Abstract
Background
This post-hoc analysis was conducted to evaluate the effect of erenumab on monthly migraine days, monthly migraine attacks, and attack duration in patients with episodic migraine to investigate whether erenumab actually prevents the occurrence of migraine attacks and/or shortens them.
Methods
We conducted a post-hoc analysis of the data from the STRIVE study, in 955 patients with episodic migraine. Relative changes from baseline to mean over months 4, 5 and 6 of the double-blind treatment phase in monthly migraine days, monthly migraine attacks and mean migraine attack duration were assessed.
Results
Erenumab reduced monthly migraine days and monthly migraine attacks compared with placebo in a similar way. Erenumab had only a minor impact on shortening the duration of migraine attacks.
Conclusion
These post-hoc analyses demonstrate that the decrease in monthly migraine days by erenumab is mainly driven by a reduction in the frequency of monthly migraine attacks and to a much lesser extent by shortening the duration of migraine attacks.
Trial registration: This study is registered at ClinicalTrials.gov (NCT02456740)
Introduction
Erenumab is a fully human monoclonal antibody that selectively targets and blocks the canonical calcitonin gene-related peptide receptor with no affinity for any related receptors (1). The efficacy, tolerability and safety of erenumab 70 mg and 140 mg administered once per month by subcutaneous injection have been demonstrated in both episodic migraine and chronic migraine (2–6). In the randomized clinical trials (RCTs) evaluating erenumab for prevention of episodic migraine, the primary efficacy outcome was based on monthly migraine days (MMD), be it the reduction in MMD from baseline (3,4), or the proportion of patients achieving a ≥50% reduction in MMD from baseline (5). This is in line with the new clinical trial guidelines for the prevention of episodic migraine, which recommend a reduction in MMD from baseline as the primary outcome, and the ≥50% responder rate for the reduction of migraine days as an alternative primary endpoint (7).
The decrease in MMD after administration of erenumab observed in RCTs in patients with episodic migraine is often assumed to be a consequence of a decrease in the number of monthly migraine attacks (MMA). However, given that migraine attacks can last 2–3 days, the decrease in MMD could theoretically also be a consequence of reduction in attack duration, with no impact on the number of MMA. In this case we should see a disconnect between the decrease in number of MMD and number of MMA. A reduction in MMA would demonstrate a real preventive effect by preventing the occurrence of migraine attacks and not only shortening the duration of attacks. The effect of erenumab on MMA, and the relative effects of erenumab on MMD and MMA has not been previously evaluated. In the current post-hoc analysis of STRIVE study (4), we therefore evaluated and compared the relative effect of two doses of erenumab (70 mg and 140 mg) vs placebo on MMD and MMA as well as duration of migraine attacks in patients with episodic migraine.
Methods
We conducted a post-hoc analysis on data from the STRIVE study (NCT02456740), which was the largest randomized, double-blind, placebo-controlled, Phase 3 study of erenumab in patients with episodic migraine (N=955). In this study, patients were randomly assigned to receive a subcutaneous injection of either erenumab 70 mg or 140 mg, or placebo once monthly for 6 months. The detailed study design (Figure 1), patient inclusion and exclusion criteria have been published previously (4).

Study design.4
A migraine day was defined as any calendar day on which the patient had onset, continuation, or recurrence of a qualified migraine headache as recorded in the electronic diary. Any calendar day on which acute migraine-specific medication (i.e. triptan or ergotamine) was used was also counted as a migraine day. A qualified migraine headache was defined as a migraine (with or without aura) lasting at least 30 minutes and meeting at least one of the following criteria (a and/or b): a) ≥2 of the following pain features: unilateral, throbbing, moderate to severe, exacerbated with exercise/physical activity; b) ≥1 of the following associated symptoms: nausea and/or vomiting, photophobia and phonophobia.
A migraine attack was defined as an episode of any qualified migraine headache or migraine specific medication intake. A migraine attack that was interrupted by sleep, or temporarily remitted, and then recurred within 48 hours was considered as one attack. Also, an attack treated successfully with medication but with relapse within 48 hours and a migraine attack lasting more than 48 hours was counted as one attack.
The monthly average migraine attack duration was expressed in days and was calculated by dividing MMD by MMA for every individual patient (if both MMD and MMA are zero, then the migraine attack duration was set to zero).
Outcome measures assessed included change from baseline (4-week baseline phase) to the last 3 months of assessment (mean over Months 4, 5 and 6) in MMD, MMA and monthly average migraine attack duration. Pre-specified exploratory (change from baseline in MMA) or post-hoc analyses (change from baseline in monthly average migraine attack duration) were conducted using the efficacy analyses set. The efficacy analysis set included patients who received at least one dose of erenumab or placebo and had at least one post baseline measurement for migraine days per month during the double-blind treatment phase (DBTP), analyzed according to randomly assigned trial regimen (4). To facilitate the comparison between MMD and MMA which was the primary comparison in our analysis, change from baseline to the last 3 months (mean over months 4, 5, and 6) in MMD were also reported here. The detailed statistical analyses for reporting the primary, secondary and exploratory endpoints have already been published (4). For the post-hoc endpoints, the adjusted analysis of least square mean (LSM) difference versus placebo utilized a generalized linear mixed model which included treatment, visit, treatment by visit interaction, stratification factors region and prior/current treatment with migraine prophylactic medication, and baseline value as covariates and assumed a first-order autoregressive covariance structure. P-values are nominal without multiplicity adjustment.
Results
During the DBTP, statistically significant greater reduction in MMD, MMA and monthly average migraine attack duration were observed in erenumab groups as compared with placebo (Table 1). The mean MMD at study baseline was 8.3 days. The absolute and percent change from baseline in MMD was −1.8 days (−22%) with placebo, −3.2 days (−39%) with erenumab 70 mg, and −3.7 days (−44%) with erenumab 140 mg over the final 3 months of the DBTP (Table 1). The mean MMA at baseline was 5.1 for placebo, 5.2 for the erenumab 70 mg, and 5.2 for the erenumab 140 mg group. The absolute and percent change from baseline in MMA was −1.3 attacks (−26%) with placebo, −1.9 attacks (−40%) with erenumab 70 mg, and −2.2 attack (−43%) with erenumab 140 mg over the final 3 months of the DBTP (Table 1). The average migraine attack duration at baseline was 1.7 days for placebo, 1.6 for the erenumab 70 mg, and 1.7 for the erenumab 140 mg group. The absolute and percent change from baseline in average migraine attack duration was −0.01 day (−1%) with placebo, −0.11 day (−7%) with erenumab 70 mg, and −0.13 day (−8%) with erenumab 140 mg over the final 3 months of the DBTP (Table 1).
Clinical responses for MMD and MMA over the final 3 months of the double blind treatment phase (mean over Months 4–6)a.

aThe analysis included patients who underwent randomization, received at least one dose of the randomly assigned trial regimen, and had at least one post-baseline measurement for migraine days per month during the double-blind treatment phase (efficacy analysis set). bLeast-squares mean changes from baseline in MMD, MMA and migraine attack duration during the DBTP are shown. cThe adjusted analysis utilizes a generalized linear mixed model which includes treatment, visit, treatment by visit interaction, stratification factors region and prior/current treatment with migraine prophylactic medication, and baseline value as covariates and assuming a first-order autoregressive covariance structure. dP-values are nominal without multiplicity adjustment.Abbreviations: CI, confidence interval; DBTP, double-blind treatment phase; LSM, least square mean; MMA, monthly migraine attacks; MMD, monthly migraine days, SD, standard deviation; SE, standard error.
The quite similar percentage reduction from baseline in MMD and MMA are as follows: for 70 mg erenumab the change in MMD is −39% versus a −40% change for MMA; and for 140 mg erenumab the change in MMD is −44% versus a −43% change for MMA (Figure 2). The reduction from baseline in average migraine attack duration was relatively small with −7% and −8% reduction compared to baseline for erenumab 70 mg and erenumab 140 mg, respectively.
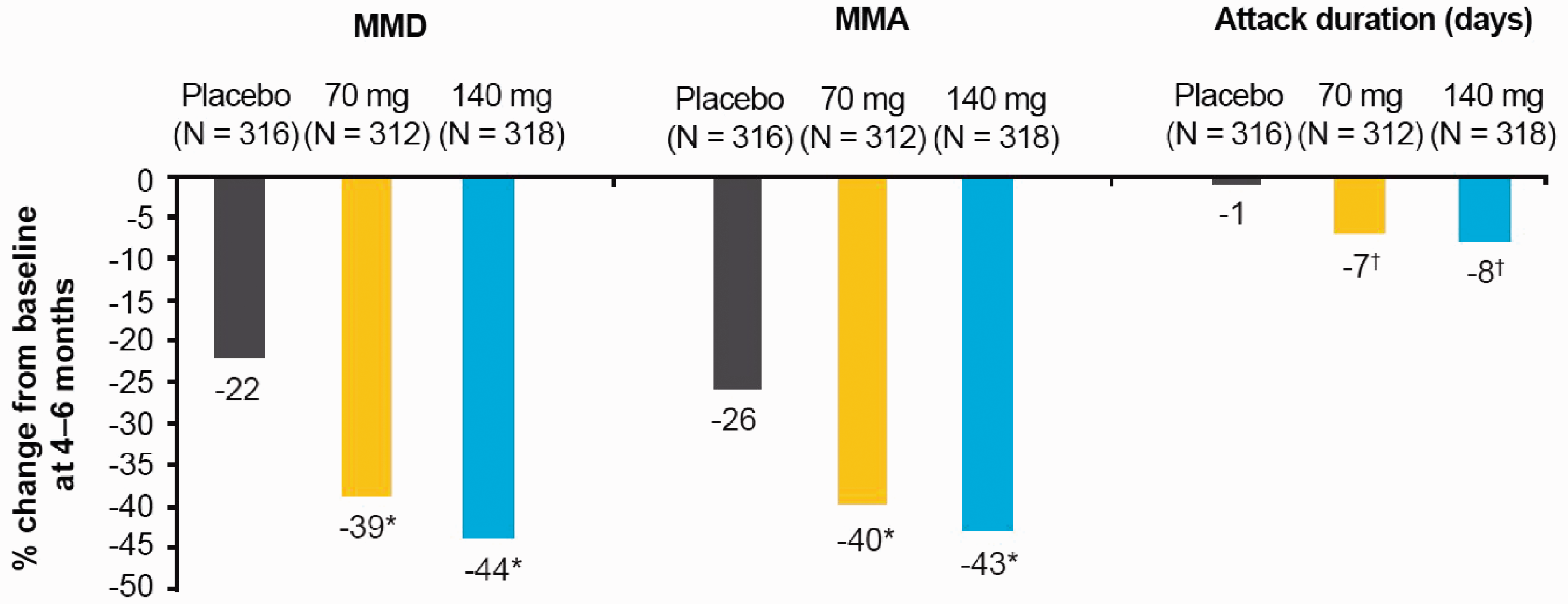
Percentage reduction from baseline in mean MMD, MMA and monthly average migraine attack duration (days) over the last three months of the DBTP (efficacy analysis set).
Discussion
The results of our post-hoc analysis of the STRIVE study in patients with episodic migraine, showed that erenumab decreases both MMD and MMA in a similar manner. Migraine attacks may last longer than a migraine day, and the definition of migraine attacks in the erenumab studies included those which were interrupted by sleep or treated successfully with a triptan. In our analysis we observed a similar reduction in migraine days and migraine attacks with erenumab compared to placebo. We also observed only a minor effect of erenumab on the duration of migraine attacks. This indicates that in prevention of episodic migraine, erenumab exerts an impact on mechanisms underlying the generation of migraine attacks, i.e. a true migraine preventive mode of action, and to a much lesser extent an effect on the duration of attacks as seen with acute migraine attack treatments.
It will be interesting to see the results of an analysis of MMD versus MMA in the randomized trial investigating the efficacy of atogepant and other gepants in the prevention of episodic migraine to see if they also have an effect on reducing MMA (8).
The strength of this analysis is the large patient number and the stringent way in which migraine days and migraine attacks were defined as per protocol. A limitation of this study is the fact that this is in part a post-hoc analysis. The assessment of migraine attack duration was not a predefined exploratory endpoint of this trial, we therefore used both MMD and MMA to calculate the attack duration in days for each individual patient. It has to be noted that assessing migraine attack duration has several obvious limitations, as it is difficult to assess accurately because some attacks will start or resolve in the night or when a patient is asleep, in addition the attack duration will be impacted by the use of acute headache treatment. Given the limitations, caution is needed in interpreting the results on migraine attack duration. Dedicated studies are needed to assess the effect of erenumab on headache duration using better defined criteria for headache duration (9).
Analyses from other monoclonal antibodies could support our findings. Of note are the results of a Phase 2 study with galcanezumab 150 mg (once every two weeks treatment for 12 weeks) which reported a decrease in monthly headache days and MMA versus placebo by LSM (standard error) of 1.2 (0.41) and 0.8 (0.29) respectively (10).
Conclusion
The current post-hoc analysis of a large RCT with erenumab suggests that the MMD and MMA decrease in parallel, which supports that erenumab prevents the occurrence of migraine attacks and not only shortens the duration of migraine attacks.
Clinical implications
This post-hoc analysis of the randomized, double-blind, placebo-controlled, parallel-group, Phase 3 STRIVE study was conducted to evaluate the effect of erenumab on the frequency of migraine days, migraine attacks and duration of migraine attacks in patients with episodic migraine. Over the last three months of the double-blind phase, both monthly migraine days and monthly migraine attacks were reduced by a similar magnitude. The duration of migraine attacks was only reduced by a small amount. These findings suggest that erenumab has a preventive action in episodic migraine by preventing the occurrence of migraine attacks.
Footnotes
Acknowledgements
We thank the study investigators and patients for their participation and commitment to this work. Medical writing support was provided by Nisha Narayanan and Preethi Bheereddy, Novartis Healthcare Pvt. Ltd, India.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Amgen Inc., Thousand Oaks, CA, USA. Erenumab is co-developed by Novartis and Amgen.
Previous presentations
The data was presented at the American Headache Society (AHS), Virtual Scientific Meeting, June 13, 2020
