Abstract
Background
Real-world data on the effectiveness of calcitonin gene-related peptide monoclonal antibodies (CGRP mAbs) in migraine patients are needed.
Methods
We performed a single-center, real-world study with an observation period of up to 12 months (mean 7.5 ± 3.4 months) after CGRP mAb administration. A total of 228 Japanese patients with episodic or chronic migraine (age, 45.9 ± 13.2 years; 184F; 45 erenumab; 60 galcanezumab; 123 fremanezumab) who were treated with CGRP mAbs for at least three months were ultimately included in this study.
Results
In the total cohort, after CGRP mAb treatment, mean monthly migraine days decreased by 7.2 ± 4.8, 8.3 ± 4.7, and 9.5 ± 5.0 at three, six and 12 months, respectively. The ≥50% monthly migraine day reduction rates at three, six and 12 months were 48.2%, 61.0% and 73.7%, respectively. In the logistic regression analysis, the presence of osmophobia and fewer baseline monthly migraine days contributed to ≥50% responders at three, six and 12 months. The ≥50% responders at three or six months were useful in predicting ≥50% responders at 12 months. In subgroups of patients with difficult-to-treat migraine (those with medication overuse headache or psychiatric comorbidities) and previous CGRP mAb users, monthly migraine days were substantially reduced over 12 months. There was no difference in monthly migraine day reduction over 12 months among three different CGRP mAbs. Adverse reactions were observed in 28 (12.3%) patients, with injection site reactions being the most common (n = 22) though generally mild in severity.
Conclusion
This real-world study confirmed the efficacy and safety of three different CGRP mAbs for prophylactic treatment of patients with migraine.
Introduction
Migraine, characterized by moderate to severe recurrent pulsating headaches accompanied by nausea, photophobia, phonophobia and osmophobia (1), is a neurological disorder that affects more than one billion people worldwide with a prevalence of 15% per year (2). Among various neurological disorders, migraine has been reported to be the one with the greatest number of years lived with disability in a wide range of age groups (3). The important discovery that migraine attacks involve the release from trigeminal nerve endings of calcitonin gene-related peptide (CGRP), which has vasodilatory properties, has led to the development of disease-specific therapeutic agents targeting CGRP, such as CGRP monoclonal antibodies (mAbs), which include erenumab, fremanezumab, galcanezumab, and eptinezumab (4). Currently, in Japan, three CGRP monoclonal antibodies, erenumab, fremanezumab, and galcanezumab, are licensed for migraine and can be used in clinical practice. Randomized clinical trials (RCTs) have provided reliable evidence regarding the efficacy, safety, and tolerability of CGRP mAbs in treating patients with migraine (5–10). In addition, RCTs of CGRP mAbs (erenumab, fremanezumab, and galcanezumab) performed among patients with migraine in Japan (11–13) showed comparable efficacy and safety compared with global RCTs conducted in the USA and Europe (8–10). However, real-world data on the effectiveness of CGRP mAbs for patients with various clinical backgrounds and comorbid conditions are still warranted. Additionally, only a few studies have addressed the variations in efficacy between different types of CGRP mAbs (14,15).
We aimed to evaluate the clinical efficacy and safety of CGRP mAbs, to determine the contributing factors to ≥50% responders at one, three, six and 12 months, and finally to evaluate the relative efficacy of three different types of CGRP mAbs in a real clinical single-center observational setting.
Methods
Study design
This is a retrospective, observational, single-center, real-world study with an observation period of up to 12 months after CGRP mAb administration. The Institutional Review Board of Dokkyo Medical University approved this study. In accordance with the Declaration of Helsinki, all participating patients were informed about this observational study in an outpatient setting and had the opportunity to decline participation in the study. Based on the retrospective and observational nature of this study, our Institutional Review Board waived the requirement for patient signatures on the informed consent forms.
Patients
Clinical records of 236 migraine patients aged 18 years and older (age, 45.9 ± 13.2 years; 190F) who attended our headache outpatient clinic from April 2022 to March 2023 and were administered CGRP mAbs (erenumab, galcanezumab or fremanezumab) were reviewed. After excluding incomplete data (n = 2) and a follow-up period <3 months after CGRP mAb administration (n = 6), we finally included 228 patients with migraine (age, 45.9 ± 13.2 years; 184F; 45 erenumab; 60 galcanezumab; 123 fremanezumab) in this study (Figure 1). The mean follow-up period was 7.5 ± 3.4 months. Sixty patients treated with galcanezumab received an initial loading dose of 240 mg, followed by monthly doses of 120 mg. Forty-five patients treated with erenumab consistently received a monthly dose of 70 mg. For fremanezumab, 73 patients were treated with monthly (225 mg) doses, and 50 patients were treated with quarterly (675 mg) doses. At 12 months, 59 patients were included in the sensitivity analysis. Seventy patients had been previously treated with CGRP mAb, and 158 patients were naïve to CGRP mAbs.
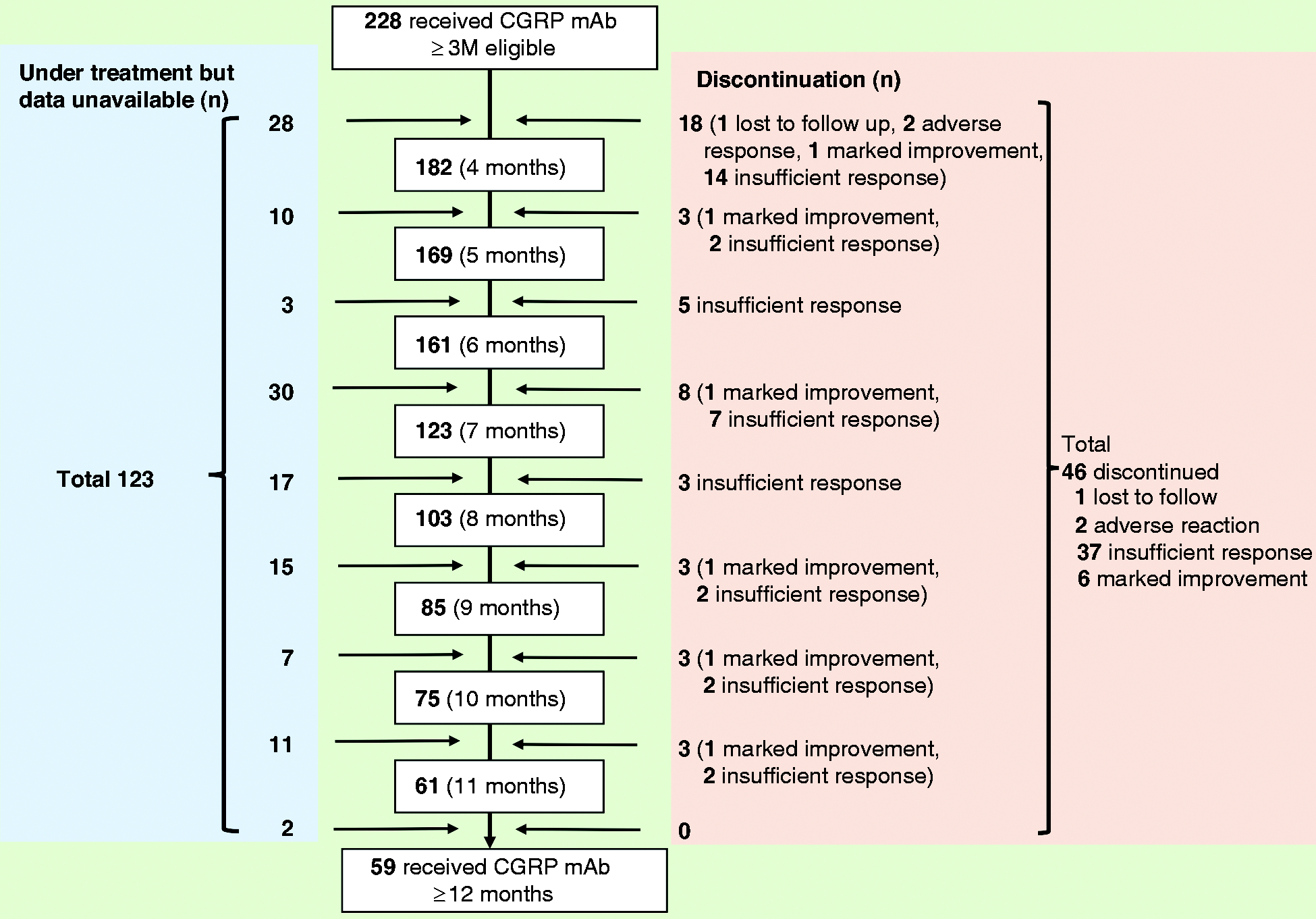
Study flowchart.
Diagnosis of migraine
All patients met the diagnostic criteria of the International Classification of Headache Disorders 3rd edition (ICHD-3) for migraine with and without aura (16). Chronic migraine (CM) was defined as headache occurring 15 or more days per month for at least three months, of which at least eight per month exhibited migraine features; episodic migraine (EM) was defined as headache occurring four to 14 days per month. Medication overuse headache (MOH) was diagnosed according to the ICHD-3 criteria (16).
Clinical evaluation
The number of monthly migraine days (MMDs) before and one to 12 months after CGRP mAb treatment was obtained from paper headache diaries. The patients were instructed to keep a daily headache diary. A reduction of <30%, 30% to <50%, 50% to <75%, and 75% to 100% in MMDs was calculated based on headache diaries. Clinical characteristics, including the duration of migraine, comorbidities, previous prophylactic medications used, previous use of CGRP mAbs, presence or absence of aura and accompanying symptoms such as photophobia, phonophobia, osmophobia, nausea and allodynia, MOH, body mass index, and migraine characteristics (pain location and type), were obtained from clinical records. Clinical characteristics between the nonresponders (<50% MMD reduction) and responders (≥50% MMD reduction) were compared at months 1, 3, 6, and 12. Adverse reactions were also collected from clinical records. A subanalysis was also conducted on the effect of CGRP mAbs in patients with daily headache (no day without headache at baseline), prior use of CGRP mAbs (those receiving second or third types of CGRP mAbs with insufficient response to the first or second types of CGRP mAb treatment) and difficult-to-treat (DTT) migraine (those with MOH or psychiatric comorbidities) (17).
Statistical analysis
Sample size calculations were not performed because of the real-world, observational setting of this study. For two-group comparisons, the Mann–Whitney U test or Student's t test was used for continuous variables as appropriate, and the chi-square test was used for categorical variables. A generalized linear mixed-effects model (GLMM) followed by Bonferroni's post hoc test was applied to assess whether the MMDs were significantly different from baseline to each month following CGRP mAb treatment. The efficacy of three different CGRP mAbs (erenumab, galcanezumab and fremanezumab) on MMD reduction was analyzed by two-way ANOVA using GLMM followed by a global test. Subanalysis was performed only for 158 patients naïve to CGRP mAbs. Each variable was compared between nonresponders and responders at one, three, six and 12 months. Logistic regression analysis was performed to determine contributing factors for ≥50% responders at one, three, six and 12 months using the likelihood ratio forward selection method with clinical factors (Table 1) as independent variables; age and sex were corrected for by the forced entry method. The prediction of ≥50% responders at month 12 using ≥50% responders from month 1 to 3 was analyzed by a multiple logistic regression model after adjustment for sex and age. We also performed sensitivity analysis for 59 patients who completed 12 months of follow-up. Two-tailed p < 0.05 was considered indicative of statistical significance. IBM SPSS Statistics version 28 (IBM SPSS, Tokyo, Japan) was used for all statistical analyses. GraphPad Prism for Mac (Version 8; GraphPad Software, San Diego, USA) and Microsoft Excel version 16.18 were used to create figures.
Clinical factors related to responders at one, three, six and 12 months.

CGRP mAbs: calcitonin gene-related peptide monoclonal antibodies; M: month; MOH: medication overuse headache; MMDs: monthly migraine days.
Significant differences in p values are indicated in bold.
Results
Baseline characteristics
Among the 228 patients in this study, 46 discontinued treatment (20.2%) during the entire study period, which included a loss to follow-up (n = 1, 0.4%), insufficient responses (n = 37, 16.2%), marked improvements (n = 6, 2.6%) and adverse responses (n = 2, 0.9%) (Figure 1). The data at 12 months were available for 59 patients. The baseline characteristics of 228 patients with migraine are shown in Table 2. The EM group had no MOH complications, and 42.1% of the CM group had MOH. Aura was more common in the EM than the CM group. Regarding pain characteristics, patients in the CM group showed less pulsating and more pressing pain than patients in the EM group. Pain characteristics did not differ between the EM and CM groups. Baseline MMDs and the number of preventive medications used previously were greater and prior use of CGRP mAb and comorbidities were more frequent in the CM group than in the EM group. Migraine with aura was associated with a higher rate of EM (51.3% vs 33.3%) and fewer baseline MMDs (15.8 ± 6.7 vs. 18.5 ± 6.7) compared to migraine without aura. Other clinical parameters did not differ between the two groups (Online Supplementary Table 1).
Baseline characteristics of patients with migraine.
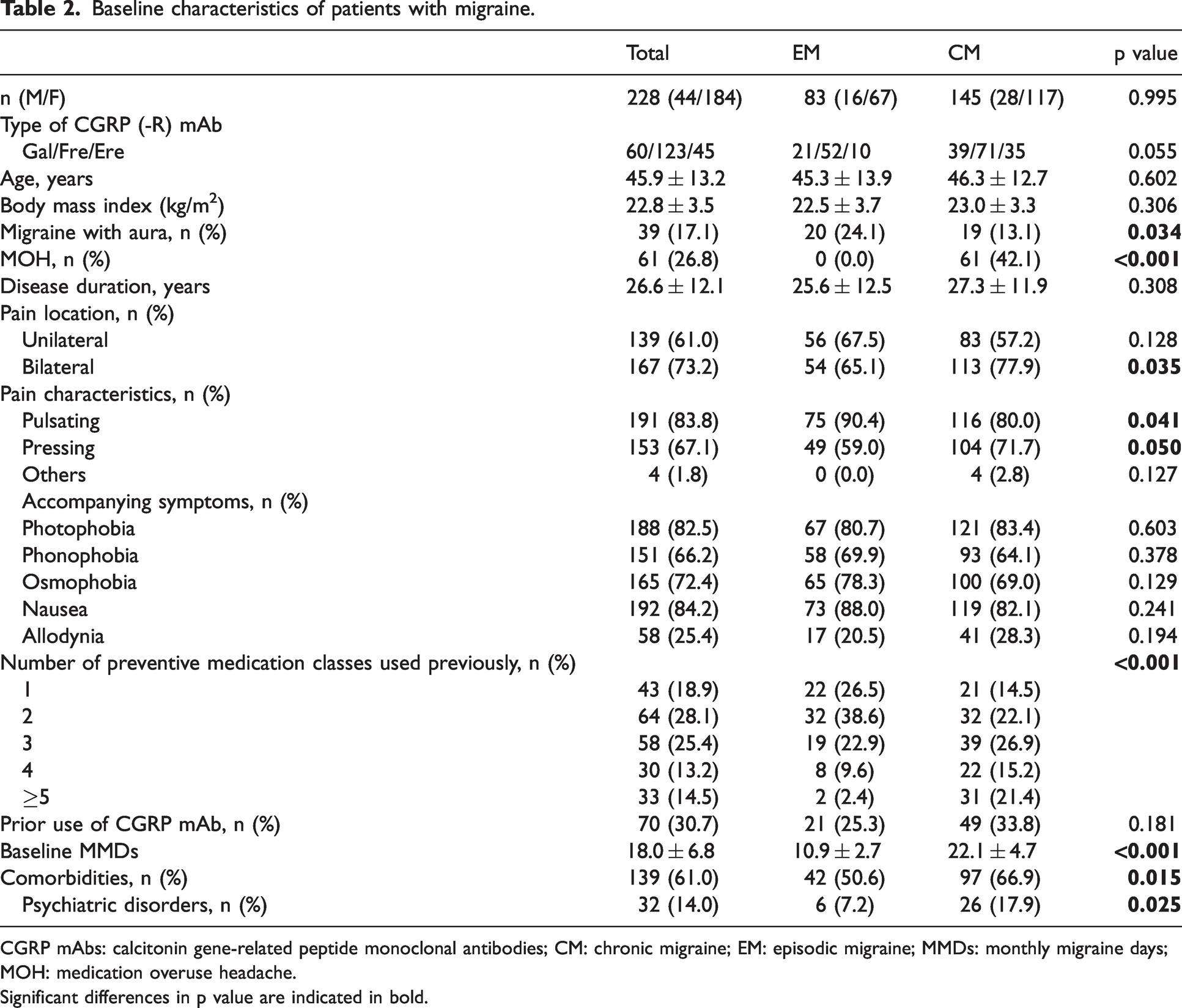
CGRP mAbs: calcitonin gene-related peptide monoclonal antibodies; CM: chronic migraine; EM: episodic migraine; MMDs: monthly migraine days; MOH: medication overuse headache.
Significant differences in p value are indicated in bold.
Efficacy
Figure 2 shows changes in MMDs from baseline. Significant mean MMD reductions from baseline to each time over 12 months was observed in the total, EM and CM cohorts using GLMM followed by Bonferroni's post hoc test. In the total cohort, the mean change in MMDs was −5.7 ± 4.6 at month 1, −7.2 ± 4.8 at month 3, −8.3 ± 4.7 at month 6, and −9.5 ± 5.0 at month 12. In the EM group, the mean MMD change was −5.4 ± 2.9 at month 1, −6.0 ± 3.0 at month 3, −6.4 ± 2.5 at month 6, and −6.7 ± 2.6 at month 12. In the CM group, the mean MMD change was −5.9 ± 5.3 at month 1, −7.8 ± 5.5 at month 3, −9.5 ± 5.4 at month 6, and −11.5 ± 5.3 at month 12. The sensitivity analysis that included 59 patients who completed 12 months of treatment was performed using a GLMM followed by Bonferroni’s post hoc test. The results showed significant mean MMD reductions from baseline to each time point over 12 months (Online Supplementary Figure 1).
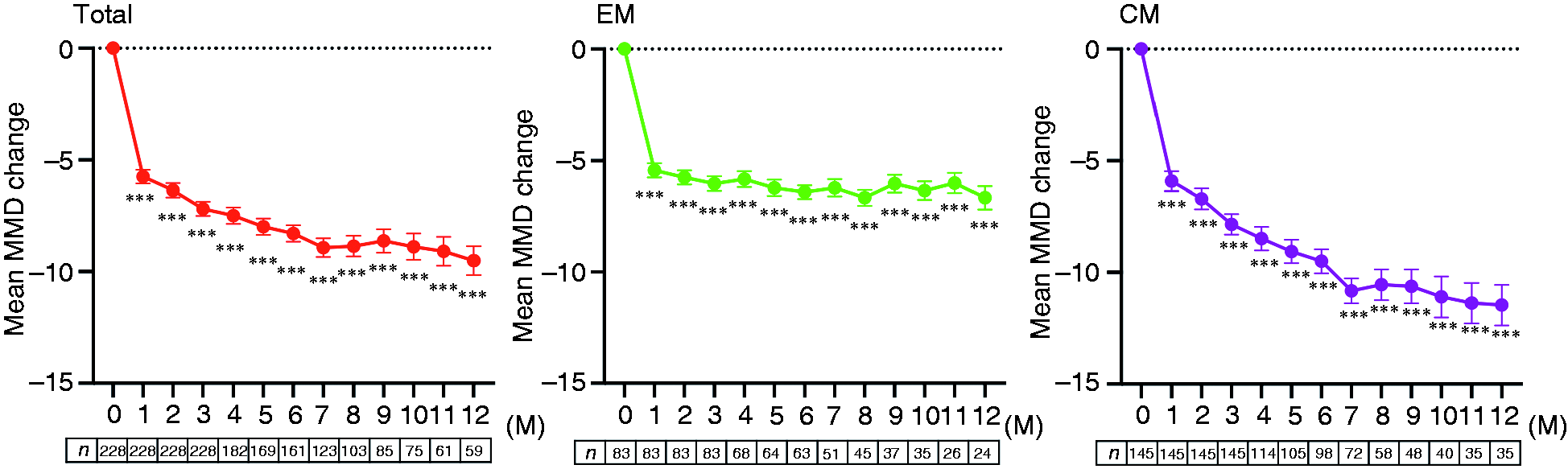
Changes in monthly migraine days from baseline after CGRP mAb administration.
The proportion of responders at each time point is shown in Figure 3. In the total cohort, the ≥50% response rates at one, three, six and 12 months were 36.0%, 48.2%, 61.0% and 73.7%, respectively. In the EM group, the ≥50% response rates at one, three, six and 12 months were 61.4%, 66.3%, 81.0% and 91.7%, respectively. In the CM group, the ≥50% response rates at one, three, six and 12 months were 22.1%, 39.3%, 49.0% and 62.9%, respectively. The results from the sensitivity analysis of 59 patients regarding the proportion of responders by time trend after CGRP mAb administration were also comparable to the total cohort results (Online Supplementary Figure 2).

Proportion of responders by time trend after CGRP mAb administration.
Efficacy of three different types of CGRP mAbs
The effects of three different types of CGRP mAbs on MMD reduction were analyzed by two-way ANOVA using GLMM. Figure 4 shows mean changes from baseline in MMDs in patients receiving erenumab, galcanezumab or fremanezumab. There was a significant difference in time (F = 102.7, p < 0.001), but there was no difference between CGRP mAb type (F = 1.889, p = 0.154) or interaction for medication type by time (F = 0.733, p = 0.822) (Figure 4a). We also performed a subanalysis including only 158 patients who were naïve to CGRP mAb. The results showed that there was a significant difference in time (F = 72.94, p < 0.001), but there was no difference between CGRP mAb type (F = 0.013, p = 0.987) or interaction for the medication type by time (F = 0.459, p = 0.989) (Figure 4b). In addition, mean changes from baseline in MMDs among 59 patients who completed 12 months of follow-up were not significantly different among the three groups (Online Supplementary Figure 3).
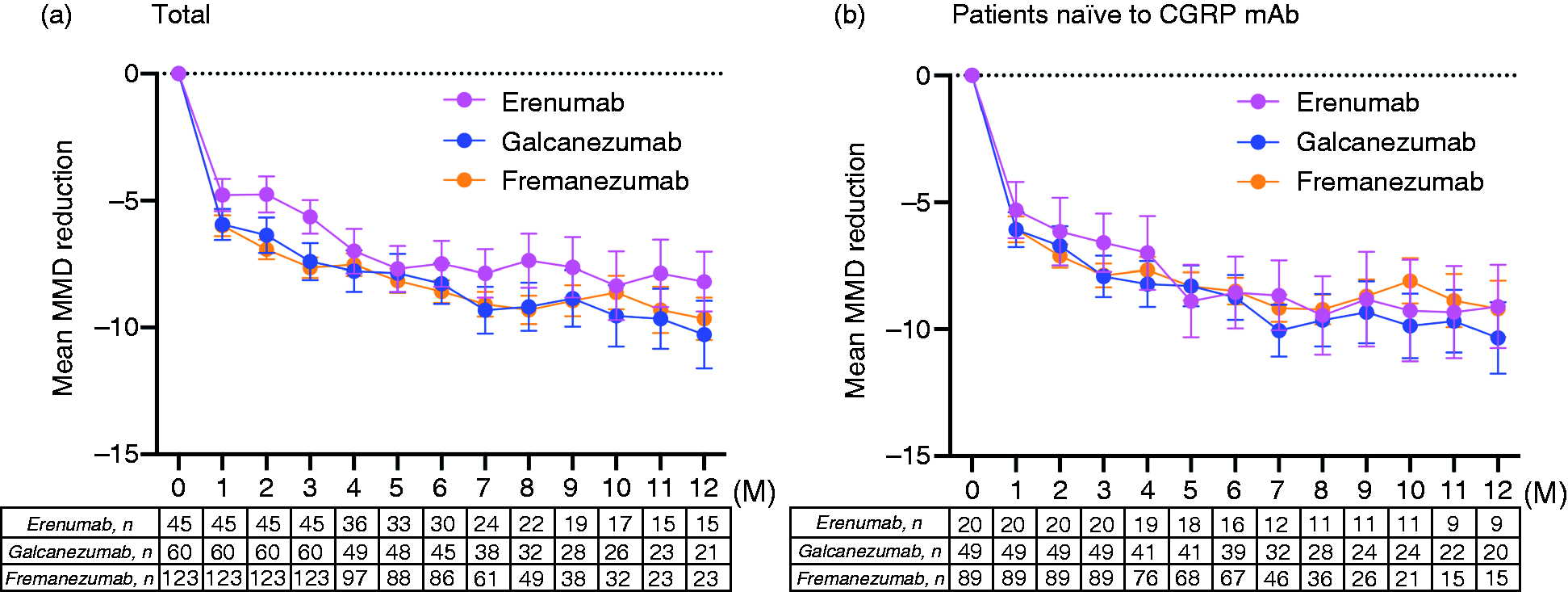
Mean changes from baseline in monthly migraine days in patients receiving different CGRP mAb. (a) Total and (b) Patients naïve to CGRP mAb
Clinical factors contributing to ≥50% responders
To determine the clinical factors contributing to the ≥50% response, baseline clinical characteristics were compared between nonresponders and responders at one, three, six and 12 months (Table 1). At one month, compared with nonresponders, responders had less MOH, longer disease duration, more unilateral localization, more pulsatile nature, fewer previous prophylactics used, and fewer baseline MMDs. Additionally, osmophobia tended to be higher in responders. At three, six and 12 months, responders showed less MOH comorbidity, more unilateral localization, more pulsatile nature and fewer baseline MMDs than nonresponders. Less prior use of CGRP mAbs and fewer previous prophylactics used were related to responders at three and six months. In the logistic regression analysis of responders using likelihood ratio forward selection, fewer baseline MMDs, longer disease duration and a smaller number of preventive medication classes used previously were related to responders at one month (Table 3). The presence of osmophobia, fewer baseline MMDs and fewer previous prophylactics used were related to responders at three months. At six and 12 months, the presence of osmophobia and fewer baseline MMDs contributed to responders.
Logistic regression analysis of responders at one, three, six and 12 months.

Using likelihood ratio forward selection. aOR, odds ratio adjusted for sex and age; 95% CI, 95% confidence interval.
Relationship between ≥50% responders at one, three and six months and ≥50% responders at 12 months
A separate analysis of 59 patients to assess whether responders at one, three and six months could predict responders at 12 months was performed with a multiple logistic model after adjustment for sex and age (Table 4). The results showed that ≥50% responders at three months (72.7% sensitivity and 73.3% specificity) and six months (86.4% sensitivity and 80.0% specificity) predicted ≥50% responders at 12 months. Persistent responders at three months, defined as a continuous ≥50% reduction in MMDs from month 1 to 3, also contributed to predictors of ≥50% responders at 12 months with a high specificity of 93.3% but had low sensitivity (38.6%). Using at least one ≥50% response rate achieved in any month 1–3 was also a predictor of a ≥50% response rate at 12 months (79.5% sensitivity and 60.0% specificity). By contrast, a ≥50% response at one month was not a significant predictor for 50% responders at 12 months.
Prediction of ≥50% responders at month 12 using ≥50% responders from month 1 to 3.

≥50% 1 M, ≥50% reduction in MMDs at month 1; ≥50% any 1–3 M, at least one ≥50% reduction in MMDs from month 1 to 3
≥50% 3 M, ≥50% reduction in MMDs at month 3; ≥50% persistent 1–3 M, continuous ≥50% reduction in MMDs from month 1 to 3
≥50% 6 M, ≥50% reduction in MMDs at month 6; cOR, crude odds ratio, aOR, odds ratio adjusted for sex and age; 95% CI, 95% confidence interval.
Significant differences in p values are indicated in bold.
Efficacy in subgroups of patients with DTT migraine, prior use of CGRP mAbs and daily headache
There were 80 (35.1%) patients with DTT migraine and 40 (17.5%) patients with daily headache. Seventy patients (30.7%) had prior CGRP mAb treatment: eight patients were switched to a third CGRP mAb, and 72 were switched to a second CGRP mAb because of insufficient efficacy. Among patients with DTT migraine and prior use of CGRP mAbs, significant changes in MMDs from baseline to one to 12 months were observed using a GLMM followed by Bonferroni's post hoc test (Figure 5). In the daily headache group, significant MMD changes from baseline were observed at one to seven months. Figure 6 shows a proportion of responders by time trend after CGRP mAb administration. Among patients with DTT migraine and past CGRP mAb users, the proportions of ≥50% responders at three, six and 12 months were 31–36%, 44–46% and 60%, respectively. In the daily headache group, the proportions of ≥50% responders at three, six and 12 months were 12.5%, 31.6% and 33.3%, respectively.

Changes in monthly migraine days from baseline after CGRP mAb administration in subgroups of difficult-to-treat migraine, prior CGRP mAb use and daily headaches. (a) Difficult-to-treat migraine (n = 80). (b) Prior CGRP mAb use (n = 70) and (c) Daily headaches (n = 40).
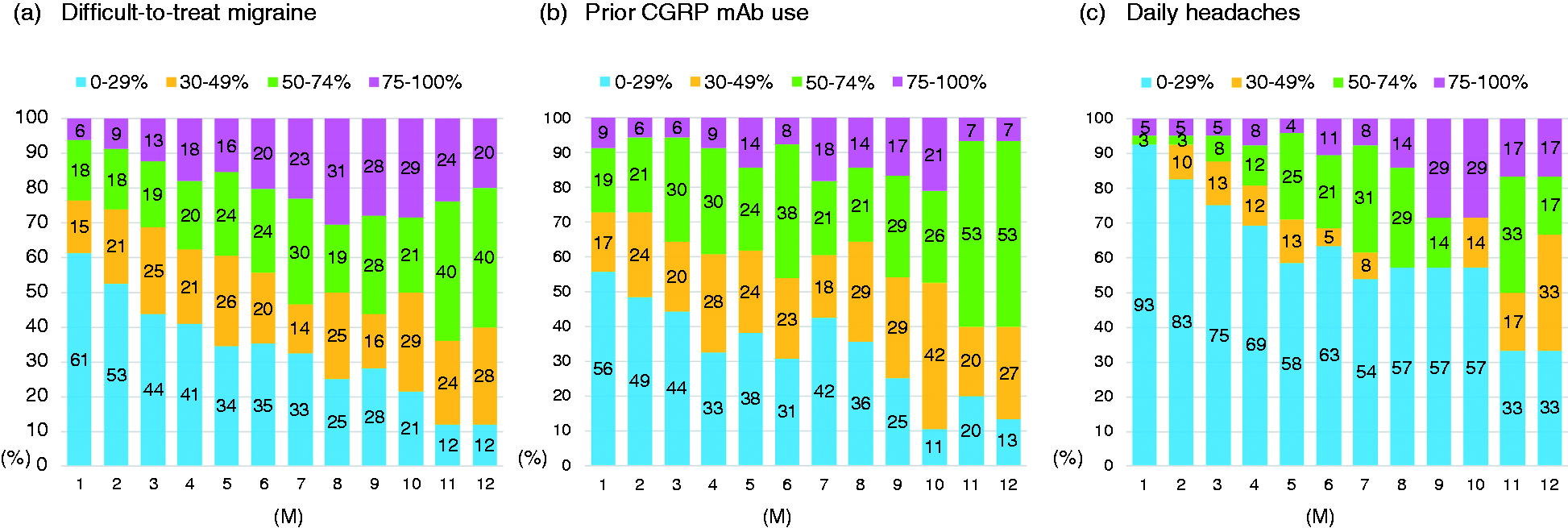
Proportion of responders by time trend after CGRP mAb administration in subgroups of difficult-to-treat migraine, prior CGRP mAb use and daily headaches. (a) Difficult-to-treat migraine (n = 80). (b) Prior CGRP mAb use (n = 70) and (c) Daily headaches (n = 40).
Adverse reactions
Adverse reactions were observed in 28 (12.3%) patients. Injection site reaction was the most common adverse reaction (n = 22, 9.6%). Constipation occurred in six patients (2.6%): four who were taking erenumab, one who was taking galcanezumab and one who was taking fremanezumab. Two patients (0.9%) discontinued use due to adverse reactions. In the other patients, adverse reactions were generally mild, requiring no additional treatment or procedures.
Discussion
We evaluated the efficacy of CGRP mAbs in migraine patients in this single-center, real-world study over a period of up to 12 months. The main findings of our study were as follows: 1) CGRP mAbs significantly reduced MMDs compared to baseline over 12 months in the overall, EM, and CM cohorts; 2) the overall ≥50% response rate was clinically sufficient, being 48.2% at three months, 61.0% at six months, and 73.7% at 12 months; 3) the presence of osmophobia and fewer baseline MMDs were related to responders at three, six and 12 months; 4) the three types of CGRP mAbs were comparably effective in reducing MMDs; 5) adverse reactions were generally low in frequency and mild in severity; 6) the ≥50% responders at three or six months could predict the ≥50% responders at 12 months; and 7) CGRP mAbs also showed significant improvement in subgroups of DTT migraine patients and past CGRP mAb users. The effect of CGRP mAbs on daily headache patients seemed to be inferior to that of the overall cohort, although the sample size was small.
In our study, among three CGRP mAbs, fremanezumab was the most frequently used. In Japan, self-injection of CGRP mAbs at home has only recently been approved. Therefore, patients who did not prefer frequent hospital visits or injections likely chose to use fremanezumab, which can be administered quarterly. In fact, 50 patients in this study received quarterly dosing of fremanezumab. Our study included past users of CGRP mAb to approximate the real-world clinical situation. On the other hand, botulinum toxin was not approved for migraine in Japan, so no patient used it.
Previous real-world studies evaluating two or more types of CGRP mAbs with three to six-month follow-up showed ≥50% response rates of 48.5–56.3% at three months (14,18–20) and 35.4–64.9% at six months (14,18,20). These response rates were comparable to those in the present study. Few real-world studies have evaluated the effects of two or more CGRP mAbs over 12 months. The ≥50% response rate at 12 months in our study was 73.7%, which was higher than the ≥50% response rate of 50% at 12 months in the real-world study that looked at the effect of three types of CGRP mAbs over one year (21). In our study, changes in MMDs from baseline to three, six and 12 months, especially in CM patients, were comparable to those in a study consisting of 203 CM patients (21). Our study from Japan showed a similar efficacy of CGRP mAbs to that in real-world studies from Europe and the USA (14,18,20,21).
We assessed which clinical factors could contribute to ≥50% responders at one, three, six and 12 months. We found that the smaller number of preventive medication classes used previously was related to responders at one and three months. Additionally, the presence of osmophobia and low baseline MMDs were related to responders at three, six and 12 months. This result is supported by the fact that the efficacy of CGRP mAbs in the daily headache group appeared to be lower than that in the overall cohort. In a multicenter study consisting of 864 patients with high-frequency EM or CM who received three types of CGRP mAbs for six months, it was found that the presence of unilateral pain, unilateral cranial autonomic symptoms and allodynia were predictors of responders (18). In a study of 260 patients with migraine receiving CGRP mAbs, typical migraine characteristics such as unilateral localization, pulsating character, photophobia or nausea were associated with ≥75% responders at three months, while CM, MOH, and concomitant depression were related to nonresponders (22). Older age, lower MMDs and fewer previous treatment failures were also associated with >50% responders at three months in a real-world study (19). Similar to previous studies, unilateral localization, pulsatile nature (18,22), less comorbid MOH (22), fewer prophylactics used previously, and fewer baseline MMDs (19) were related to responders in our study. Regarding the effect of aura on clinical efficacy, in logistic regression analysis, the presence of aura did not remain a predictor of responders, but it was associated with ≥50% responders at six and 12 months. This could have been due to the possibility of a higher percentage of EM and fewer baseline MMDs among patients with migraine with aura.
Unexpectedly, we found that osmophobia was a predictor of ≥50% responders at three, six and 12 months, a finding not previously reported. However, other sensory hypersensitivity symptoms or accompanying symptoms were not significant predictors for ≥50% responders. Although the diagnostic criteria for migraine in the latest ICHD-3 do not include osmophobia among migraine-specific features, its presence may be useful in the diagnosis of migraine and predicting headache severity (23). As in previous studies, a relatively high rate of osmophobia (68.3%), possibly related to interracial differences between Asian and Western populations (24), was observed in this study. In migraine patients with osmophobia during intermittent periods of headache attacks, higher cerebral blood flow was observed in the left piriform cortex and anterior superior temporal gyrus in both olfactory stimulation and nonolfactory stimulation conditions compared to controls (25). Stankewitz and May (26) found activation of the network of olfactory-related brain areas, including the amygdala, insular and piriform cortices and the rostral pons, following olfactory stimulation in interictal migraine patients. Results from another study utilizing positron emission tomography demonstrated that 5-HT1A receptor availability was increased in the pontine raphe during odor-triggered migraine attacks (27). These observations may suggest an association between olfactory-related brain regions and the trigeminal vascular systems of the pons in the pathogenesis of migraine.
Central or peripheral CGRP levels increase in response to central (repeated cortical spreading depression) or peripheral signals (changes in the trigeminal vascular systems), resulting in peripheral and central sensitization, leading to sensory hypersensitivity, including osmophobia (28). The trigeminal nerves expressing CGRP project into the olfactory bulb, and olfactory sensory neurons, which project axons to the olfactory bulb, express CGRP receptors (29). However, complicatedly, CGRP released from trigeminal sensory fibers in response to irritable stimulation inhibits the odor response of olfactory receptor neurons (29). Thus, the role of CGRP in osmophobia remains relatively undetermined.
In a real-world prospective observational study including 238 migraine patients receiving galcanezumab, the absence of any accompanying symptoms, such as nausea, vomiting, and photo/phonophobia, predicted nonresponders at three months. Since gastrointestinal symptoms, including nausea, may be attributable to increased CGRP (30) and photophobia induced by CGRP administration was attenuated by triptans in a mouse model (31), the development of these accompanying symptoms may be related to the CGRP pathway. Additionally, according to the results of a real-world study, the presence of cutaneous allodynia could be a predictor of responsiveness to erenumab (32). In 113 migraineurs, 38.1% had preictal olfactory hypersensitivity, which is associated with longer duration of illness and migraine-related burden, suggesting that osmophobia may be associated with increasing central sensitization in the progression of migraine (33). Taken together, the presence of osmophobia in our study may be an important non-headache symptom predictive of response to CGRP mAb treatment, possibly reflecting the involvement of the CGRP pathway and central sensitization.
An association of osmophobia with depression, anxiety (34) and headache severity (35) in patients with migraine has been described. Differences in clinical characteristics accompanying osmophobia may contribute to the finding that osmophobia is a predictor of favorable CGRP mAb responsiveness in this study. The responder group at months 3, 6, and 12, in which patients exhibited more osmophobia, showed greater baseline MMDs and MOH comorbidity. However, osmophobia was involved in the responders, while increased baseline MMDs and greater comorbid MOH were involved in the nonresponders. Delussi et al. (35) found that olfactory hypersensitivity was related to migraine severity at baseline but was unrelated to drug efficacy at follow-up. Further studies are needed on the association between CGRP mAb responsiveness and osmophobia.
A network meta-analysis of RCTs including 3052 migraine patients showed that CGRP mAbs (galcanezumab, eptinezumab, and fremanezumab) were superior to CGRP receptor mAbs (erenumab) in reducing MMDs (36). In a real-world setting, Schiano et al. (20) reported that the number of 50%-100% responders in general was similar between the CGRP mAb and CGRP-receptor mAb groups; however, ≥75 to 100% responders were more prevalent in patients treated with CGRP mAb (galcanezumab and fremanezumab) compared to those treated with CGRP-receptor mAb (erenumab). In contrast, our study did not show a difference in MMD reduction at any time point in the total cohort using two-way ANOVA with the GLMM. Quintana et al. (15) analyzed 123 migraine patients (56 with erenumab, 29 with fremanezumab, and 38 with galcanezumab) who received CGRP mAbs for at least six months. The results showed similar efficacy (MMD reduction from baseline by 14 to 17 days) for all three CGRP mAbs, but a description of the statistics regarding between-group comparisons was not found. Unlike our study, several previous real-world studies examining the efficacy of CGRP mAbs have excluded patients with a history of CGRP mAb use (18,19,37). Because the efficacy of the three CGRP mAbs may be affected in patients with a history of CGRP mAb use when refractory patients are included, we also performed a subanalysis of the effect of the three CGRP mAbs only in CGRP mAb-naïve patients. The subanalysis results showed that the difference in efficacy of the three CGRP mAbs was further diminished, demonstrating equivalent efficacy of the three different CGRP mAbs.
The effectiveness of a single CGRP mAb or CGRP receptor mAb is well described in RCTs conducted in very select populations because the trials have many inclusion and exclusion criteria and exclude patients with comorbidities, which may compromise their generalizability to the general population affected by the disease (38). Therefore, these RCTs are not representative of the actual overall population; the results of RCTs need to be supported by real-world clinical situations. Therefore, our clinical data showing the efficacy of three CGRP mAbs in a total of 228 patients with CM or EM up to 12 months are of great clinical importance.
In a real-world setting study including 77 CM patients receiving erenumab, fremanezumab or galcanezumab, the three types of CGRP mAbs were well tolerated. Among the 90 trials of CGRP mAbs, the most frequent adverse effects included constipation (26.7%) and injection site reactions (10.0%) (14). Similarly, our study showed adverse responses in 12.3% of patients, with injection site reactions being the most common though generally mild in severity. Only 0.9% of patients discontinued CGRP mAbs due to adverse reactions. Because the CGRP molecule is involved in regulating the immune and cardiovascular systems, there was concern that clinical studies involving patients with various systemic diseases would show a higher incidence of adverse reactions than RCTs (39), but this was not the case in our study.
One of the limitations of our study was that the number of patients was not constant over a 12-month period due to discontinuation and reduction in available patient data at each visit. We instructed patients to keep a daily headache diary to minimize recall bias and found that most patients complied. However, we cannot rule out the possibility that a few patients failed to keep a daily headache diary if their headache continued for two or three days. We also did not include acute medication use per month or the number of headache days per month; instead, we focused on changes in the number of monthly migraine days, which better reflects the degree of disability caused by migraine.
Conclusion
This 12-month, real-world study confirmed the efficacy and safety of three different types of CGRP mAbs (erenumab, galcanezumab and fremanezumab) for prophylactic treatment of patients with EM or CM. The relationship between osmophobia and CGRP mAb efficacy requires further study.
Clinical implications
CGRP mAbs significantly reduced MMDs compared to baseline over 12 months. The overall ≥50% response rate was high, being 48.2% at three months, 61.0% at six months, and 73.7% at 12 months. The presence of osmophobia and fewer baseline MMDs contributed to ≥50% responders at three, six and 12 months. The three types of CGRP mAbs (erenumab, galcanezumab and fremanezumab) were comparably effective in reducing MMDs. CGRP mAbs were also effective in subgroups of patients with DTT migraine and previous CGRP mAb users.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231177649 - Supplemental material for Effectiveness of three calcitonin gene-related peptide monoclonal antibodies for migraine: A 12-month, single-center, observational real-world study in Japan
Supplemental material, sj-pdf-1-cep-10.1177_03331024231177649 for Effectiveness of three calcitonin gene-related peptide monoclonal antibodies for migraine: A 12-month, single-center, observational real-world study in Japan by Keisuke Suzuki, Shiho Suzuki, Tomohiko Shiina, Muneto Tatsumoto, Hiroaki Fujita, Yasuo Haruyama and Koichi Hirata in Cephalalgia
Footnotes
Acknowledgments
The authors thank Drs. Kei Funakoshi, Tomoko Komagamine, and Hiroki Onuma, Department of Neurology, Dokkyo Medical University, for their assistance with this study. The authors thank Ms. Sanae Tani and Ms. Kaori Soma, Department of Neurology, Dokkyo Medical University, for their help with this study.
Author contributions
All authors contributed to the acquisition and interpretation of data for this study. KS drafted the manuscript. KS and YH performed the statistical analysis. SS, TS, MT, HF, YH and KH performed a critical review of important intellectual content. All authors approved the final manuscript.
Availability of data and materials
The datasets from this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. KS received lecture fees from Eli Lilly Japan, Daiichi Sankyo, Amgen and Otsuka Pharmaceutical Co., Ltd., during the conduct of the study. SS received lecture fees from Eli Lilly Japan, Daiichi Sankyo, Amgen and Otsuka Pharmaceutical Co., Ltd., during the conduct of the study. TS received lecture fees from Eli Lilly Japan, Daiichi Sankyo, Amgen and Otsuka Pharmaceutical Co., Ltd., during the conduct of the study. MT received lecture fees from Eli Lilly Japan, Daiichi Sankyo, Amgen and Otsuka Pharmaceutical Co., Ltd., during the conduct of the study. HF and YH have nothing to disclose. KH received lecture fees from Eli Lilly Japan, Daiichi Sankyo, Amgen, Sawai Pharmaceutical Co., Ltd. and Otsuka Pharmaceutical Co., Ltd. during the conduct of the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
