medications. However, there is a considerable inter-patient variability in the tolerability and efficacy of the available preventive treatment options (5,6).
Currently available oral migraine preventive medications are associated with low adherence rates due to lack of efficacy and/or poor tolerability (6–8). In a retrospective claims analysis of 8707 patients in the USA, poor persistence (25%) was observed after the first 6 months of treatment, and it further declined to 14% by 12 months (9). In clinical practice, patients who tolerate and achieve a clinically meaningful response to treatment are likely to continue treatment, whereas those not achieving an adequate response or who experience adverse events are likely to discontinue treatment. Therefore, it is clinically relevant to understand the actual degree of benefit in patients achieving certain thresholds of response because those achieving benefit are more likely to remain on treatment.
Erenumab is a fully human monoclonal antibody that selectively targets and blocks the canonical calcitonin gene-related peptide receptor (10). It is approved as a preventive treatment for adults with migraine in the US and the EU (11,12). Patients with CM treated with erenumab (70 and 140 mg) demonstrated significant reductions in the primary endpoint of monthly migraine days (MMD) in a 12-week, pivotal, placebo-controlled study (NCT02066415) (baseline: 18.0 days; reduction of 6.6 days for both 70 and 140 mg versus 4.2 days for placebo, [both p < 0.001]). Compared with placebo, a greater proportion of patients treated with erenumab 70 and 140 mg achieved ≥50% reduction in MMD from baseline (secondary endpoint) (13), which is considered a clinically meaningful response (14,15).
Here, we report response rates at the ≥50%, ≥75% and 100% thresholds (as defined by relative reduction from baseline in MMD) as well as nonresponse (≤0%) from this study (13) and contextualize the actual benefit achieved in the subgroups of patients responding at the above thresholds.
Methods
Patient eligibility criteria
Patient eligibility criteria have been described previously (13). Briefly, men and women aged 18–65 years with a history of CM (≥15 headache days per month, of which ≥8 were migraine days; with or without aura) were included in the study. Patients with overuse of triptans, ergot derivatives, simple analgesics, and combinations of drugs (any combination of those above, combination analgesics, or opiates) were permitted to participate in the study (13).
Patients aged ≥50 years at migraine onset and a history of cluster headache or hemiplegic migraine, or CM with continuous pain (i.e. no pain-free periods of any duration during the 1 month before screening) were excluded. Patients with no therapeutic response to more than three categories of preventive treatment were also excluded.
Study design
This was an exploratory analysis of data from a multicenter, randomized, double-blind placebo-controlled study (NCT02066415) of patients with CM conducted in 69 headache and clinical research centres in North America (Canada and the USA) and Europe (Czech Republic, Denmark, Finland, Germany, Norway, Poland, Sweden, and the UK). The study comprised an initial screening phase (up to 3 weeks), baseline (4 weeks), a double-blind treatment phase (DBTP; 12 weeks), and a safety follow-up phase (12 weeks) (13). Eligible patients were randomly assigned (3:2:2) to receive placebo, erenumab 70 mg, and erenumab 140 mg, once every 4 weeks for the 12-week DBTP (13).
An independent ethics committee or relevant institutional review board for each study center approved the final study protocol. The study was conducted in accordance with International Conference on Harmonization Good Clinical Practice regulations/guidelines, and in accordance with the ethical principles set forth in the Declaration of Helsinki. All centers complied with local regulations. All patients provided written informed consent before study initiation. Summaries of data by unmasked treatment assignment were prepared by an independent biostatistician group and reviewed by an independent data monitoring committee.
Endpoints and assessments
Results for the primary endpoint, change from baseline in MMD to the last 4 weeks (month 3) of the 12-week DBTP, have been reported previously (13). The responder rates were derived from percentage change from baseline in MMD at each time point; that is, to month 1, month 2 or month 3 (primary time point).
Waterfall plot analysis of change in MMD from baseline
Waterfall plots were generated to show the change in MMD from baseline to Month 3 for individual patients in each treatment group (bars above baseline indicate worsening and bars below baseline indicate reduction in MMD [improvement]).
Responder rate analyses
The responder rate analyses included achievement of ≥50% (pre-specified secondary endpoint), ≥75% (post hoc analysis), and 100% (post hoc analysis) reduction in MMD from baseline to Month 3. In addition, the percentage of patients with no response to treatment, defined as no change or worsening of MMD, was assessed (post hoc analysis).
Efficacy and patient-reported outcomes in subgroups based on various thresholds of MMD response
A post hoc analysis was also conducted to contextualize the actual treatment benefit among patients achieving or not achieving response at the ≥50%, ≥75%, and 100% thresholds. Outcome measures assessed were change from baseline to month 3 in MMD, migraine-specific medication treatment days (MSMD), the Headache Impact Test (HIT-6) scores, and Migraine Disability Assessment (MIDAS) total scores.
Statistical analysis
The proportions of patients with ≥50%, ≥75%, and 100% reduction in MMD were calculated from baseline to month 3. Odds ratios (ORs) and associated p-values for each erenumab dose versus placebo were obtained from the Cochran-Mantel-Haenszel test using data including placebo and the corresponding erenumab dose group alone. Missing data was imputed using non-responder imputation (NRI). The efficacy analyses set included patients who received either placebo, erenumab 70 mg, or 140 mg and had at least one post-baseline monthly eDiary measurement. For ≥50% reduction from baseline in MMD, statistical significance was determined based on a pre-specified hierarchical gate-keeping procedure adjusted for multiplicity. Statistical significance for post hoc endpoints of ≥75% and 100% reduction in MMD were determined by comparing nominal p-values with a significance level of 0.05. Analysis of efficacy in responder subgroups at the ≥50%, ≥75%, and 100% thresholds was based on descriptive statistics alone.
Results
Patient disposition and baseline characteristics
Patient disposition and baseline characteristics have been reported previously (13). In summary, a total of 667 patients were randomized. The mean age of the patients was 42.1, 41.4, and 42.9 years in the placebo, erenumab 70 mg, and 140 mg treatment groups, respectively. The majority of patients were women (placebo: 79.0%, erenumab 70 mg: 86.9% and erenumab 140 mg: 84.2%). The mean (standard deviation [SD]) MMD at baseline was 18.2 (4.7), 17.9 (4.4), and 17.8 (4.7) days in the placebo, erenumab 70 mg, and 140 mg groups, respectively. The mean (SD) monthly acute migraine-specific medication use at baseline was 9.5 (7.6), 8.8 (7.2), and 9.7 (7.0) days in the placebo, erenumab 70 mg, and 140 mg groups, respectively. The proportion of patients who failed ≥1 prior preventive treatment due to lack of efficacy or poor tolerability were 69.9%, 66.5%, and 66.3% in the placebo, erenumab 70 mg, and 140 mg groups, respectively.
Waterfall plot analysis of change in MMD from baseline
In the overall DBTP population, a higher number of patients showed reduction in MMD from baseline in the erenumab 70 and 140 mg groups versus placebo (patients showing reduction in MMD: placebo, n = 191; erenumab 70 mg, n = 149; erenumab 140 mg, n = 144; patients with no change or worsening: placebo, 66; erenumab 70 mg, n = 26; erenumab 140 mg, n = 35; Figure 1).
Waterfall plot of change in MMD from baseline versus baseline MMD (efficacy analysis set), (a) placebo (b) erenumab 70 mg (c) erenumab 140 mg.
Spectrum of response
As presented in Figure 2, regardless of threshold used for the responder definition, a higher percentage of erenumab-treated patients achieved responses compared with patients receiving placebo. The proportion of patients achieving ≥50%, ≥75%, and 100% response is presented in Figure 2. Overall, 28.1%, 16.3%, and 20.9% of patients demonstrated no change or worsening in the placebo, erenumab 70 mg, and 140 mg groups, respectively.
Distribution of response rate, based on relative change from baseline in MMD at month 3.
≥50% responder rate
At month 3, 39.9% (n = 75/188) and 41.2% (n = 77/187) of patients treated with erenumab 70 mg and 140 mg, respectively, achieved ≥50% reduction from baseline in MMD compared with 23.5% (n = 66/281) treated with placebo. The odds of achieving ≥50% reduction in MMD from baseline to month 3 were significantly greater in patients treated with erenumab compared with placebo (erenumab 70 mg: OR 2.2, 95% confidence interval [CI]: 1.5–3.3, p < 0.001; erenumab 140 mg: OR 2.3, 95% CI: 1.6–3.5, p < 0.001; Figure 3) and this effect was observed from month 1 (week 4) onwards. The proportion of patients who achieved ≥50% response in one or more of the 3 monthly treatment periods in the placebo, erenumab 70 mg, and 140 mg groups were 30.6%, 57.4%, and 54.0%, respectively.
Proportion of patients with a ≥ 50% reduction from baseline in MMD.
≥75% responder rate
At month 3, 17.0% (n = 32/188) and 20.9% (n = 39/187) of patients treated with erenumab 70 mg and 140 mg, respectively, achieved ≥75% reduction from baseline in MMD compared with 7.8% (n = 22/281) of patients treated with placebo. The odds of achieving ≥75% reduction in MMD from baseline to month 3 were significantly greater with erenumab 70 and 140 mg compared with placebo (erenumab 70 mg: OR 2.4, 95% CI: 1.4–4.3, p = 0.002; erenumab 140 mg: OR 3.1, 95% CI: 1.8–5.5, p < 0.001; Figure 4). The proportion of patients who achieved ≥75% response in one or more of the 3 monthly treatment periods in the placebo, erenumab 70 mg, and 140 mg groups were 12.8%, 25.0%, and 27.3%, respectively.
Proportion of patients with a ≥ 75% reduction from baseline in MMD.
100% responder rate
Achievement of 100% reduction from baseline in MMD is a high bar for a CM population. The proportion of patients who achieved a 100% response at month 3 in the placebo, erenumab 70 mg, and 140 mg groups were 0.4% (n = 1/281), 4.3% (n = 8/188), and 2.7% (n = 5/187), respectively.
Efficacy and patient-reported outcomes in subgroups based on response at various thresholds
Among the ≥50% responders, the baseline MMD was 17.1 and 17.0 for erenumab 70 mg and erenumab 140 mg, respectively (compared with 18.0 and 17.8 for the overall erenumab 70 mg and erenumab 140 mg groups, respectively; Table 1). For the subgroup of patients with response at a ≥50% threshold, mean reductions from baseline of −12.2 (70 mg) and −12.5 (140 mg) MMD were achieved (Figure 5).
Mean change in MMD from baseline to month 3. Baseline characteristics of response subgroups. Data are mean (SD) unless indicated; please refer to Tepper et al. 2017 (13) for complete demographics and baseline characteristics of the double-blind treatment phase. Yes or No indicates response/no response at given threshold. Overall population represents all erenumab-treated population at respective doses. HIT-6: Headache Impact Test (higher score indicates worse outcomes); MIDAS: Migraine Disability Assessment (higher score indicates worse outcomes); MMD: monthly migraine days; MSMD: migraine-specific medication treatment days; N: total number of patients; SD: standard deviation.

Greater reductions in MSMD, HIT-6, and MIDAS total scores were observed for both doses of erenumab in the responder subgroups compared with the overall erenumab-treated population (Figures 6–8). For the subgroup of patients with a response at the ≥75% and 100% thresholds an even greater reduction in MMD was observed than in the overall erenumab-treated population (Figure 5). The change in MSMD was lower in a relatively small 100% response subgroup compared to patients who did not achieve 100% response (Figure 6), likely attributable to lower MMD and MSMD at baseline (Table 1). For the subgroups of patients who did not achieve a response at the ≥50%, ≥75%, or 100% thresholds, reductions in MMD were not greater than the overall erenumab-treated population.
Mean change in MSMD from baseline to month 3. Mean change in HIT-6 from baseline to month 3. Mean change in MIDAS total score.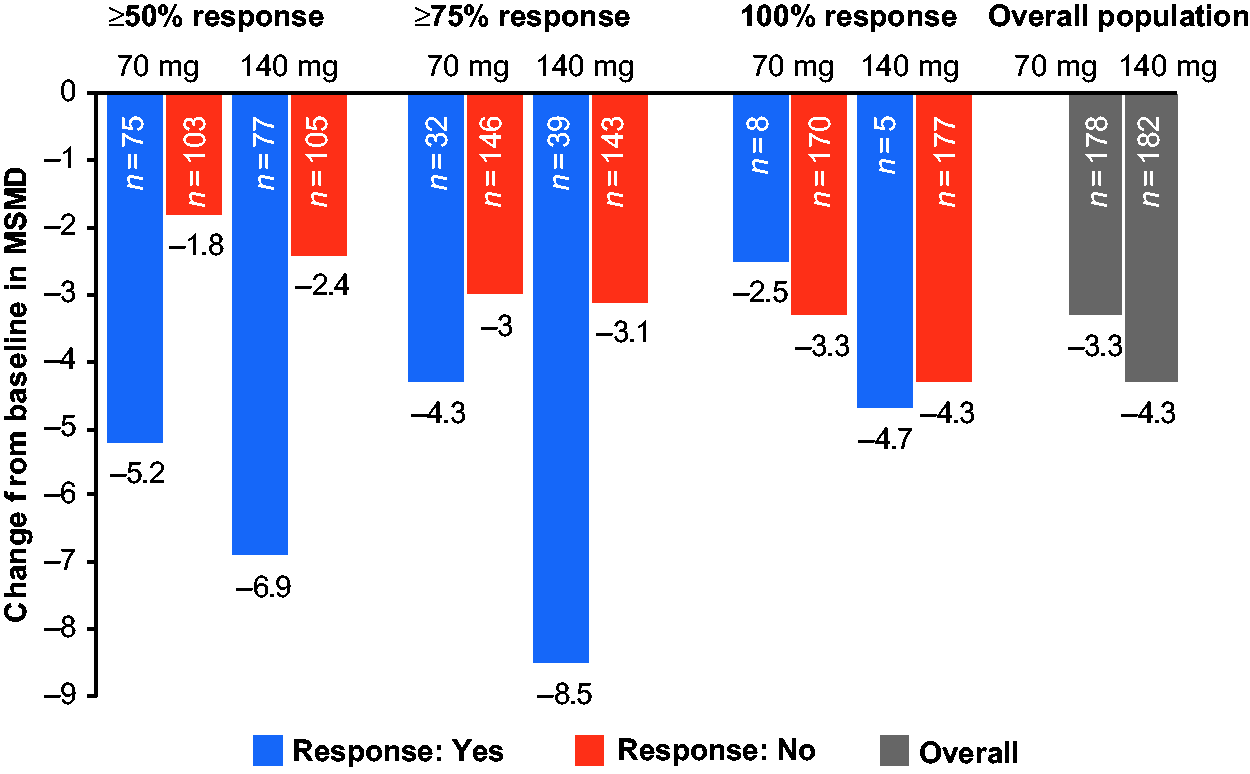


Discussion
In this post hoc analysis, erenumab showed greater efficacy in the response (≥50%, ≥75%, and 100% reduction from baseline in MMD) subgroups than in the overall-treated population. Both the spectrum of response across all patients and the overall greater reduction in MMD among patients treated with erenumab versus placebo are evident from the waterfall plots. The odds of achieving a response at any threshold was greater in patients treated with erenumab than placebo, and the odds increased for higher thresholds of response. These data complement the results of the primary endpoint, change in MMD at month 3, and underscore the greater magnitude of efficacy realized by patients who achieve 50% or greater response. Although these analyses focus on efficacy at month 3, significantly greater odds of achieving ≥50% and ≥75% reduction in MMD with both erenumab doses compared with placebo were evident from month 1 (week 4) onwards. Not all patients in the study experienced MMD reduction, and in fact ∼ 20% of patients across treatment groups did not demonstrate any improvement (change in MMD from baseline ≥0), a result that is consistent with other placebo-controlled migraine prevention trials. A challenge for future research is to identify predictors of response/nonresponse to a given preventive treatment.
In clinical trials, treatment effect is assessed by measuring differences in the overall study population based on treatment received. However, response to treatment varies, and not all patients experience the same level of benefit. Hence, an exploratory analysis to better understand the spectrum of treatment benefit among patients, assessing efficacy of erenumab based on different thresholds of response was performed. In this analysis, across all outcome measures at the ≥50% threshold, the magnitude of change was substantially better in responders than in the overall erenumab-treated population and in patients who did not achieve a ≥50% response. This highlights the rather wide spectrum of response to treatment and emphasises that mean results for an overall study population will underestimate the actual benefit achieved in the subgroup of patients who are deemed responders (e.g. using 50% as a commonly applied response threshold). Similarly, at the ≥75% and 100% thresholds, a greater effect was observed at both doses across all outcome measures in responders compared with the overall erenumab-treated population. In an additional analysis performed in subgroups of CM patients with prior prophylactic treatment failure (0, ≥1, ≥2 and ≥3) and medication overuse, erenumab 70 mg and 140 mg demonstrated greater efficacy than placebo (16,17). These results support that patients who respond to erenumab treatment based on the traditional MMD-related responder definition can show substantial treatment benefits on other clinical and functional outcomes. In clinical practice, patients achieving a sufficient response to treatment are more likely to continue the treatment. Hence, these findings are important from a clinical practice perspective as they describe the actual benefits among patients classified as responding to erenumab treatment.
A slight imbalance in baseline characteristics was observed, with lower baseline MMD and MSMD in patients achieving a response versus those not achieving a response at a given threshold. These differences may be attributable, at least in part, to the relatively small number of patients in the responder subgroups at the higher thresholds, particularly at the 100% threshold. Intuitively, achievement of a higher-level response would appear to be more likely in patients with a lower frequency of events, and this is indeed reflected in the baseline MMD for responders versus those who did not achieve response in both dose groups, with larger differences in baseline MMD at the ≥75% and 100% thresholds (Table 1). Similarly, baseline MSMD were generally lower in the responder subgroups, again suggesting that lower migraine frequency/severity in a CM population may be associated with a greater likelihood of a high-level response. Interestingly, baseline HIT-6 scores, a measure of the impact of headache on patients’ QoL, was comparable in the subgroups achieving/not achieving responses at a given threshold. Baseline MIDAS scores (a measure of headache-related disability) were higher among patients not achieving a response (more clearly seen at the ≥50% and ≥75% thresholds), suggesting that patients who were most disabled by their migraines, which may be linked to greater frequency of events, may be less likely to achieve a high level of response.
Achievement of ≥50% response is a generally accepted threshold in clinical trials and in clinical practice (14,15). The threshold of response that is deemed clinically meaningful, in terms of relative reduction in migraine days, is likely to be dependent on individual patient characteristics, such as baseline frequency, and likewise a clinically meaningful treatment effect may also depend on other outcomes, such as improvement in pain severity and reduced impact of migraine on functioning (14,15). Furthermore, the response thresholds used are arbitrary and dichotomous, and patients not achieving a given level of response may still experience a treatment benefit. Although there is a clear spectrum of response to treatment, currently, identification of predictors of response represents an important scientific gap. In this analysis, the response rate was assessed over 3 months; future studies should investigate responses over the longer term. This study included patients with medication overuse, history of preventive treatment failure, and comorbidities such as anxiety and depression. However, patients with daily headache, cluster headache, hemiplegic migraine and other comorbidities (e.g. fibromyalgia and poorly controlled hypertension) were excluded from this study and may limit the generalizability of the results to broader populations.
In conclusion, patients treated with erenumab had greater MMD response rates versus placebo at month 3, regardless of the threshold used to define response. Patients achieving ≥50%, ≥75%, and 100% responses based on change in MMD showed substantially greater reductions not only in migraine days, but also in the use of migraine-specific medication, headache impact, and disability as assessed by the HIT-6 and MIDAS scores, compared with the overall erenumab-treated population. These findings may help to provide context for setting realistic patient expectations regarding the spectrum of response that may be attained at the individual patient level during treatment with erenumab, and for those who respond to treatment, the greater therapeutic gain that may be achieved.
Clinical implications
This post-hoc analysis of a double-blind, placebo-controlled study of erenumab in patients with CM was conducted to contextualize the actual treatment benefit in subgroups of patients achieving specific response thresholds (≥50%, ≥75%, and 100% reduction in MMD from baseline). At month 3, a greater proportion of patients achieved ≥50% and ≥75% reduction in MMD with erenumab than with placebo. Compared with the overall erenumab-treated population, greater reductions in MMD and MSMD were observed amongst ≥50% and ≥75% responders versus those who did not achieve these response thresholds. These findings provide context for setting realistic expectations regarding actual treatment benefit experienced by patients responding to treatment.
Footnotes
Acknowledgements
We thank the study investigators and patients for their participation and commitment to this work. The medical writing support was provided by Dinesh T Makhija, Novartis Healthcare Pvt. Ltd, India.
Contributors
All authors participated in the study design, implementation, and/or conduct of the study. All authors contributed to the review of the protocol and approved the final manuscript.
Ethical approval and conduct
The study protocol was reviewed by the Independent Ethics Committee or Institutional Review Board for each center. The study was conducted according to the ethical principles of the Declaration of Helsinki.
Patient information and consent
Informed written consent was obtained from each subject before conducting any procedures. Additional information on the study was provided verbally by the study investigator or in a written format.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JLB has received research grants from Allergan, Teva, Amgen, Alder, Biohaven, Zosano, Colucid, and Eli Lilly and Company and has served on advisory boards/lecturer for Eli Lilly and Company, Teva, Amgen, Promius, Supernus, Valeant, and Avanir.
HCD has received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Alder, Allergan, Amgen, Autonomic Technology, Bristol-Myers Squibb, CoLucid, Electrocore, Ipsen, Lilly, Medtronic, MSD, Novartis, Pfizer, Schaper and Brümmer, Teva and Weber & Weber. Received financial support for research projects by Allergan, Electrocore, MSD and Pfizer. Headache research at the Department of Neurology in Essen is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union. He has no ownership interest and does not own stocks of any pharmaceutical company. He serves on the editorial boards of Cephalalgia and Lancet Neurology. He chairs the Clinical Guidelines Committee of the German Society of Neurology and is a member of the Clinical Trials Committee of the IHS.
DD has received consulting fees, and speaking and/or teaching fees from Allergan, Amgen, Biogen Idec, Novartis, Bayer, and Teva.
MCF has served on advisory boards and/or has consulted or received research support from Alder, Allergan, Avani, Dr. Reddy’s Laboratories, Eli Lilly, Scion, and Teva.
PJM has received consulting fee from Alder and research support from Amgen, Biohaven, Eli Lilly, and Teva.
PW is an Investigator in clinical trials sponsored by Teva, Amgen, Genentech, Novartis, Allergan, AstraZeneca, Biogen Idec, Ipsen and Lilly, has participated in advisory boards for Teva, Amgen, Avanir, Novartis, Allergan, Supernus and Lilly, and has been on a speaker’s bureau for Allergan, Amgen, Avanir, Lilly, Promius, Novartis, and Supernus.
JK, SW and SR are full-time employees of Novartis. SC, FZ, RAL, and DDM are full-time employees of Amgen.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Amgen, Thousand Oaks, CA, USA, and Novartis Pharma AG, Basel, Switzerland.
